Abstract
Background
In our previous research, we addressed the issues of poor stability and water solubility of scutellarin by developing scutellarin nanoparticles. Our findings indicated that these nanoparticles exhibited enhanced anti-colon cancer activity compared to scutellarin alone. However, the underlying mechanisms of scutellarin’s anti-colon cancer effects require further investigation. Scutellarin is known to suppress the proliferation of different tumor cells. In this study, we explored whether scutellarin, encapsulated in nanoparticles, could suppress the proliferation of colon cancer cells by modulating ferroptosis.
Objectives
In this study, we explored whether scutellarin, encapsulated in nanoparticles, could suppress the proliferation of colon cancer cells by modulating ferroptosis.
Materials and Methods
SW480 and HCT-116 cells were divided into (a) control group, scutellarin group, and scutellarin nanoparticles group; and (b) control group, scutellarin nanoparticles group, ferrostatin-1 group, and scutellarin nanoparticles + ferrostatin-1 group. First, we determined the half-maximal inhibitory concentration of colon cancer cells by Cell Counting Kit-8 (CCK-8) assay. Then, the ferroptosis of cells in each group was detected, and the levels of oxidative stress-related factors, superoxide dismutase (SOD) and malondialdehyde (MDA), were detected by biochemical kits. Cell death was detected by terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay, cell invasion was detected by Transwell assay, and reactive oxygen species (ROS) levels were detected by flow cytometry. Western blotting was used to detect ferroptosis-related glutathione peroxidase 4 (GPX4), solute carrier family 7 member 11 (SLC7A11), and ferroportin 1 (FPN1) expression levels.
Results
We found that in SW480 and HCT-116 cells, the IC50 of scutellarin nanoparticles cells was lower, cell apoptosis and invasion were more obvious, the oxidative stress level was also higher, Fe2+ content was increased, and the expression of GPX4, SLC7A11, and FPN1 was decreased. These results were changed after the addition of ferrostatin-1.
Conclusion
Scutellarin nanoparticles have a better anti-tumor effect than the same dose of scutellarin. Scutellarin nanoparticles can inhibit the proliferation, migration, and invasion of cancer cells by promoting ferroptosis, as well as cell apoptosis.
Introduction
Worldwide, colon cancer ranks third in terms of malignant tumors and fourth in terms of cancer deaths (Vogel et al., 2022). At present, the primary treatment of colon cancer is surgery combined with radiotherapy or chemotherapy, and only 65.2% of patients survive 5 years after diagnosis, which is a poor prognosis (Fabregas et al., 2022). The current treatment methods do not have a good control of colon cancer cells. Therefore, it is necessary to find new effective treatment methods. Scutellarin is a flavonoid extracted from the chrysanthemum plant Erigeron brevis, which has good pharmacological effects such as anti-oxidation, anti-platelet, liver protection, and anti-inflammation (Wang, Bao, et al., 2023). It can significantly improve the clinical symptoms of patients with cardiovascular and cerebrovascular diseases and also has anti-tumor effects (Vesaghhamedani et al., 2023; Xie et al., 2023). Its role in the occurrence and development of colon cancer and related mechanisms is rarely reported. The aim of this study is to explore whether scutellarin regulates the development of colon cancer through ferroptosis in order to provide an effective method for the clinical treatment of colon cancer patients.
In addition to its low bioavailability, scutellarin is poor in its ability to dissolve in water and absorb. Drug-carrying nanoparticles can enhance the permeability of biofilms (Sahab-Negah et al., 2020). The high dispersion and large surface area of nanoparticles enhance contact between drugs and biofilms and increase the drug’s bioavailability and absorption at the absorption site (Liu, Yang, et al., 2020). The systemic toxicity of a drug can be greatly reduced by reducing its local concentration, thus decreasing its concentration in the rest of the body (Ali, 2023). After coating scutellarin, we found its absorption rate to increase significantly (Wang et al., 2021).
An imbalance in intracellular iron metabolism leads to ferroptosis, a new type of programmed cell death characterized by iron accumulation and lipid peroxidation. It mainly catalyzes lipid peroxidation of intracellular lipids in the presence of intracellular iron, thereby inducing cell death. When ferroptosis occurs, the highly expressed polyunsaturated fatty acids in cell membranes dehydrogenate, triggering the peroxidation chain reaction and producing a large number of oxygen free radicals, leading to cell death. Intracellular iron overload (Su et al., 2020) and lipid peroxidation (Li et al., 2021) are prerequisites for ferroptosis in cells. In this study, we used scutellarin nanoparticles as carriers to investigate their inhibitory effect on colon cancer by regulating ferroptosis.
