Abstract
In this study we used biosynthesis methods to create bimetallic nickel cobalt nanoparticles (Ni-Co NPs) utilizing seaweed. Before exposure to target cells, the characterization of Ni-Co NPs is done by UV-Vis spectrophotometry, EDX, SEM, TEM, the shape of g Ni-Co NPs are polygonal form and its size is measured 38.27 ± 3 nm. The cytotoxic effect of g Ni-Co NPs on HuH7 and HCT cells were determined by MTT and NRU assays. The cytotoxicity of NPs increased in a concentration dependent manner and it showed high cytotoxic effect on HCT-116 cells than HuH-7 cells. We determined IC50 24 h for HuH-7 and HCT -116 cells at 24 h, it was 65.84 and 24.73 μg/mL, respectively. ROS was elevated at higher concentration of Ni-Co NPs. LPO was increased at 16 μg/mL in HuH-7 cells and 19 μg/mL in HCT-116 cells. CAT was reduced in HCT-116 cells than HuH-7 cells high concentration of NPs. JC-1 staining, the loss of MMP in control, Ni-Co NPs exposed cell were evaluated. In HuH-7 and HCT-116 cells, maximum apoptotic cells were observed at high concentration. Apoptotic genes were expressed in both type cells. The above findings highlight the significance of Ni-Co NPs and useful in a number of cancer treatments.

Introduction
Comparing green production of nanoparticles with other comparable approaches, the use of living cells through biological processes is more efficient and produces a larger mass. In order to create green nanoparticles, plants provide a number of components and biochemicals that can act as reducing and stabilizing agents. Comparing the green synthesis procedures to conventional biological, physical, and chemical methods, they are more stable, non-toxic, economical, and environmentally friendly. 1 Emerging uses for green produced metal and metal oxide nanoparticles in the biomedical field include biosensing platforms, immunotherapy, wound healing, tissue repair, regenerative medicine, and dentistry. Different biological activities, environmental remediation, photocatalysis, catalytic reduction, sensing, energy storage, and several organic transformations like coupling, reduction, and multicomponent reactions are just a few of the functions of plant-mediated cobalt oxide nanoparticles made from different plant extracts.2,3 According to Korde et al., 4 green produced selenium nanoparticles can enhance the performance of solar cells, photocopiers, xerography, rectifiers, antioxidants, catalysis, and anticancer devices. Potential photocatalytic dye degradation, antioxidant, antidiabetic, anticancer, antibacterial, and antifungal capabilities are all exhibited by green produced cerium oxide nanoparticles. 5 The safety and exposure route of nanoparticles are still being investigated. The toxicity of materials at the nanoscale is a topic of particular discussion due to the size difference. Cobalt nickel nanoparticles are now being considered for the antitumor effect on human breast cancer cells. 6 However, the so-recent report indicated that there was a lack of systematic assessment of the DNA damaging and carcinogenic potential of bimetallic NPs in spite of their extensive use in nanotechnological applications. 7 Based on insufficient data in people and sufficient evidence in experimental animals, the International Agency for Research on Cancer has carcinogenic hazard of chemicals to humans. 8
Human liver cancer (HuH-7) and colon cancer cells have been a key model system in the study of mechanism and the function of the cancer cells against xenobiotic materials. 9 Cancer cells are of special interest in nanotoxicological and/or nanomedicine research for the primary reason that cancer therapy heralds a new era marked by improvements in the specificity, effectiveness, and tolerability of cancer treatments. 10 Also, some of the NPs are intended to target the blood vessels, it has been suggested that the interactions between endothelial cells and NPs should be carefully assessed to better understand the potential in vivo effects of NPs. 11
Seaweeds are increasingly used in European cuisines due to their nutritional value. Many algal constituents, such as polyphenols, are important antioxidants and thus considered beneficial to humans. 12 However, many seaweed species can accumulate heavy metals and exhibit potential health risks upon ingestion. 12 Particle size, content, and associated reactivity all affect the toxic effects on cancer cells. 13 Experimental study has revealed a larger mutagenesis potential of the Ni-Co mixture when compared with its individual components. 14 Therefore, we investigated the cytotoxicity and apoptotic properties of Ni-Co NPs on HuH-7 and HCT cells over 24 h. The overproduction of ROS in live tissue under stress is one among the several mechanisms underlying NPs-mediated toxicity. Cells primarily produce reactive oxygen species from mitochondria, and the electron transport chain is where most of the ROS is produced. Oxidative stress, apoptotic responses and genotoxicity reactions are the principal mechanisms of toxicity in Ni-Co NPs. The objective of this study was to investigate the toxic effects of Ni-Co NPs on HuH-7 and HCT cells. Furthermore, our results will be useful in assessing the environmental friendliness and safety of gNi-Co NPs use in industry.
Materials and Methods
Chemicals and Reagents
The chemicals 4,5-dimethyl-2-thiazolyl)-2,5 diphenyl-2H-tetrazolium bromide (MTT), dimethyl sulfoxide (DMSO), Hoechst 33258 fluorescent dye, 2′,7′-dichlorodihydrofluorescein diacetate (H2DCFDA), Dulbecco’s Modified Eagle Medium (DMEM), Phosphate Buffered Saline (PBS), Fetal Bovine Serum (FBS), Trypsin- EDTA solution 1X etc, were purchased from Sigma-Aldrich. Other chemicals related to the current experiment were bought at local markets.
Synthesis of Nickel Cobalt Nanoparticle from Algae Sargassum tenerrimum
In this we have synthesized nickel cobalt nanoparticle (Ni-Co NPs) from algae Sargassum tenerrimum according (Almutairi et al., 2024) method. 15 Sargassum tenerrimum is common seaweed algae collected from the sea coast of Dammam Saudi Arabia. It was washed and dried at 50°C for a stable dry weight. The dried algae will be grinded into a powder by using a grinder (Ikon, China). 50 gm of algae powder will be mixed with 100 mL of Milli-Q deionized water in a 250 mL glass conical flask, the mixture will be put at 4°C for 48 h before being warmed at 100°C for 60 min. To separate the aqueous extract and remove the heavy biomaterials, it will be centrifuged at 13 000 r/min for 15 min and filtered using Whatman filter paper. To prepare Ni-Co NPs using a precipitation reaction technique, 30 mL of extract of algae will be mixed with 10 mL of nickel chloride and cobalt sulfate in a 100 mL conical flask. After, it will be constant mixing, the color of the extract will be changed to brown as a result of the reduction of Ni and Co ions. To stabilize the process, drop-by-drop mixtures of ascorbic acid (0.1 M) solutions will be added to the conical flask. After the reaction is finished, the suspension will be mixed for 24 h at 30°C at 500 r/min. The precipitate material will be cleaned three times with distilled water. The precipitate materials will dry for 24 h and 4 h on a hot plate at 100°C. The black-brownish powder is a green Ni-Co NPs.
