Abstract
Musculoskeletal (MSK) infections encompass acute and chronic osteomyelitis, subacute lesions and soft tissue infections, including cellulitis, pyomyositis, abscesses, tenosynovitis, septic bursitis and necrotising fasciitis. Early recognition is critical, as clinical findings may be nonspecific and delayed diagnosis can result in deformity, chronic sinus tracts or sepsis. Imaging is pivotal for diagnosis and management. Radiographs are first-line but often detect late changes. Computed tomography (CT) is valuable for cortical destruction, sequestra, and gas. Magnetic resonance imaging (MRI) is highly sensitive for marrow and soft tissue involvement. Key imaging clues improve diagnostic confidence: The penumbra sign in Brodie’s abscess, the ghost sign in diabetic foot osteomyelitis, and paradiscal marrow involvement with smooth-walled paravertebral abscesses in spinal tuberculosis (TB). Nuclear medicine and fluorodeoxyglucose positron emission tomography/CT (FDG PET/CT) complement structural imaging in detecting active disease and monitoring therapy. Soft tissue infections require a multimodal approach. Ultrasound (US) rapidly detects superficial collections and guides drainage. CT delineates fascial thickening, gas and deep abscesses. MRI precisely defines muscular, fascial and tendon involvement, allowing accurate characterisation of cellulitis, pyomyositis, abscesses, tenosynovitis and necrotising fasciitis, while detecting joint or bone extension. This article provides a concise overview of imaging in MSK infections, highlighting diagnostic pearls that enhance accuracy and guide timely management.
Keywords
Introduction
Musculoskeletal (MSK) infections, including osteomyelitis, septic arthritis, soft tissue infections and prosthetic joint infections, pose significant diagnostic and therapeutic challenges. Imaging plays a central role in early detection, disease characterisation and treatment monitoring.
Magnetic resonance imaging (MRI) has emerged as the modality of choice due to its high sensitivity in detecting bone marrow and soft tissue changes.[1] Diffusion-weighted imaging (DWI) and contrast-enhanced sequences improve diagnostic accuracy, especially in differentiating necrosis and abscess. Computed tomography (CT) remains valuable for detecting gas, cortical destruction and guiding interventions.[2] Ultrasound (US) is useful in superficial infections, while positron emission tomography/CT (PET/CT) and PET/MRI offer promising functional imaging for complex or recurrent cases.[3]
Despite these advances, several issues remain. Diagnostic criteria and imaging protocols are inconsistent, limiting comparability across studies. Data is limited in specific populations (e.g., diabetic foot, children) and for newer techniques such as PET/MRI. Many studies are retrospective with small sample sizes. Challenges persist in distinguishing infection from sterile inflammation or postoperative changes, especially in the presence of metallic hardware.
Debates continue regarding optimal imaging pathways, the value of newer PET tracers, the role of non-contrast MR techniques and standardisation efforts such as the MSK Infection Reporting and Data System (MSKI-RADS).[4] Emerging issues include the validation and clinical utility of MSKI-RADS and the integration of hybrid imaging modalities.[3]
Future research should focus on multicenter prospective studies with standardised protocols, development of quantitative imaging biomarkers, validation of PET/MRI and new tracers, and AI-assisted tools for image interpretation. Emphasis must also shift toward patient-centred outcomes, ensuring imaging improvements leading to better clinical care, reduced morbidity and cost-effective management.
Methodology
This narrative review was conducted to summarise the current role of MSK radiology in the evaluation of bone and soft tissue infections, highlighting advances, ongoing debates, gaps in knowledge and future research directions. A comprehensive literature search was performed using databases including PubMed, Embase, and Google Scholar, covering articles published between 2010 and 2025. Keywords and MeSH terms included:
‘MSK infections’, ‘osteomyelitis’, ‘soft tissue infection’, ‘MRI’, ‘CT’, ‘US’, ‘PET/CT’, ‘PET/MRI’, ‘diabetic foot infection’, ‘imaging in infection’, ‘MSKI-RADS’ and ‘molecular imaging in MSK infections’.
Articles were selected based on relevance to MSK radiology and infection, including original studies, systematic reviews, meta-analyses and expert consensus statements. Priority was given to high-quality studies, prospective data and recent advances. Studies not in English or unrelated to MSK infection imaging were excluded.
MRI is a key modality in MSK radiology due to its excellent soft tissue contrast and ability to detect early changes such as bone marrow oedema, abscesses and soft tissue involvement. It is particularly useful in complex infections such as diabetic foot and spinal osteomyelitis.[5] However, MRI is relatively expensive, time-consuming and contraindicated in some patients with metallic implants or claustrophobia.
CT plays an important complementary role, especially in identifying cortical bone destruction, sequestra, and gas within soft tissues.[6] It is fast, widely available and useful for image-guided procedures. Despite these strengths, CT offers limited soft tissue contrast and involves radiation exposure.
US is commonly used for evaluating superficial soft tissue infections, joint effusions and guiding aspirations or drainages. It is safe, portable and cost-effective, with no radiation exposure. However, its utility is limited in deep or intraosseous infections and is highly operator-dependent.
Positron emission tomography, often combined with CT or MRI (PET/CT or PET/MRI), provides metabolic information that aids in the diagnosis of chronic or complex infections, including those involving prosthetic devices.[7] It can identify sites of occult infection and distinguish between active and inactive disease. Nonetheless, PET imaging can be limited by high cost, limited availability, radiation exposure (in PET/CT) and difficulty in differentiating infection from sterile inflammation.
Together, these imaging modalities offer complementary strengths and are selected based on clinical scenario, anatomical location, patient factors and the need for anatomical versus functional information.
Discussion
Bone Infections
Osteomyelitis refers to an infectious inflammatory process involving the bone marrow, typically caused by microbial pathogens. The term is derived from the Greek words: Osteo (bone), myelo (marrow) and itis (inflammation). Osteomyelitis is categorised by duration into acute (<2 weeks), subacute (2–6 weeks), and chronic (>6 weeks) forms.
In children, hematogenous spread predominates, most often due to S. aureus. In adults, contiguous spread and direct inoculation are more common and infections are frequently polymicrobial. Less common organisms include Salmonella (particularly in sickle cell disease), Pseudomonas aeruginosa (notably in intravenous drug users and post-traumatic cases) and Mycobacterium tuberculosis (TB) (causing skeletal TB).
The disease demonstrates a male predominance, with incidence rising with advancing age.[8,9] This increase is thought to be related to the higher prevalence of comorbidities such as diabetes mellitus and peripheral vascular disease in older men.
Acute osteomyelitis predominantly affects children and younger adults, whereas chronic osteomyelitis is more often seen in older individuals. The tibia is the most frequently affected site due to its subcutaneous location and susceptibility to trauma. Other commonly involved bones include the femur, phalanges and calcaneum.
Acute Osteomyelitis
Osteomyelitis imaging findings vary by infection route. Hematogenous spread affects children’s long bone metaphyses and adults’ spines, showing medullary lucencies and periosteal reactions. Contiguous spread, commonly seen in adults, especially in cases such as diabetic foot ulcers, typically begins with soft tissue changes such as cellulitis or abscess formation and gradually progresses to involve the bone and medullary cavity.
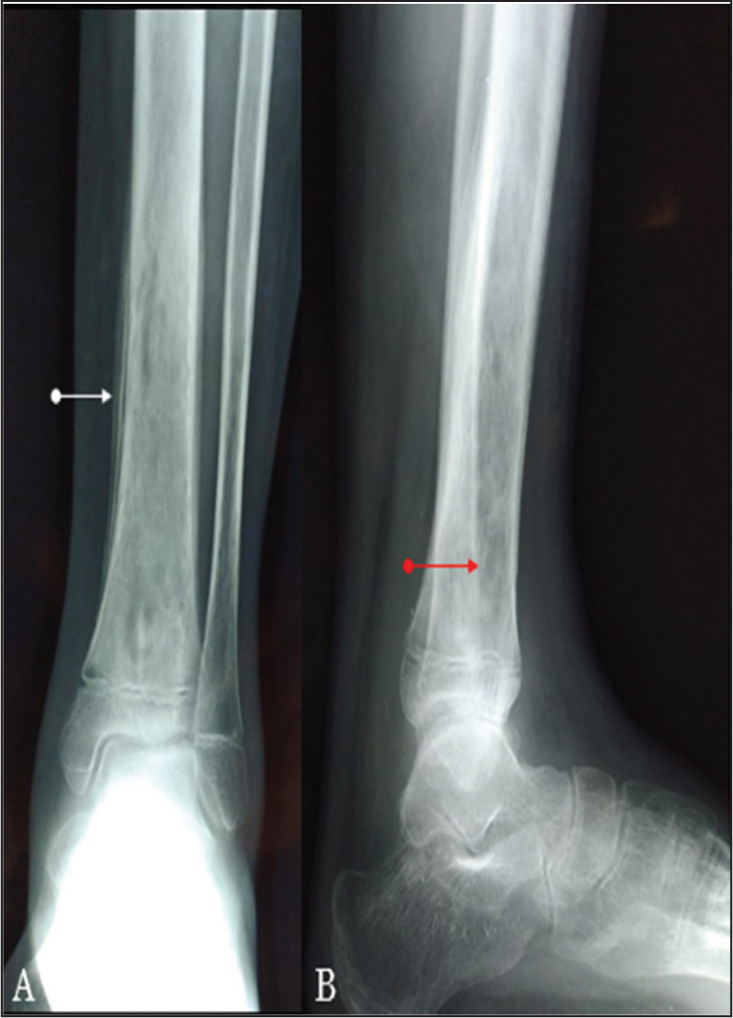
Plain radiographs are the initial imaging choice for suspected osteomyelitis due to availability and cost, though early sensitivity is low. Soft tissue swelling, subcutaneous gas or foreign bodies may appear within days. Bony changes such as cortical destruction, permeative marrow lucency, and periosteal reaction typically appear after 1–2 weeks [Figure 1], requiring 30%–50% bone loss.[10] In children, thin cortices may lead to subperiosteal abscess formation.