Materials and Methods
Cell Experimental Groups
We routinely cultured the human colon cancer cells HCT-116 (Wuhan Pricella Biotechnology Co., Ltd., CL-0096) and SW480 (Wuhan Pricella Biotechnology Co., Ltd., CL-0223B) in DMEM low-glucose medium supplemented with 10% fetal bovine serum (FBS). Two experiments were performed for each of the two cell lines. In experiment 1, the cells were divided into scutellarin group (25, 50, 75, and 100 µmol/L scutellarin) and scutellarin nanoparticle group (25, 50, 75, and 100 µmol/L scutellarin nanoparticles). In experiment 2, the cells were divided into control group (no treatment), scutellarin nanoparticle group (35.7 µmol/L or 38.5 µmol/L scutellarin nanoparticles), IC50 scutellarin group (35.7 µmol/L or 38.5 µmol/L scutellarin), and nanoparticle no-loaded group (35.7 µmol/L or 38.5 µmol/L nanoparticle no-loaded), ferrostatin-1 group (1 µmol/L), and IC50 scutellarin nanoparticle combined with ferrostatin-1 group (35.7 µmol/L or 38.5 µmol/L scutellarin nanoparticles + 1 µmol/L ferrostatin-1). The nanoparticle encapsulation technology is subject to the invention patent application (CN 112656776A).
Cell Counting Kit-8 (CCK-8) Assay
Logarithmic cells were inoculated into 96-well plates at a density of 5 × 103/well, 100 µL/well, at 37°C with 5% CO2. Following 60–70% confluence of the cells, different concentrations of drugs were added for 48 h, depending on the group. CCK-8 dye (10 µL) was added to each well (Biosharp Biotechnology Co., Ltd.) 48 h after inoculation and cultured for 1 h at 37°C. Measurement of absorbance at 450 nm was carried out using a microplate reader (BioTek Instruments, Inc.).
Transwell Assay
We used a Transwell assay to detect the invasion of each group. The cells in the logarithmic growth phase were washed with phosphate-buffered saline (PBS) and collected after trypsin digestion, centrifuged at 250 g for 5 min, and the supernatant was removed by suction. An appropriate amount of medium was added to make it a cell suspension, and the cell density was adjusted to 2 × 105 cells/well. The corresponding concentrations of drugs were added according to the experimental protocol and incubated at 37℃ with 5% CO2. We added 600 µL of 10% FBS medium to the lower chamber. A methanol–glacial acetic acid stationary solution (3:1) was used to fix cells that had migrated to the membrane’s lower surface, and Giemsa staining was performed after 10 min. Cell migration was microscope-counted (LEICA DMI1) at a magnification of 100 in four randomly selected fields.
Reactive Oxygen Species (ROS) Assay
To measure ROS levels, we utilized a flow cytometry-based method with the ROS assay kit (S0033S, Beyotime Biotechnology). The cells were then incubated with 1 mL of this DCFH-DA solution (1:1,000, 10 µmol/L) at 37°C in a 5% CO2 incubator for 20 min. Post-incubation, the cells were washed three times with a serum-free culture medium. ROS levels were subsequently quantified using flow cytometry.
The Contents of Fe2+, Superoxide Dismutase (SOD), and Malondialdehyde (MDA) Were Detected by Kits
The cells in each group were collected. The contents of Fe2+ (Beijing Xolaibio Technology Co., Ltd., BC5415), SOD, and MDA were detected according to the instructions of the kit.
Western Blotting
In each group, GPX4, SLC7A11, and FPN1 expression levels were assessed by Western blotting. The cells were collected and lysed with 100 mL of RIPA (Beyotime Biotechnology Co., Ltd.) for 10 min, and the supernatant was centrifuged. The BCA method (Beyotime Biotechnology Co., Ltd.) was used for measuring protein concentrations. 20 µg of total protein was separated using sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) (12% gel, BL502A, Biosharp) and transferred to Immobilon-PSQ PVDF (ISEQ00010; Sigma–Aldrich). Previous protocols have been described and published for GPX4 (abclonal, A1933, 1:1,000), SLC7A11 (affbiotech, DF12509, 1:2,000), FPN1 (abclonal, A14884, 1:5,000), and β-actin (abclonal, AC026, 1:50,000). After washing, ECL luminescent solution was added, and the bands were scanned using a chemiluminescent gel imager (Shanghai Tanon Technology Co., Ltd., China).