Physiochemical Characterization of Ni-Co NPs
The physical characterization of Ni-Co NPs was done by UV-Vis Spectroscopy, SEM, EDX TEM, dynamic light scattering (DLS).
UV-Vis Spectroscopy
UV-Vis spectroscopy is used to characterize and confirm the presence of reduced Ni-Co ions as Ni-Co NPs in the algal culture. The wavelength range used for taking absorbance is from 200 nm to 700 nm. Both the UV range and the visible range are present in UV-Vis spectroscopy.
Scanning Electron Microscopy (SEM)
According to Shi et al, 16 a scanning electron microscope (SEM), provides imaging and characterization of nanomaterials and nanostructures in real-time with nano-meter resolution and a wide scanning area Ni-Co NPs were put onto a carbon-coated copper grid and observed using SEM (JEOL model 2100F, Tokyo, Japan).
Transmission Electron Microscope (TEM)
It has been determined the size and shape of Ni-Co NPs determined by using Alarifi et al. method. 17 Ni-Co NPs has been suspended in water at a concentration of 1 mg/mL. The stock suspension was probe-sonicated at 40 W for 15 min. Ni-Co NPs was layered on a carbon-coated copper grid for analysis of the size of the particle. The films on the TEM grids allowed to dry before capturing the image by using TEM (JEOL model 2100F, Tokyo, Japan).
X-Ray Diffraction (XRD)
It is a technique for obtaining detailed information on the physicochemical and crystalline properties of nanomaterials. The X-ray diffraction (XRD, Rigaku, Tokyo, Japan) of the Ni-Co NPs was performed at 9 kW and coupled with Smart lab guidance software (Smart Lab Studio II package software).
Dynamic Light Scattering (DLS)
The mean hydrodynamic size and zeta potential of Ni-Co NPs in culture medium and distilled water were observed by dynamic light scattering (DLS) (Nano-Zeta Sizer-HT, Malvern Instruments, Worcestershire, UK). 17 The Ni-Co NPs suspended in water and culture medium at a concentration of 1 mg/mL. Then, the suspension was and probe-sonicated at 40 W for 15 min at room temperature to measure the hydrodynamic size and zeta potential.
Cell Culture
Human liver
Treatment of Ni-Co NPs
Ni-Co NPs were suspended in a cell culture medium and diluted to appropriate concentrations (0, 2, 10, 15, 30, 60, 100, and 150 µg/ml) to exposure for HuH-7 and HCT-116 cell lines for 24 h. The exposed cells were harvested to determine cytotoxicity, oxidative stress, apoptotic, and gene expression. Cells were not treated to Ni-Co NPs served as a control in each experiment.
MTT Test and Determination of IC50 24 h of Ni-Co NPs
The MTT test was applied to find out mitochondrial function as described by Ali et al. 18 HuH-7 and HCT-116 cell lines were seeded into a 96-well plate (8 × 104 cells/well) in the complete medium at the volume of 100 µl/well. After overnight incubation, the medium was removed and added 100 µL culture media containing different concentrations of Ni-Co NPs (0, 2, 10, 15, 30, 60, 100, and 150 µg/ml) for 24 h. The medium was replaced by MTT solution (5 mg/mL), at a volume of 20 µl to each well and incubated for 4 h at 37°C in a dark. After incubation, the MTT solution was removed, and formazan crystals formed by mitochondrial reduction of MTT were solubilized in (100 µl/well) DMSO and gently shacked for 15 minutes. The plates were read using a microplate reader (Synergy-H1; BioTek, United States) at a wavelength of 570 nm. The assay was performed in triplicate with 10 replicates per sample. On the basis of IC50-24 h value, the three sublethal concentrations were determined for HuH-7 cells Sub lethal I (1/4th of IC50 = ∼16.46 µg/ml), Sub lethal II (1/2nd of IC50 = ∼32.92 µg/ml) and Sub lethal III (3/4th of IC50 = ∼49.38 µg/ml). The three sublethal concentrations for HCT-116 cells Sub lethal I (1/4th of IC50 = ∼6.18 µg/ml), Sub lethal II (1/2nd of IC50 = ∼12.36 µg/ml) and Sub lethal III (3/4th of IC50 = ∼18.55 µg/ml).
Neutral Red Uptake Test
The g- Ni-Co NPs cytotoxicity was evaluated using the NRU test in according with Ali et al 19 method.
Determination of Intracellular Reactive Oxygen Species (ROS)
Production of ROS was measured in HuH-7 and HCT-116 cells after exposure NPs by using DCFH-DA dye as a fluorescence agent. 20 The HuH-7 and HCT-116 cells were seeded in black 96 wells plate at density of 8 ×104 cells/well at 37°C and allowed to adhere for 24 h in a CO2 incubator at 37°C. Next, cells were exposed to different concentration of g Ni-CO NPs to HUH-7 and HCT-116 cells followed by incubation for 24 h in the CO2 incubator at 37°C. On completion of their respective exposure periods, incubation media was discarded and replaced with 100 µl of pre-prepared diluted H2DCF-DA dye solution in the dark, the plates were returned to the incubator for 60 min. The absorbance was measured at wavelengths of 485 and 530 nm for using a microplate reader (Microplate Reader-Gen5™ BioTek Cytation 5™, USA).
