Abstract
Background
Colon cancer, a prevalent form of gastrointestinal malignancy, poses a significant public health issue globally. The onset of colon cancer is a complex mechanism implicating genetic and environmental factors, including dietary habits and lifestyle choices.
Objectives
The current study was undertaken to investigate the anti-tumor effects of artemetin against colon cancer cells.
Materials and Methods
The influence of artemetin on the proliferation of HCT-116 cells was evaluated with an MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide) test. Also, the extent of apoptosis in both untreated and artemetin-exposed HCT-116 cells was studied using a dual staining technique. The activities of the caspase enzymes (caspase-3, -8, and -9) in both untreated and artemetin-exposed HCT-116 cells were evaluated using a commercial diagnostic kit.
Results
The administration of several dosages of artemetin significantly suppressed the HCT-116 cells dose-dependently. Furthermore, artemetin treatment significantly induced apoptosis in HCT-116 cells, which is evidenced by dual staining assay. Artemetin treatment also elevated the caspase-3, -8, and -9 activities in the colon cancer cells.
Conclusion
The present study revealed that artemetin possesses anti-cancer activities against colon cancer, reducing cell viability and inducing caspase-mediated apoptosis in HCT-116 cells. Consequently, the present findings indicate artemetin as a favorable anti-tumor candidate for future colon cancer therapy.
Introduction
Colon cancer, or colorectal cancer, is a devastating disease that impacts millions of individuals globally. It ranks as the third most prevalent cancer globally, with almost 1.80 million new incidences and 862,000 fatalities documented globally. The alarming prevalence and mortality rates of colon cancer have made it a subject of significant concern and extensive research (Paragomi et al., 2024). The causes of colon cancer are diverse, implicating a complex interaction of genetic, environmental, and behavioral determinants in its progression. Genetic predispositions, such as familial adenomatous polyposis and Lynch syndrome, can elevate an individual’s risk of colon cancer. Additionally, environmental factors, including dietary habits, physical activity, and exposure to certain toxins, can also play a crucial role in the etiology of the condition (Morgan et al., 2023). Risk factors for colon cancer are numerous and well-documented. Increasing age, obesity, an inactive lifestyle, a diet rich in red and processed meats, and a history of inflammatory bowel illnesses are all acknowledged as major risk factors (Wang et al., 2024). Comprehending the complexity of colon cancer and its diverse risk factors is essential for formulating potential preventative and treatment approaches. Ongoing research in areas such as dietary modifications, gut microbiome modulation, and targeted therapies holds promise for improving outcomes for people affected by this devastating condition (Sawicki et al., 2021).
Apoptosis, a critical process of cell death mechanism, plays a vital role in the control of tissue balance and the subsequent elimination of injured or abnormal cells. However, the dysfunction of apoptosis has participated in the onset of numerous ailments, including cancer. In colon cancer, the dysfunction of apoptosis can result in the uncontrolled growth and survival of tumor cells, ultimately resulting in the onset of the disease (Abaza et al., 2022). Extrinsic and intrinsic signaling are the two major mechanisms through which apoptosis is regulated in mammalian cells. The extrinsic mechanism of apoptosis is initiated by extracellular death receptor signals, while the intrinsic apoptotic mechanism is triggered by intracellular stress signals that disrupt mitochondrial membrane integrity. In colon cancer, the dysregulation of these pathways can lead to the evasion of apoptosis, a hallmark of cancer (Neophytou et al., 2021). One of the key processes by which colon cancer cells escape apoptosis is through the overexpression of anti-apoptotic genes Bcl-2 and Bcl-xL. These genes block the activation of pro-apoptotic proteins, inhibiting the initiation of the apoptotic cascade. Additionally, colon cancer cells may downregulate the pro-apoptotic genes, like Bax and Bak, further tipping the balance in favor of cell survival (Tong et al., 2022). Targeting the dysregulation of apoptosis has developed as a favorable technique for treating colon cancer.
