Abstract
The reactive oxygen species take role in pathogenesis of many diseases including hypoxia, hypercholesterolemia, atherosclerosis, nephropathy, hypertension, ischemia–reperfusion damage, and heart defects. The aim of this study was to evaluate whether crocin administration could protect kidney injury from oxidative stress in streptozotocin-induced diabetic rats. The rats were randomly divided into 3 groups each containing 10 animals as follows: group 1, control group; group 2, diabetes mellitus (DM) group; and group 3, DM + crocin group. At the end of the study, trunk blood was collected to determine the plasma levels of blood urea nitrogen (BUN) and creatinine (Cr). The kidney tissue was removed, and biochemical and histological changes were examined. Diabetes caused a significant increase in malondialdehyde (MDA) and xanthine oxidase (XO) activities and a decrease in glutathione (GSH) contents ( p < 0.01) when compared with control group in the rat kidneys. Crocin given to DM rats significantly decreased MDA ( p < 0.01) and XO ( p < 0.05) activities and elevated GSH ( p < 0.05) contents when compared with DM group. Plasma levels of BUN and Cr were significantly higher in the DM group when compared with the control group ( p < 0.01). Pretreatment of the DM animals with crocin decreased the high level of serum Cr and BUN. Control group was normal in histological appearance, but congestion, severe inflammation, tubular desquamation, tubular necrosis, and hydropic degeneration in tubular cells were observed in the DM group. Histopathological changes markedly reduced, and appearance of kidney was nearly similar to control group in DM + crocin group. Our results show that crocin could be beneficial in reducing diabetes-induced renal injury.
Introduction
Oxidative stress occurs when oxygen free radicals are generated in excess through a reduction of oxygen or when antioxidant defense systems are impaired. Free oxygen radicals are highly toxic on all the cell components, especially in the cellular membrane. Increased free radical production exerts cytotoxic effects on the membrane phospholipid, resulting in the formation of toxic products such as malondialdehyde (MDA). 1 The reactive oxygen species (ROS) take role in pathogenesis of many diseases including hypoxia, hypercholesterolemia, atherosclerosis, hypertension, ischemia–reperfusion damage, and heart defects. 2,3 In addition, oxidative stress has an important role among complications of diabetes. 4,5 It has been asserted that hyperglycemia supports the oxidative stress with both enzymatic and nonenzymatic mechanisms. 5
Diabetes mellitus (DM) is the metabolic disorder with high prevalence rate. Prevalence of DM is extremely high around the globe and is assumed to be around 6% up to 2025. 6 DM is characterized with hyperglycemia and is related to chronic disorders of carbohydrate, protein, and lipid metabolisms. Absolute (type 1) and relative (type 2) insulin deficiency are observed in diabetes. 7 If diabetes is not cured in time, it causes some complications such as atherosclerosis, nephropathy, retinopathy, and neuropathy. 8 –10 It was observed in patients with DM that oxidative stress was increased, antioxidant defense system was spoiled, and therefore, complications relating diabetes developed and proceeded. 11 It has been clinically and experimentally proven that ROS increase in both types of diabetes. ROS causes depletion of cellular antioxidants such as glutathione (GSH). Normally, the level of oxidative stress is regulated by antioxidant defense system. 12 The antioxidant defense systems, both the nonenzymatic free radical scavengers (e.g. vitamin E and vitamin C) and the antioxidants scavenging enzymes (catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase (GSH-Px)), offer protection to cells and tissues against oxidative injury. 13
Oral antihyperglycemic drugs such as insulin, biguanidine, and sulfonylurea are the milestones in the treatment of diabetes, but they may lead to adverse effects, cannot regulate blood glucose always, and cannot inhibit the complications of diabetes.