For the qualitative analysis (microscopic fluorescence imaging), the cells were seeded in 6 wells plate at a density of 8 × 104 cells/well and incubated for 24 h at 37°C. Then cells were exposed to different concentration of g Ni-CO NPs for 24 h. After incubation media was discarded and replaced with 1 mL of pre-prepared diluted DCF-DA dye solution in the dark, then plates were returned to the incubator with for 60 min the dye solution was removed and the cells were washed 3-5 times with PBS. Fluorescent images were taken by a fluorescence microscope (DMLB, Leica, Germany).
Oxidative Stress Biomarkers
HuH-7 cells (8 ×104 cells) were seeded in 25 cm2 culture flasks at different concentration of Ni-Co NPs 16.46 µg/ml, 32.92 µg/ml ,49.38 µg/ml and HCT-116 cells were seeded in 25 cm2 culture flasks at different concentration of gNi-Co NPs 6.18 µg/ml, 12.36 µg/ml, 18.55 µg/ml for 24 h. Next, cells were rinsed with chilled PBS and buffer solution was added according to each experiment methodology and scraped by scraper (Fisher Brand Cell scrapers, Fisher Scientific, USA) and collected in a glass tube after scraping. Then, the cell suspension was sonicated for 10 min at 4°C by a Q700 sonicator. After sonication, lysis buffer was mixed in scrapped cells and centrifuged at 4500 r/min for 15 min at 4°C and the supernatant (cell lysate) was put on ice to determine LPO level and CAT activity.
Measurement of LPO Level
The extent of membrane LPO was estimated by measuring the formation of malondialdehyde (MDA) using the ‘Cayman’s lipid hydroperoxide experiment kit’. according to the manufacturer. absorbance was read at 500 nm using a microplate reader (Synergy-H1; BioTek, United States).
Measurement of Catalase Activity
The CAT activity was determined using the commercially available enzyme assay kit Cayman’s. According to the manufacturer’s protocol. Absorbance was read at 540 nm using a microplate reader (Synergy-H1; BioTek, United States).
Determination of MMP
After exposure of gNi-Co NPs to HuH-7 and HCT-116 cells, the MMP of cells were evaluated by using JC-1 Cayman’s kit (Cayman chemical kit 10009172) as per the manufacturer’s protocol. The cells were grown in cover slips in 6 well plates and 96 well black bottom plates at a density of 8 × 104 then kept in a 5% CO2 incubator for 24 h at 37°C. After incubation, cells were exposed to gNi-Co NPs for 24 h. The culture medium was removed, and then the cells were washed with PBS, and stained with dye JC-1 and incubated with JC-1 dye for 60 min in 5% CO2 at 37°C. After incubation, the black 96 well plates fluorescence absorbance was measured at excitation wavelengths (485 nm) and emission excitation (530 nm) using microplate reader-Gen5™ BioTek Cytation 5™, USA). In in 6 well plates, the cells were washed 3-5 times with PBS and then cover slips were shifted to microscopic slides. Images were captured by a confocal microscope (CRCL’s LSM 780 NLO).
Determination of Caspase-3 Enzyme
After exposure of Ni-Co NPs to HuH-7 and HCT-116 cells, the caspase-3 enzyme of cells were evaluated by using caspase-3 Cayman’s kit as per the manufacturer’s protocol.
Gene Expression by RT-PCR
RNA Isolation and Synthesis of cDNA
RNA extraction of both treated and untreated cells where, HuH-7 and HCT-116 cancer cell lines were seeded in 25 mL flask at density of 8 × 104 cells/well incubated at 37°C and 5% CO2 for 2 h. cells were exposure of Ni-Co NPs for 24 h, at 37°C and 5% CO2. after that was discard the media and wash the cell with cool PBS, 1 mL of trizol was added to each flask and incubated on ice (or in freezer) for 5 min. Cells were collected in Eppendorf tubes (1 mL/tube) and then 200 µl of cold chloroform was add to each tube. Tubes were shaken by hand for 15 sec then centrifuged at 12000 r/min (4200 rcf) for 15 min at 4°C. 500 μL of supernatant (third layer) was taken and put in new cool Eppendorf tube.500 μL of iso-propanol alcohol was added to each tube, followed by gently shaking and then incubated on ice for 10 min. After incubation tubes were centrifuged at 12000 r/min for 10 min at 4°C. The supernatant was discarded carefully and then 1000 μL of cold absolute ethanol was added to each tube and shaked gently by hand for 15 sec. Tubes were centrifuged at 9000 r/min for 5 min at 4°C. The supernatant was discarded carefully (pellet was noticed) and tubes were allowed to dry for 10-15 min. 30 μL of DEPC water or nuclease-free water was added. RNA concentration and purity were measured using Nanodrop 8000 spectrophotometer (Thermo Fisher Scientific, USA). The ratio of absorbance (260 A/280 A = 1.5-2) was used as an index for RNA purity. Extracted RNA was stored at −80°C.
Complementary DNA (cDNA) Synthesis
The Component of cDNA Master Mix

Thermal Cycler Setup

The Contents of the PCR Master Mix

The Contents of the RT-PCR Master Mix for Each Gene Preparation

The Sequences of Primers

RT-PCR Protocol Setup

The RT-PCR data were analyzed using the relative gene expression (ΔΔCt) method.
22
The fold change in the level of target genes between treated and untreated cells, adjusted for the amount of GAPDH, was calculated as follows:

Statistical Analysis
Statistical analysis was done by using one-way ANOVA through GraphPad Prism Software (version 8.0.1, La Jolla California USA). Differences were considered significant at *P < 0.05. Minimum 3 independent experiments were done in duplicate for each experiment.
Results
Characterization of Ni-Co NPs
Figure 1A represents the UV vis spectrum of Ni-Co NPs and the SEM-EDS results confirmed the presence of nickel, cobalt in Ni-Co NPs and of other elements (Figure 1B). Co at 0.5 keV and Ni at 1 keV to confirm the formation of Ni-Co nanoparticles (Figure 1C). The XRD result showed to formation of Ni-Co NPs (Figure 1D). The normal size of Ni-Co NPs is 38.27 ± 3 nm (Figure 1E and F). Characterization of Ni-Co NPs (A) UV Spectrum and (B) Image of Ni-Co NPs by Scanning Electron Microscope (C) EDX Spectrum (D) X-Ray Diffraction Spectra of Ni-Co NPs. (E) Image of Ni-Co NPs by Transmission Electron Microscope (JEM 1011). (F) Distribution of Ni-Co NPs in Water Suspension
DLS and zeta potentials were used to confirm the particle size and stability of Ni-Co NPs in water and culture media suspension. The size of Ni-Co NPs was determined at 173 ± 2 nm zeta potential of the NPs in aqueous solution was −0.901 mV.
