Abstract
Background
Colorectal cancer constitutes a major global health issue, defined by the unregulated growth of atypical cells in the large intestine or rectum. This intricate disease, resulting from a combination of genetic, environmental, and lifestyle causes, presents a significant risk to persons globally.
Objectives
This work was undertaken to study the anti-cancer effects of sophoricoside on colon cancer cells.
Materials and Methods
An MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) test was employed to study the influence of sophoricoside on the growth of HCT-116 cells. The apoptotic levels in the untreated and sophoricoside-treated HCT-116 cells were evaluated using 4′,6-diamidino-2-phenylindole (DAPI) and dual staining techniques. The concentrations of oxidative stress indicators in the experimental cells were evaluated using the corresponding test kits.
Results
The administration of various dosages of sophoricoside significantly suppressed HCT-116 cell proliferation in a dose-dependent manner. Sophoricoside treatment significantly enhanced apoptosis in HCT-116 cells, which is evidenced by the DAPI and dual staining assays. It also elevated oxidative stress marker levels by reducing the antioxidants in HCT-116 cells.
Conclusion
The present investigation illustrated that sophoricoside possesses anti-cancer activities against colon cancer by reducing cell viability and inducing oxidative stress-dependent apoptosis in HCT-116 cells. Consequently, the present findings indicate sophoricoside as a promising and beneficial alternative for future colon cancer therapy.
Introduction
Colorectal cancer, commonly referred to as colon cancer, represents a significant global health issue, characterized by the uncontrolled growth of cells within the large intestine or rectum. This complex disease, arising from a confluence of genetic, environmental, and lifestyle causes, poses a substantial threat to individuals worldwide. Colon cancer, characterized by a diverse array of neoplastic cells developing within intricate microenvironments, impacts both genders and ranks as the second-highest cause of cancer-related mortality (Siegel et al., 2023). Annually, about 1 million individuals are impacted, resulting in over 500,000 fatalities globally, with its prevalence escalating at a concerning pace. Recent data indicate that colon cancer ranks as the third most frequently detected cancer in males and the second in females on a global scale (Marcellinaro et al., 2023). The burden of colon cancer is not static; projections indicate a concerning upward trajectory, with estimates suggesting an elevate to 3.2 million new incidences and 1.6 million mortalities by the year 2040. Significantly, 20% of patients present with metastatic colon cancer, while up to 50% of those with early-stage disease experience relapse later, even undergoing curative-intent surgery, adjuvant chemotherapy, and/or radiotherapy (Xi & Xu, 2021).
The etiology of colon cancer is multifaceted, with a complex interplay of genetic predisposition, environmental exposures, and lifestyle choices participating in its progression. While the precise mechanisms remain under investigation, several key factors have been identified as significant contributors. Obesity, a significant global public health issue, is known as a primary cause of colon cancer (Ionescu et al., 2023). It has already been demonstrated that obesity induces chronic inflammation, disrupts sex hormone control, and modifies insulin signaling, thereby facilitating the onset of colon cancer. Alterable risk variables, including dietary practices, physical exercise, and tobacco consumption, significantly affect an individual’s vulnerability to colon cancer. Diet is thought to be responsible for around 70% of colorectal cancer cases, with high utilization of red and processed meats, saturated fats, and refined carbohydrates associated with high risk (Rawla et al., 2019). Conversely, a diet rich in fiber, fruits, and vegetables is connected with a protective effect. Beyond lifestyle choices, certain diseases, like inflammatory bowel disease and diabetes mellitus, have been implicated as independent risk factors for colon cancer. About 5%–10% of colorectal cancer incidences are attributable to hereditary causes, including familial adenomatous polyposis and Lynch syndrome (Roshandel et al., 2024).
Current treatment modalities for colon cancer generally involve a combination of surgery, chemotherapy, and radiation therapy, but these approaches are often associated with significant challenges. Surgical resection remains the primary treatment option for localized colon cancer, offering the potential for cure (Puzzo et al., 2025). However, complete surgical removal may not always be possible, especially in cases with advanced disease or involvement of critical structures. Chemotherapy, using medications like 5-fluorouracil, oxaliplatin, and irinotecan, aims to eliminate tumor cells that may have spread beyond the primary tumor site, but it can also cause systemic toxicities affecting normal cells (Kumar et al., 2023). Radiation therapy is employed to shrink tumors before surgery, but it can also damage surrounding tissues and lead to long-term complications. Despite advancements in treatment strategies, a substantial proportion of patients with colorectal cancer experience disease recurrence or metastasis, underscoring the need for novel therapeutic approaches with improved efficacy and reduced toxicity (Romero-Zoghbi et al., 2025).
