Abstract
Background
Diabetes mellitus (DM) is a prolonged endocrine syndrome recognized by defective metabolism of biomolecules on account of flaws in insulin resistance. Diabetic nephropathy (DN) is a central microvascular complication of DM with inadequate treatment alternatives. Wogonin (WOG) is a pharmacologically active flavonoid isolated from the roots of Scutellaria baicalensis Georgi, which exerts strong renoprotection. However, the defensive mechanism of WOG against DN is not completely illuminated.
Objectives
This study investigates the role of WOG in streptozotocin (STZ)-induced DN in high-sucrose-high-fat fed rats.
Materials and Methods
DM was induced in experimental rats by a solo dosage of STZ (40 mg/kg) administered intraperitoneally. All rats were allocated into five sets, namely, normal, diabetic control (DC), metformin (150 mg/kg), and WOG (25 and 50 mg/kg) treated orally for 2 months.
Results
The antidiabetic and renoprotective effects of WOG were assessed by analyzing lipid profiles, glucose, inflammatory mediators, oxidative stress markers, renal functional parameters, gain in body weight, and histopathology of the kidney. In the current study, WOG could improve DM-induced metabolic alterations, dyslipidemia, kidney dysfunction, and inflammatory and oxidative prominence over its antidiabetic, antioxidative, antihyperlipidemic, and anti-inflammatory actions.
Conclusion
Thus, WOG is employed as a nephroprotective agent in DC rats.
Introduction
Diabetes mellitus (DM) is an endocrine chronic illness described by insistent levels of huge amounts of glucose in the blood, with the metabolism of proteins, carbohydrates, and lipids impaired as a consequence of insulin resistance or insulin secretion flaws (Pareek et al., 2009; Paneni et al., 2014). Nearly 463 million grown-ups grieve from DM, which can be able to develop 700 million by 2045 (Saeedi et al., 2019). Universally, DM is the early reason for death after cardiovascular disease (CVD) (Sun et al., 2022). Hyperglycemia leads to DM that develops membrane destruction and lipid peroxidation owing to the extreme generation of oxidative free radicals, which enhances the progression of further impediments. The most common pathological features of DM secondary complications are diabetic nephropathy (DN), neuropathy, CVD, and retinopathy.
DN is a serious microvascular complication linked with DM. DM patients with insistent hyperglycemia may develop around 30–40% of cases of DN and it is recognized as the primary cause of renal failure (Donate-Correa et al., 2020; Varghese & Jialal, 2023). Accumulating suggestions established that several hemodynamic, metabolic, and inflammatory signaling, specifically fibrosis, hyperglycemia, oxidative stress, and stimulation of renin–angiotensin, are included throughout the DN pathology (Akarshini & Aruna, 2014; Toth-Manikowski & Atta, 2015). It is a medical condition represented by durable proteinuria, constant drop in glomerular filtration rate, glomerular and tubular cell hypertrophy, excessively recruiting the component of extracellular matrix (ECM), mesangial expansion, inflammatory response, oxidative stress, tubulointerstitial fibrosis, and glomerular sclerosis that ultimately resulting in functional kidney letdown (Varghese & Jialal, 2023; Zhu et al., 2018). Therefore, it is an urgent requirement to find a further possible medication for DM-allied DN treatment (Nasiry et al., 2019).
Chinese herbal remedies are extensively used for DM treatments and its related complications (Liu et al., 2020; Meng et al., 2020). Among them, wogonin (WOG) is a pharmacologically active flavonoid derived from the roots of Scutellaria baicalensis Georgi, which has strong renoprotective actions (Du et al., 2019; Zheng et al., 2020). In a range of illnesses, WOG has exposed numerous pharmacological actions, for instance, antioxidative, anti-inflammatory, antiapoptotic, and cell cycle regulation (Hong et al., 2020). Meanwhile, WOG is chiefly recognized as an anticancer agent for its apoptotic, antitumor, and antioxidant activities (Yao et al., 2014; Zhao et al., 2010). The anticancer deeds of WOG are mainly owing to its capability to rummage oxidative free radicals to reduce nuclear factor kappa B (NF-κB) activity and to constrain many genes essential for the control of the cell division triggering cytostatic effects (Navale et al., 2018). Earlier, it has been documented that WOG administration diminished receptor-interacting protein kinase (RIPK)-facilitated necroptosis after cisplatin-prompted acute kidney injury in rodent models (Meng et al., 2018; Zhang et al., 2017). However, the fundamental mechanism of the renoprotective effect of WOG desires to be assessed. Hence, the current research work was intended to establish if WOG could avert the streptozotocin (STZ)-induced high-sucrose-high-fat (HSHF)-fed rat’s DN.