14,15
Therefore, alternative antidiabetic solutions that have higher benefit
It has been reported that saffron (C. sativus L.) has hypolipemic, antiinflammatory, antioxidant, and anticarcinogenic effects. 17 –20 Crocin (crocetin glycoside), crocetin, and safranal are the main active constituents of saffron. 21 In addition, saffron comprises protein, sugars, vitamins, flavonoids, amino acids, vital minerals, and other chemical compounds. 22,23 Crocin, which is the water-soluble and active constituent of saffron, is effective in protecting against ischemia–reperfusion damage and eliminates oxidative stress in rats. 24 It has also been reported that saffron extract, crocin, and safranal have remarkable radical scavenging activities and this activity arises from antioxidant properties. 25
The aim of this study was to evaluate whether crocin administration could protect kidney injury from oxidative stress in streptozotocin (STZ)-induced diabetic rats.
Materials and methods
Experimental animals
In this study, 30 female Wistar albino rats (Inonu University Animal Research Center, Malatya, Turkey), weighing between 150 and 200 g, were housed in individual cages for 21 days in a well-ventilated room with a 12-h:12-h light/dark cycle at 21°C. Animals were fed with standard rat chow and tap water ad libitum. The experiments were performed in accordance with the guidelines for animal research from the National Institute of Health and were approved by the Committee on animal Research at Inonu University, Malatya, Turkey.
Experimental design
At the onset of the study, each rat was weighed and blood samples were taken from tail veins for the measurement of blood glucose levels. The rats were randomly divided into 3 groups each containing 10 animals as follows: group 1, control (nondiabetic rats); group 2, DM (STZ-induced untreated diabetic rats); and group 3, DM + crocin (STZ-induced diabetic rats treated with crocin). Type 1 DM was induced in female Wistar rats by a single intraperitoneal (i.p.) injection of STZ (Sigma Chemical Co., St Louis, Missouri, USA). STZ dissolved in 0.01 M sodium citrate buffer (pH 4.5) was administered at a single dose of 50 mg/kg body weight via i.p. injection. In control group, however, 1 ml sodium citrate buffer was injected via i.p. injection. Three days after administration of STZ, tail vein blood glucose levels were measured with a glucometer (Accu-Check Active, ROCHE, Germany) using reagent strip (Accu-Check Active Glucose test strips, ROCHE, Germany). Those rats with blood glucose levels within normal limits are categorized in the control group, while the rats with blood glucose levels >270 mg/dl were categorized under DM and DM + crocin groups and considered as diabetic. Normal saline was administered in the control and DM groups; and crocin (Sigma Chemical Co.) dissolved in normal saline was administered in DM + crocin group at a dose of 20 mg/kg/day. 26 All the administrations were performed at the same hour and with a volume of 5 ml/kg body weight/day via gavage and continued for 21 days. At the end of the study, all animals were killed under ketamine anesthesia. Trunk blood was collected to determine the plasma levels of blood urea nitrogen (BUN) and creatinine (Cr). The kidney tissue was removed rapidly and decapsulated. It was divided into equal two pieces. One of the pieces was placed into 10% formaldehyde for routine histopathological examination. The other piece was stored at −80°C for the measurement of MDA, GSH levels, and xanthine oxidase (XO) activity.
Biochemical analysis
The kidney tissue was homogenized in ice within 1 M tris(hydroxymethyl)aminomethane–hydrochloric acid buffer (pH 7.5; includes protease inhibitor, phenylmethylsulfonyl fluoride, 1 mM) via homogenizator (IKA Ultra Turrax T25 basic, IKA Labotechnik, Staufen, Germany) at 16,000 r/min and at +4°C for 3 min. The homogenates were used for the measurement of MDA, GSH levels, and XO activities. All the procedures were performed at +4°C. The main principle of the analysis is based on the fact that MDA in the medium reacts when heated with thiobarbituric acid and creates a pink chromogen. The intensity of the pink color is in direct proportion to MDA concentration. MDA levels were assayed spectrophotometrically at 535 and 520 nm according to the method described by Ohkawa et al. 27 The results were expressed as nanomoles per gram wet tissue.
GSH levels were measured by the method followed by Ellman. 28 GSH is reacted with 5,5-dithiobis-2-nitrobenzoic acid resulting in the formation of a product which has a maximal absorbance at 410 nm. Results were expressed as nanomoles per gram wet tissue.
