Abstract
Coccidioidomycosis (CM) is a fungal disease that results from inhalation of spores of Coccidioides immitis and C posadasii. If symptomatic, disease primarily manifests as community-acquired pneumonia; however, additional pulmonary manifestations such as pleural effusion, empyema, and cavitation may occur. Diabetic patients have an increased risk of severe and cavitary CM. Cavitary disease may erode vasculature and pulmonary parenchyma leading to further complications. Furthermore, chronic cavities can become colonized as well and develop superimposed infections. This is a case of cavitary CM in uncontrolled diabetic nonadherent to treatment presenting with hemoptysis and mycetoma.
Case Presentation
A 48-year-old Hispanic male with diabetes mellitus, untreated pulmonary Coccidioidomycosis (CM), and recent infection with COVID-19 presented with hemoptysis, night sweats, and a 45-lb unintentional weight loss. He was diagnosed with CM 7 years prior to this presentation but was unable to obtain prescribed medication. Three years before presentation, he was admitted to an outside facility with sudden onset shortness of breath. Imaging revealed a right lower lobe (RLL) cavity measuring 4.6 × 3.7 cm with right-sided pneumothorax and bronchopulmonary fistula (Figures 1-3). He underwent a video-assisted thoracotomy with pleurodesis. His coccidioidal complement fixation (CF) titer was 1:32. He was started on 600 mg of fluconazole and discharged home with outpatient follow-up. For the next 2 years, the patient reported intermittent adherence to his medication. He frequently visited outside facilities complaining of persistent, nonproductive cough; his CF titer remained at 1:4. Computed tomography (CT) scan of chest showed residual RLL cavitary lesion (Figure 4). One year later, he presented to an outside facility and was diagnosed with COVID-19, without the need for supplemental oxygen and steroids. X-rays obtained at that time showed RLL infiltrates (Figure 5).

Chest x-ray, 3 years prior to presentation showing right pneumothorax, right hilar adenopathy, and right lower lobe cavitary lesion with air fluid level.

Chest CT scan, 3 years prior to presentation showing right lower lobe cavity measuring 4.6 × 3.7 cm with right-sided pneumothorax and bronchopulmonary fistula.
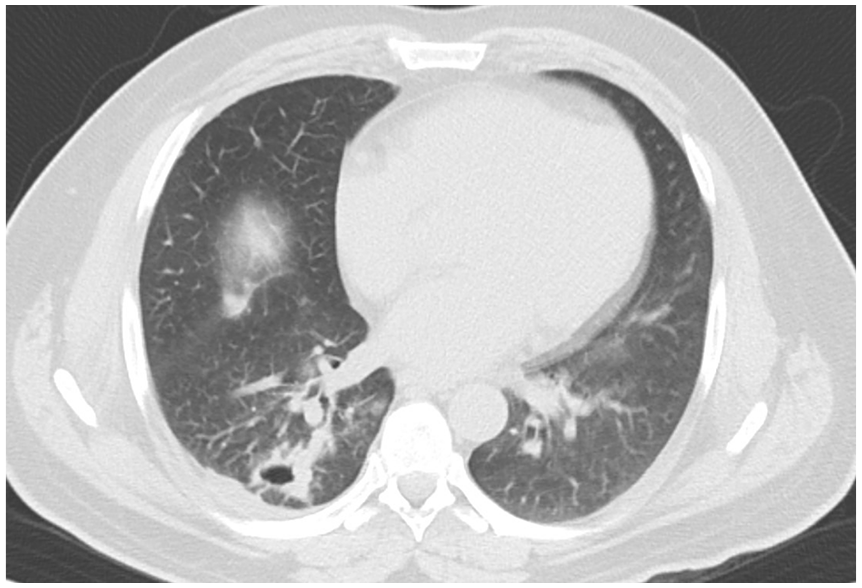
Chest CT scan, 3 years prior to presentation showing residual cavitary lesion after video-assisted thoracotomy with pleurodesis.

Chest CT scan, 2 years prior to presentation showing residual right lower lobe cavity.

