Abstract
Background
Diabetic nephropathy (DN) is a common microvascular complication of diabetes and its currently the leading cause of end-stage renal disease worldwide.
Reducing its incidence and delaying its progression are urgent medical priorities around the world.
Objectives
This study set out to evaluate the effects of using licoricidin (LCD) to treat diabetic nephropathy (DN) in male Wistar rats with diabetes that had been induced by streptozotocin (STZ).
Materials and Methods
Four sets of 24 male Wistar rats were created. The control group 1 animals in the first group were fed the standard diet. Following a single intravenous injection of STZ to induce diabetes, the remaining 18 rats were split evenly into three groups: group 2 was assigned to the diabetic control group; group 3 was given an oral dose of 20 mg/kg of LCD, while the fourth group received oral glibenclamide (GLB) at a dose of 25 mg/kg. Upon conclusion of the treatments, the body weight, blood glucose, insulin level, and kidney function markers in the experimental rats were assessed. The concentrations of inflammatory cytokines and oxidative stress-related marker levels were assessed. The kidneys of the experimental rats underwent histological examination.
Results
The administration of LCD significantly increased body weight and insulin levels and subsequently decreased glucose and renal dysfunction indicators in rats with STZ-induced DN. The LCD significantly decreased inflammatory response and oxidative stress by enhancing antioxidant levels in rats with DN. The histological examination results corroborated the therapeutic efficacy of LCD in treating DN.
Conclusion
LCD treatment improves renal function by reducing inflammation and stress in animal models caused by STZ, according to the study’s findings.
Introduction
A condition known as diabetes is characterized by hyperglycemia while fasting or after meals. Diabetes mellitus (DM), chronic hyperglycemia, is linked to end-organ failure, damage, and malfunction in the kidney, retina, nerves, heart, and blood arteries, among other organs and tissues (Alam et al., 2014). Insulin-dependent (type I) and non-insulin-dependent (type II) are the two categories of DM. An autoimmune disease, type I diabetes, is characterized by an inflammatory response that occurs near the islets, followed by the specific cell death that produces insulin. This contrasts with type II diabetes, which is characterized by resistance to insulin secretion and decreased production of insulin (Deshmukh et al., 2015).
Diabetic nephropathy (DN) is one of the most common microvascular complications of DM and remains a leading cause of chronic kidney disease (CKD) and end-stage renal disease (ESRD) globally. According to a 2024 global analysis, approximately 30%–40% of individuals with diabetes develop some form of nephropathy, with increasing incidence particularly in low- and middle-income countries (Saeed et al., 2024). The global burden is exacerbated by poor glycemic control, hypertension, and delayed diagnosis, making DN a major contributor to morbidity and healthcare costs. A recent meta-analysis published in Frontiers in Nephrology (2025) estimated the pooled prevalence of DN at 28.2% among adults with diabetes worldwide, with significant regional variation (Zhou et al., 2025). In India, where the diabetic population exceeds 77 million, DN prevalence ranges from 25% to 35%, depending on urban versus rural settings and the duration of diabetes (Chatterjee et al., 2024). These findings highlight the urgency of early diagnosis, preventive strategies, and novel therapeutic interventions for DN (Sagoo & Gnudi, 2019).
The oxidative stress caused by hyperglycemia is connected to several diabetic complications, including DN. The association between oxidative stress and the inflammatory reaction in the development of DN has been extensively studied. Long-term hyperglycemia causes renal tissues to produce a large amount of reactive oxygen species (ROS) and oxidative stress (Qi et al., 2020). The pathophysiology of DM and, therefore, DN involves both immune system activation and persistent inflammation. According to some research, immune cells, including monocytes, lymphocytes, and macrophages, as well as cytokines, growth factors, chemokines, adhesion molecules, and nuclear factors, are all implicated in the pathophysiology of DM and, of course, have a significant impact on its consequences (Duran-Salgado & Rubio-Guerra, 2014).
As rapid screening technology advances quickly, natural compounds with a broad range of bioactive effects have gained public interest as potential novel medications (Hu et al., 2021). It is known that several phytochemicals produced from plants have antioxidant and anti-hyperglycemic properties (Bhutkar & Bhishe, 2011). Additionally, diabetes and its consequences are commonly treated with traditional herbal treatments. Therefore, the herbal medications that have been used traditionally may help prevent or cure kidney impairment brought on by diabetes (Ankita et al., 2015). An isoflavonoid called licoricidin (LCD) is isolated from Glycyrrhiza glabra roots. Numerous biological actions of LCD, such as anti-cancer properties and a potent anti-bacterial impact against Helicobacter pylori, have been extensively demonstrated (Ma et al., 2022). A key ingredient in liquorice extract is LCD, an isoflavone with two isoprenyl groups. Significant hepatoprotective, anti-viral, anti-carcinogenic, anti-inflammatory, anti-allergy, and estrogenic properties are exhibited by liquorice root. Like liquorice, LCD exhibits strong anti-cancer, anti-inflammatory, and anti-bacterial properties (Shan et al., 2019). The main objective of the current study is to assess the efficacy of LCD against diabetes in Wistar rats induced by STZ by looking at the biochemical and histological indicators. In both untreated and treated rats, the effects of LCD were investigated on the levels of body weight, FBG, insulin, antioxidants, proinflammatory indicators, and kidney functional markers.