AP (A) and lateral (B) radiograph of both bones of the lower limb showing permeative bone destruction (red arrow) with lamellated periosteal reaction (white arrow) with a wide zone of transition in a case of acute osteomyelitis
US detects soft tissue abnormalities earlier than radiographs. In children, periosteal thickening is easily visualised. It also guides drainage of collections and biopsies, but is limited to superficial tissues due to bony shadowing.
CT better visualises bony changes, especially cortical destruction and identifies rare intramedullary air foci, a highly specific sign of osteomyelitis. It delineates infection extent and guides aspiration or biopsy. Contrast-enhanced CT improves soft tissue assessment.
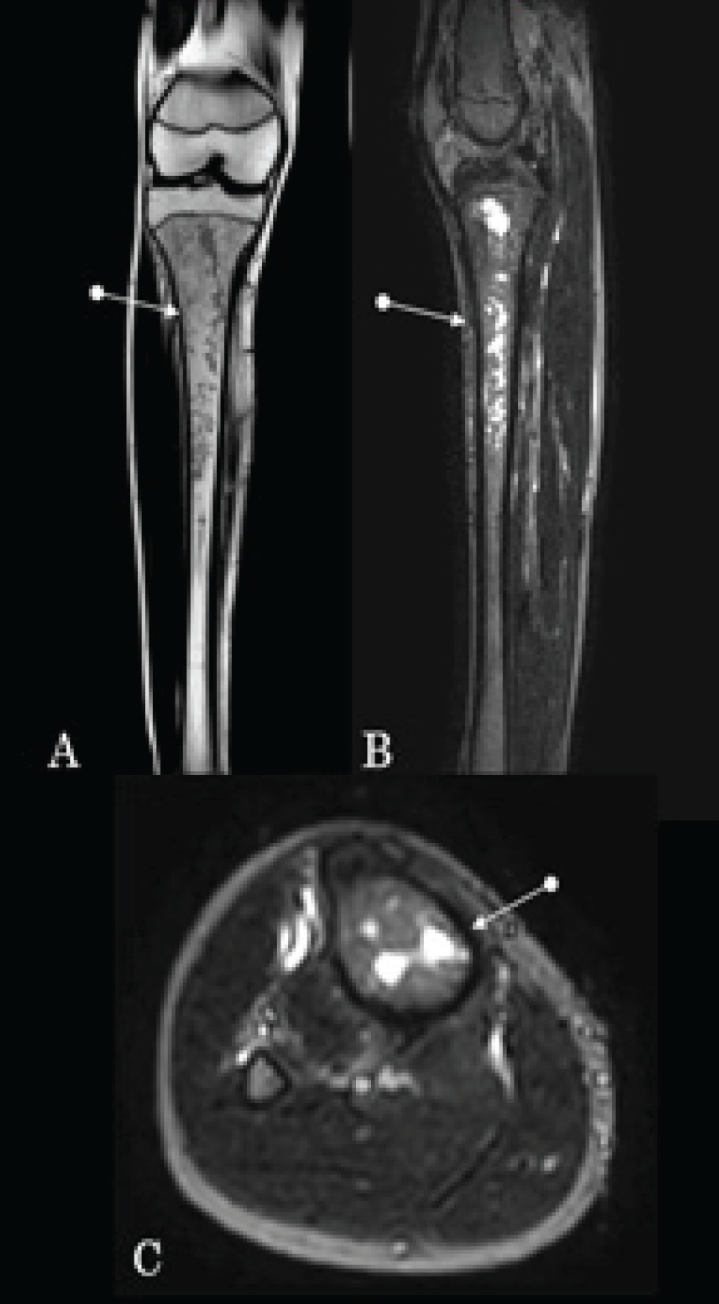
MRI is the most sensitive early imaging modality for osteomyelitis, often within the first week. T1-, T2- and fat-suppressed/ Short tau inversion recovery (STIR) sequences show marrow oedema (T2/STIR hyperintense, T1 hypointense). MRI delineates sinus tracts, abscesses and necrotic zones with contrast [Figure 2].[11]
Coronal T1 (A), sagittal (B) and axial (C) STIR images depicting marrow signal changes involving the upper and mid part of the tibia with areas of liquefactive necrosis consistent with acute osteomyelitis in the given clinical picture in a different patient
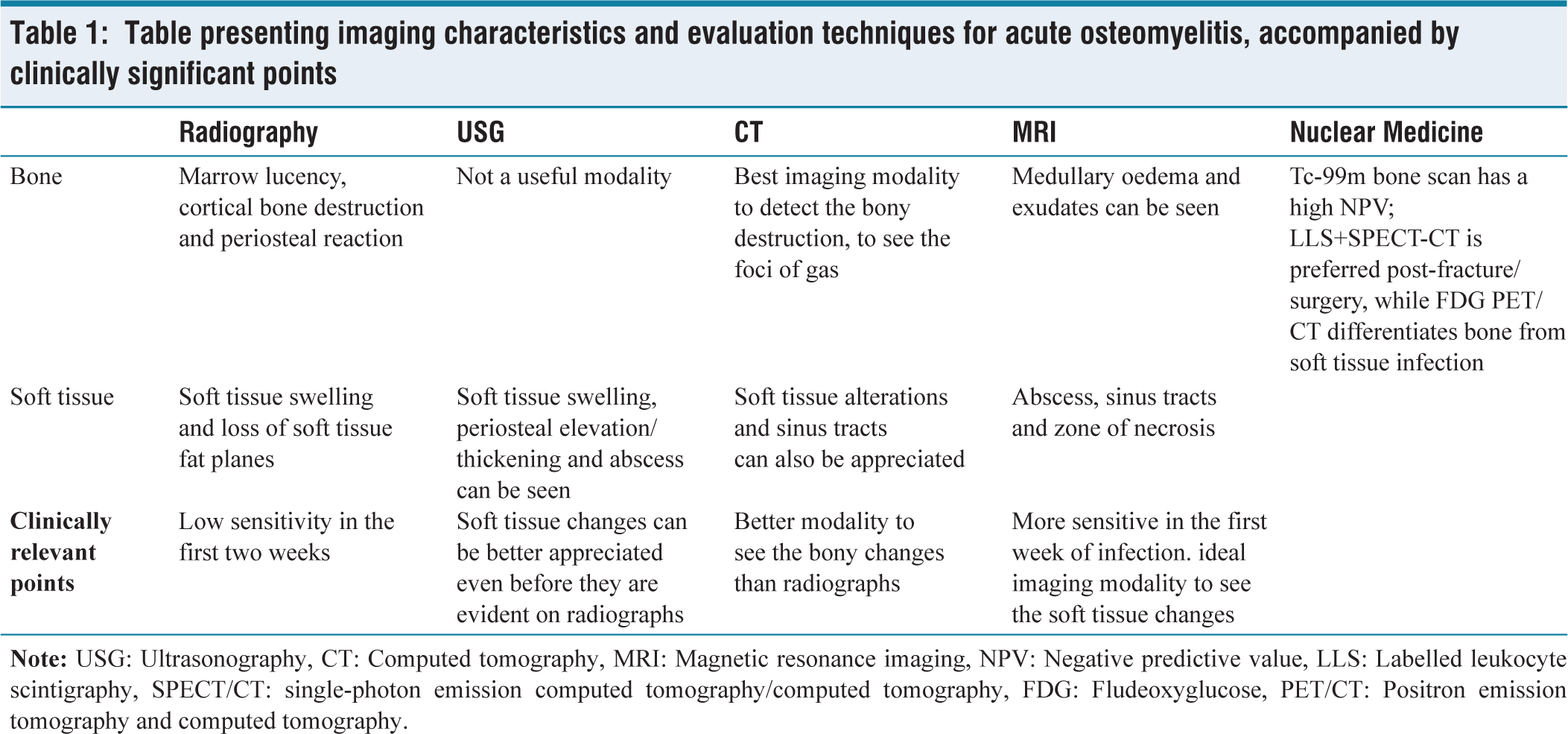
Radionuclide bone scintigraphy can detect osteomyelitis within 24–48 hours of symptom onset and has a high negative predictive value, making it useful in equivocal cases. Common techniques include Tc-99m bone scans, gallium citrate, labelled leukocyte scans and single-photon emission CT, each offering functional assessment of infection. Fluorodeoxyglucose (FDG) PET/CT combines metabolic and anatomical information, improving differentiation between infectious and noninfectious processes and aiding precise localisation. While bone scans are highly sensitive, they lack specificity due to uptake in fractures, arthritis or degenerative changes. Hybrid PET/CT further enhances diagnostic confidence by correlating metabolic activity with exact anatomical structures [Table 1].
Table presenting imaging characteristics and evaluation techniques for acute osteomyelitis, accompanied by clinically significant points
Chronic Osteomyelitis
The progression of acute osteomyelitis to chronic disease usually results from delayed or inadequate treatment. Chronic osteomyelitis is characterised by necrosis of bone due to impaired vascular supply and the subsequent formation of sequestrum, involucrum and cloaca.
Plain radiographs often demonstrate cortical thickening and sclerosis, with adjacent lytic lesions involving the marrow. A lucent sinus tract may occasionally be identified. The US is valuable for evaluating soft tissue complications such as abscesses, fistulae or sinus tracts in chronic disease.