Chest x-ray, 1 year prior to presentation showing bilateral haziness and right lower lobe cavity during diagnosis of COVID-19.
On his current presentation, he arrived febrile to 38.8°C, tachycardic, tachypneic, and hypoxic for which started on 2 L nasal cannula. Presenting x-ray of the chest revealed a large cavitary lesion. The cavity was further defined by CT scan, measuring 11 × 7.0 × 8.3 cm with central filling mass. It was suspected that 2 cavities from the right upper lobe (RUL) posterior segment and RLL superior segment (Figures 6-8) combined into 1 cavity. Coccidioidal CF titers had increased to 1:32 (Table 1) and glycosylated hemoglobin A1c was 12.5%. He was started on 800 mg of fluconazole. Bronchoscopy revealed multiple mucoid nodules on the medial aspect of the left mainstem bronchus starting approximately 1 cm below the main carina (Figure 9) and mild bronchitis with evidence of old blood emanating from the RLL posterior segment/superior segment. Bronchial lavage confirmed CM mycetoma via direct stain and fungal culture (Table 2).

Chest x-ray on initial presentation showing large cavity extending from right lower lobe to right upper lobe with central filling mass.

Chest CT scan upon presentation showing large cavitary lesion 11 × 7.0 × 8.3 cm with central filling mass. Perhaps a result of combined 2 cavities from the right upper lobe posterior segment and RLL superior segment.

Chest CT scan upon presentation showing large cavitary lesion 11 × 7.0 × 8.3 cm with central filling mass. Perhaps a result of combined 2 cavities from the right upper lobe posterior segment and RLL superior segment.
Coccidioides immitis Laboratory Studies.

Performed at Kern County Public Health Laboratory.

Bronchoscopy showing multiple mucoid nodules on the medial aspect of the left mainstem bronchus starting approximately 1 cm below the MC.
Microbiology Results.

Abbreviations: AFB = Acid-fast bacilli; KOH = Potassium hydroxide; BAL = Bronchoaleveolar lavage.
During his admission, he continued to experience up to 300 mL hemoptysis daily. Interventional radiology successfully performed arterial embolization and coiling of the right tracheobronchial and intercostal bronchial artery to control hemoptysis (Figures 10 and 11). Due to the presence of a tree-in-bud pattern in his chest CT and positive QuantiFERON Gold test, coinfection with mycobacterium tuberculosis was suspected. Further history obtained revealed a family history of tuberculosis before immigration to the United States. Three consecutive morning sputum samples and all bronchoscopic samples stained negative for acid-fast bacilli. The patient had clinical improvement in cough and shortness of breath and his hemoptysis resolved. He was discharged home on fluconazole 800 mg.

Fluoroscopy showing arterial embolization and coiling of the right intercostal bronchial artery.
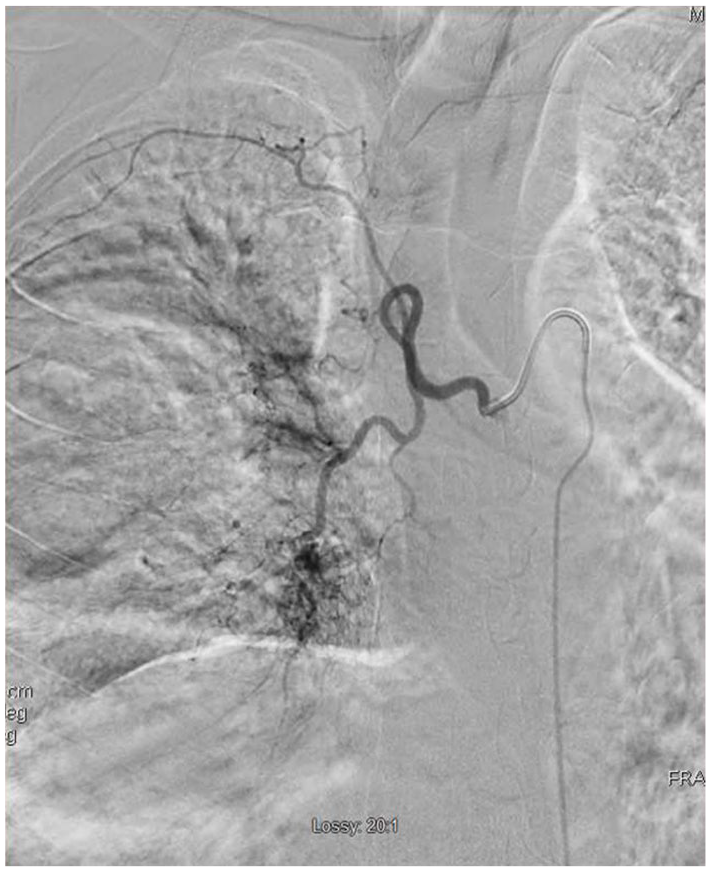
Fluoroscopy showing arterial embolization and coiling of the right tracheobronchial.
One week later he returned febrile, in respiratory distress, and hypoxemic. Vital signs were significant for a temperature of 39.1°C, heart rate of 114 beats per minute, respiratory rate of 31 breaths per minute, blood pressure of 144/90 mm Hg, and oxygen saturation on pulse oximetry of 86% while on room air. Laboratory studies found elevated white blood cell count, erythrocyte sedimentation rate, and C-reactive protein (Table 3). Imaging revealed new left lower lobe consolidations (Figure 12). Due to initial concern for progressive severe pulmonary CM now in the left lower lobe, he was started on intravenous liposomal amphotericin B. Within 2 days, his symptoms and need for supplemental oxygen requirement improved. Due to speed of recovery, it was surmised that he aspirated the residual content of his cavity into the contralateral lung, rather than had developed severe pulmonary CM. His antifungal treatment was switched to posaconazole 400 mg daily orally. His condition continued to improve, and he was discharged home on oral posaconazole.
Laboratory Results.