Materials and Methods
Chemicals
WOG, STZ, and biochemicals were acquired from Gibco (CA, USA). The HSHF diet was procured from BeiGene Pharma (China). Sun Pharmaceutical Industries Ltd. (Shanghai, China) provided metformin (MET). Elabscience Biotechnology (Wuhan, China) provided enzyme-linked immunosorbent assay (ELISA) assay kits.
Trial Animals
Wistar male rats, a total number of 50 having a body weight (BW) of 150–200 g, were selected. Animals were preserved in hygienic polypropylene cages in specified research laboratory environments. A typical pellet diet and clean water were given to the entire animals. They were adapted 7 days before the trials. All treatment protocols were piloted according to the Animal Ethics Committee.
Induction of DM
All rats were arbitrarily allocated to receive the normal control (NC) rats (n = 10) as the typical pellet diet and given the HSHF diet remaining rats (n = 40). After 14 days on HSHF chow and an intraperitoneally administered STZ solution that was freshly produced using pH 4.5 citrate buffers at a fixed dosage of 40 mg/kg, all animals were fasted for 12 hours. Two weeks later, the fasting blood glucose (FBG) from the tail veins was estimated. Animals having FBG ≥11.1 mmol/L were regarded as the diabetic control (DC) (Dubey et al., 2013).
Experimental Protocol
The rats were divided into a control group and five diabetic clusters, each including six rats. They were separated into five groups (n = 6): Group I: NC; Group II: DC; Group III: DC + MET (150 mg/kg/day, orally); Group IV: DC + WOG (25 mg/kg, orally); and Group V: DC + WOG (50 mg/kg, orally). All animals were given a dose of 5 mL for 60 days. Following the conclusion of the trial, all animals were starved for 12 hours and sedated with pentobarbital sodium (i.p., 45 mg/kg). Blood was taken via heart puncture and then sacrificed. A portion of the blood was preserved in ethylenediaminetetraacetic acid (EDTA) tubes, while the remaining amount was centrifuged to extract the serum. The kidney was weighed, cut up, and fixed in a 4% formalin-neutral buffer.
Estimation of BW and FBG Level
BW and FBG were determined weekly when fasting for 10 h. A solo dosage of glucose (2.5 g/kg) was treated in rats once fasting for 12 h. The level of BG at 0, 0.5, 1, and 2 h after glucose gavage was analyzed. The assessment of glucose area under the curve (AUC) was determined as follows: AUC = (0 h BG) × 0.25 + (0.5 h BG) × 0.5 + (1 h BG) × 0.75 + (2 h BG) × 0.5.
Estimation of Fasting Insulin (FINS), Homeostasis Model Assessment of Insulin Resistance (HOMA-IR), and Glycosylated Hemoglobin (HbA1c)
The determination of FINS was performed based on the manufacturer’s guidelines. HOMA-IR was determined according to the formula: HOMA-IR = FBG × FINS/22.5. EDTA tubes containing blood samples were employed for the assay of HbA1c estimated by a hemoglobin meter.
Measurement of Lipid Profiles
Lipid profiles in serum such as triglycerides (TG), total cholesterol (TC), low-density lipoprotein-cholesterol (LDL-C), and high-density lipoprotein-cholesterol (HDL-C) were investigated according to the company’s guidelines.
Analysis of Renal Function
On the 56th day, urine samples of rats were collected and kept in metabolic cages for 1 day. After the kidneys were segregated, the kidney/BW ratio was determined. A biochemical automatic analyzer assessed urine protein, serum creatinine (Scr), and blood urea nitrogen (BUN).
Analysis of Inflammatory Mediators
Inflammatory mediators comprising interleukin (IL)-1β, IL-6, and tumor necrosis factor-alpha (TNF-α) were examined based on the manufacturer’s guidelines.
Assay of Oxidative Stress Markers
In serum, oxidative stress mediators comprising malondialdehyde (MDA), catalase (CAT), superoxide dismutase (T-SOD), and glutathione peroxidase (GSH-Px) were analyzed according to ELISA kit directions.
Analysis of Kidney Histopathology
For histopathological examination, the kidney was separated, tissues fixed with formalin (10%), and paraffin fixed. Sections of 5 µm were cut using a rotary microtome, followed by hematoxylin and eosin (H&E) staining procedures, and observed through a light microscope.