Plant bioactive compounds have garnered increasing attention as potential therapeutic agents for colon cancer due to their diverse pharmacological activities and relatively low toxicity profiles (Delgado-Gonzalez et al., 2023). Sophoricoside, an isoflavone glycoside, is a major bioactive compound derived from the dried fruit of Sophora japonica, a traditional Chinese medicine. It has been reported that sophoricoside has demonstrated anti-dermatitis (Kim & Lee, 2021), anti-asthma (Kim & Lee, 2021), anti-hepatic steatosis (Zhang et al., 2020), anti-sepsis (Wu et al., 2021), hepatoprotective (Chen et al., 2023), and anti-arthritic (Liu et al., 2024) properties. Furthermore, a recent report from Wang et al. (2025) has demonstrated that sophoricoside has shown anti-cancer effects against glioblastoma. Nonetheless, its anti-cancer efficacy against colon cancer has not been evaluated to date. Therefore, this work was undertaken to study the anti-cancer properties of sophoricoside on colon cancer cells.
Materials and Methods
Chemicals
The major chemicals utilized in this study, including sophoricoside, antibiotics, fetal bovine serum (FBS), and so on, were procured from Sigma–Aldrich, USA. The biochemical indicators were assessed using standard kits obtained from Abcam, USA.
Maintenance of Cells
The HCT-116 colon cancer cells were acquired from ATCC, USA, and cultivated in Dulbecco’s Modified Eagle Medium (DMEM) media enriched with 10% FBS and 1% anti-mycotic mixture in a 5% CO2 incubator. The mature cells were harvested upon reaching 80% confluency and utilized for further fluorescence staining and biochemical tests.
MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) Assay
The growth of HCT-116 cells was examined using the MTT test (Mosmann et al., 1983). Cells were cultivated on a 96-well plate for 24 h and subsequently exposed to sophoricoside at various doses (1, 5, 10, 20, 40, 80, and 160 µM) for an additional 24 h. Subsequent to the treatment, MTT (20 µL) solution was combined with DMEM (100 µL) in the wells for a duration of 4 h. Following the dissolution of the generated formazan deposits with dimethyl sulfoxide (DMSO) (100 µL), the absorbance of the plate was taken at 570 nm.
4′,6-Diamidino-2-phenylindole (DAPI) Staining
The DAPI fluorescent staining technique was employed to examine the nuclear morphology of apoptotic cells in HCT-116 cells. After culture of HCT-116 cells in a 24-well plate, the cells were administered with sophoricoside at a dosage of 20 µM for 24 h. The cells were fixed in 4% paraformaldehyde for 30 min and then stained with DAPI (200 µg/mL) for 20 min. The influence of sophoricoside on apoptosis in HCT-116 cells was further studied using a fluorescence microscope.
Assay of Oxidative Stress Markers
HCT-116 cells administered with sophoricoside and control groups were lysed using cell lysis buffer, and the cell lysate was produced to assess the concentrations of oxidative stress indicators. The concentrations of thiobarbituric acid reactive substances (TBARS), superoxide dismutase (SOD), and glutathione (GSH) were measured in the untreated and sophoricoside-administered HCT-116 cells utilizing the respective kits as per the manufacturer’s specifications (Abcam, USA).
Statistical Analysis
The statistical assays were done using GraphPad Prism software (version 10.1.2), and the results are presented as the Mean ± Standard Deviation (SD) of triplicates. One-way analysis of variance (ANOVA) and Tukey’s post hoc tests were utilized to study the values, with p < .05 as significant.