Conventional treatment options for colon cancer, such as surgical intervention, radiation, and chemotherapy, while effective to an extent, often come with substantial side effects and limitations such as nausea, fatigue, and compromised immune function, which can adversely affect the patient’s quality of life (Anand et al., 2023). Consequently, there is more attention on discovering alternative therapeutic approaches, especially those obtained from natural sources like plants, to address the challenges associated with current colon cancer treatments (Victoir et al., 2024). One promising approach is the utilization of bioactive compounds found in plants, known as phytochemicals. These natural compounds have shown remarkable potential in targeting various pathways implicated in colon cancer progression, like cell cycle regulation, apoptosis, and antioxidant modulation (López-Gómez & Uranga, 2024). Recently, extensive research has been performed to explicate the underlying processes by which phytochemicals exert their anti-cancer effects, with the aim of developing more effective and safer therapeutic options (Cheng et al., 2025). Artemetin is a 5-hydroxy-3,6,7,3′,4′-pentamethoxyflavone compound, which is found extensively in numerous plants, including Artemisia absinthium plant (De Almeida et al., 2016). It has already been reported that artemetin has numerous biological activities such as antioxidant, anti-microbial, anti-malarial, anti-atherosclerotic, hepatoprotective, anti-inflammatory, and hypotensive properties (de Souza et al., 2011; Martim et al., 2021; Patel & Patel, 2024). Furthermore, it has been well suggested that artemetin demonstrated anti-cancer properties against breast and liver cancer cells (Vo et al., 2022). Nevertheless, the anti-tumor effects of artemetin against colon cancer have not been systemically explored yet. Consequently, the current work was performed to assess the anti-tumor effects of artemetin on colon cancer cells.
Materials and Methods
Chemicals and Reagents
The primary chemicals and reagents, such as artemetin, Dulbecco’s modified Eagle’s Medium (DMEM), antibiotics, and fetal bovine serum (FBS), were acquired from Sigma-Aldrich, USA. The biochemical markers were assessed using the commercial kits obtained from Elabscience, USA.
Collection and Maintenance of Cell Culture
The HCT-116 cells were purchased commercially from ATCC, USA, and cultivated in DMEM media supplemented with FBS (10%) in an incubator with 5% CO2. The cultured cells were harvested at achieving 80% confluency and utilized for further studies.
MTT (3-[4,5-Dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide) Cytotoxic Assay
The proliferation of untreated and artemetin-treated HCT-116 cells was examined using the MTT test (Mosmann, 1983). Cells were loaded onto a 96-well plate and subsequently exposed to artemetin at varying doses (2.5, 5, 7.5, 10, 12.5, 25, and 50 µg/mL) for a further 24, 48, and 72 h periods. After the treatment, MTT (20 µL) solution and DMEM (100 µL) were combined in the wells for 4 h (Rosa et al., 2022). Following the dissolution of the formed formazan deposits with dimethyl sulfoxide (DMSO), the absorbance of the plate was measured at 570 nm.
Apoptosis Analysis by Dual Staining Technique
The dual staining studies were performed on the HCT-116 cells to assess their apoptosis level (Baskić et al., 2006). The HCT-116 cells were cultured in a 24-well plate and subsequently treated with IC50 value of artemetin and/or doxorubicin (DOX) for a further 24 h. Subsequently, acridine orange/ethidium bromide (AO/EB) (100 µg/mL) stains were introduced into the wells for 5 min in a dark place to assess apoptosis in HCT-116 cells utilizing a fluorescence microscope.
Analysis of Caspase Enzyme Activities in the HCT-116 Cells
Both untreated and artemetin-exposed HCT-116 cells were obtained, and cell lysate was produced by lysing the cells with cell lysis buffer to assess the caspase activities. The caspase-3, -8, and -9 activities were studied in the cell lysates of untreated and artemetin-exposed HCT-116 cells using appropriate commercial diagnostic kits as per the manufacturer’s specifications (Elabscience, USA).
Statistical Analysis
The statistical studies were conducted using GraphPad software (version 9), and findings are given as a mean ± SD of triplicate values. One-way analysis of variance (ANOVA) and Tukey’s post hoc tests were utilized to study the values, with a significance of p < 0.05.