Statistical Analysis
The statistical trial was accomplished by GraphPad Prism software version 8.0.1, which was employed to perform an analysis of variance (ANOVA) and, subsequently, Duncan’s test. The results were stated as mean ± SD. To assess the statistical significance, a Student’s t-test was conducted and considered at p < 0.05.
Results
Influence of WOG on BW and AUC
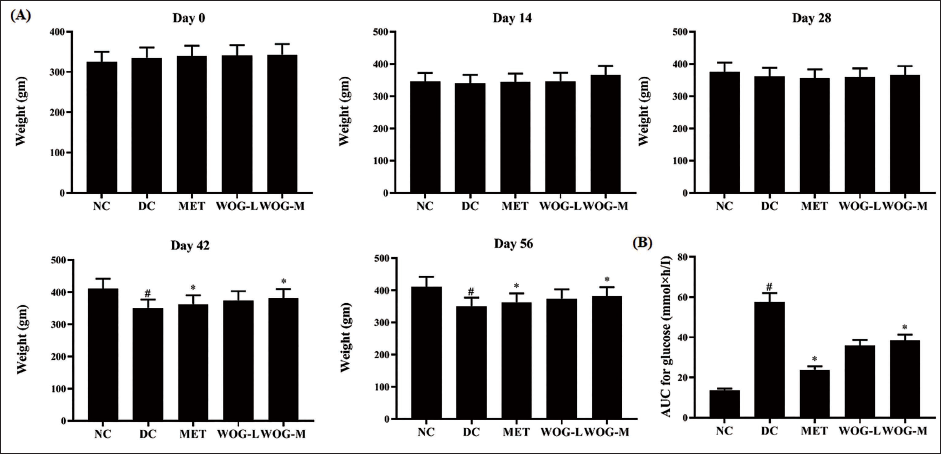
DC rats showed a significantly lower BW increase (p < 0.05) and higher AUC compared to NC rats. Administration of MET and WOG (25 and 50 mg/kg) significantly (p < 0.05) increased BW growth while decreasing AUC during the trial. The effect was higher in MET-treated DC rats than in WOG treatment (Figure 1).
Influence of Wogonin (WOG) on (A) Body Weight (BW) and (B) Area under the Curve (AUC) of Control and Diabetic Control (DC) Rats. Data were Denoted as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 and #p < 0.01 against Normal Control (NC).
Effect of WOG on the FBG Level
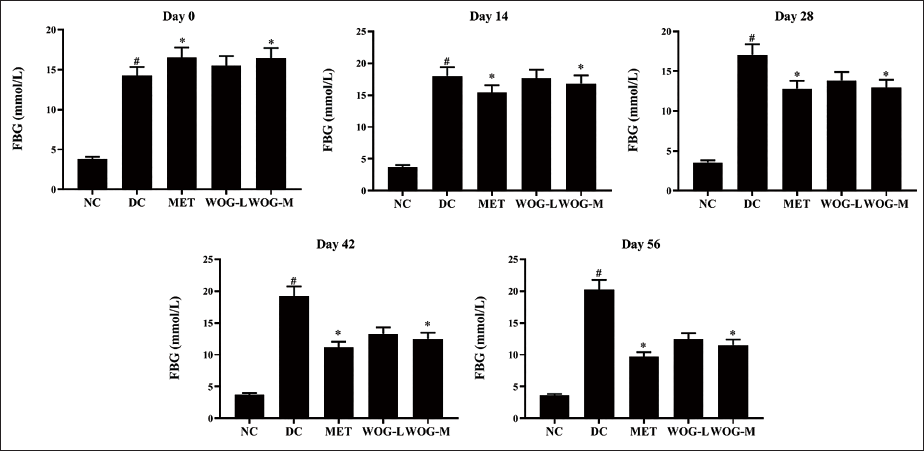
DC rats showed significantly higher levels of FBG (p < 0.05) compared to NC rats. On the 14th day, MET resulted in a significant decrease (p < 0.05) in FBG levels, while WOG (50 mg/kg) on the 28th and 25 mg/kg on the 42nd day. Results established that MET, WOG (25 mg/kg), and WOG (50 mg/kg) avert FBG in DC rats expressively (Figure 2).
Influence of Wogonin (WOG) on the Fasting Blood Glucose (FBG) Level of Control and Diabetic Control (DC) Rats. Data were Denoted as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 and #p < 0.01 against Normal Control (NC).