Abbreviation: EOS, eosinophil; WBC, white blood cell.

CT chest upon second admission showing emptied right-sided cavitary lesion and new left lower lobe infiltration.
Discussion
Coccidioides is a dimorphic fungus that forms arthroconidia during the mycelial phase. Inhalation of arthroconidia initiates the parasitic phase by transforming into spherules that are found in infected tissue. However, parasitic mycelial structures, such as hyphae, have been also identified mostly inside the lung cavity or sinuses which demonstrates chronic infection and the long evolution of the disease. Poorly controlled diabetic individuals with pulmonary CM are already at increased risk for chronicity and development of cavitation. 1 Mycetoma, or fungus ball, is usually defined as a mass within a cavity and has been reported as early as 1971. 2 For the case presented, pulmonary CM was the primary etiology of the cavitary lesion; however, the filling mass inside the cavity could have been a mycetoma from aspergillus, mucormycosis, CM itself, candida, or other fungal opportunistic infections and malignancy.
Coccidioidal pulmonary cavities are typically thin-walled, gas-filled space within the zone of consolidation and necrosis that communicates with the bronchial tree. Up to 50% of cases are known to resolve spontaneously; however, this depends on many host and pathogen factors. 3 Our patient has a history of uncontrolled diabetes and likely presented with 2 separate cavities in the right lung. Imaging demonstrated cavity formation starting in the posterior segment of the RUL and the superior segment of the RLL. Together they formed a very large cavity 11.0 × 7.0 × 8.3 cm containing mycetoma and blood clots. It is difficult to establish whether the contents of the individual cavities have ruptured across the major fissure creating a contained bronchopulmonary fistula that sealed into one large cavity. An alternative is that there was an incomplete major fissure, and the cavity is a continuation of a RUL posterior segment and RLL posterior segment.
The bronchoscopy showed mild to moderate bronchitis with presence of submucosal and mucosal nodules. These findings are similar to previously reported findings by our group.4,5 In addition, endobronchial lesions were visualized on the left mainstem bronchus suggestive of active disease. Biopsy was not performed at the time due to the uncertainty of sudden onset of massive bleeding from the right lung and possibly difficult to control bleeding from the left lung. Bronchoalveolar lavage with gram stain and culture demonstrated evidence of hyphae and mycelia of CM, but no spherules. This form of the organism was most likely growing as a mycetoma and affecting the mucosa in the cavity causing it to bleed.
Upon the patient’s return 1 week later, new infiltrates found in the posterior segment of the left lower lobe suggested that he might have aspirated the contents of the mycetoma from the contralateral lung. However, with the presence of the endobronchial lesions, it is possible that the disease was already beginning to spread. The first case of coccidioidal laryngotracheobronchitis was reported in 1966 in a female toddler. 6 A case series reviewed 38 cases with coccidioidal endotracheal and endobronchial disease. They determined it was associated with parenchymal and disseminated disease and represents a chronic form of the disease. Infection can occur in both immunocompromised and immunocompetent hosts and acquired directly or via extension into the trachea or bronchial tree from affected lymph nodes. Additional complications include bronchitis and bronchiolitis of the bronchial tract, mucosal ulceration, erosion, and obstruction into the trachea and large airways. 7
The complications of pulmonary CM include persistent symptoms, enlarging cavities, rupture of cavity, and persistent hemoptysis despite therapy. Although the exact incidence is unknown, cavitary lesions can require resection and decortication. 8 Hemoptysis has been reported in up to 30% to 50% of patients with pulmonary CM. 9