Cells Viability
Total cell viability is proportional to the number of viable cells, which are metabolically active cells that change the tetrazolium salt (MTT) to a purple formazan crystal. The HuH-7 and HCT-116 cells were treated with different concentrations (0, 2, 10, 15, 30 ,60, 100, 150 μg/mL) of Ni-Co NPs for 24 h. The MTT assay results showed that the cell viability was decreased in dose dependent manner. Data indicated the percentage viability of HuH 7 cells are100, 94, 83, 78, 69, 62, 24, and 14 %, and the viability of HCT-116 are 100, 69, 66, 61, 51, 15, 10, 8 %, respectively compared with the control (Figure 2A). Cells Viability of HuH-7 and HCT-116 Cells after Exposure to Ni-Co NPs in for 24 h, Based on the (A) MTT Assay (B) NRU Assay. Each Value Represents the Mean ± SE of Three Experiments. n = 3, *P < 0.05, **P < 0.01, Versus Untreated Control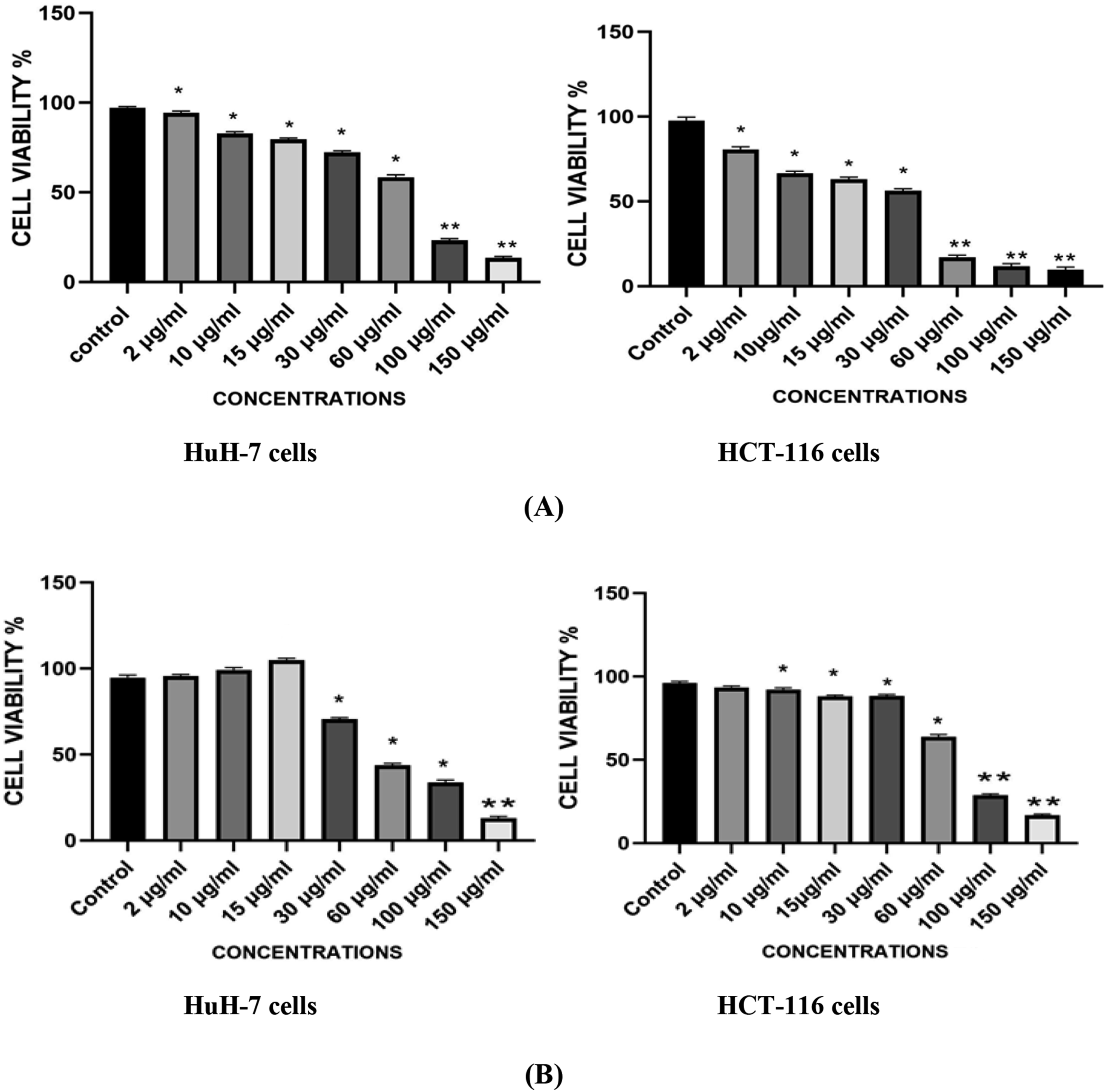
The neutral red intake assay was used to evaluate the viability of cells using the technique described by Alarifi et al 23 and the results demonstrated that exposure of the Ni-Co NPs reduced cell viability in a dose-dependent manner at 24 h as compared with control (Figure 2B).