Influence of WOG on HOMA-IR, FINS, and HbA1c
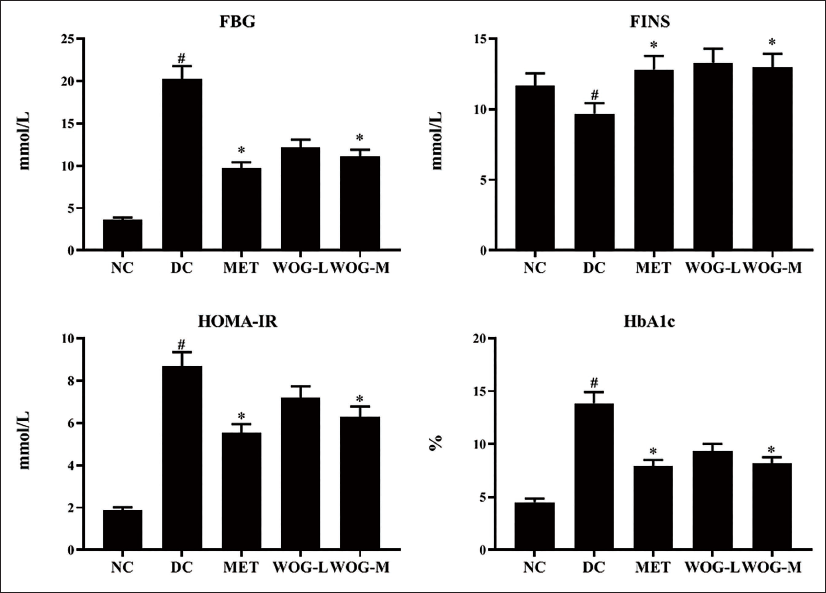
In DC rats, FINS was significantly lower (p < 0.05) compared to NC rats, although HOMA-IR and HbA1c levels were higher. In contrast, rats treated with MET or WOG showed a significant increase (p < 0.05) in FINS and a decrease (p < 0.05) in HOMA-IR and HbA1c in DC groups (Figure 3).
Influence of Wogonin (WOG) on Fasting Blood Glucose (FBG), Fasting Insulin (FINS), Homeostasis Model Assessment of Insulin Resistance (HOMA-IR), and Glycosylated Hemoglobin (HbA1c) Level of Control and Diabetic Control (DC) Rats. Data were Denoted as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 and #p < 0.01 against Normal Control (NC).
The Stimulus of WOG on Lipid Profiles
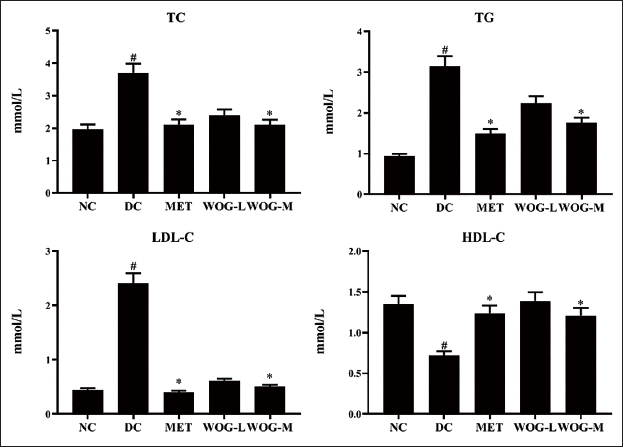
DC rats showed significantly higher levels of TG, TC, and LDL-C (p < 0.05) and lower levels of HDL-C (p < 0.05) when compared to NC rats. Administration of MET and WOG groups upgraded HDL-C pointedly and reduced TG, TC, and LDL-C more than DC. These data designated that MET and WOG, particularly WOG (50 mg/kg BW), could improve the metabolism of lipids in DC animals (Figure 4).
The Stimulus of Wogonin (WOG) on Lipid Profiles (Total Cholesterol (TC), Triglycerides (TG), Low-density Lipoprotein-Cholesterol (LDL-C) and High-density Lipoprotein-Cholesterol (HDL-C)) of Control and Diabetic Control (DC) Rats. Data were Denoted as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 and #p < 0.01 against Normal Control (NC).