Materials and Methods
Materials
Before the start of the study, all of the chemicals, reagents, kits, and equipment were purchased commercially.
Animal Housing
Following clearance by the Institutional Animal Ethical Committee, male Wistar rats aged 6–8 weeks, with a body weight of 220 ± 20 g, were obtained from the animal facility. Throughout the study duration, all rats were given free access to a standard diet and purified drinking water. The rats were acclimated in a carefully monitored lab setting by maintaining the temperature at 25°C, sustaining 55% relative humidity for a week, and making sure there was a 12-h light and dark cycle. The rats were given regular rat chow and constant access to fresh water after they had acclimated. The ethics committee approved all experimental procedures used in this work, and the rats were treated with the highest care and attention.
Experimental Animals and Methodology
In this study, male Wistar rats (n = 24) were randomly assigned to four groups (n = 6 per group). All procedures involving animals were approved by the Institutional Animal Ethics Committee and conducted following the guidelines for the care and use of laboratory animals.
Diabetes was induced in all animals except the control group using streptozotocin (STZ) at a dose of 55 mg/kg body weight, freshly prepared in 0.05 M sodium citrate buffer (pH 4.5), and administered intraperitoneally. Rats with fasting blood glucose (FBG) levels exceeding 200 mg/dL were considered diabetic and included in the study.
Animals were divided into four experimental groups:
Group 1 (control): Received vehicle only and standard laboratory diet. Group 2 (diabetic control): STZ-induced diabetic rats without treatment. Group 3 (LCD-treated): Diabetic rats treated orally with LCD (20 mg/kg body weight) dissolved in 1% DMSO, administered once daily. Group 4 (GLB-treated): Diabetic rats treated with glibenclamide (GLB) at 25 mg/kg, administered twice weekly.
The selection of the 20 mg/kg dose of LCD was based on previous studies that demonstrated its efficacy in improving glycemic parameters and organ protection in diabetic rat models without inducing toxicity (Alrefaei & Elbeeh, 2025). Pharmacokinetic studies of LCD have shown that it possesses good oral bioavailability and reaches peak plasma concentrations within 1–2 h post-administration, with a half-life of approximately 4–6 h, making it suitable for once-daily dosing (Alrefaei & Elbeeh, 2025). To minimize bias, animals were randomly assigned to treatment groups using a computer-generated randomization schedule. All personnel involved in data collection and histological analysis were blinded to the treatment groups. At the end of the treatment period, animals were euthanized under anesthesia, and kidney tissues were collected for histological evaluation.
Tissue Preparation
At the end of the experimental period, intracardiac blood samples were obtained under ketamine/xylazine anesthesia, and the rats were killed by cervical dislocation. Kidney tissues were immediately taken out, rinsed with cold normal saline (0.9%), and stored in a freezer at −80°C. The samples of kidney tissue were thawed and suspended in cold phosphate buffer. Parameters such as kidney function markers and oxidative stress marker levels were all assessed using the supernatants.
Estimation of Body Weight
Measurements of the rats’ body weights were made in each of the four groups to examine for variations in their total weight, and the findings were interpreted appropriately.
Estimation of FBG and Insulin
All experimental rats had their FBG measured following an overnight fast. A common glucometer was used to test FBG after blood samples were drawn from the tail vein. After the experimental design, the insulin level was measured.
Estimation of Kidney Function Marker
The amounts of urea and creatinine in the serum were estimated using an enzymatic colorimetric kit using the procedures outlined by Bartels et al. (1972) and Fawcett and Scott (1960).
Estimation of Oxidative Stress Marker
After homogenizing the kidney tissue in cold PBS (pH 7.4), it was centrifuged for 15 min at 2,000 rpm. Enzyme-linked immunosorbent assay (ELISA) kits (SenBejia Biotech, Nanjing, Jiangsu, China) were used to measure the levels of advanced oxidation protein products (AOPP) and malondialdehyde (MDA) in the supernatant.