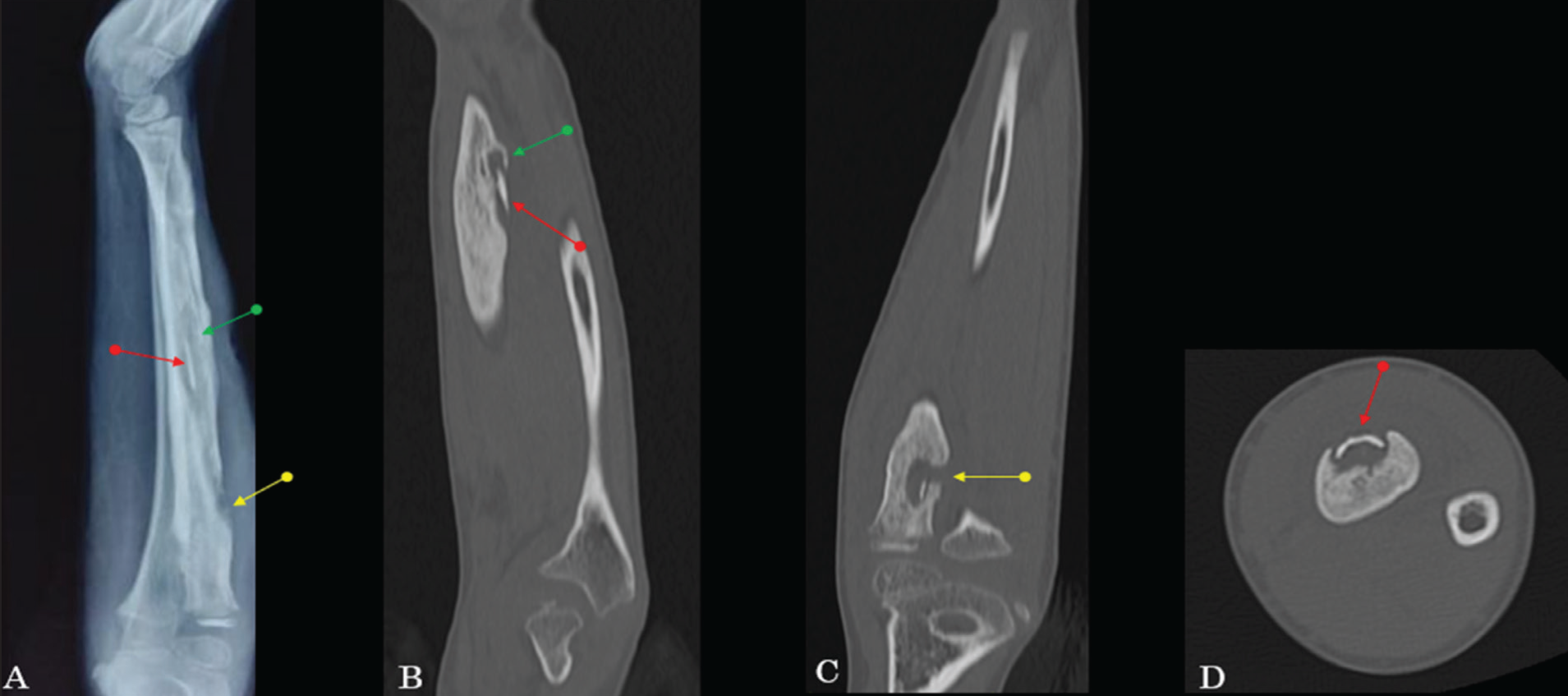
CT plays a major role in identifying sequestra, which may be obscured on radiographs. It also delineates the involucrum, a dense shell of periosteal bone encasing the infection. Cloacae, which represent openings in the involucrum allowing pus or necrotic material to drain externally, can also be well demonstrated [Figure 3]. When cloacae extend to the skin surface, they form sinus tracts.
Lateral radiograph (A), sagittal (B), Coronal (C) and axial (D) CT of forearm showing sequestrum (red arrow), involucrum (green arrow) and cloaca (yellow arrow) in chronic osteomyelitis involving radius
On MRI, sequestra appear hypointense on T1 and T2. Linear T2 hyperintense tracks may represent cloacae extending from the involucrum to the skin, forming sinus tracts. Contrast-enhanced MRI helps assess infection extent, localise sequestra and distinguish active infection from fibrous scar tissue, aiding differentiation of acute on chronic versus inactive chronic osteomyelitis.[12,13]
Radionuclide imaging is useful in differentiating chronic osteomyelitis from soft tissue infections and in detecting active disease in complex or post-surgical cases [Table 2, Figure 4].
Table outlining key imaging characteristics and the role of various modalities in assessing chronic osteomyelitis

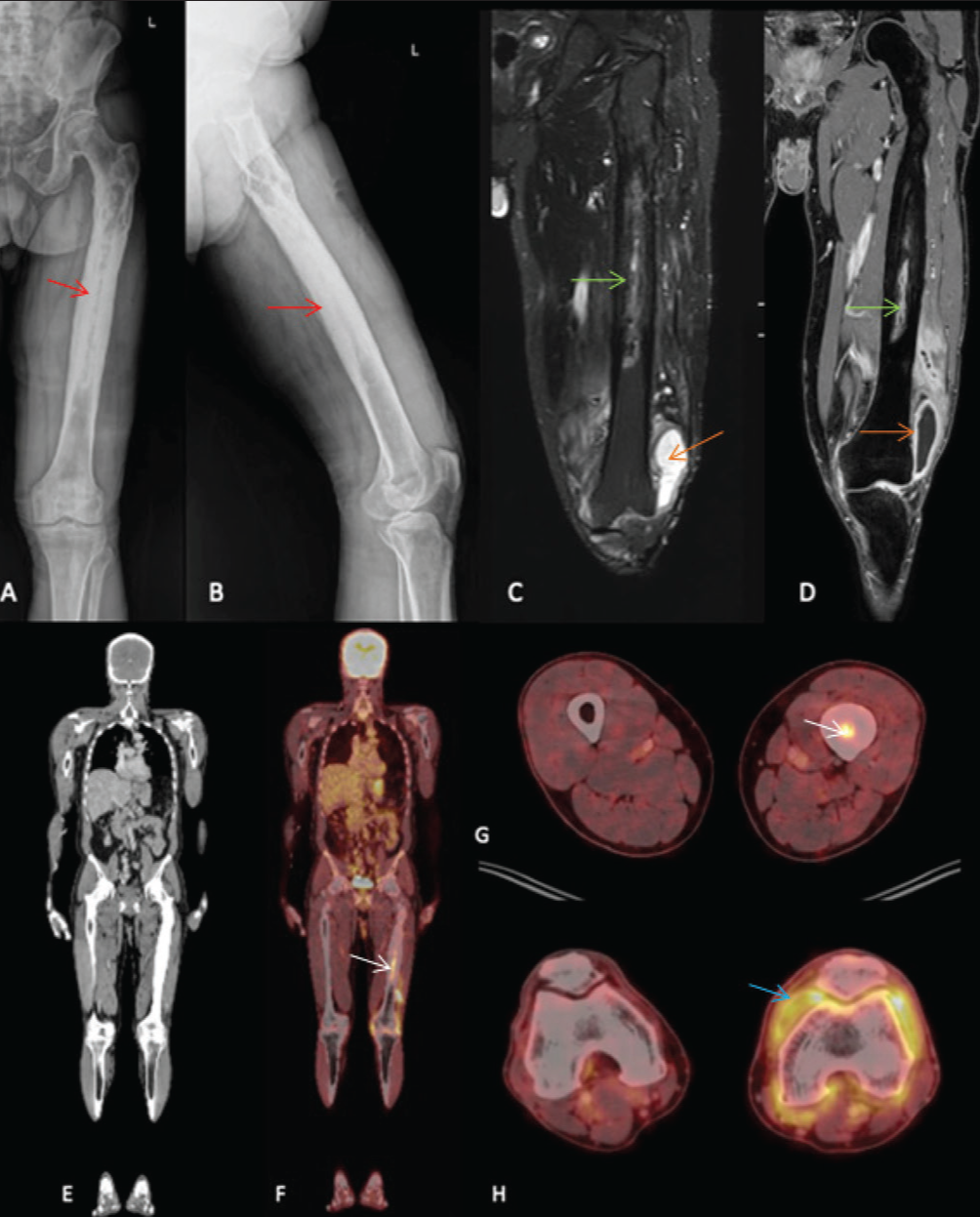
Frontal and lateral radiographs (A, B) of the left femur in a patient with thigh and knee pains showing diffuse cortical thickening and medullary sclerosis (red arrows), suggesting chronic osteomyelitis. Coronal STIR and T1 fat sat post contrast MRI images (C, D) show patchy intramedullary oedema with a thin collection (green arrows). An associated synovial collection (orange arrows) is noted at the knee, showing peripheral enhancement on contrast study, suggesting infective synovitis. FDG PET CT was performed. Coronal whole body CT (E), Fused PET CT coronal (F) and axial images (G, H) show active linear area of increased FDG avidity within the marrow region of mid shaft of left femur (white arrows), FDG non avid extensively thickened and irregular cortex of shaft of left femur and FDG avid diffuse synovial thickening (blue arrow) in left knee joint, suggesting acute on chronic osteomyelitis in left femur with acute infective synovitis of left knee joint
Complications of untreated or poorly managed osteomyelitis include osteonecrosis, septic arthritis from joint extension and growth disturbances in children due to physeal involvement. Chronic sinus tracts may rarely undergo malignant transformation (Marjolin’s ulcer or squamous cell carcinoma, incidence 0.3%–1.6%).
Subacute Osteomyelitis (Brodies Abscess)
Brodie’s abscess is a localised intraosseous collection within granulation tissue and sclerotic bone, usually causing minimal systemic response. Radiographs show an oval or multiloculated lytic lesion along the long axis with reactive sclerosis and minimal periosteal reaction; a lucent tract toward the physis is pathognomonic [Figure 5A].