Results
Effect of Sophoricoside on the HCT-116 Cell Viability
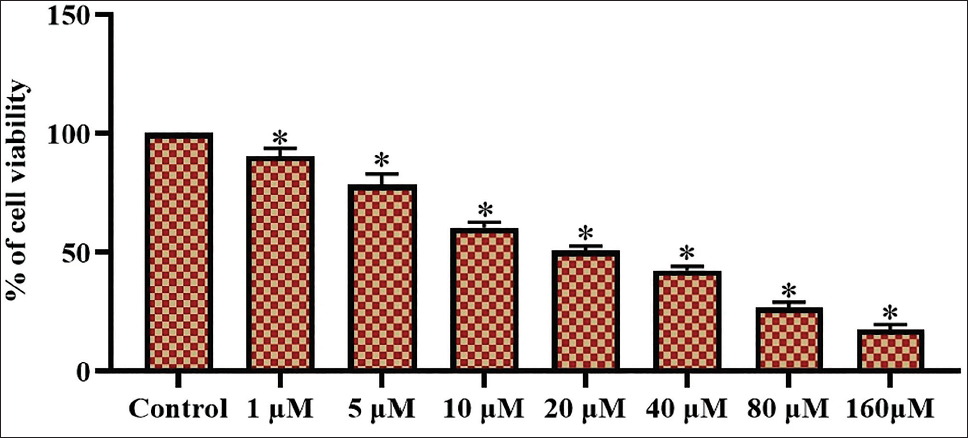
Figure 1 illustrates the findings of the MTT study, depicting the influence of sophoricoside on the growth of HCT-116 cells. Sophoricoside treatment markedly diminished the growth of HCT-116 cells at varied doses (1–160 µM). The elevated levels of sophoricoside exhibited a marginal inhibitory effect on the proliferation of HCT-116 cells, as illustrated in Figure 1. The IC50 concentration of sophoricoside was recorded at 20 µM against HCT-116 cells, and this dosage was selected for the following investigations.
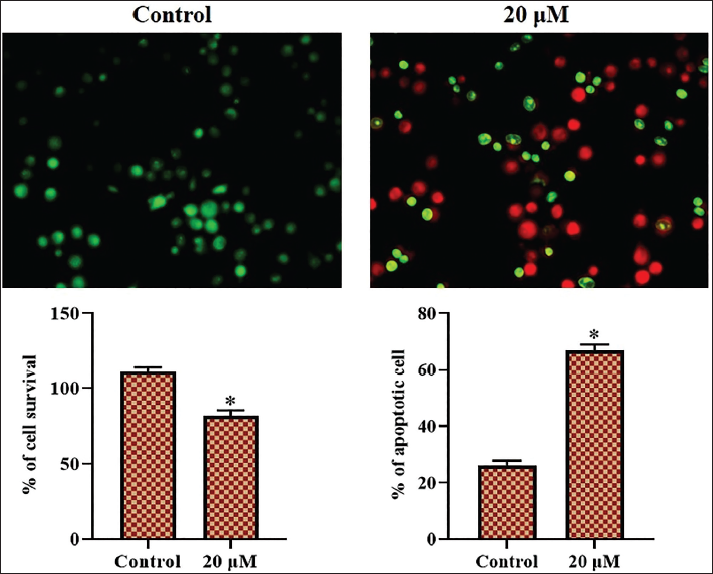
Effect of Sophoricoside on Apoptosis in HCT-116 Cells
The dual staining technique was performed to assess apoptosis in the untreated and sophoricoside-administered HCT-116 cells, with the results presented in Figure 2. The HCT-116 cells exhibited increased yellow/orange fluorescence following treatment with 20 µM of sophoricoside, indicating the onset of apoptosis (Figure 2).
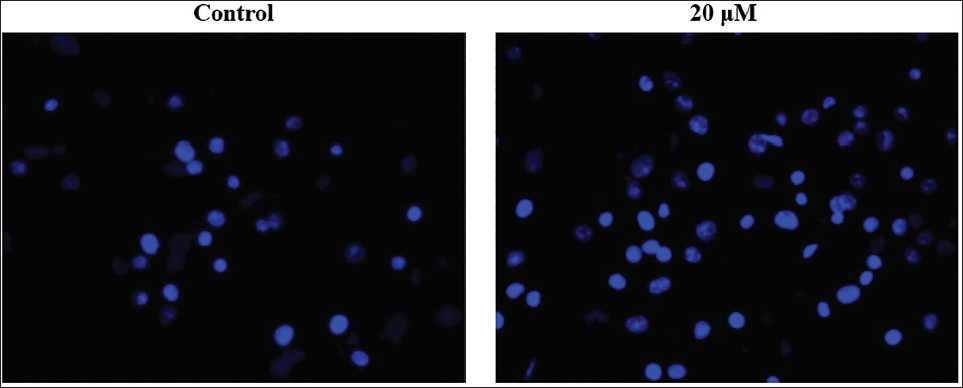
Effect of Sophoricoside on Apoptosis in HCT-116 Cells
The occurrence of apoptotic cell death in untreated and sophoricoside-exposed HCT-116 cells was studied using DAPI staining, with findings illustrated in Figure 3. The HCT-116 cells subjected to sophoricoside treatment at a dosage of 20 µM exhibited heightened instances of apoptosis, which is evidenced by the intense fluorescence, cell injury, and the development of apoptotic bodies.