IC50 Value of Ni-Co NPs
The IC50 value 24 h for Ni-Co NPs to HuH-7 and HCT-116 cells were evaluated by the dose-response curve graph using the program Origen Pro 8.5 (Figure 3) and it was determined on the basis of the data of MTT test. IC50 value 24 h for Ni-Co NPs to HuH-7 is 65.84 µg/ml (Figure 3A) and HCT-116 cells is 24.72 µg/ml (Figure 3B). (A) The Half Maximal Inhibitory Concentration (IC50) Value 24 h Calculated for Ni-Co NPs in Treated Cells of HuH-7 Cell Line Based on MTT Results. (B) The Half Maximal Inhibitory Concentration (IC50) Value 24 h Calculated for Ni-Co NPs in Treated Cells of HCT-116 Cell Line Based on MTT Results. n = 3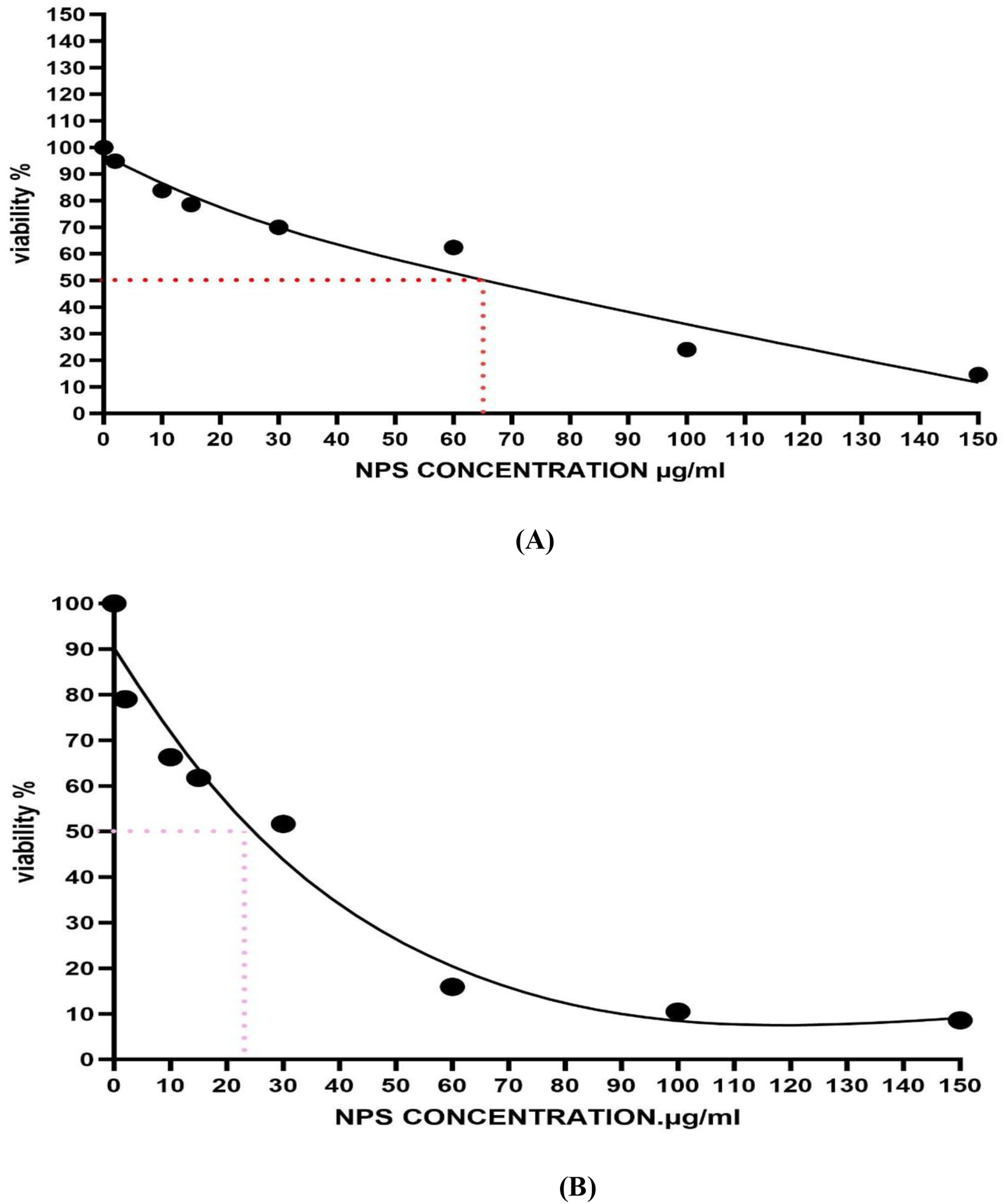
Reactive Oxygen Species
The obtained data showed a significant (P < 0.05) increase in ROS level after treatment with Ni-Co NPs compared to control. The higher the concentration of Ni-Co NPs, the higher the ROS generation. Figure 4A and B shows the production of ROS in HuH-7 cells as stained with the fluorescent dye DCFH-DA. Figure 4A and C shows the production of ROS in HCT-116 cells as stained with the fluorescent dye DCFH-DA. (A) Percentage Change in Intracellular ROS Generation in the in HuH-7 and HCT-116 Cells (B) Induction of ROS Levels in HuH-7 Cells (C) Induction of ROS Levels in HCT-116 Cells after Exposure With Ni-Co NPs for 24 h. Each Value Represents the Mean ± SE of Three Experiments. n = 3, (*P < 0.05) Versus Untreated Control, Scale bar = 200 µm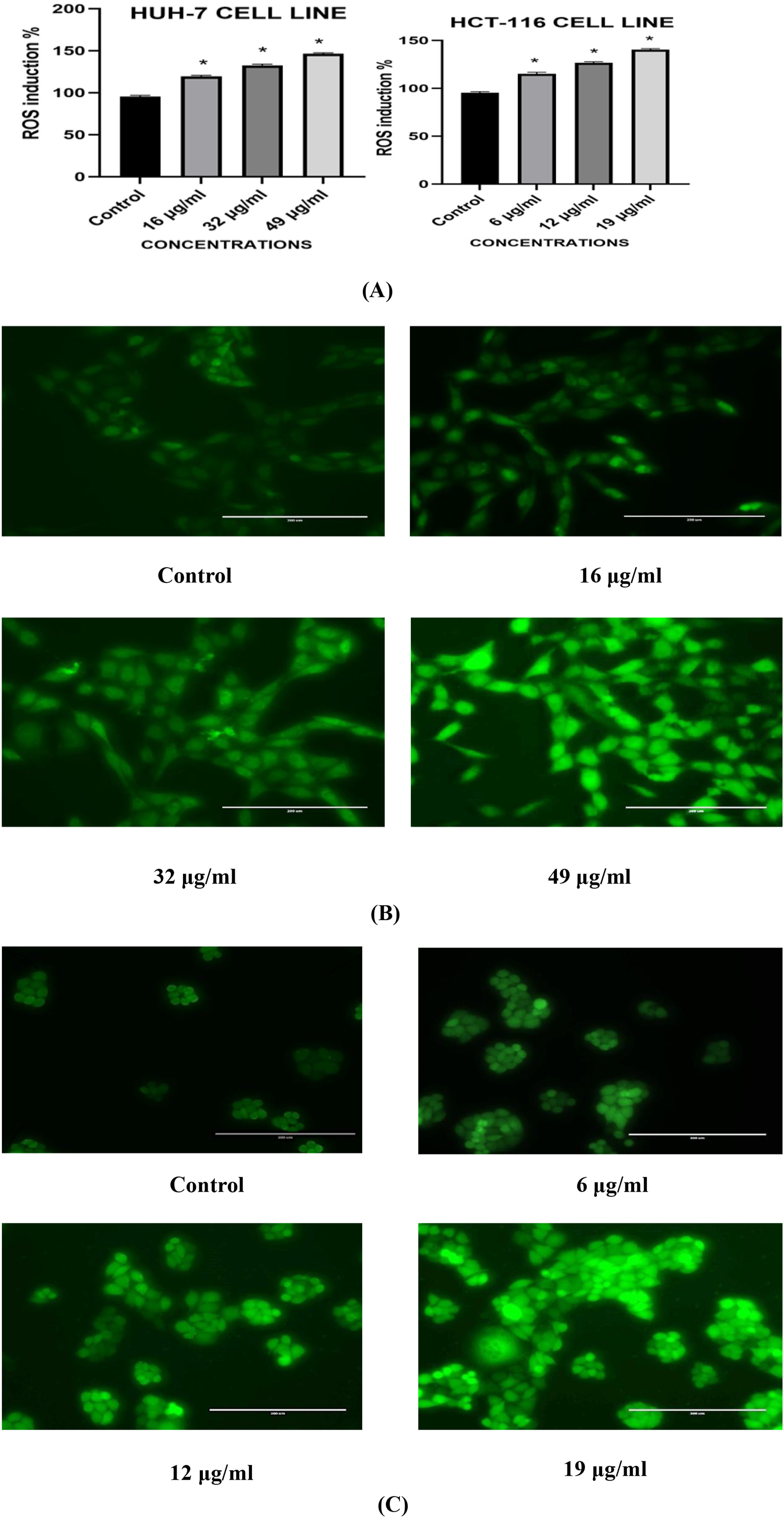
Oxidative Stress Assay
Lipid peroxidation, is a complex chemical process that leads to oxidative degradation of lipids, resulting in the formation of peroxide and hydroperoxide derivatives. It occurs when free radicals, specifically ROS, interact with lipids within cell membranes. This reaction leads to the formation of lipid radicals, referred to as lipid