The first step in management is to determine whether the hemoptysis is life-threatening. Prior definitions varied from 150 to greater than 300 mL over 24 hours; however, approximating the amount of expectorated blood may be challenging and inaccurate. Clinical factors such as patency of airway, gas exchange abnormalities, or hemodynamic instability are more important variables to define life-threatening hemoptysis.
The next step is to determine the source of the site of bleeding. 10 Chest x-ray, when placing the patient into a dependent position, is known to only localize bleeding in 46% of cases. 11 Multidetector CT scan can identify the origin of bleeding in 70% of cases, an underlying cause up to 77% of cases, compared with 48% of cases with bronchoscopy. 12 The combination of bronchoscopy and CT scan increased diagnostic etiology to 83%. 10 Management is aimed at airway stabilization and isolation of the hemorrhage. Bronchoscopy is utilized to identify the source of bleeding, provide local tamponade on, and extract the clot. Definitive therapy is often performed with bronchial artery embolization with success rates varying from 70% to 99%; however, the recurrence rate for bleeding is high and estimated up to 58% within 30 days. 13 This case’s bronchoscopy revealed old blood from both the RUL posterior segment and the RLL superior segment supporting the fact that this was an old cavity.
Treatment of pulmonary CM with cavitation is mainly with azoles. At Valley Fever Institute, 14 we recommend a high dose of fluconazole (600-800 mg). Other azoles such as itraconazole, posaconazole, and isavuconazole are reserved for refractory cases or intolerance. Voriconazole is avoided due to toxicity and association with cutaneous malignancies. The role of amphotericin B is unknown perhaps due to penetration to the cavities. The duration of therapy is prolonged and depends on the management of underlying diseases such as diabetes. Chronicity, with a lack of serological and/or radiological response, is not uncommon.
In this case, antifungal sensitivities were obtained (Table 4), and fluconazole was changed to posaconazole due to lack of response and providing wider coverage for other potential superimposing mycetoma such as aspergillosis in near future. When symptoms progress to involve hemoptysis, further diagnostic testing is required to treat the underlying infection as well as to identify and localize the source of bleeding. Temporary measures include bronchoscopy and medications such as tranexamic acid. Persistent or massive bleeding requires consideration for additional interventions. Interventional radiology–guided embolization of select pulmonary arteries was employed to treat our patient’s hemoptysis.
Coccidioides immitis Antifungal Sensitivity Studies.

Conclusion
Patients presenting with massive hemoptysis and cavitary lesions require management through a multidisciplinary approach. We highlight a case where pulmonary, infectious diseases, thoracic surgery, and interventional radiology were required to provide appropriate management. It is known that diabetic individuals are at increased risk of CM. However, this case demonstrates formation of a giant cavitary lesion from 2 smaller cavities with mycetoma inside and endobronchial lesions. Nonadherence to medical therapy further increases the risk of recurrence via the same process, additional infectious agents, bleeding, and aspiration of contents.
Footnotes
Authors’ Note
This case is selected to be presented as a poster presentation at the 2022 Western Medical Research Conference on January 20, 2022, Carmel, California, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to report this case was obtained from the Kern Medical Institutional Review Board; approval ID: 21088.
Informed Consent
Informed consent was obtained from the patient for their anonymized information to be published in this article.