Statistical Analysis
In order to determine statistical significance, we used Student’s t-tests and one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test, to analyze the data after three repetitions of each experiment. GraphPad Prism was used for data analysis and graph production.
Results
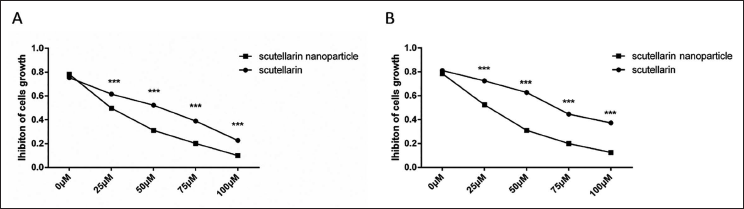
The IC50 values of scutellarin nanoparticles and scutellarin on SW480 and HCT-116 cells were detected by CCK-8, and the results showed that the cell proliferation inhibition rate increased with the increase of drug concentration. In the SW480 cell line, IC50 values of scutellarin and scutellarin nanoparticles were 70.8 µmol/L and 36.7 µmol/L, respectively (Figure 1A). In the HCT-116 cell line, the IC50 values of scutellarin and scutellarin nanoparticles were 90.1 µmol/L and 38.5 µmol/L, respectively (Figure 1B). Building on these results, subsequent experiments will utilize 40 µmol/L of scutellarin or scutellarin nanoparticles.
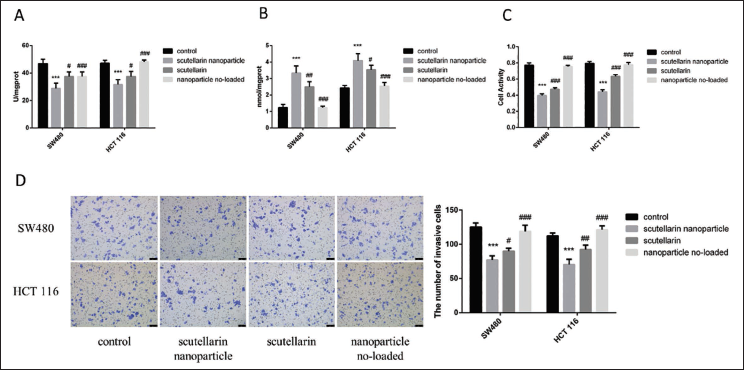
SOD and MDA levels were determined across the two cell lines using biochemical kits. As illustrated in Figure 2A and B, scutellarin nanoparticles significantly reduced SOD levels compared to other groups (p < 0.05), while MDA levels were markedly elevated (p < 0.05). Additionally, cell viability and invasion assays indicated that the scutellarin nanoparticles group had significantly fewer surviving cells and reduced cell invasion than those observed in the other groups across both cell lines (p < 0.05) (Figure 2C and D).
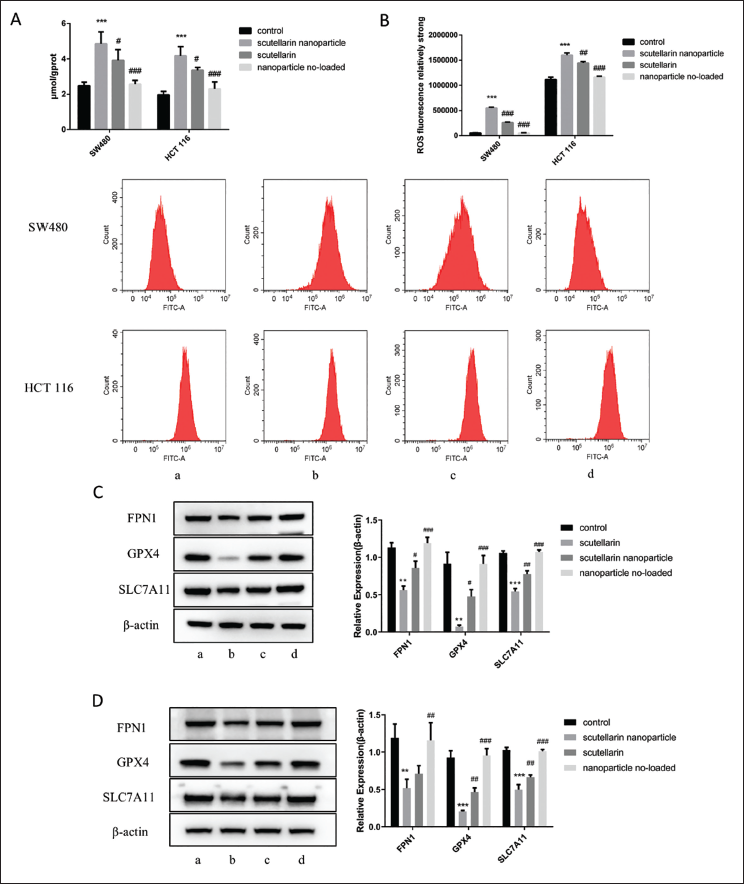
Next, we assessed the Fe2+ content in the two cancer cell lines. As shown in Figure 3A, Fe2+ levels were notably elevated in the scutellarin nanoparticle group relative to the other groups (p < 0.05). Similar results were observed in flow cytometry measurements of cellular ROS levels (Figure 3B). Western blot analysis of relevant proteins revealed that the expression of GPX4, SLC7A11, and FPN1 was significantly reduced in cells treated with scutellarin nanoparticles compared to other groups (Figure 3C and D, p < 0.05).