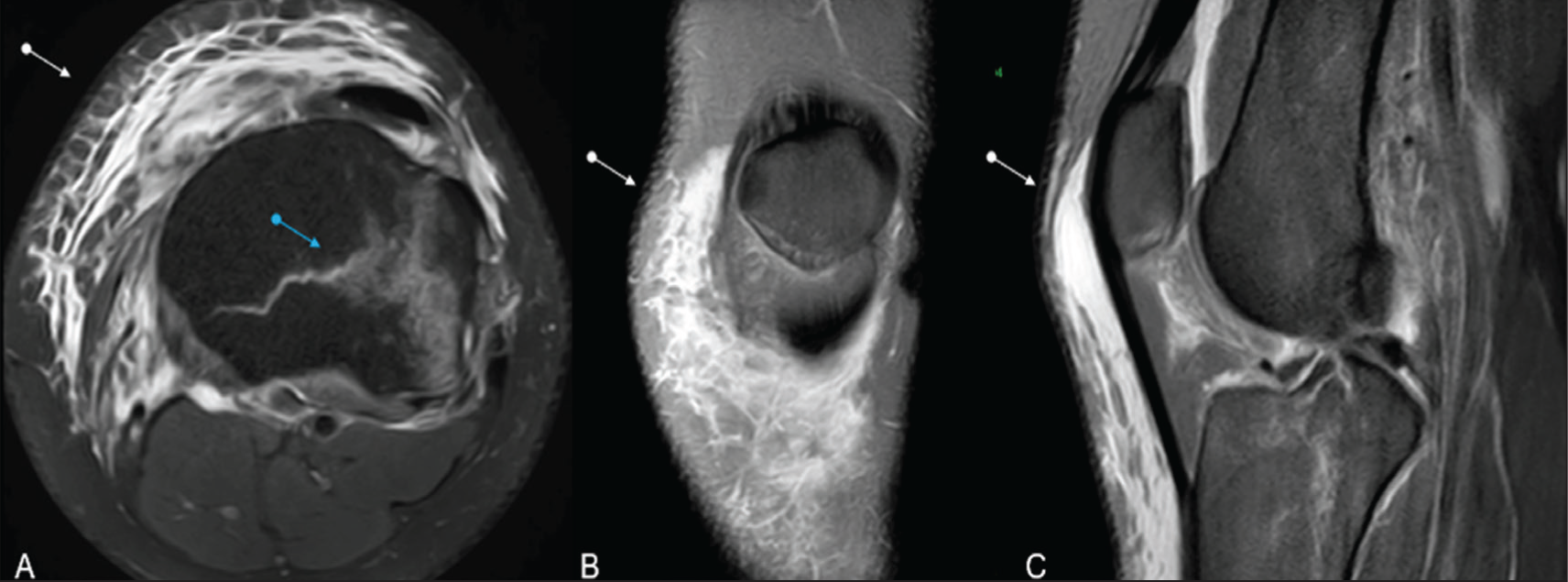
Frontal radiograph (A) of the knee depicting a lytic lesion with a sclerotic rim involving the proximal tibia (white arrows). Coronal STIR (B and C) images showing intra-osseous abscess with extension to the adjacent muscular plane (yellow arrows) depicting Brodie’s abscess
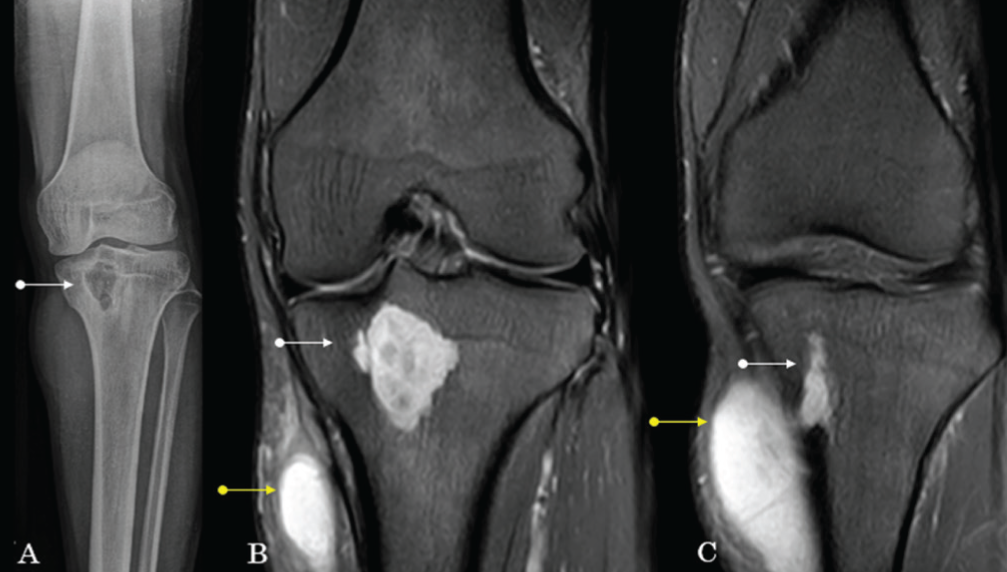
CT delineates margins and detects sequestra, showing a hypodense cavity with dense ossified rim and possible periosteal reaction. MRI is most informative, with the T1 penumbra sign: Necrotic core, granulation tissue, sclerotic rim and peripheral oedema. T2 shows concentric high, low and low signal, while post-contrast images reveal peripheral ring enhancement [Figure 5B and 5C]. Bone scintigraphy demonstrates increased but nonspecific uptake.[14]
Several conditions can mimic osteomyelitis clinically and on imaging, including gout, Charcot’s arthropathy, Ewing’s sarcoma, cellulitis, septic arthritis, bone metastases and TB.
Diabetic Foot Osteomyelitis (DFO)
In diabetics, osteomyelitis typically arises from contiguous spread via chronic ulcers, commonly affecting the calcaneum, first and fifth metatarsals and first distal phalanx. Differentiating diabetic foot osteomyelitis (DFO) from Charcot arthropathy, which affects the midfoot without infection, is a key diagnostic challenge.
Radiographs show soft tissue defects or skin irregularities from ulcers, with possible gas tracking into bone, cortical erosion or focal osteopenia [Figure 6]. CT highlights cortical erosions, bone loss and marrow density changes, aiding diagnosis and defining infection extent.[15]
Oblique and AP radiograph of foot (A, B) showing neuropathic type of involvement of third and fourth metatarsophalangeal joints (white arrows) with atrophic pattern (red arrow) and reactive sclerosis involving the proximal phalanges (Yellow arrows). AP and lateral radiographs (C, D) of the foot showing retrocalcaneal soft tissue swelling (white arrows), florid periosteal reaction, sclerosis and new bone formation in the posterior half of the calcaneum (orange arrow). Skin irregularity representing an ulcer (green arrow). Corresponding MRI images, coronal PD SPIR (E), axial DWI (F) (b value 400), sagittal STIR (G), show bone marrow oedematous changes involving the calcaneum (White arrows), which shows restricted diffusion (F) with associated ulcer (green arrow)
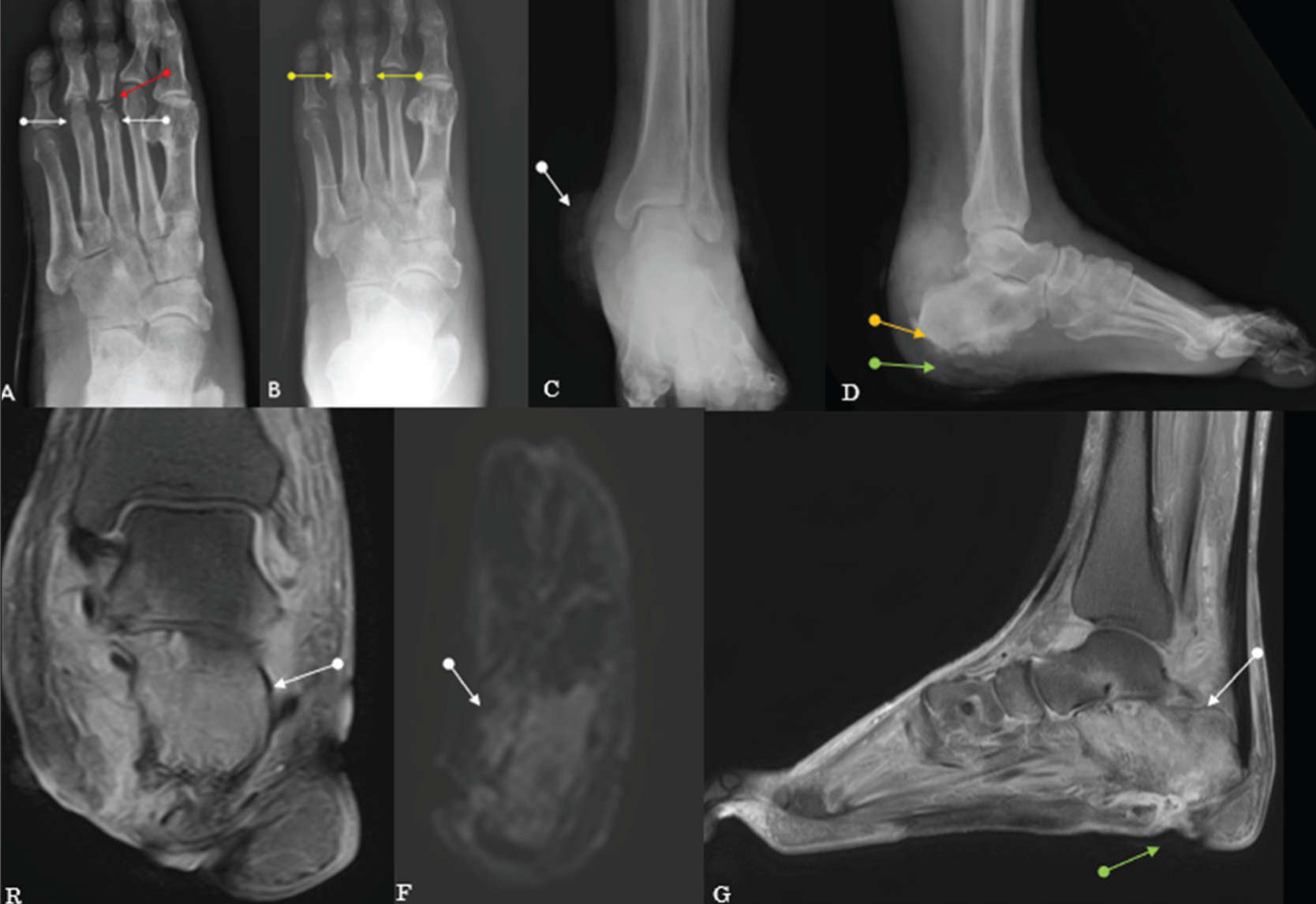
MRI is the most sensitive modality for diagnosing DFO, with a reported sensitivity of approximately 90% and specificity of around 79%. However, specificity decreases in patients with prior surgery, trauma or coexisting Charcot’s arthropathy. Bone biopsy with histopathology and microbiology remains the gold standard for diagnosis. On MRI, bone marrow oedema appears hyperintense on T2-weighted and STIR sequences [Figures 6E, 6F and 6G].