XO activity was determined spectrophotometrically according to the method described by Prajda and Weber based on the formation of uric acid from xanthin which increases absorbance at 292 nm (ε M 9.2 × 103). 29 One unit of activity is defined as one micromole of uric acid formed per minute, and data are presented as units per gram protein.
Protein levels were measured by the Bradford method. 30 The absorbance was recorded at 595 nm using an ultraviolet–visible spectrophotometer. Bovine serum albumin was used as the protein standard.
Blood samples for the measurement of plasma BUN and Cr levels were drawn into ethylenediaminetetraacetic acid-containing tubes and immediately placed on ice. All tubes were centrifuged within several minutes of collection and stored at −70°C until assay. Plasma BUN and Cr levels were performed using commercially available Architect c 1600 automatic analyzer kits (Abbott, Abbott Park, Illinois, USA).
Histological determination
For the light microscopic examination, kidneys were fixed in 10% formaldehyde and embedded in paraffin. Tissue blocks were cut at 5 μm, mounted on slides, stained with hematoxylin–eosin (H-E). The tissue sections were evaluated for severity of kidney injury such as congestion, inflammation, tubular desquamation, tubular necrosis, and hydropic degeneration in tubular cells. For this analysis, kidney injury was semiquantitatively graded as follows: (0) normal, (1) mild, (2) moderate, and (3) severe, for each criterion. The maximum score was 15. All sections were examined in random order under blindfold conditions using a Leica DFC280 light microscope and a Leica Q Win and Image Analysis system (Leica Micros Imaging Solutions Ltd, Cambridge, UK).
Statistical analysis
Data were analyzed using Statistical Package for Social Sciences for Windows version 13.0 (SPSS Inc., Chicago, Illinois, USA) statistical program. Histopathological and biochemical results were expressed as mean ± SE. Normality for continued variables in groups was determined by the Shapiro–Wilk test. The variables did not show normal distribution ( p < 0.05). Kruskal–Wallis and Mann–Whitney U tests were used for comparison of variables among the studied groups. The value of p < 0.05 was considered statistically significant.
Results
Biochemical results
Table 1 presents the tissue levels of MDA, GSH, XO, BUN, and Cr levels. Briefly, diabetes caused a significant increase in MDA, the end product of lipid peroxidation and XO activities, and a decrease in GSH contents ( p < 0.01) when compared with the control group of rats kidneys. Crocin given to DM rats significantly decreased MDA ( p < 0.01) and XO ( p < 0.05) activities, and elevated GSH ( p < 0.05) contents when compared with DM group in the kidney tissues (Table 1).
The tissue levels of MDA, GSH, XO, BUN, and Cr levels.a

MDA: malondialdehyde; GSH: glutathione; XO: xanthine oxidase; BUN: blood urea nitrogen; Cr: creatinine; DM: diabetes mellitus.
aData are given as mean ± SE.
b p < 0.01: compared with the control rats.
c p < 0.01: compared with diabetic rats.
d p < 0.05: compared with diabetic rats.
e p < 0.05: compared with control rats.
Plasma levels of BUN and Cr were significantly higher in the DM group, when compared with the control group ( p < 0.01). Pretreatment of the DM animals with crocin decreased the high level of plasma Cr and BUN but this decrease was not statistically significant ( p > 0.05).
Histological results
Control group was normal in histological appearance (Figure 1(a)). On the other hand, congestion, severe inflammation, tubular desquamation, tubular necrosis, and hydropic degeneration in tubular cells were observed in the DM group. Some of the glomeruli appeared to be deformed. Increased mesangial cell proliferation and matrix expansion were observed in DM group (Figure 1(b) and (c)). Histopathological changes markedly reduced and appearance of kidney was nearly similar to the control group in DM + crocin group, (Figure 1(d)). When DM group and DM + crocin group were compared, a statistically significant difference was detected ( p = 0.001). The histopathological damage score is shown in Table 2.