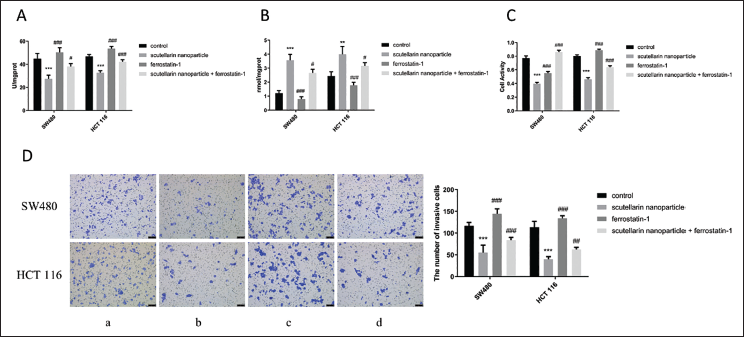
In the previous research, we found that scutellarin nanoparticles significantly reduced the proliferation and viability of SW480 and HCT-116 cells and induced ferroptosis. To further investigate if scutellarin nanoparticles influence colon cancer cells through ferroptosis, we introduced ferrostatin-1, a ferroptosis inhibitor, in subsequent experiments. The results showed that the combination of scutellarin nanoparticles and ferrostatin-1 significantly increased SOD levels (p < 0.05) and decreased MDA levels (p < 0.05) compared to the scutellarin nanoparticles group alone (Figure 4A and B). Additionally, cell viability and invasion assays revealed that the scutellarin nanoparticles + ferrostatin-1 group showed significantly higher levels of cell apoptosis and reduced cell invasion compared to the scutellarin nanoparticles group (p < 0.001) (Figure 4C and D).
Fe2+ content measurements revealed that the scutellarin nanoparticles combined with ferrostatin-1 significantly reduced Fe2+ levels compared to the scutellarin nanoparticles alone (p < 0.01) (Figure 5A). Similar results were observed for ROS levels (Figure 5B). Western blot analysis of Fe2+-related proteins revealed that, in the scutellarin nanoparticles + ferrostatin-1 group, the expressions of GPX4, SLC7A11, and FPN1 were significantly upregulated compared to the scutellarin nanoparticles group (p < 0.05) (Figure 5C and D).