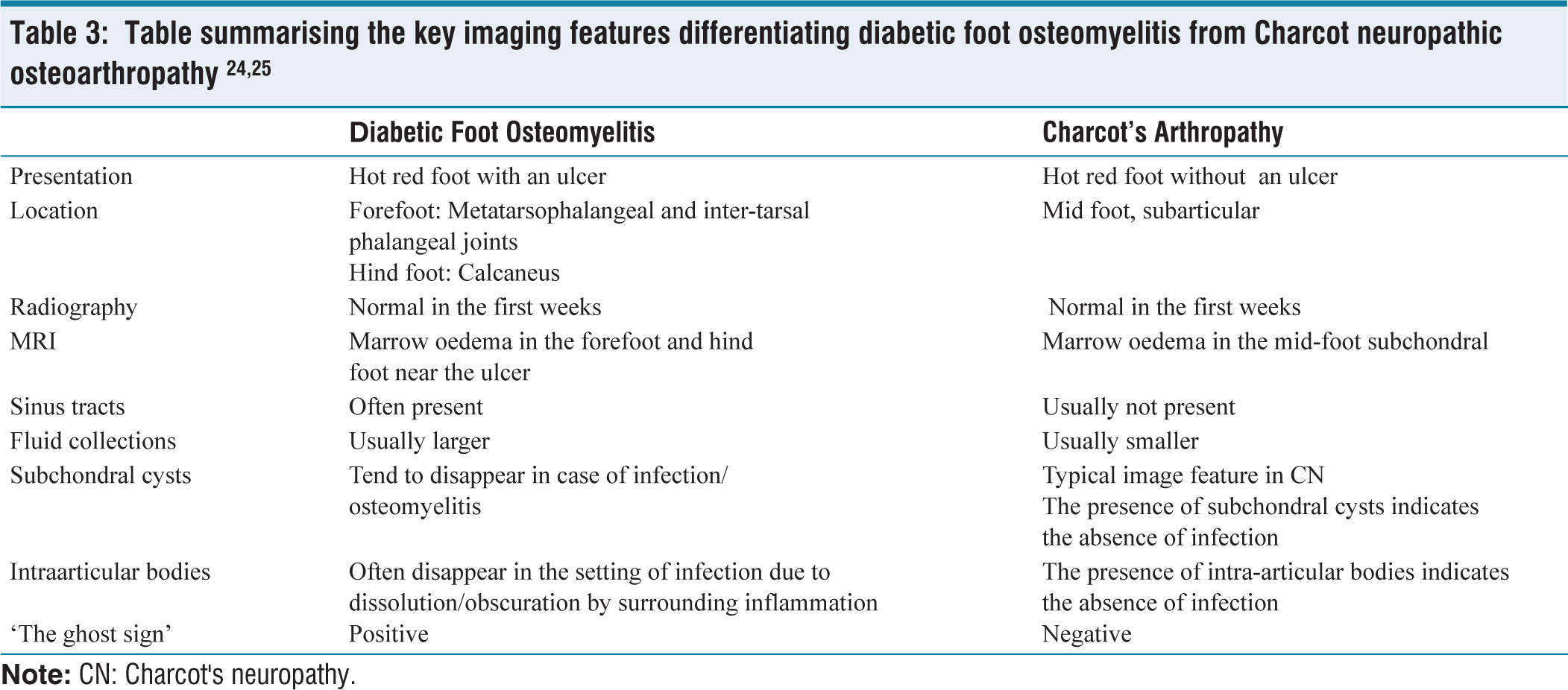
The ‘ghost sign’ refers to the apparent disappearance of bone due to marrow infiltration, cortical erosion and hypo-intensity, causing the bone to blend with surrounding soft tissues on T1-weighted images, while residual structure may still be seen on T2-weighted images. On apparent diffusion coefficient (ADC) maps, infected bone loses its normal hypointense ‘black bone’ signal and appears diffusely hyperintense relative to adjacent healthy bone. It is highly suggestive of osteomyelitis in the diabetic foot and helps differentiate it from neuropathic (Charcot) arthropathy[16] [Table 3].
Table summarising the key imaging features differentiating diabetic foot osteomyelitis from Charcot neuropathic osteoarthropathy 24,25
The role of FDG PET in DFO is evolving, with studies suggesting potential utility, though its precise role is yet to be firmly established.
Tubercular Spondylitis
Spinal TB, caused by Mycobacterium TB, spreads hematogenously to vertebral cancellous bone. Arterial dissemination leads to paravertebral involvement, while venous Batson’s plexus spread may produce central or skip lesions. The thoracolumbar junction is most commonly affected in spinal TB, though cervical and sacral regions may also be involved. Patterns include paradiscal (most common), central (via venous plexus), anterior (subligamentous) and posterior elements (rare).
Clinically, patients present with chronic back pain, neurological deficits from cord compression and occasionally fever, night sweats or weight loss.[17]
Radiographs show changes late (~30%–40% bone loss), including endplate irregularity, disc space narrowing, paravertebral shadows, cortical destruction and kyphosis; useful for baseline and follow-up (AP + lateral). CT better depicts cortical destruction, including fragmentary, osteolytic, subperiosteal and sclerotic patterns and detects paravertebral/psoas calcifications suggestive of TB, while guiding biopsy; soft tissue evaluation is limited.
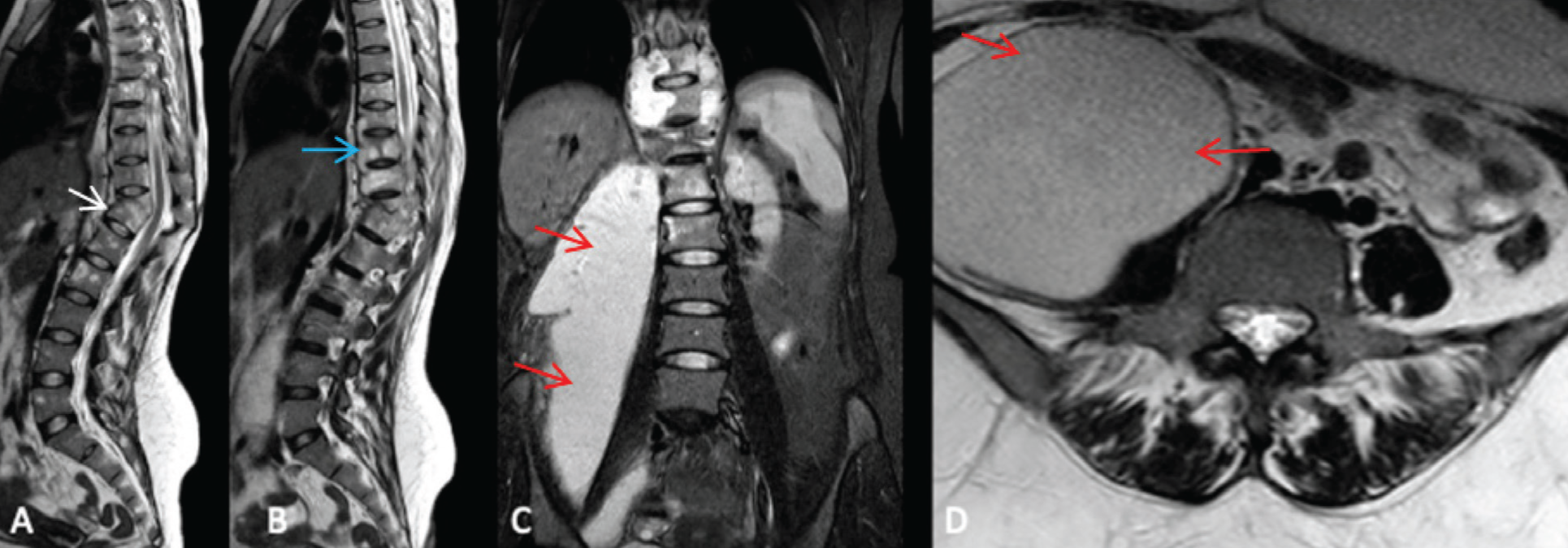
MRI (sensitivity ~96%, specificity ~93%) detects early marrow involvement, skip lesions, epidural/paraspinal abscesses and cord compression [Figure 7]. Findings include T1 hypointensity, T2/STIR hyperintensity, reduced disc height with T2 hyperintense disc signal and enhancement, large smooth-walled abscesses (often with calcifications) and less disc destruction than pyogenic infections. Atypical forms include isolated abscesses, posterior element disease, concentric collapse and ivory vertebra. Whole-spine MRI is essential to identify multifocal or skip lesions and occult neural compression; post-contrast imaging confirms active disease and abscess extent.[18]
Sagittal T2 (A, B), coronal STIR (C) and axial T2 (D) images depicting infective spondylitis at T11-T12 level (white arrow) with endplate destruction, focal kyphosis, anterior epidural component causing mild indentation over lower thoracic cord, anterior subligamentous spread of infection (blue arrow) with linear collection extending from T6 to L1 levels, associated perivertebral soft tissue collections and large right psoas abscess (red arrows), in a case of tubercular spondylitis
Bone scans and FDG PET/CT detect multifocal/atypical disease and assess treatment response, but specificity is limited.