(a) Control group: histological appearance is normal. H-E; ×40. (b) DM group: histopathological changes are observed such as tubular necrosis and hydropic degeneration (arrows) in tubules. H-E; ×40. (c) DM group: congestion (star), inflammation (thick arrows), and tubular desquamation (thin arrows) are also detected in this group. In addition, increased mesangial cell proliferation and matrix expansion are observed. H-E; ×40. (d) DM + crocin group: histopathological changes are reduced. H-E; ×40. H-E: hematoxylin–eosin; DM: diabetes mellitus.
The histopathological kidney damage score of all groups.a
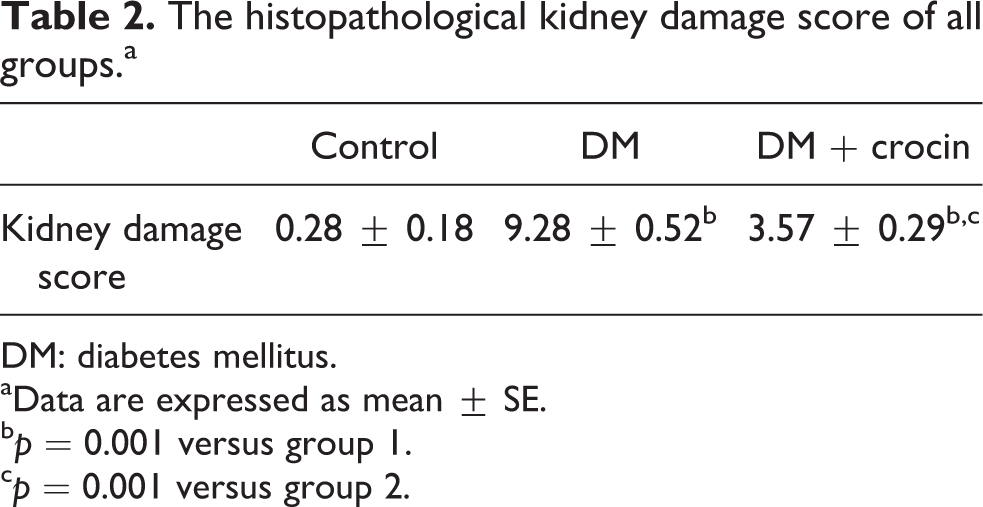
DM: diabetes mellitus.
aData are expressed as mean ± SE.
b p = 0.001 versus group 1.
c p = 0.001 versus group 2.
Discussion
In the present study, we evaluated the elimination of renal damage: (i) renal hemodynamics, (ii) detailed oxidant–antioxidant status, and (iii) kidney histopathology.
Our first finding is that crocin has protective effects on kidney hemodynamics. In the present study, plasma Cr and BUN levels increased in DM group, but the increased plasma Cr and BUN levels were decreased by crocin. The protective effect of crocin on Cr and BUN levels arises from its antioxidant properties, 25 although it has been found that ROS contributes to the spoilage of glomerular filtration rate (GFR). 31 Recently, some studies have shown that for humans, serum Cr with other clinical characteristics could be a more accurate measurement of GFR than the Cr clearance. 32 The spoilage in glomerular function leads to increase in BUN levels. Serum Cr concentration is more important than BUN levels in the early periods of kidney diseases. On the other hand, BUN begins to increase significantly just after the renal parenchymal damage. 33 Lu-Jia and Li-Jing reported that serum Cr and BUN levels were increased in diabetic rats. 34 Verma and Bordia 35 showed that the increased kidney functions were the signs of kidney dysfunctions when the diabetic rats were compared with control group. In the present study, plasma Cr and BUN levels increased significantly in DM group when compared with the control group. Uladimir suggested that defects or dysfunctions in several organs, especially in kidneys, were related to the long-term damage of hyperglycemia. 36 Recently, Jarald et al. 37 have reported that Cr and urea levels increased significantly in diabetic rats. They have showed that kidney dysfunctions of diabetic rats may arise from the increase in ROS which is the reason for tissue damages and the increase in lipid peroxidation. Additionally, Shah et al. 38 have reported that oxidative stress increase in diabetics and the decrease in cellular antioxidants lead to gradual loss in kidney functions, kidney tubular injury, and proteinuria. Elgazar et al. 39 have reported that the increased BUN and Cr levels decreased when saffron extract was administered to rats in several doses in parallel with the dose administered. This study also supports our study. In the present study, pretreatment of the DM animals with crocin decreased the high level of serum Cr and BUN, but this decrease was not statistically significant. Our results show that the high level of Cr and BUN will be close to the levels of control group when the dose of crocin is increased.