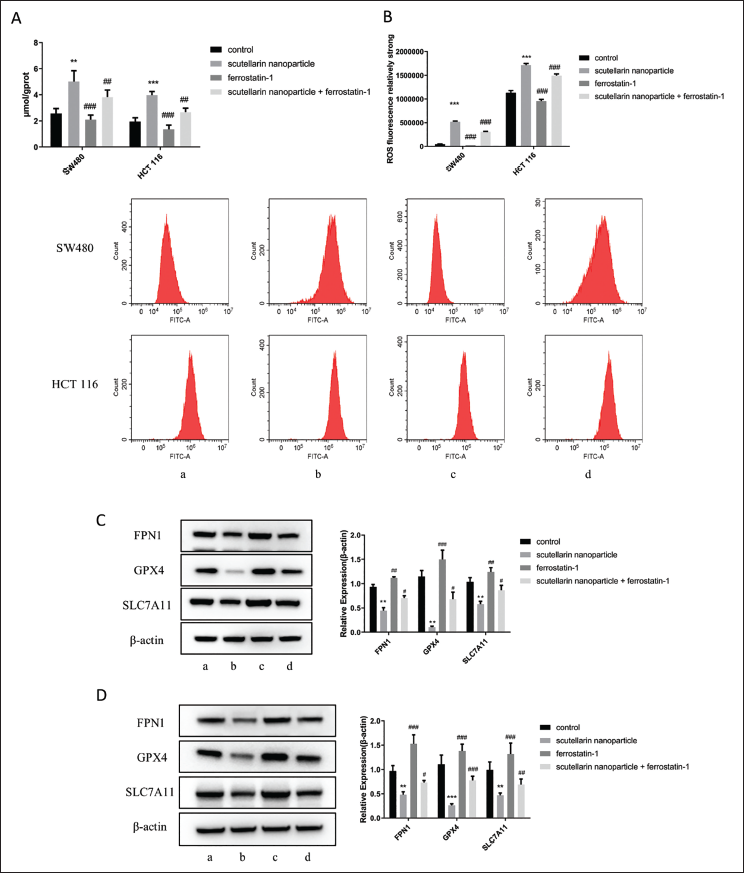
Discussion
With the significant increase in the morbidity and mortality of tumors, it is essential to find new drugs with effective targets and anti-tumor effects (Sung et al., 2021). Aside from inhibiting tumor cell growth, proliferation, invasion, and metastasis (Wang, Liu, et al., 2023), scutellarin can also increase chemotherapy sensitivity (Ge & Zhong, 2024). Most of its anti-cancer mechanisms are related to the apoptosis pathway, but it also involves tumor angiogenesis, cell cycle, ferroptosis, and other aspects, which are expected to become a new generation of anti-tumor drugs (Shi et al., 2019).
Compared with scutellarin itself, scutellarin nanoparticles have advantages regarding drug stability and bioavailability. By encapsulating in nanocarriers, scutellarin nanoparticles form a relatively closed environment, which can effectively prevent the destruction of the drug by external factors, thereby improving the chemical stability of the drug. This encapsulation can also prevent aggregation and sedimentation between the drug particles, maintain the uniform dispersion state of the drug in the preparation, further ensure the stability of the drug, and enhance its bioavailability. In addition, scutellarin has poor water solubility and weak permeability in small intestinal epithelial cells, resulting in low oral bioavailability. Nanoparticle technology can improve the bioavailability of drugs and promote their absorption and transport by improving solubility and permeability. In this study, colon cancer cells were used as the research subjects. CCK-8 assay was used to detect the IC50 values of different concentrations of scutellarin nanoparticles and scutellarin on SW480 and HCT-116 cells. This could provide clearer observation results and data analysis based on these concentrations in the experiment. Within a certain concentration range, the effect of breviscapine on cells showed a dose-dependent manner, which means that as the concentration increased, its effect on cells also changed. These concentrations of 25, 50, 75, and 100 µmol/L were selected for the study so that the effects of different concentrations of scutellarin on cells could be observed to evaluate its biological activity more accurately. At the same time, these concentration gradients help draw the concentration–effect curve to understand the mechanism and effect of breviscapine more intuitively. The results showed that the inhibition rate of cell proliferation increased with increased drug concentration. This indicated that the colon cancer cells were less sensitive to breviscapine alone, but after coating with breviscapine nanoparticles, the sensitivity of cells to breviscapine was enhanced, and the colon cancer cells were significantly inhibited by breviscapine nanoparticles.
An enlargement of the cancerous site can aggravate the inflammatory response and oxidative stress in patients with colon cancer (Saleh et al., 2023). Changes in MDA content can reflect the degree of oxidative stress through the content of oxygen free radicals in tissues and tissue damage (Jelic et al., 2021). It can remove free radicals from the body, protect cells from damage, and play a crucial role in maintaining the balance between oxidation and antioxidation (Muro et al., 2024). We used biochemical kits to measure SOD and MDA levels in both cell lines. Our results showed that treatment with scutellarin nanoparticles increased MDA content and decreased SOD content, and inhibited the growth of colon cancer cells. This suggests that scutellarin nanoparticles can inhibit cancer cell growth by regulating oxidative stress. Additionally, cell viability and invasion assays indicated that the scutellarin nanoparticles group had significantly fewer surviving cells and reduced cell invasion than those observed in the other groups across both cell lines. Subsequently, we examined the changes in ROS, Fe2+, and ferroptosis-related proteins in cells. We found that scutellarin nanoparticles could increase the levels of ROS and Fe2+ in cells. Downregulation of GPX4, SLC7A11, and FPN1 protein expression. This indicated that scutellarin nanoparticles significantly reduced the proliferation and viability of SW480 and HCT-116 cells and induced ferroptosis.