Complications of tubercular osteomyelitis include kyphosis and gibbus deformity, epidural abscess causing cord compression and neurological deficits, spinal instability with chronic pain and rare malignant transformation of chronic sinuses.
Spine (spondylitis) is the most common (~50%) site for osteoarticular TB, followed by peripheral joints (hip, knee, ankle), osteomyelitis, tenosynovitis and bursitis.[19] Radiologic findings in osteoarticular TB evolve from early soft tissue swelling, juxta-articular osteoporosis and effusion to advanced joint space narrowing, peripheral erosions and bone destruction. MRI is most sensitive, showing marrow oedema, synovial thickening, abscesses and sinus tracts. CT detects cortical destruction and calcified abscesses; radiographs remain useful for chronic changes.[20]
Treatment of bone infections includes broad-spectrum antibiotics for acute osteomyelitis (with clindamycin or vancomycin for Methicillin-resistant Staphylococcus aureus [MRSA]), surgical debridement for chronic cases, conservative antibiotic therapy for Brodie’s abscess and standard multidrug antitubercular chemotherapy for spinal TB.
Soft Tissue Infections
Soft tissue infections are common, but physical examination may not always reveal their severity or extent, such as spread to deeper tissues, bone or joints; in these cases, radiological imaging is essential for accurate diagnosis and assessment. The role of MSK radiology in diagnosing and managing various soft tissue infections is outlined below.
Cellulitis
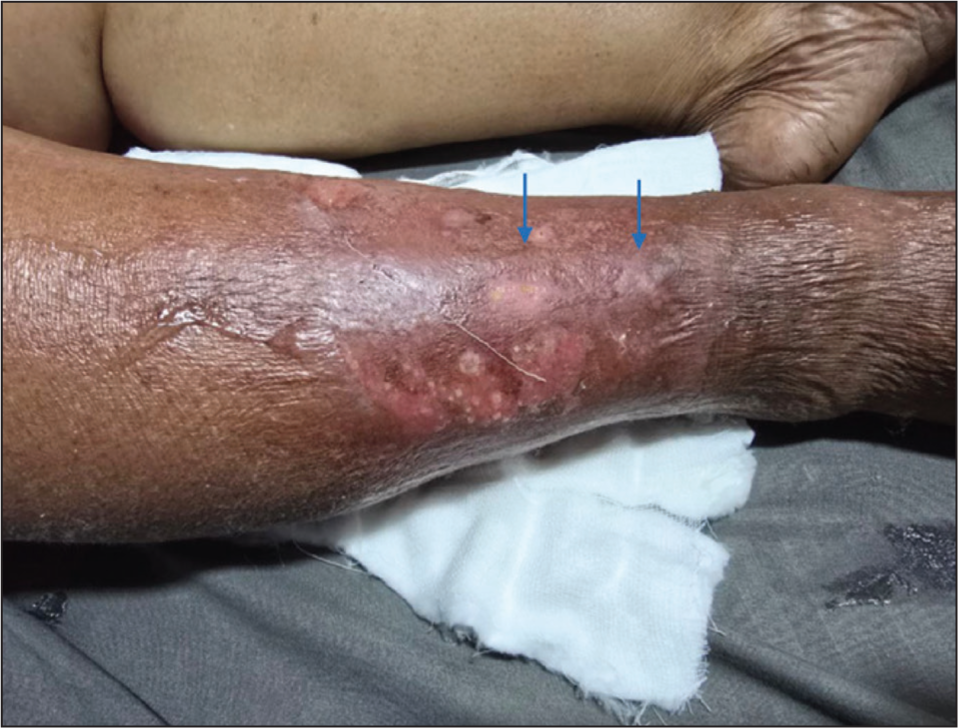
This condition refers to a non-necrotising infection of the subcutaneous tissue and superficial fascia, without involvement of the deep fascia or underlying muscular structures. Common risk factors include soft tissue ulcers in patients with diabetes or vascular insufficiency, as well as retained foreign bodies. Clinically, patients typically present with localised erythema, tenderness, swelling and warmth, often accompanied by systemic features such as fever and chills [Figures 8 and 10].
Shows a clinical picture of a case of cellulitis-erythema and swelling involving the lower half of the right leg
Radiographs may demonstrate nonspecific findings such as diffuse soft tissue oedema, obliteration and displacement of fat planes and occasionally the presence of a radiopaque foreign body (if present).
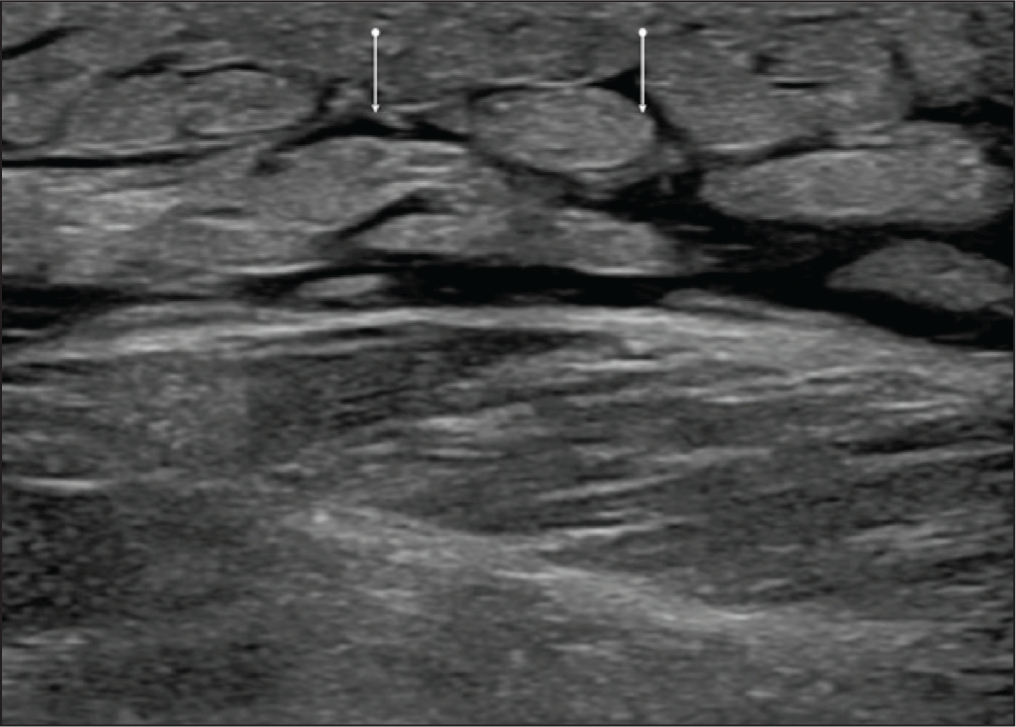
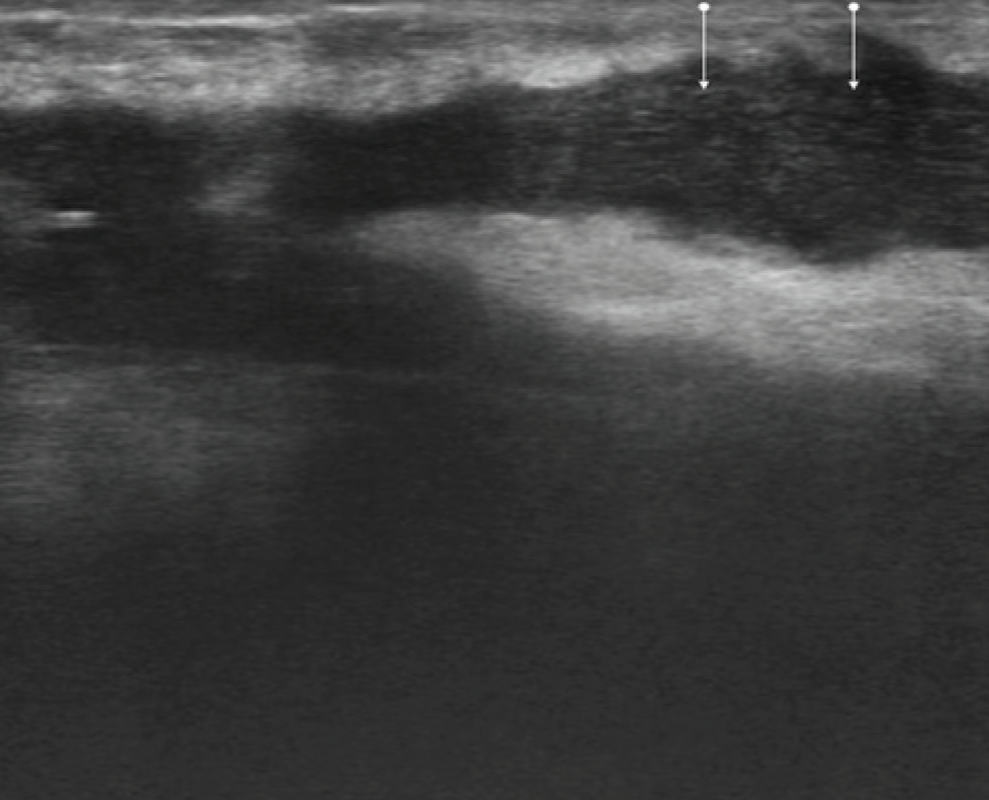
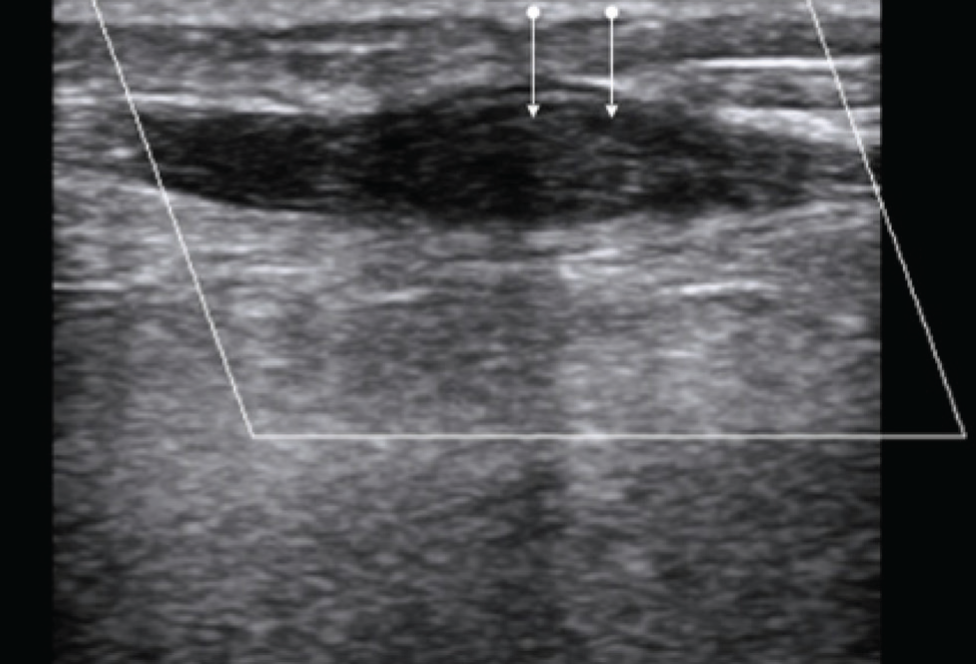
USG reveals subcutaneous tissue oedema with hypoechoic stranding insinuating between echogenic fat lobules, producing the characteristic ‘cobblestone’ appearance [Figure 9]. On colour Doppler evaluation, increased vascularity is observed, which is more specific for an underlying inflammatory process.[21]
Ultrasound image in a case of cellulitis, showing characteristic cobblestone appearance
Axial (A), coronal (B) and sagittal (C) PD STIR MRI images of the knee in a patient with no recent injury, showing significant interstitial subcutaneous soft tissue oedema in the supra-patellar region representing cellulitis changes
Infectious Myositis (Pyomyositis)