peroxides or lipid oxidation products, which in turn react with other oxidizing agents, leading to a chain reaction that results in oxidative stress and cell damage. The results showed that activity of lipid peroxidation increased in of HUH-7 at 16 µg/ml Ni-Co NPs and HCT-116 each in a dose dependent manner as shown in Figure 5A. (A) Effect of Ni-Co NPs on Lipid Peroxidation Activity in HUH-7 and HCT-116 Cell Line for 24 h and (B) Catalase Levels in HUH-7 and HCT-116 Cell Lines after Treatment With Ni-Co NPS for 24 h. Each Value Represents the Mean ± SE of Three Experiments. n = 3, *P < 0.05, Versus Control
Catalase is one of the important antioxidant enzymes that alleviate oxidative stress to a significant amount by degradation of hydrogen peroxide into water and oxygen. It is mainly located in peroxisomes. The results showed that activity of catalase after treatment with different concentrations of Ni-Co NPs in HUH-7 cell line were significantly increased compared to the control group, but in HCT-116 cell line the increasing was only in 6 μg/mL and decreased in concentration 12, 19 μg/mL compared to the control group (Figure 5A).
Assessment of MMP in HuH-7 and HCT-116 Cells
Mitochondrial membrane permeability (MMP) was measured using the accumulation of JC1 dye. Dye is a sensitive marker for mitochondrial membrane potential change. where, JC-1 gives red fluorescence, but in case of mitochondrial membrane potential change, the dye gives green fluorescence. The results showed that the treatment with Ni-Co NPs for 24 h led to a gradual loss of MMP as the concentration increased. However, the normal MMP was found in control cells as JC-1 dye penetrated into the cells and produced J aggregates with deep red fluorescence, while treated cells with Ni-Co NPS decreased their MMP and did not incorporate JC-1 stain (green fluorescence).