Estimation of antioxidant Enzymes
The kidney tissue was centrifuged for 15 min at 2,000 r/min after being homogenized in cold PBS (pH 7.4). Catalase (CAT), glutathione peroxidase (GPx), and superoxide dismutase (SOD) were estimated in the supernatant using ELISA kits.
Determination of Inflammatory Cytokine Levels
The levels of interleukin-6 (IL-6), tumor necrosis factor (TNF)-α, and interleukin-1β (IL-1β) in kidney homogenate were assessed using commercially available ELISA kits.
Histopathological Examination
Following animal sacrifice, kidney tissues were preserved in saline, processed in 10% formalin, treated as usual, and then coated with paraffin. For microscopic analysis, slices that were 5 µm thick were produced and stained with hematoxylin and eosin (H&E) dye. Using an Olympus light microscope with a digital camera, the stained sections were inspected and captured on video.
Statistical Analysis
All statistical analyses were performed using GraphPad Prism version 10.0 (GraphPad Software, San Diego, CA, USA). Data are presented as mean ± standard deviation (SD). Before conducting a one-way analysis of variance (ANOVA), data were tested for normality using the Shapiro–Wilk test to confirm the assumption of a Gaussian distribution. Homogeneity of variances was also verified using Levene’s test. Following ANOVA, Dunnett’s post hoc test was applied to compare each treatment group with the diabetic control group. A p value of less than .05 was considered statistically significant, with stronger significance accepted at p < .01.
Results
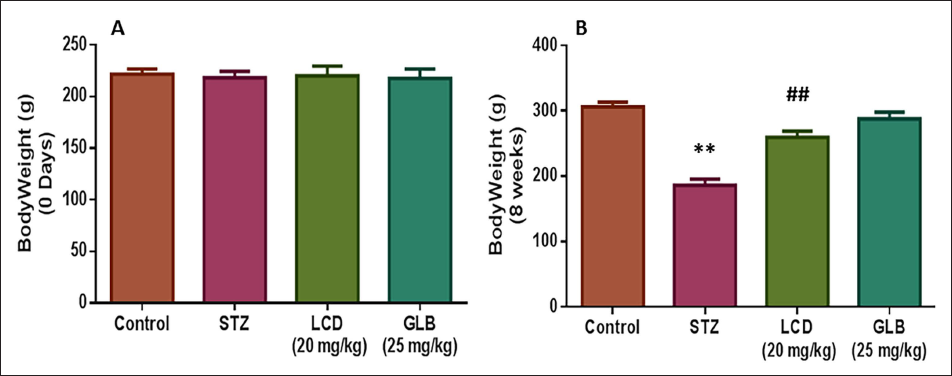
Effect of LCD on Body Weight in Diabetic Rats
Figure 1 depicts the impact of ingesting LCD on rats’ body weight was investigated. Compared to rats without diabetes, diabetic control rats had a drop in body weight. The rats in the diabetic control plus GLB, normal control, and diabetic control plus LCD groups did not significantly lose body weight at the end of continuous therapy.
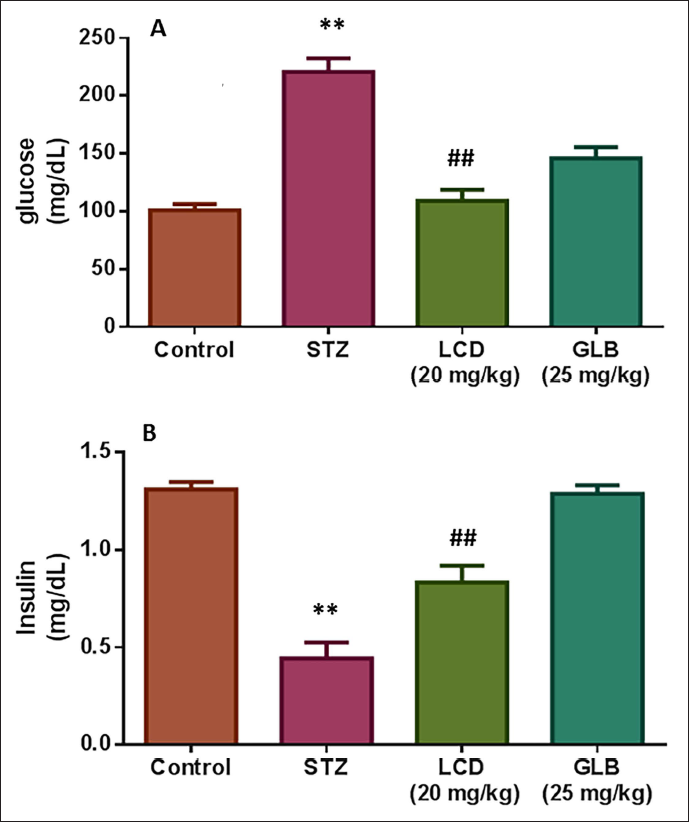
Effect of LCD on FBS and Insulin Level
Figure 2 shows the effect of LCD on FBS and insulin levels. When compared to normal control rats, the diabetic control rats showed low insulin levels and elevated FBG levels. Additionally, the LCD-treated diabetic control rats had elevated insulin and reduced FBG levels. Based on these results, however, it was shown that LCD is essential for preventing kidney pathogenesis by controlling insulin and glucose levels.