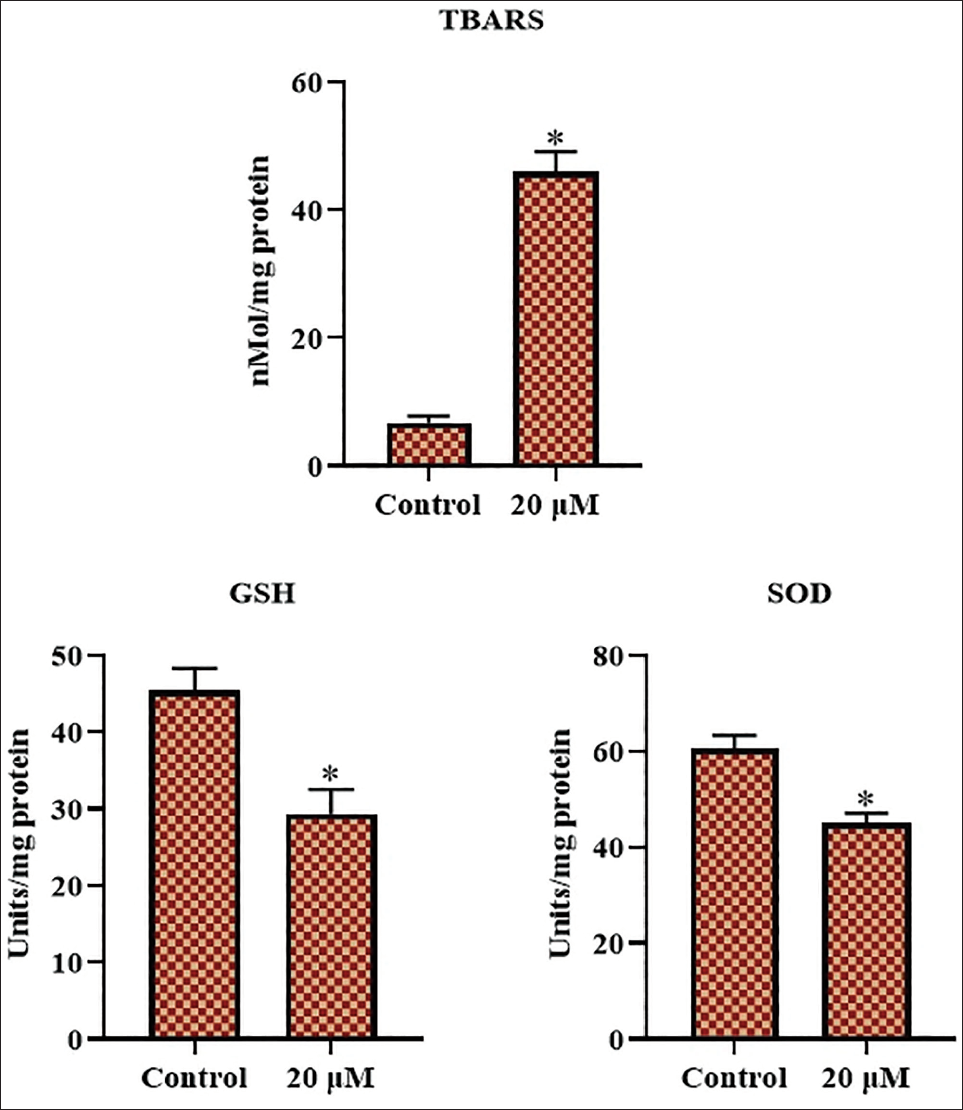
Effect of Sophoricoside on Oxidative Stress Markers in HCT-116 Cells
Figure 4 illustrates the concentrations of TBARS, GSH, and SOD, which are assessed in both untreated and sophoricoside-administered HCT-116 cells. Elevated levels of GSH and SOD, together with decreased TBARS, were observed in the control cells. Concurrently, HCT-116 cells administered with 20 µM of sophoricoside exhibited significantly elevated TBARS levels and reduced GSH and SOD levels in comparison to the control (Figure 4). The data indicate that sophoricoside promotes oxidative stress in HCT-116 cells.