This was evident in both types of cells HUH-7 and HCT-116 (Figure 6). Results showed a significant increase in the number of apoptotic cells compared to the control untreated cells (Figure 6A–C). NPs treatment illustrated a higher number of apoptotic cells as compared to the control, as shown in Figure 6A–C. (A) Induction of MMP in HuH-7 and HCT-116 Cell Line When Treated With Different Treatments of Ni-Co NPs for 24 h. Fluorescence Microphotograph Showed the Effect of Different Concentrations of Ni-Co NPs on the Potential of Mitochondrial Membrane Permeability in (B) HuH-7 and (C) HCT-116 Cell Line after 24 h. n = 3, *P < 0.05 Versus Untreated Control
Caspase-3 Enzyme Activity
Apoptosis is a type of programmed cell death that is managed by the members of the caspase family of cysteine proteases. Procaspases are activated in response to the diverse cell death stimuli. The results showed that activity caspase 3/7 in HuH-7and HCT-116 cell lines were increased in cells as the dose of treatment increased compared to the control group and the Ni-Co NPs induced apoptosis by activating the caspases 3/7 activity (Figure 7). Caspase 3/7 Activity in HUH-7 and HCT-116 Cell Lines after Treatment With Ni-Co NPS for 24 h. Each Value Represents the Mean ± SE of Three Experiments. *P ≤ 0.05 1 Versus Control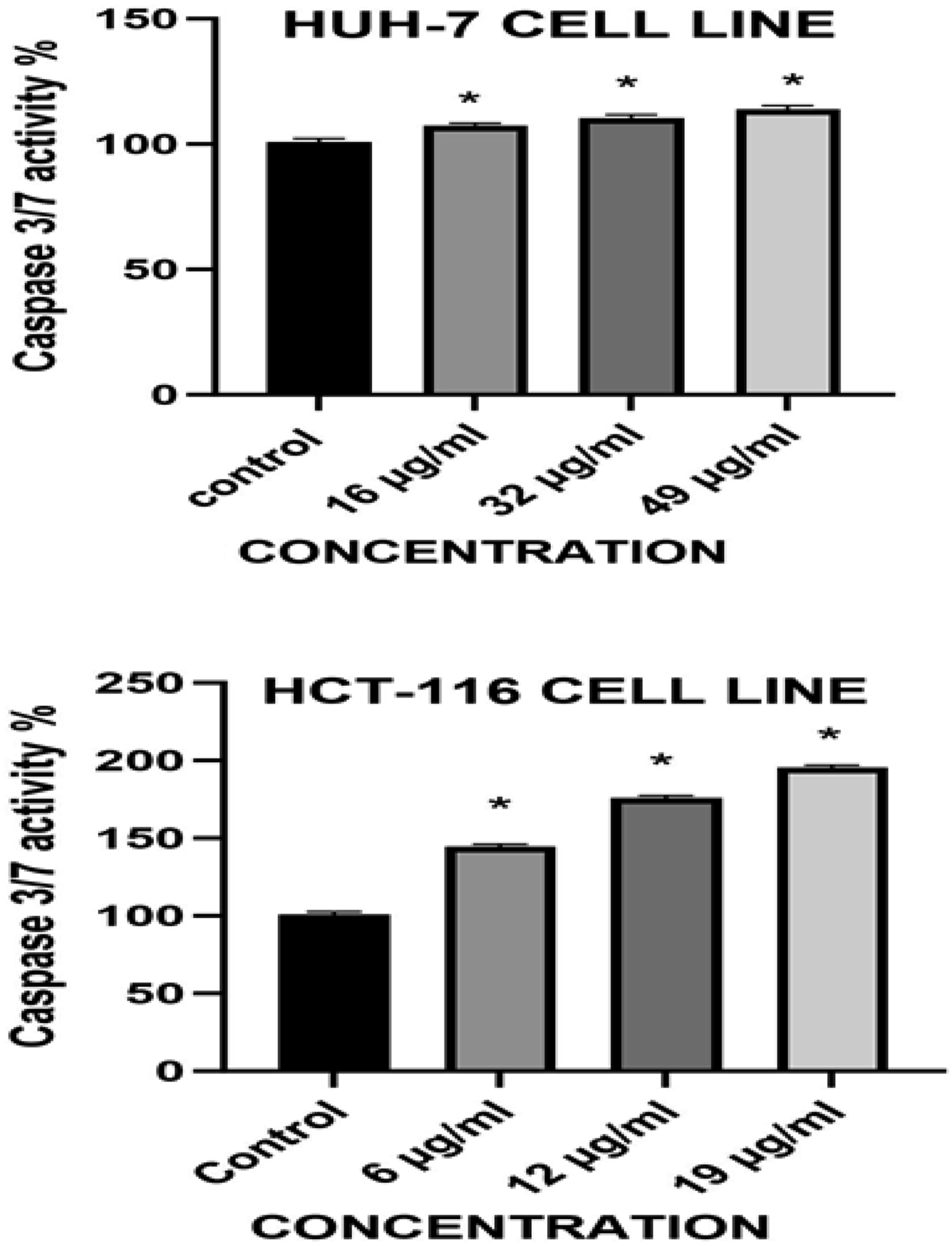
Gene Expression
In HUH-7 cells present data revealed downregulation bax in concentration 16, 32 μg/mL and increase in concentration 49 μg/mL compared with untreated cells (Figure 8A). However, HCT-116 treated cells for 24 h showed significantly upregulation in concentration 6, 19 μg/mL and downregulation in concentration 12 μg/mL compared to control (Figure 8A). Obtained data illustrated the expression of bcl-2 as an anti-apoptotic gene was significantly downregulated in concentration 12, 32 μg/mL HuH-7 cells and increased in concentration 49 μg/mL treated with Ni-Co NPs compared to the control (Figure 8B). Furthermore, we note HCT-116 cells was increase in concentration 6 μg/mL compared to the control group and decrease in concentration 12, 19 μg/mL compared to the control (Figure 8B). mRNA Levels in HUH-7 and HCT-116 Cells after Treatments of Ni-Co NPs for 24 h (A) Bax (B) Bcl2 (C) Caspase-3 (D) p53. n = 3, *P < 0.05, Versus Untreated Control
The expression of caspase-3 was assessed in the HuH7 cells after 24 h exposure. The expression of caspase-3 was significantly upregulated after treating at a concentration 49 μg/mL Ni-Co NPs (Figure 8C). In contrast, the expression of the caspase-3 gene was significantly downregulated after treating with 16, 32 μg/mL from Ni-Co NPs compared with the control (Figure 8C).
Furthermore, in HCT-116 cells, it was significantly increased after treating at a concentration 6, 19 μg/mL Ni-Co NPs and a decrease in level 12 μg/mL concentration compared with the control (Figure 8C). The expression of p53 gene was assessed in the HuH7 cells treated with Ni-Co NPs after 24 h. The level of p53 was significantly decreased after treatment with concentration 16.49 μg/mL and increased significantly with concentration 32 μg/mL according to control group (Figure 8D). In contrast the HCT-116 cells treated with Ni-Co NPs the level of p53 was significantly decreased after treatment with concentration 6 and12 μg/mL and increased significantly with concentration 19 μg/mL according to control group (Figure 8D).