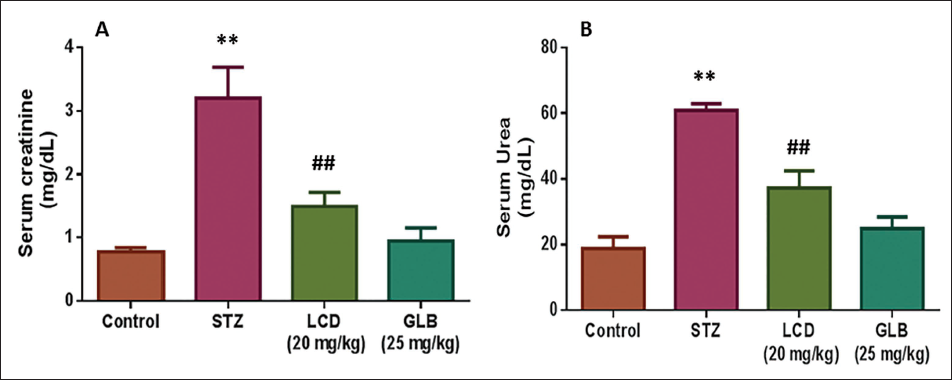
Effect of LCD on Kidney Function Markers
The experimental animals in each group had their blood levels of urea and creatinine tested as depicted in Figure 3. Serum urea and creatinine levels significantly increased after STZ treatment in comparison to normal control rats (p < .05). Nonetheless, the diabetic rats provided with LCD exhibited a considerable drop in their urea and creatinine levels.
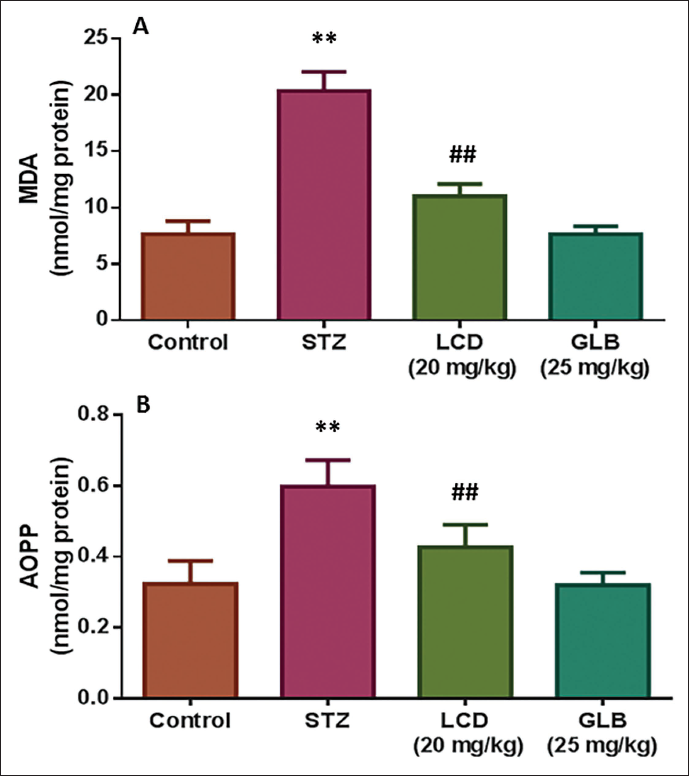
Effect of LCD on Oxidative Stress Markers
The effect of LCD on oxidative stress markers is seen in Figure 4. The level of MDA and AOPP was significantly up-regulated in the STZ-administered animals in comparison to normal control rats. The animals treated with LCD showed a considerable decrease in the oxidative stress markers, indicating the antioxidant property of LCD.