Results
Effect of Artemetin on the Proliferation of HCT-116 Cells
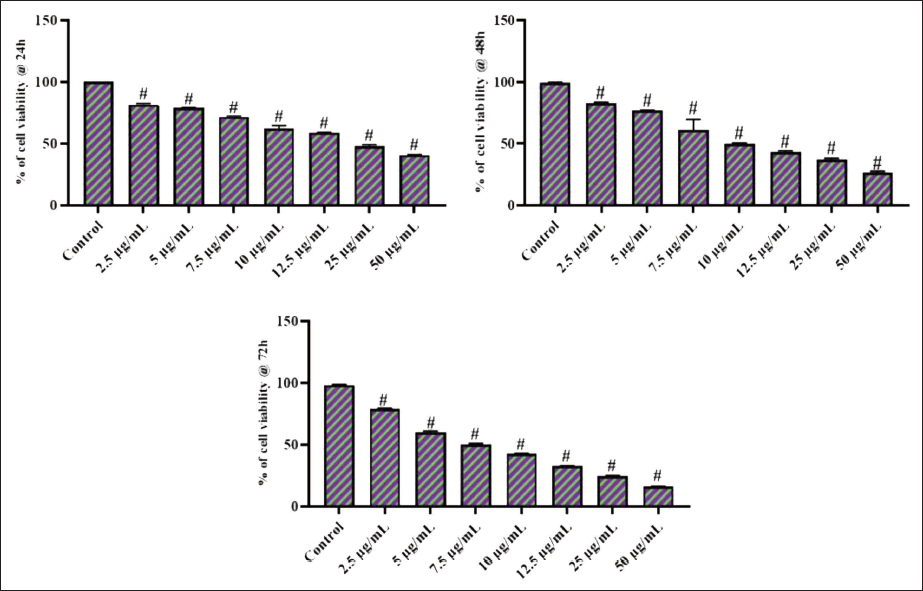
Figure 1 illustrates the findings of the MTT cytotoxicity assay of both untreated and artemetin-treated HCT-116 cells. The artemetin treatment considerably diminished the proliferation of HCT-116 cells at a dose range of 2.5–50 µg/mL, which indicates its cytotoxic properties against colon cancer cells. The elevated concentrations of artemetin remarkably inhibited the HCT-116 cell growth. The IC50 concentration of artemetin was noted to be 5 µg/mL for HCT-116 cells, and this dosage was chosen for subsequent studies.
Effect of Artemetin on the Viability of Colon Cancer HCT-116 Cells. The Artemetin Treatment Significantly Diminished the Viability of Colon Cancer HCT-116 Cells at Concentrations of 2.5–50 µg/mL, Which Indicates Its Cytotoxic Properties Against Colon Cancer Cells. The Values were Studied Using GraphPad Software and the Results are Presented as a Mean ± SD of Triplicate Values. One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Tests were Employed to Analyze the Data. The Findings are Given as a Mean ± SD of Triplicates. “#” Signifies that Values Differ Significantly at p < .05 from the Control Group.