To further investigate if scutellarin nanoparticles influence colon cancer cells through ferroptosis, we introduced ferrostatin-1, a ferroptosis inhibitor, in subsequent experiments. Intracellular Fe2+ is required for tumor cell growth and proliferation (Lei et al., 2022). Studies have shown that tumor cells contain more Fe2+ than normal cells because transferrin binds to Fe3+ in peripheral blood, and Fe3+ enters cells through endocytosis. Then, Fe3+ is reduced to Fe2+ under the action of intracellular reducing substances and then participates in cellular activities such as deoxyribonucleic acid (DNA) synthesis (Wu et al., 2023). Consequently, Fe2+ generates ROS through the catalyzed Fenton reaction, which destroys tumor cells (Du et al., 2019; Yang et al., 2014).
After adding ferrostatin-1, MDA content and SOD levels in cancer cells were increased, and scutellarin nanoparticles no longer inhibited cell viability and invasion ability. At the same time, Fe2+ and ROS levels were decreased in cancer cells. This suggests that scutellarin nanoparticles can inhibit the growth of tumor cells by regulating ferroptosis.
It has been demonstrated that oxidative stress can induce apoptosis, necrosis, and ferroptosis (Aycan et al., 2023), and that ferroptosis is mostly characterized by iron-dependent lipid ROS accumulation (Jiang et al., 2021). GPX4 catalyzes the conversion of lipid peroxides to non-toxic lipids (Xue et al., 2023). When GPX4, a key regulator of ferroptosis, is inactivated, lipid peroxides are susceptible to reduction by free Fe2+, resulting in excess ROS. Besides mediating cystine uptake (Liu, Xia, et al., 2020), SLC7A11 is also involved in cell growth, proliferation, metabolism, and oxidative stress response (Jyotsana et al., 2022). SLC7A11 is overexpressed in a variety of tumors, and its expression level is closely related to the proliferation, invasion, metastasis, and tumor microenvironment of tumor cells. Its upregulation represents drug tolerance and a low survival rate (Liu et al., 2021). SLC7A11 plays an important role in the Gpx4 regulatory pathway and lipid metabolism pathway that starts the ferroptosis process and is closely related to ferroptosis (Liu et al., 2019; Sakamoto et al., 2020). The ferroptosis pathway is the main signaling pathway regulating iron accumulation or iron overload, and FPN1, as an important target in the ferroptosis pathway, is the only known iron transfer protein, membrane ferroportin (Zou et al., 2022). Therefore, we analyzed the expression of the above proteins by Western blot to verify whether the ferroptosis pathway was activated in cancer cells. The results showed that ferrostatin-1 inhibitors could upregulate the protein expression of GPX4, SLC7A11, and FPN1. This indicated that the ferroptosis pathway in cancer cells was activated, and scutellarin nanoparticles could inhibit tumor cell growth by regulating ferroptosis. At the same time, the test results also show that scutellarin nanoparticles have better therapeutic effects than scutellarin itself, which provides certain theoretical support and a basis for the clinical application of scutellarin nanoparticles.
Conclusion
In conclusion, breviscapine nanoparticles exhibit unique novelty and great therapeutic potential in the treatment of colon cancer by targeting the ferroptosis signaling pathway to inhibit colon cancer. Its unique mechanism of action and the application of nanotechnology make it broad application prospects and in-depth research value in the treatment of colon cancer.
Footnotes
Abbreviations
ANOVA: Analysis of variance; BCA: Bicinchoninic acid; CCK-8: Cell Counting Kit-8; FPN1: Ferroportin 1; GPX4: Glutathione peroxidase 4; MDA: Malondialdehyde; RIPA: Radioimmunoprecipitation assay; ROS: Reactive oxygen species; SDS-PAGE: Sodium dodecyl sulfate–polyacrylamide gel electrophoresis; SLC7A11: Solute carrier family 7 member 11; SOD: Superoxide dismutase; TUNEL: Terminal deoxynucleotidyl transferase dUTP nick-end labeling.
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Commercial cell lines SW480 and HCT-116 are used, which do not involve animal experiments and primary cell extraction, therefore no ethical approval required.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Scientific Research Project of the Science and Technology Department of Sichuan Province (No. 2022YFS0408).