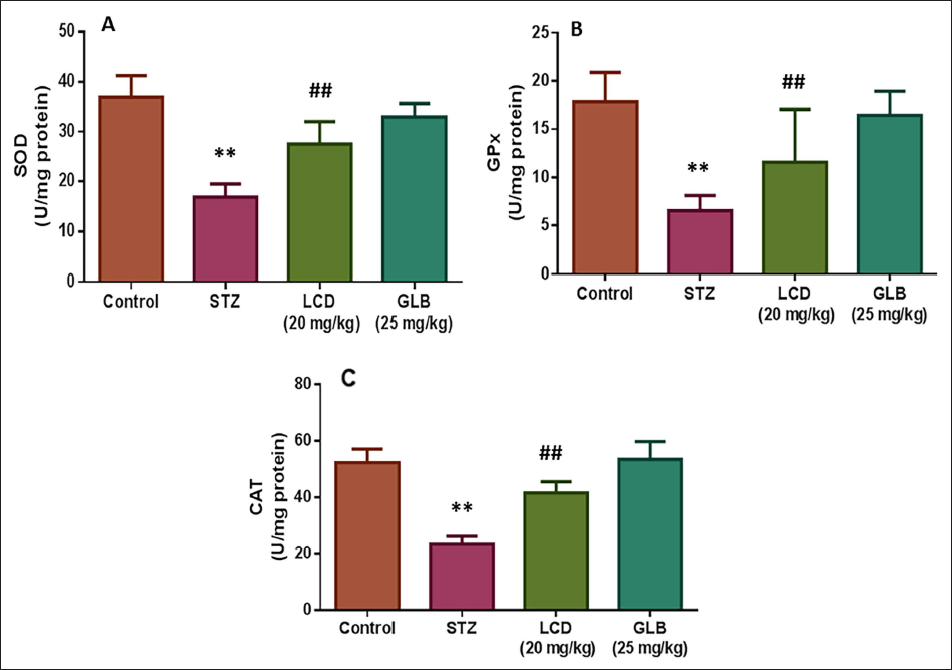
Effect of LCD on Antioxidant Enzymes
LCD antioxidant capability was evaluated by determining the amounts of the antioxidant enzymes CAT, GPx, and SOD (Figure 5). The findings demonstrated that the CAT, GPx, and SOD enzyme levels were lower in diabetic rats. LCD, however, had a notable protective impact on kidney function in diabetic rats. Significantly elevated amounts of the enzymes CAT, SOD, and GPx were found in the data.
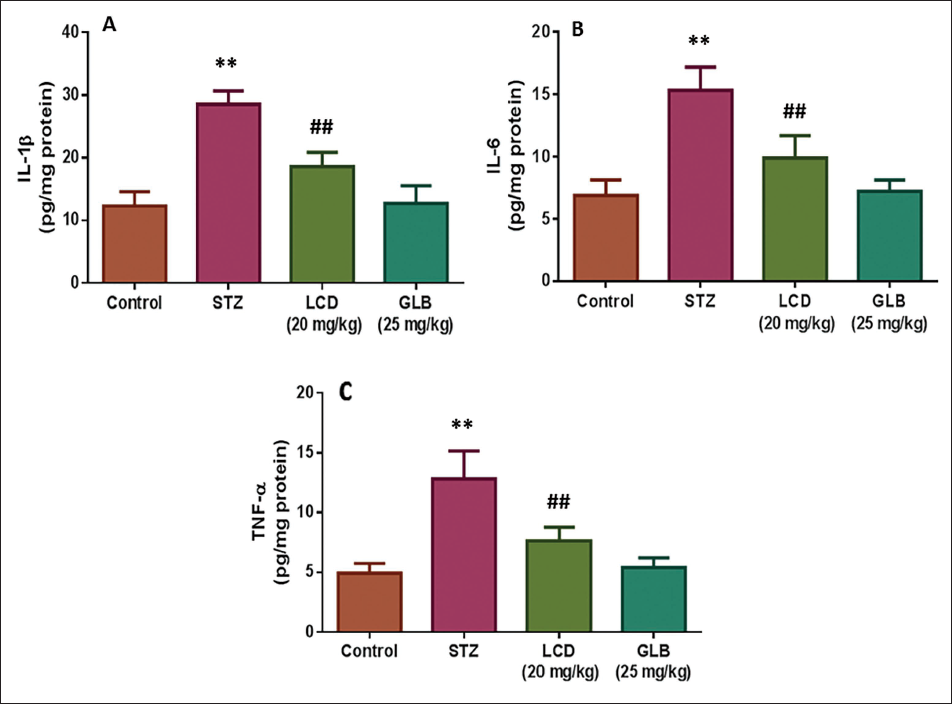
Effect of LCD on Inflammatory Cytokines
The kidney tissues of both control and treated rats were examined for levels of IL-1β, TNF-α, and IL-6. The results are displayed in Figure 6. In comparison to the control rats, the diabetic control rats had considerably higher amounts of IL-1β, TNF-α, and IL-6 in their renal tissues. LCD-administered diabetic rats demonstrated a considerable reduction in inflammatory cytokines.