Pyomyositis refers to the formation of an intramuscular abscess. Clinically, it typically presents initially with fever and, if left untreated, may progress to septicaemia and multi-organ failure.
CT demonstrates heterogeneous attenuation within an enlarged muscle, along with effacement of the fat planes between the affected muscle and adjacent soft tissues. MRI reveals an enlarged muscle with a hypointense signal on T1-weighted and hyperintense signal on T2-weighted sequences, associated with focal fluid collection or abscess. Additional findings may include overlying skin and fascial thickening, as well as subcutaneous oedema. Post-contrast images typically show irregular peripheral enhancement of the abscess wall. Significant diffusion restriction on DWI is also noted.
Soft Tissue Abscess
This condition refers to a focal collection of pus within soft tissues resulting from the immune response to pathogenic microorganisms.
USG demonstrates a focal cystic lesion containing internal echoes, with no vascularity on Doppler imaging [Figure 11]. CT reveals a fluid-density soft tissue mass, which may contain gas. In chronic abscesses, peripheral calcifications can occasionally be seen. MRI depicts a homogeneous or heterogeneous hypointense signal on T1-weighted sequences and a hyperintense signal on T2-weighted sequences, with variable wall characteristics. DWI shows restricted diffusion, with ADC values typically ranging from 0.30 to 0.80 × 10⁻³ mm²/s [Figure 12]. Post-contrast images demonstrate thick, irregular wall enhancement of the abscess, along with surrounding oedema.
Ultrasound image in a case of soft tissue abscess, linearly oriented collection with internal echoes
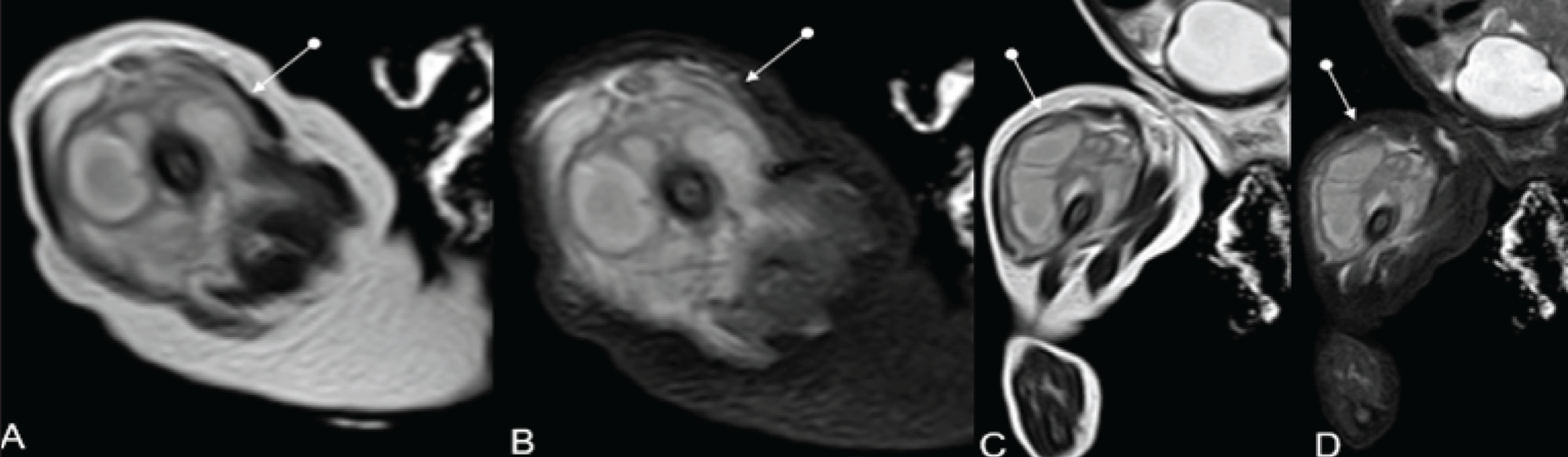
Axial T2, STIR (A, B), coronal T2 and STIR (C, D) MRI images of the right leg of a child depicting a multi-lobulated collection with internal septations involving the muscular plane of the right thigh, representing an abscess
Infectious Tenosynovitis
It refers to thickening and inflammation of the tendon and synovial tissue surrounding it. Radiographs show soft tissue swelling in the region of the tendon. USG shows fluid and oedema in the tendon sheath. MRI shows tendons and synovium show hyper-intense signal on T2W. In case of long-standing infections, tendons may rupture. In a few cases, debris may be present in tendon sheaths. Contrast study shows thickening and enhancement of the synovium surrounding the fluid within the tendon sheath.
Septic Bursitis
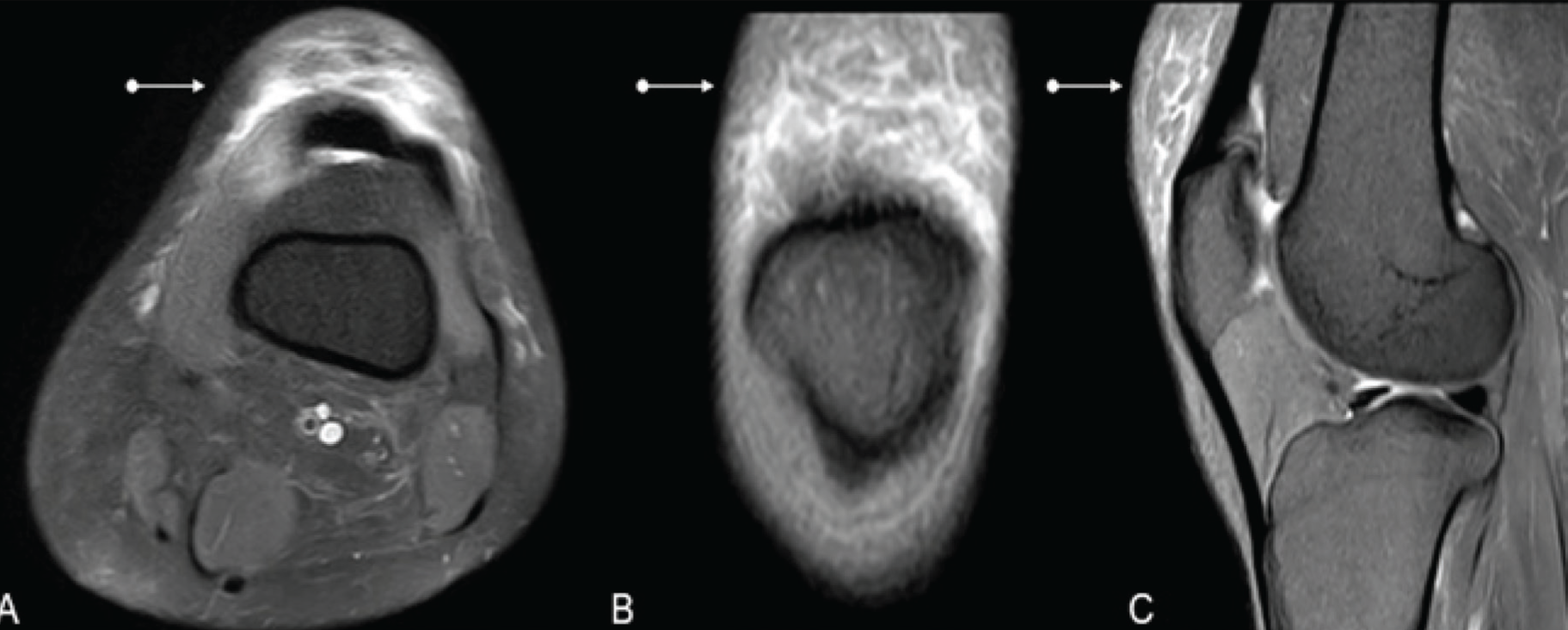
This condition refers to infection involving the synovial-lined bursae. USG is useful in detecting bursal fluid collections and serves as a reliable modality for image-guided aspiration. MRI often demonstrates a distended bursa filled with fluid, appearing hypointense on T1-weighted images and hyperintense on T2-weighted images, similar to fluid signal characteristics. Associated findings may include underlying bone erosion or marrow oedema [Figure 13]. Post-contrast sequences typically reveal a thick, enhancing rim surrounding the bursal fluid, with or without internal debris. It is most commonly encountered at the prepatellar, iliac fossa, ischio-gluteal, trochanteric, subdeltoid, subacromial and olecranon bursae.