Effect of Artemetin on Apoptosis in HCT-116 Cells
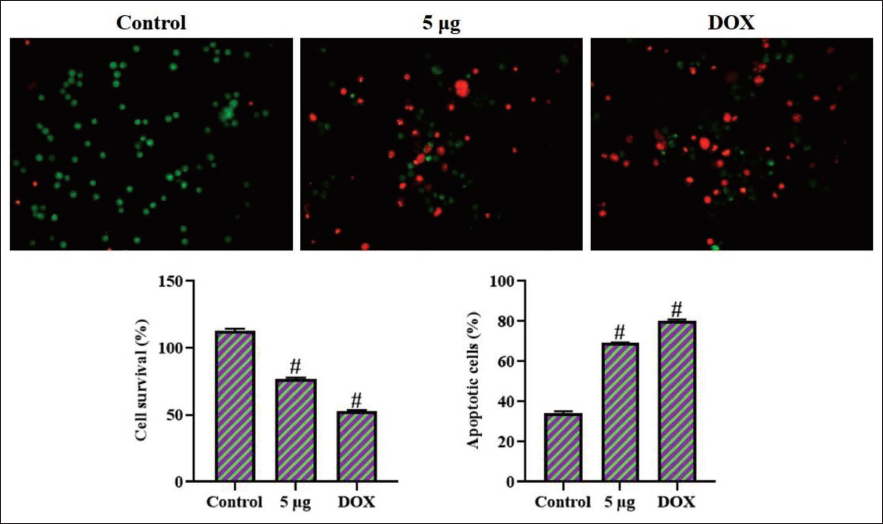
The dual staining procedures were performed to assess the apoptosis level in the untreated and artemetin-treated HCT-116 cells, with the results presented in Figure 2. The HCT-116 cells exhibited strong yellow and orange fluorescence following exposure to 5 µg/mL of artemetin, indicating the presence of early and late apoptosis, respectively. Similarly, the outcomes of DOX 2 µg treatment indicated an increased presence of cells exhibiting bright yellow/orange fluorescence, thus confirming the initiation of apoptosis (Figure 2).
Effect of Artemetin on the Apoptotic Cell Death in the Colon Cancer HCT-116 Cells. The Colon Cancer HCT-116 Cells Exhibited Increased Yellow and Orange Fluorescence After Treatment with 5 µg/mL of Artemetin, Which Suggests the Presence of Both Early and Late Apoptotic Cell Deaths, Respectively. The Values were Studied using GraphPad Software and the Results are Presented as a Mean ± SD of Triplicate Values. One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Tests were Employed to Analyze the Data. The Findings are Given as a Mean ± SD of Triplicates. “#” Signifies that Values Differ Significantly at p < .05 from the Control Group. DOX: Doxorubicin.
Effect of Artemetin on Caspase Enzyme Activities in HCT-116 Cells
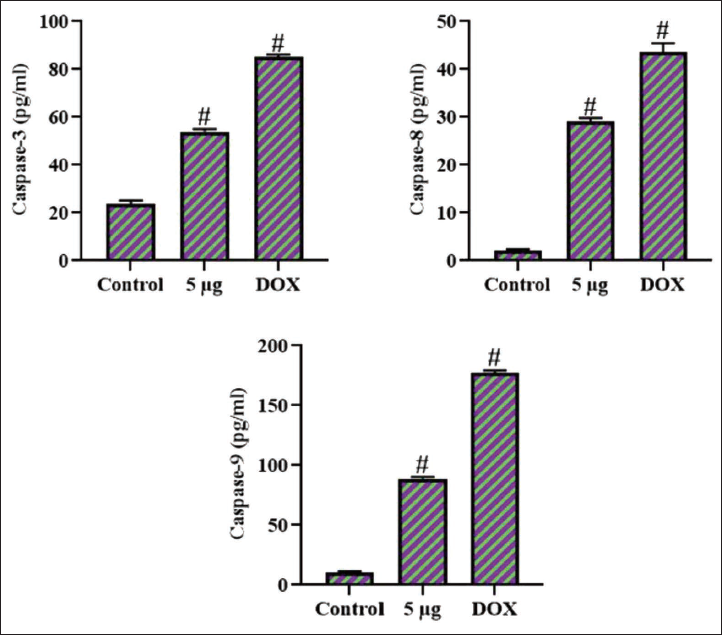
The caspase enzyme activities in cell lysates of both untreated and artemetin-exposed HCT-116 cells were investigated, with the results presented in Figure 3. The caspase-3, -8, and -9 activities were found to be diminished in the untreated cells. Whereas the 5 µg/mL of artemetin treatment resulted in a remarkable increase in all three caspase enzymes (caspase-3, -8, and -9) in the colon cancer cells when compared with the control. These results were further corroborated by the findings of DOX treatment, which likewise elevated the caspase enzyme activities in the HCT-116 cells.