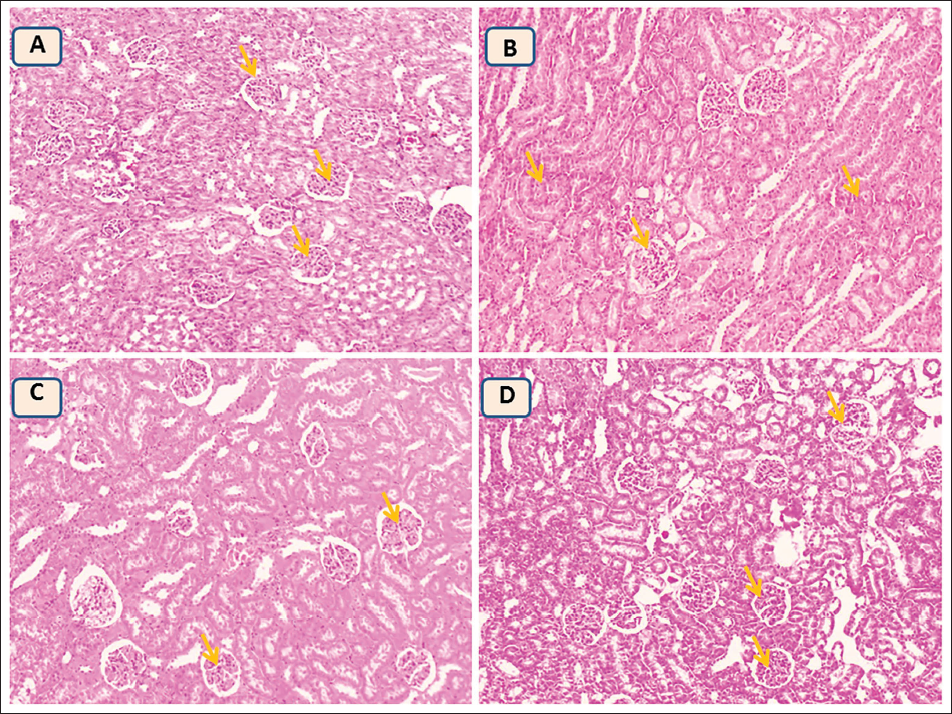
Effect of LCD on Histopathological Architecture
H&E staining were used to analyze renal morphology to assess the impact of LCD on renal structural alterations in all experimental animals (Figure 7). Healthy kidney architecture, featuring a normal proximal and distal tubule with normal epithelium and glomerulus, was revealed by histological analysis of normal control groups (group 1). Kidney damage was evident in the histological sections of the kidneys of diabetic rats, which displayed altered congestion, glomerular architecture, and lymphocyte infiltration (group 2). However, compared to the diabetes control group, the pathological alterations of the renal tissues in the diabetic groups who received LCD revealed less damage since there was reduced congestion and a smaller amount of inflammatory cells (groups 3 and 4).
Discussion
The decline in renal function observed in individuals with type 1 and type 2 DM over an extended period is known as DN or diabetic kidney disease. Glycemic and blood pressure regulation are connected to the disease’s development, which is believed to proceed in phases. In the initial 10–20 years following the initiation of diabetes, the typical prevalence of DN is significant (3% annually) (Sulaiman, 2019). The onset and development of DN include several directions and mediators, which include oxidative stress and inflammatory processes, all of which are thought to be substantial in recent years. Knowing the salient characteristics of the inflammatory progressions that contribute to the onset and progression of DN also makes it easier to find new possible targets and create creative anti-inflammatory treatment plans. The increased ROS caused by higher blood sugar levels is a major contributor to the pathophysiology of DN. It is believed that hyperglycemia-induced oxidative stress causes proinflammatory protein levels to rise through invasive macrophages, which then release inflammatory cytokines that cause inflammation on both a local and systemic basis (Samsu, 2021).
A significant source of nutritious foods is traditional herbs and spices. Many of them have demonstrated significant impacts on biological activity and disease processes, and they include antioxidants. Moreover, it has been shown that a variety of antioxidants obtained from food and medicinal plants enhance the body’s capacity for detoxification and antioxidant defense by activating redox-sensitive transcription factors (Al-Waili et al., 2017). The current study focused on examining the effects of a naturally occurring chemical called LCD on the development of diabetes-related issues in rats that were stimulated with STZ. LCD’s anti-diabetic effects were compared with those of GLB, a popular drug.