Discussion
Colon cancer, a common tumor of the digestive tract, exhibits a considerable global health issue due to its elevated incidence and fatality rates. The intricate interaction of genetic predisposition, environmental influences, and lifestyle choices facilitates the onset and progression of colon cancer. Environmental factors such as lifestyle, cultural and social practices, alcohol intake, and smoking, along with family history, have been identified as risk factors in the etiology of colon cancer. Despite advancements in diagnostic techniques and treatment modalities, colon cancer continues to present substantial challenges in clinical management, including recurrence, metastasis, and drug resistance (Lewandowska et al., 2022). Current treatment strategies for colon cancer generally involve a combination of surgery, chemotherapy, and radiation therapy. Surgery remains the primary intervention for resectable tumors, aiming to remove the cancerous tissue and surrounding lymph nodes. Chemotherapy, utilizing cytotoxic drugs, plays a crucial role in eradicating residual cancer cells after surgery and managing advanced-stage disease (Chakrabarti et al., 2020). Targeted therapy and immunotherapy are more recent treatment options. However, conventional chemotherapeutic agents often exhibit systemic toxicities, affecting both cancerous and healthy cells, leading to adverse effects and compromised life quality of patients. These unpleasant side effects drive the need to develop safer and more effective tumor-inhibiting agents (Underwood et al., 2024). Moreover, the development of drug resistance remains a significant obstacle, limiting the long-term efficacy of chemotherapy regimens. Despite breakthroughs in radiotherapy for monitoring local tumor growth and reducing adverse effects, controlling distant lesions beyond the irradiated region continues to pose a significant problem. Despite the initial success of surgery and adjuvant therapies, a majority of patients experience disease recurrence and metastasis, underscoring the need for novel therapeutic approaches (Ciardiello et al., 2022).
The search for alternative and complementary therapies has gained momentum, with a focus on exploring the potential of natural compounds derived from plants. Plant bioactive compounds have demonstrated promising anti-cancer effects, including the capacity to inhibit tumor cell growth, trigger apoptosis, and suppress metastasis. They frequently operate via various routes of action, complicating the ability of cancer cells to build resistance (Fernandez-Muñoz et al., 2025). Many of these compounds demonstrate selective cytotoxicity to tumor cells while sparing non-malignant cells, offering a potential advantage over conventional chemotherapeutic agents. The diversity of plant-derived compounds provides a rich reservoir for drug discovery, with ongoing research focused on identifying and characterizing novel compounds with potent anti-cancer activity. Phytotherapy appears as a potential alternative for discovering new drugs to fight cancer and drug resistance (Gavrilas et al., 2022).
The in vitro cytotoxicity assays, particularly the MTT assay, are vital for assessing the potential of plant bioactive compounds as anti-cancer agents. The MTT test, based on the reduction of MTT to formazan crystals by metabolically active cells, provides a quantitative measure of cell viability and proliferation. The cell viability assay explains the cellular response to toxic materials and provides information on cell death, survival, and metabolic activities (Pantea et al., 2023). By measuring the absorbance of the formazan product, researchers can determine the cytotoxic effects of test compounds on cancer cells, providing valuable insights into their potential therapeutic efficacy. This method offers a simple, reliable, and high-throughput approach for screening large libraries of compounds and identifying promising drug candidates. Overall, the MTT assay serves as a cornerstone in preclinical drug development, facilitating the identification and characterization of novel anti-cancer agents with the potential to improve outcomes for patients with cancer (Yao et al., 2023). In this research, the results have depicted that sophoricoside successfully reduced the HCT-116 cell viability. These findings suggest the cytotoxic nature of the sophoricoside on the colon cancer cells.