Effect of Artemetin on the Caspase Enzyme Activities in the Colon Cancer HCT-116 Cells. The Activities of the Caspase-3, -8, and -9 Enzymes were Remarkably Increased in the Artemetin-treated Colon Cancer HCT-116 Cells. The Values were Studied Using GraphPad Software and the Results are Presented as a Mean ± SD of Triplicate Values. One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Tests were Employed to Analyze the Data. The Findings are Given as a Mean ± SD of Triplicates. “#” Signifies that Values Differ Significantly at p < .05 from the Control Group. DOX: Doxorubicin.
Discussion
Colorectal cancer, also called colon cancer, is a prevalent type of cancer that arises from the large intestinal linings. The onset of colon cancer is a highly complex process involving genetic and environmental factors, including dietary habits and lifestyle choices (Kumar et al., 2023). The pathophysiology of colon cancer is complex, with a series of genetic and epigenetic modifications driving the adenoma-carcinoma sequence. These changes can include mutations in key genes implicated in cell proliferation, differentiation, and apoptosis. Additionally, environmental factors, such as excessive consumption of red and processed meats, as well as a sedentary lifestyle, were connected with an elevated risk of colon cancer development (Abreu Lopez et al., 2024). The HCT-116 cells have become an extensively utilized in vitro model for studying the underlying mechanisms and potential therapeutic interventions in colon cancer (Cruz et al., 2018). The HCT-116 cell line has become a valuable tool in colon cancer research, as it retains many of the characteristics of human colorectal carcinoma. This cell line has been extensively used to investigate the molecular mechanisms underlying colon cancer development, including cell proliferation, migration, invasion, and drug responsiveness (Chitiva et al., 2024). Furthermore, HCT-116 cells have been employed to study the role of cellular mechanisms, like growth, differentiation, and apoptosis, in the case of colon cancer development. By utilizing this in vitro model, scientists can gain important insights into the pathophysiology of colon cancer and explore potential therapeutic strategies to address this significant public health concern (Robson et al., 2023).
The MTT cytotoxicity test is a widely used method in cancer research to assess the viability of tumor cells in response to various treatments, including chemotherapeutic agents, novel drug candidates, and other potential cytotoxic compounds. This versatile assay has become a staple tool in the in vitro evaluation of anti-cancer agents, as it offers a rapid and quantitative assessment of cellular metabolic activity, which is directly proportional to the number of viable cells (Ghasemi et al., 2021). The fundamental premise of the MTT assay is that metabolically active cells transform the yellow tetrazolium salt MTT into purple formazan crystals, which can be solubilized and measured using spectrophotometry (Pantea et al., 2023). This process is facilitated by the activity of mitochondrial dehydrogenase enzymes, which are essential for cellular respiration and energy production. By measuring the absorbance of the formazan product, the relative viability of cells exposed to different treatments can be determined, enabling the evaluation of cytotoxic effects and the potency of anti-cancer compounds (Eder et al., 2022). The MTT assay has been widely adopted in cancer research due to its simplicity, sensitivity, and reproducibility. It has been used to assess the in vitro cytotoxicity of numerous chemotherapeutic compounds, natural products, and novel drug candidates against several tumor cells (Lewandowski et al., 2022). In this work, the MTT test was employed to examine the cytotoxic potentials of artemetin against HCT-116 cells. The findings clearly proved that the artemetin treatment remarkably decreased the HCT-116 cell growth in different periods dose-dependently.