When animals are given STZ, they develop diabetes, which is associated with tissue protein loss, increased muscle atrophy from hyperglycemia, and excessive weight loss from hypoinsulinemia (Mestry et al., 2017). Following LCD administration, the STZ-diabetic rats’ body weight showed a substantial rise, suggesting that the hyperglycemia-induced muscle tissue deterioration had been prevented. STZ, an anti-biotic that kills pancreatic islet β-cells, is frequently employed in studies to simulate DM, as a reduction in β-cell activity, which ultimately results in insulin insufficiency and hyperglycemia (Furman, 2021). In this study, rats in the diabetic control group received 55 mg/kg of STZ to cause diabetes; as a result, their blood glucose levels were noticeably higher than those of the control group. The rats that were provoked with STZ showed a substantial diminution in their fasting sugar level after taking LCD medicine. This observation demonstrates LCD anti-diabetic efficacy.
Serum urea and creatinine, two biomarkers of DN, are known to increase with hyperglycemia in uncontrolled diabetics and often correspond with the degree of kidney damage (Bamanikar et al., 2016). The liver produces urea as a waste product during the metabolism of proteins. After being released into the bloodstream, urea is filtered by healthy kidneys and eliminated as urine. Reduced renal function is indicated by a higher blood urea level. Phosphocreatine in muscle is broken down non-enzymatically to produce creatinine. On an everyday basis, the body converts around 2% of its creatinine to creatinine. A significantly elevated blood creatinine level is linked to inadequate renal elimination of creatinine, which has led to the discovery that creatinine is a reasonably accurate measure of the condition of the kidneys (Campion et al., 2017). In the current investigation, STZ-administered animals were observed to have high levels of serum creatinine and urea, indicating kidney damage. Animals provoked with STZ when treated with LCD exhibited a decline in the level of serum creatinine and urea. The results were comparable to the positive group animals (group 4).
A disparity between antioxidants and oxidants is known as oxidative stress. It depends on how oxidant radicals are produced and built up in cells and tissues, as well as how well a biological system is capable of eliminating these reactive substances. In addition to being a major contributor to the onset of type 1 and type 2 diabetes, oxidative stress also plays a major part in the onset of diabetic complications, such as DN. Alterations in renal hemodynamics and metabolism are associated with oxidative stress. Lipid peroxidation is the process by which free radicals oxidize polyunsaturated fatty acids (linoleic acid, arachidonic acid, and so on), which can seriously harm tissue. Lipid peroxidation and the formation of prostaglandin and thromboxane produce MDA. Higher MDA levels indicate renal damage (Vodošek Hojs et al., 2020). One potential indicator of oxidant-mediated protein degradation is AOPP. Patients with DM, cardiovascular disorders, hypertension, and atherosclerosis have been found to have elevated levels of AOPPs. The capacity of AOPPs to produce proinflammatory cytokines and sticky molecules indicates that they have biological functions and are chemically similar to advanced glycation end-products (AGEs) (Conti et al., 2019). Similarly, in our present study, the level of MDA and AOPP was elevated in STZ-induced animals, indicating the DN condition. On treatment with LCD, the level of MDA and AOPP drastically declined and was comparable to the positive control group.
There is mounting data that people with diabetes have a reduced antioxidant capacity. Studies on animals and humans have demonstrated that antioxidant therapy works in lowering DN. Both type 1 and type 2 diabetic groups, with and without nephropathy, showed significantly reduced amounts of CAT, GPx, and SOD, as well as total antioxidant capacity (TAC) when compared to normal healthy individuals (Ranjbar et al., 2016). Certain enzymes, including SOD, GPx, and CAT, are generated within the tissues during different metabolic processes and are part of the defense system. These enzymes can safely stop free radicals in their tracks. GPx, one of the main enzymatic antioxidants, reduces the growing state of oxidative stress and transforms peroxides from harmful to non-toxic substances. This helps to prevent problems from diabetes (Altuhafi et al., 2021). According to the current study, taking LCD elevated the levels of antioxidant enzymes such as CAT, SOD, and GPx. This illustrates LCD’s antioxidant properties. This suggests that LCD may be an effective treatment for DN.
IL-1β, TNF-α, and IL-6 are examples of proinflammatory cytokines that play a crucial role in the onset and development of DN. DN animal models have considerably higher renal levels of IL-1β, TNF-α, and IL-6 in comparison to healthy animals (Hu et al., 2022). Among the cytokines that are crucial to the pathophysiology of DN are IL-6 and IL-1β, which have an impact on invading cells and renal residents (Araújo et al., 2020). Through a number of pathways, such as reduced GFR, vasoconstriction carried on by elevated endothelin-1 (ET-1) production, poor glomerular filtration barrier, and proteinuria, TNF-α aids in the onset of DN. By activating NADPH, elevated TNF-α production can also cause oxidative stress in mesangial cells. TNF-α is also believed to have direct cytotoxic effects on glomerular cells and to be involved in apoptosis (Samsu, 2021). Following LCD administration, diabetic rats’ levels of these inflammatory mediators dramatically dropped, suggesting the medication may have anti-inflammatory effects.