Axial (A), coronal (B) and sagittal (C) MRI PD SPIR images depicting subcutaneous collection and adjacent oedema involving the anterior aspect of the knee. Aspiration and fluid analysis confirmed an infective nature, representing infective bursitis. Fracture seen involving the lateral tibial plateau (blue arrow)
Necrotising Fascitis
It refers to inflammation involving the fascia with associated necrotising changes [Figure 14]. USG demonstrates thickening of fascial planes with associated turbid fluid collection and the presence of gas, seen as echogenic foci with posterior dirty shadowing. On MRI, the deep fascia appears thickened, showing hypointense signal on T1-weighted and hyperintense signal on T2-weighted images. Gas is often poorly detected on MRI. Post-contrast sequences typically demonstrate enhancement of the adjacent inflamed soft tissues.[22]
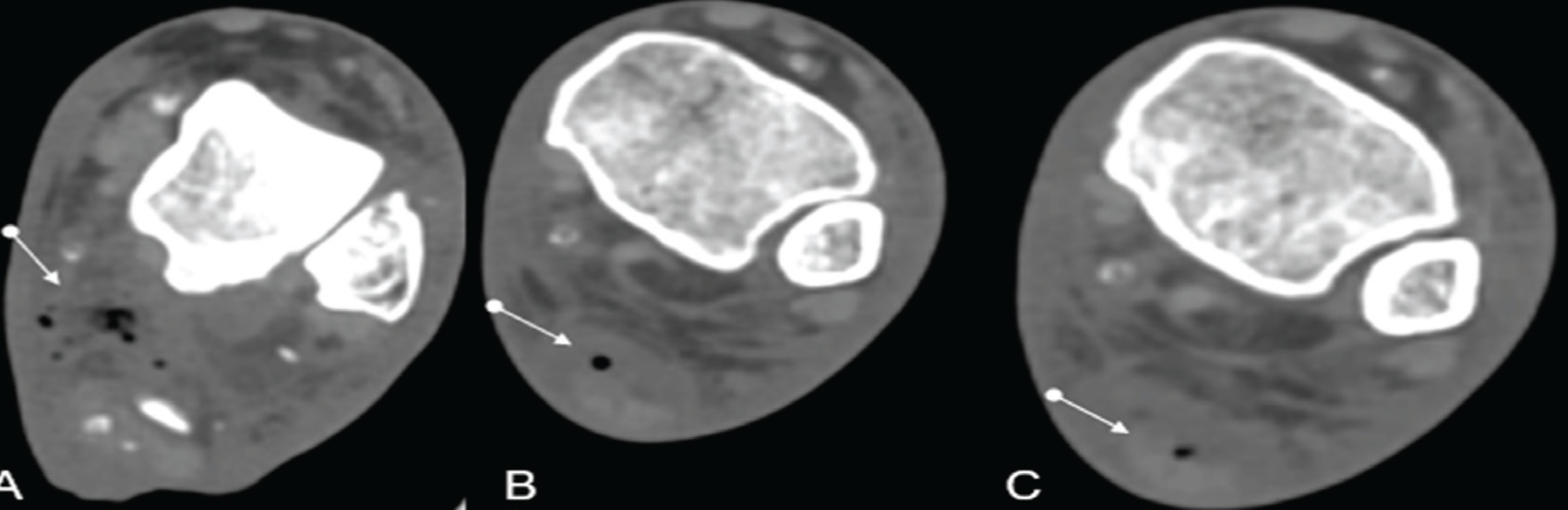
CT axial sections of lower limb angiogram (non-traumatic patient) showing necrotising fasciitis evident by multiple air foci with soft tissue oedema along the tendo-achilles and adjacent soft tissues of the left leg. This patient exhibited clinical signs of infection in the leg and foot
Soft Tissue Infection Mimics
Mimics of soft tissue infections are diverse non-infectious conditions and other conditions that can appear similar to skin and soft tissue infections, such as deep vein thrombosis, gout, allergic reactions, drug reactions and various forms of inflammation or injury, all of which can cause symptoms such as redness, swelling, warmth, and pain. Rarely, tumours can also mimic soft tissue infections.
Superficial thrombophlebitis refers to an inflammatory disorder of the superficial veins with coexistent venous thrombosis. It most commonly involves the lower limbs, particularly the great saphenous vein (60%–80%) or, less frequently, the small or short saphenous vein (10%–20%). Clinically, patients present with a reddened, warm, inflamed and tender area along the course of a superficial vein. A palpable cord is often detected and some patients may also demonstrate surrounding oedema or associated pruritus, which can mimic cellulitis. On colour Doppler imaging, the affected superficial vein appears dilated and distended with hypoechoic thrombus and shows a lack of compressibility [Figure 15].[23]
Doppler image of left great saphenous vein (GSV) showing dilated GSV distended with hypoechoic thrombus, wall thickening and absent vascularity on Doppler, suggestive of superficial venous thrombosis with thrombophlebitis
Limitations of the Study
This narrative review is limited by the inherent constraints of its design, including the lack of systematic inclusion criteria and quantitative synthesis. While a comprehensive literature search was conducted, there is a possibility of selection bias, as some relevant studies may have been missed due to publication language restrictions or indexing limitations. Furthermore, much of the available literature is heterogeneous in methodology, imaging protocols and diagnostic criteria, making direct comparisons challenging. Many of the cited studies are retrospective, single-centre or based on small sample sizes, which may limit generalisability. The rapid evolution of imaging technology, particularly in PET/MRI and artificial intelligence applications, also means that findings and recommendations may quickly become outdated. Additionally, due to space constraints, this review may not fully cover emerging imaging techniques or less commonly used modalities in specific subpopulations such as paediatrics or immunocompromised patients.
Conclusion
MSK radiology plays a critical role in the diagnosis and management of bone and soft tissue infections. MRI remains the cornerstone modality due to its superior soft tissue resolution and early detection capabilities, while CT, US and nuclear imaging techniques offer important complementary information. Despite technological advances, challenges persist in standardising imaging protocols, interpreting findings in complex clinical scenarios and distinguishing infection from sterile inflammation or postoperative changes. Emerging modalities such as PET/MRI and the introduction of structured reporting systems such as MSKI-RADS represent promising steps forward but require further validation. Future research should focus on multicenter prospective studies, the development of quantitative imaging biomarkers and the integration of artificial intelligence to enhance diagnostic accuracy and clinical decision-making. Overall, a multimodal and patient-centred imaging approach remains essential for optimising outcomes in MSK infections.
Clinical Significance for Practice
This review provides a practical overview of current imaging approaches in MSK infections, helping clinicians choose appropriate modalities based on clinical needs. While its strength lies in summarising diverse imaging options and highlighting emerging trends such as MSKI-RADS and hybrid imaging, the narrative format limits its ability to provide quantitative comparisons or systematic evidence. Despite its limitations, this review emphasises the importance of a multimodality imaging approach, highlights areas of diagnostic uncertainty and outlines future directions that may impact clinical practice, such as standardisation efforts (e.g., MSKI-RADS) and the integration of advanced hybrid imaging and artificial intelligence.
Footnotes
Acknowledgements
The authors would like to acknowledge Dr. Yadlapalli Sushmitha and Dr.Joe Thomas for their contributions and insights for this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
1291/25.
Credit author statement
Hanumanthu Manasa Mayukha: Concepts, design, manuscript preparation, manuscript editing and review, data analysis and literature search.
Vineet Wadhwa: Concepts, design, manuscript preparation, manuscript editing and review, data analysis and literature search.
Raghu Teja Sadineni: Concepts, design, manuscript editing and review, data analysis and literature search.
Godaba Pravallika Prasanna: Design, manuscript editing and review, data analysis and literature search.
Kondruganti Chandra Sekhara Rao: Manuscript editing and review, data analysis and design.
Velicheti Sandeep: Concepts, design, definition of intellectual content, manuscript editing and review, data analysis and data acquisition.
Data availability
None.
Use of artificial intelligence
Nil.