Apoptosis, a highly regulated form of cell death mechanism, has gained extensive interest in the area of cancer research because of its potential therapeutic applications. Cancer cells often exhibit dysregulated apoptotic pathways, allowing them to evade this natural cell death mechanism and proliferate uncontrollably. Induction of apoptosis in tumor cells, therefore, represents a favorable approach for cancer therapy (D’Amico & De Amicis, 2024). One of the key applications of the initiation of apoptosis in tumor cells is the development of novel anti-cancer agents. Plant-derived compounds have been shown to demonstrate anti-cancer effects by triggering apoptotic signaling in tumor cells (Naeem et al., 2022). In vitro cancer research has been instrumental in elucidating the mechanisms by which apoptosis can be induced in cancer cells. Dual staining analysis, a technique that combines two or more staining methods, has emerged as a valuable tool in this endeavor, providing a comprehensive understanding of the apoptotic process in cancer cells (Liu et al., 2015). Dual staining analysis has been instrumental in elucidating the mechanisms by which drug candidates trigger apoptosis in tumor cells. By combining staining techniques, researchers can gain a better understanding of the apoptotic process, including the involvement of specific signaling pathways, the activation of caspases, and changes in mitochondrial function (Sun et al., 2012). The activation of apoptosis in tumor cells has also been studied as a strategy for overcoming chemotherapy resistance. Cancer cells often develop intrinsic resistance to chemotherapy through the inhibition of the apoptotic pathway, such as the overexpression of anti-apoptotic genes or the less expression of pro-apoptotic genes. By targeting the apoptotic pathway, novel anti-cancer therapies aim to circumvent this resistance and enhance the effectiveness of cancer therapy (Hamilton et al., 2021). In this work, the dual staining assay results clearly demonstrated the intense yellow/orange fluorescence in the HCT-116 cells following exposure to artemetin, which witnesses the occurrence of both early and late apoptosis, respectively. Therefore, it was clear that artemetin can trigger apoptosis in colon cancer cells.
At the molecular level, apoptosis is characterized by the activation of a family of cysteine proteases (caspases), which act as the executioners of this cell self-destruction program. Among the various caspases, caspase-3, -8, and -9 have emerged as particularly important in the case of cancer treatment (Abdelghany et al., 2024). Cancer is a disease marked by the dysregulation of apoptosis, with cancer cells often exhibiting resistance to the natural cell death process. This resistance can arise by several mechanisms, including the overexpression of anti-apoptotic genes and the downregulation of pro-apoptotic genes. As such, the stimulation of caspase-dependent apoptosis in tumor cells has become a promising target for anti-cancer therapies (Kumar et al., 2022). The intrinsic or mitochondrial-mediated apoptotic pathway implicates the release of cyt-c from mitochondria, which then binds to the Apaf-1 protein, resulting in caspase-9 activation (Lopez & Bouchier-Hayes, 2022). The extrinsic or death receptor-mediated mechanism is commenced by the binding of extracellular death ligands to cell surface receptors, triggering caspase-8 activation. Both of these pathways ultimately converge on caspase-3 activation, the central executioner caspase responsible for the cleavage of hundreds of cellular proteins, leading to morphological and biochemical alterations (Li et al., 2022). Targeting the caspase-mediated apoptotic signaling in tumor cells has been the focus of extensive research. One approach is the use of plant bioactive compounds that demonstrate anti-tumor effects by the activation of apoptotic signaling (Jiang et al., 2020). Additionally, strategies to inhibit the expression of anti-apoptotic genes, like Bcl-2, can sensitize tumor cells to apoptosis-inducing therapies (Dabrowska et al., 2016). The findings of the current work suggested that artemetin can considerably elevate the caspase-3, -8, and -9 enzymes in the HCT-116 cells. These results suggest that artemetin can activate caspase-mediated apoptosis in colon cancer cells.
Conclusion
The current study revealed that artemetin possesses anti-cancer activities against colon cancer, reducing cell viability and inducing apoptosis in HCT-116 cells by upregulating caspase activities. The artemetin treatment significantly reduced cell growth and activated caspase-dependent apoptosis in HCT-116 cells. Consequently, the present findings indicate artemetin as a promising anti-tumor agent for future colon cancer therapy. Additionally, future research is strongly advised to thoroughly understand the specific therapeutic roles of artemetin’s anti-cancer actions against colon cancer.
Footnotes
Abbreviations
AO/EB: Acridine orange/ethidium bromide; DMEM: Dulbecco’s modified Eagle’s Medium; DMSO: Dimethyl sulfoxide; DOX: Doxorubicin; FBS: Fetal bovine serum; MTT: (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
None.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project supported by Clinical Study on Endoscopic Resection of Colorectal Submucosal Tumors Using Under water Combined with Metal Clip-Assisted Snare, China (Grant No. 2441ZF271).