To assess the systemic safety of LCD treatment, potential organ toxicity was evaluated by monitoring general health status, behavior, and body weight throughout the study. No mortality or abnormal clinical signs were observed in the treated groups. In addition, histopathological analysis of the liver revealed normal hepatic architecture, with no signs of necrosis, inflammation, or fatty changes in LCD-treated rats. Liver function biomarkers—including alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP)—remained within the normal physiological range, indicating no hepatocellular injury. Hematological parameters such as hemoglobin (Hb), red blood cell (RBC) count, total leukocyte count (TLC), and platelet count also showed no significant deviations from control values, suggesting that LCD did not induce hematological toxicity. These findings are consistent with previous studies on structurally similar phytochemicals or bioactive compounds used in diabetic models, which demonstrated both efficacy and a favorable safety profile at comparable doses (Sánchez et al., 2019; Zhao et al., 2021). Comprehensive toxicological profiling, including liver and blood parameters, is crucial for evaluating the therapeutic potential of any novel compound in chronic disease models (OECD, 2008).
However, the present study has certain limitations. First, it was conducted over a relatively short treatment period in an animal model, which may not fully capture the chronic progression of human DN. Second, mechanistic pathways such as inflammatory signaling, mitochondrial dysfunction, or gene expression changes were not investigated. Third, although no overt toxicity was observed, a more comprehensive long-term toxicity and pharmacokinetic profile is warranted before clinical translation. Future studies should focus on clarifying the molecular mechanisms underlying LCD’s renoprotective effects and evaluating its efficacy in other models of CKD or combination therapies.
Our histological results in STZ-treated rats revealed glomerular shrinking and tissue death, edema, neutrophil penetration, and damage to renal tubular epithelial cells. These results aligned with those from previous studies and the biochemical results. Kidney damage in the diabetic animals, which displayed congestion, glomerular architecture, and lymphocyte infiltration, was all shown to decrease with LCD administration.
Conclusion
The study found that the administration of LCD significantly reduced oxidative stress and inflammation in STZ-induced diabetic rats, and fixed all the detrimental histological and biochemical changes caused by diabetes. LCD’s potent antioxidant qualities allowed it to act safely and efficiently as an anti-diabetic agent. The study also demonstrated that LCD treatment led to a reduction in oxidative stress markers and preserved renal architecture, suggesting its potential as a therapeutic candidate for preventing or delaying the progression of DN. The study’s novelty lies in its identification as a bioactive compound capable of modulating both glycemic and oxidative pathways, with efficacy comparable to standard anti-diabetic drugs like GLB. These findings contribute to the growing body of evidence supporting plant-derived or synthetic bioactives as alternative or adjunct treatments for diabetic complications.
Footnotes
Abbreviations
AOPP: Advanced oxidation protein products; BW: Body weight; CAT: Catalase; DM: Diabetes mellitus; DN: Diabetic nephropathy; ELISA: Enzyme-linked immunosorbent assay; FBG: Fasting blood glucose; GLB: Glibenclamide; GPx: Glutathione peroxidase; GR: Glutathione reductase; GSSG: Oxidized glutathione; H&E: Hematoxylin and eosin; IL-6: Interleukin-6; IL-1β: Interleukin-1β; LCD: Licoricidin; MDA: Malondialdehyde; ROS: Reactive oxygen species; SOD: Superoxide dismutase; STZ: Streptozotocin; TNF-α: Tumor necrosis factor.
Data Availability
Data will be made available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study is approved by the Ethics Committee of The First Affiliated Hospital of Kunming Medical University (Approval Number: KMMU20221505).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Yunnan Province Clinical Research Center for CKD 202102AA100060; Yunnan Fundamental Research Kunming Medical University Projects (Grant No. 202301AY070001-161); The National Natural Science Fund of China (Grant No. 82160165); The “famous doctor” special program of Yunnan Xingdian Talent support plan in 2022 (Grant NO. RLMY20220009); The young and middle-aged academic and technical leaders reserve talented person project in Yunnan province (Grant NO. 202105AC160093); Yunnan Clinical Medical Center for Endocrine and Metabolic Diseases (Grant NO. 2024YNLCYXZX0073).
Informed Consent
The participant has consented to the submission of the article to the journal.