Discussion
One of the most popular methods for producing nanoparticles is the green synthesis process. In comparison to other techniques, the process is a useful and efficient tool that is also economical, environmentally benign, and non-toxic. Magnetic nickel and cobalt nanoparticles are used as magnetic mirror, antibacterial activities and magnetic properties. 24 Plant and algae mediated synthesized nickel and cobalt nanoparticles from various types of plants such as Vitis vinifera, Nerium oleander and Capparis zeylanica. Algae-based biosystems can be used in wastewater treatment processes in different ways, including removal of pathogenic bacteria, reduction of biological and chemical oxygen demands, and removal of nitrogen, phosphorus, and other nutrients/pollutants. 25 The fate and behavior of NiCo NPs in the environment and biological systems challenge their application in cancer therapy and depend on functional features, including size, shape, surface charge, dispersity. It has been evidenced that very small size NPs are degraded in the liver and hepatocytes and excreted in feces and urine. Gold nanoparticles smaller than 20-30 nm are swiftly eliminated from circulation by the excretory system, such as kidneys; hence, they do not accumulate in the body tissue, whereas particles bigger than 200 nm are absorbed by the phagocytic system. 26 The concentration, aggregation, and circulation period of nanoparticles all affect the effectiveness of drug delivery systems. Humans are not aware of the risk of exposure to nanoparticles, which can enter human and animal body through many routes, which is a significant and consequence of the high application of nano-based substances. In instance, nanoparticles can morph in size over a range of mediums and offer a measurable increase in surface area in comparison to mass. One of the most often utilized models of cells in vitro is the human liver (HuH-7) and colon (HCT) cancer cells. 27 In the current study we examined the effects of Ni Co NPs on HuH-7 and HCT-116 cells through different the processes. Ni Co NPs has been selected for this investigation in order to find its impact on HuH-7 and HCT-116 cells growth inhibition, genotoxic reactions, oxidative stress, and apoptotic cell death. The size distribution of Ni Co NPs was observed by the results of dynamic light scattering analysis. The size of Ni Co NPs was determined by TEM and it is 38.27 ± 3 nm. On the other hand, HuH-7 and HCT-116 cells were exposed to Ni-Co NPs and cell viability was determined by MTT and NRU assays. The mitochondrial dehydrogenase enzyme, which is only active in live cells, reduces water-soluble tetrazolium salt into an insoluble formazan, which is the basis for the MTT assay. 20 The MTT data shows that there were several disruptions in the metabolic capacities of the cells, which led to the suppression of growth. After 24 h of exposure of Ni-Co NPs, it reduced a substantial cell viability of HuH-7 and HCT-116 cells and 50% inhibition of cell growth (IC50 value) were 65.84 µg/ml for HuH-7 and 24.72 µg/ml for HCT-116 cells. The most widely recognized and developed mechanism for the potential toxicity of NPs is most likely the nanomaterial-induced oxidative stress. Curiously, no comprehensive investigation integrating of cell growth, cytotoxicity, internalization of NPs, production of ROS, and cell cycle analysis of various cell lines indicative of putative retention organs has been documented for Ni-Co NPs to yet.
These outcomes suggest that oxidative stress is probably the specific mechanism of Ni-Co toxicity, despite the fact that it has been implicated as the toxic mechanism associated with other monometallic NPs such as Co, Ni, and Mn. 28 This is reportedly linked to the cytotoxic effects of NPs on normal and cancer cells. 29 In this study cytotoxicity of Ni-Co NPs against HuH-7 and HCT-116 cells were examined and different results were found. On the basis of these finding first time it is reported that cytotoxicity of Ni-Co NPs depends upon the type of cell lines. who also demonstrated cell type differences. Our findings, which are consistent with a number of other studies, showed that the combination of Ni and Co creates a specific dangerous entity that generates more ROS. 30 Oxidative stress caused by an excess of ROS is often closely linked to oxidative damage to proteins and DNA, which is why nanomaterials are often genotoxic. According to our findings, the combination of Ni and Co NPs enhances their ability to production of ROS. Comparing the ROS level to the Ni Co NPs, exposed both cells the results demonstrated a considerable induction in ROS due to the cytotoxicity. Sargassum tenerrimum is a brown seaweed with strong potential to be used as a functional food ingredient, mainly due to its antioxidant properties. In the human cell culture models, extract of Sargassum muticum has been shown by Pinteus et al. 31 to display cytoprotective properties. The effects of Ni-Co NPs, Sargassum muticum on HuH-7 and HCT-116 cell lines generated a significant (P < 0.05) quantitative dose-dependent increase in ROS production, which was further supported by the lipid peroxide and catalase activities. Current research indicates that Ni-Co NPs reduced the viability of HuH-7 and HCT-116 cell lines, raised ROS and LPO levels, and elevated catalase levels. ROS produced as a result of physical damage to the mitochondrial membrane is one cause of mitochondrial damage. Increased ROS generation or a compromised antioxidant defense in response to ongoing nanomaterial exposure can both cause the disrupted oxidant balance. Our findings are in line with earlier findings that As2O3 dosages that induce apoptosis in BEAS-2B cells result in noticeably higher levels of gene expression in apoptotic cells. 32 By upregulating the expression of bax, caspase-3, and p53 and downregulating that of bcl-2, Ni-Co NPs clearly caused apoptosis in HuH-7 and HCT-116 cells. The anti-cytotoxic properties of Sargassum tenerrimum may mitigate the adverse effects of Ni-Co NPs. We discovered that high-concentration Ni-Co NPs induced a greater degree of apoptosis in cells that were just exposed as opposed to the control. This suggests that the degree of apoptosis induction was significantly increased by Ni-Co NPs.
Conclusion
For the first time, Ni Co NPs have been made using the green synthesis method with an aqueous extract of Sargassum tenerrimum. Two cell types, HuH-7 and HCT-116, were used in a comparison study using green-produced Ni Co NPs. Investigations using SEM, EDX, TEM, XRD, and DLS confirmed the size, shape, and composition of Ni Co NPs in addition to their production. Following a notable apoptosis, the green-produced Ni Co NPs showed cytotoxic effect when applied to HuH-7 and HCT-116 cells. The green techniques that are currently available are simple to apply, quick, non-toxic, safe for the environment, and beneficial for medical uses.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Ongoing Research Funding Program (ORF-2025–27), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This article contains all the data generated or analyzed during this study. Further enquiries can be directed to the corresponding author.