Another result of our study is that significant kidney damage has been observed in diabetic rats when compared with control group. Tissue damage has been prevented by the administration of crocin. This protection was achieved by the decreased MDA and XO activities and increased GSH contents and less tissue damage in the kidneys due to antioxidant properties of crocin. The healthy kidneys relatively and approximately consume oxygen at the rate of 10% of the total oxygen consumed by the body. Thus, the deviation in the generation of ROS is clearly observed in kidneys. In healthy rats, ROS generated by the kidneys are metabolized by appropriate scavenger mechanisms. However, the excessive ROS generation in endogenous- or exogenous-induced kidney injuries may lead to acute or improved kidney damage. 40 Therefore, it has been clearly demonstrated that renal injury is decreased by the reduced free radicals. Oxidative stress and the secondary lipid peroxidation increase in DM. 41
In the present study, MDA levels in DM group were found to be significantly higher than in the control group. Crocin also significantly decreased MDA production. MDA was used to measure the severity of lipid peroxidation, releasing the toxic effect of ROS, which destroys unsaturated fatty acids in the cell membrane. 42 MDA is the most important and studied parameter relating the lipid peroxidation in many diseases in recent years.
In this study, GSH levels were elevated by crocin in DM + crocin group when compared with DM group. Normally, the level of oxidative stress is regulated by antioxidant defense systems. 12 GSH is a vital part of nonenzymatic antioxidant system and have active roles in the antioxidant defense system of the body. As an antioxidant, it can neutralize free radicals, reduce hydrogen peroxide, and stabilize sulfhydryl groups. Hosseinzadeh et al. reported that the water extract of saffron and crocin significantly inhibited the ischemia–reperfusion-induced oxidative damage in the kidneys of rats. They also showed that the free radical-induced lipid peroxidation and MDA levels were decreased by saffron extract and crocin due to their antioxidant properties. 24
In the current study, our important result is the elevation of the XO activity in DM group when compared with the control group. Crocin also significantly decreased XO activity. Free oxygen radicals are generated from several sources in cells. It is known that the most important free radical source is XO. Under pathologic conditions, it may be generated from reversible (by means of blockage or oxidation of its thiol groups) or irreversible (by means of limiting proteolysis) process. 43 XO plays an important role in the catabolism of purines in some species, including humans. 44,45 XO enzyme is a potential superoxide source in the STZ-induced diabetic rats and this condition might be the reason of pathogenesis of diabetes complications. 46 Additionally, the formation of free radical and the decrease of oxidative stress may be ensured by administering XO inhibitors such as allopurinol in diabetics. 47
Another remarkable finding in our study is that biochemical and histological results are parallel to each other. In the present study, we examined some diabetes-induced alterations including congestion, severe inflammation, tubular desquamation, tubular necrosis, and hydropic degeneration in tubular cells. Control group was normal in histological appearance. On the other hand, these histopathological changes were observed in DM group. In addition, some of the glomeruli appeared to be deformed. Crocin also reduced significantly these histopathological changes and appearance of kidney was nearly similar to control group. Ajami et al. 48 suggested that treatment with gentamicin induced moderate to severe histological damage with dominant tubular necrosis extended to distal parts of proximal tubules and epithelial cells dissociation with cast formation, loss of brush border in large parts of proximal tubules, and tubular obstruction. When they administered 80 mg/kg saffron extract, damage was seen only in individual cells in proximal tubules with preservation of epithelial cells brush borders.
In conclusion, we demonstrate that crocin has a protective effect on diabetic nephropathy by preventing oxidative stress. Crocin shows an antioxidative effect by scavenging ROS. This antioxidative mechanisms cause MDA, BUN, and Cr levels to decrease, but GSH levels to increase after crocin treatment in diabetic rats. However, more studies are needed relating the nephroprotective efficiency of crocin.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