Apoptosis plays a crucial role in sustaining tissue homeostasis by removing abnormal cells. Apoptosis is executed through a complex cascade of molecular events, involving distinct signaling pathways, caspase activation, and ultimately, the dismantling of the cell. Dysregulation of apoptosis is a common sign of tumors, as it allows malignant cells to escape apoptosis, proliferate uncontrollably, and resist therapy (Pistritto et al., 2016). The apoptotic process is characterized by a specific sequence of morphological changes, commencing with cell shrinkage, chromatin condensation, and organelle fragmentation, which results in the loss of cell-to-cell and cell-to-matrix interactions. Subsequently, the cell membrane undergoes blebbing, resulting in the development of apoptotic bodies containing cellular components. These bodies are then consumed by phagocytic cells, preventing the release of intracellular contents and minimizing inflammation. Apoptosis is actively induced by the cell itself and is an integral part of the metabolism (Tian et al., 2024). The balance between cell death and proliferation is disrupted in tumor formation, highlighting the significance of apoptosis in preventing cancer development. Understanding the intricate mechanisms of apoptosis and its dysregulation in cancer is critical for developing effective cancer therapies that can restore the apoptotic potential of malignant cells (Jan & Chaudhry, 2019). In cancer, the apoptotic machinery is often dysregulated, permitting tumor cells to escape apoptosis and survive under conditions that would normally trigger apoptosis in healthy cells. Targeting apoptosis in cancer has developed as an useful therapeutic technique, with several approaches being developed to restore the apoptotic potential of malignant cells (Giménez-Bonafé et al., 2009). In this study, the outcomes of the fluorescent staining studies, dual staining and DAPI staining, have evidenced that the sophoricoside considerably induced apoptosis in HCT-116 cells. These results suggest that sophoricoside can inhibit colon cancer progression by enhancing apoptosis in colon cancer cells.
Oxidative stress, defined as an imbalance between ROS generation and antioxidant defenses, significantly contributes to the activation of apoptosis in cancer cells. ROS are produced during cellular metabolism and can induce oxidative damage to cell components. This oxidative damage can trigger a cascade of events leading to cellular dysfunction and ultimately apoptosis. The precise mechanisms by which oxidative stress induces apoptosis are multifaceted and involve the activation of various signaling cascades, including the mitochondrial, endoplasmic reticulum stress, and the death receptor cascades (Di Carlo & Sorrentino, 2024). The analysis of antioxidants and TBARS levels in sample drug-treated cancer cells is crucial for comprehending the mechanism of oxidative stress-mediated apoptosis. SOD is an important antioxidant that catalyzes the dismutation of superoxide radicals into hydrogen peroxide and oxygen. It is one of the crucial enzymes for the antioxidant mechanism. GSH is a tripeptide antioxidant that plays a critical role in detoxifying ROS and sustaining cellular redox balance. TBARS are a measure of lipid peroxidation, a process in which ROS damage lipids, leading to the formation of various aldehydes and other products that can react with thiobarbituric acid (Catalano et al., 2025). By measuring the levels of these three molecules, researchers can gain insights into the degree of oxidative stress and the effectiveness of drug treatments in modulating the cellular redox environment. The levels of SOD, GSH, and TBARS can provide valuable information about the mechanisms of action of drugs that induce oxidative stress-dependent apoptosis in tumor cells. Changes in these parameters can indicate whether a drug is directly increasing ROS production, inhibiting antioxidant defense mechanisms, or inducing lipid peroxidation. Reduced levels of SOD and GSH may diminish the superoxide scavenging process, rendering the tissue more vulnerable to oxidative stress (Liu et al., 2023). The present findings clearly proved that the untreated control cells showed increased GSH and SOD, with decreased TBARS. In contrast, HCT-116 cells treated with sophoricoside illustrated increased TBARS levels and decreased antioxidants in comparison to the control. These findings witnessed that the sophoricoside can enhance oxidative stress, thereby facilitate oxidative stress-dependent apoptosis in colon cancer cells.
Conclusion
The present investigation revealed that sophoricoside possesses anti-cancer activities against colon cancer by reducing cell viability and inducing oxidative stress-dependent apoptosis in HCT-116 cells. The sophoricoside treatment significantly reduced cell viability, elevated apoptosis, and heightened oxidative stress indicators in HCT-116 cells. Consequently, the present findings indicate sophoricoside as a promising and beneficial alternative for future colon cancer therapy. The present study has some limitations, such as our research was conducted in vitro, and further in vivo studies are needed to confirm the efficacy and safety of sophoricoside in a more physiologically relevant setting. Additionally, we suggest that the exact mechanisms underlying the anti-cancer effects of sophoricoside require further elucidation, and we plan to investigate these mechanisms in future studies.
Footnotes
Abbreviations
ANOVA: Analysis of variance; DMSO: Dimethyl sulfoxide; FBS: Fetal bovine serum; MDA: Malondialdehyde; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; ROS: Reactive oxygen species; SD: Standard deviation; SOD: Superoxide dismutase.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the relevant ethics committee or Institutional Review Board (IRB).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has provided informed consent for the submission of the article to the journal.