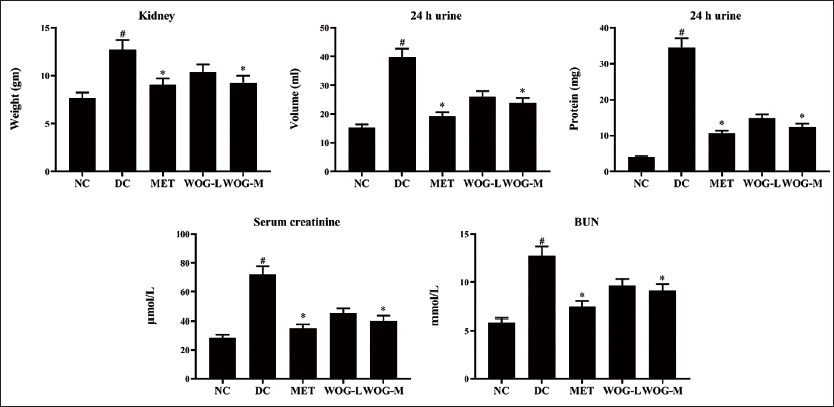
Influence of WOG on DN Factors
Figure 5 shows significant increases (p < 0.05) in kidney/BW ratio, urine protein, urine volume, BUN, and Scr in a 1-day DC sample compared to NC. Treatment with MET and OG showed a significant reduction (p < 0.05) compared to DC. MET and WOG enhanced renal function.
Influence of Wogonin (WOG) on Body Weight/Kidney Weight Ratio, and Diabetic Nephropathy (DN) Factors of Urine Protein, Urine Volume, Serum Creatinine, and Blood Urea Nitrogen (BUN) in Control and Diabetic Control (DC) Rats. Data were Denoted as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 and #p < 0.01 against Normal Control (NC).
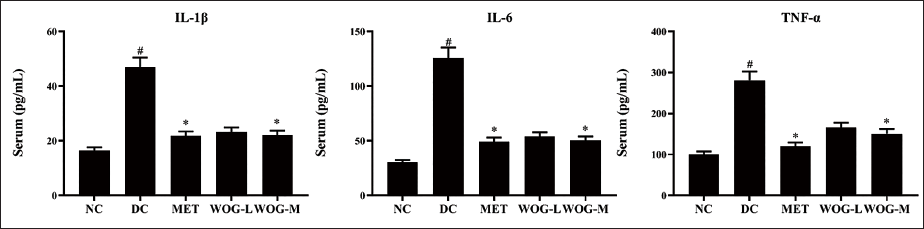
Influence of WOG on Inflammatory Mediators
Inflammatory mediators comprising TNF-α, IL-1β, and IL-6 were moderately significantly raised (p < 0.05) in DC counter to NC. Conversely, levels of these mediators were alleviated (p < 0.05) in MET and WOG groups considerably more than in DC (Figure 6).
Influence of Wogonin (WOG) on Inflammatory Mediators Interleukin (IL)-1β, IL-6, and Tumor Necrosis Factor-Alpha (TNF-α) of Control and Diabetic Control (DC) rats. Data were Denoted as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 and #p < 0.01 against Normal Control (NC).
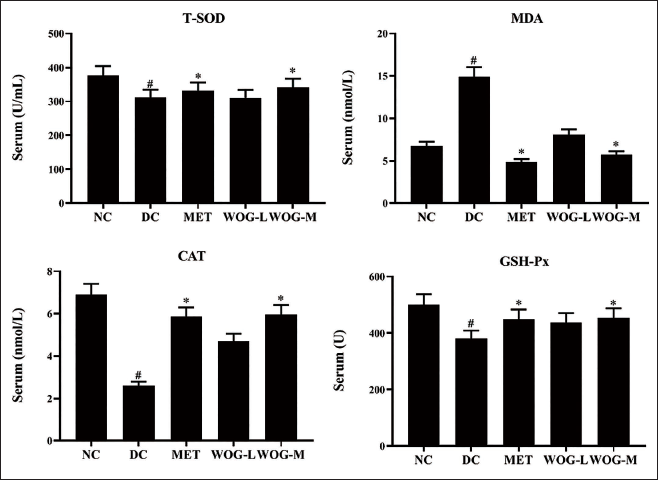
The Stimulus of WOG on Oxidative Markers
The antioxidant status of T-SOD, CAT, and GSH-Px expressively diminished (p < 0.05) in DC versus NC, but the MDA level was elevated (p < 0.05). Conversely, levels of antioxidants were pointedly elevated significantly (p < 0.05) in MET and WOG rats and reduced significantly (p < 0.05) the level of MDA counter to DC. These results mentioned that MET and WOG groups, specifically WOG (50 mg/kg BW), could attenuate oxidative stress and improve the antioxidant status in DC animals (Figure 7).
Influence of Wogonin (WOG) on Oxidative Markers Superoxide Dismutase (T-SOD), Malondialdehyde (MDA), Catalase (CAT), and Glutathione Peroxidase (GHS-Px) of Control and Diabetic Control (DC) Rats. Data were Denoted as Mean ± SD of Six Observations and the Significance is Considered as *p < 0.05 and #p < 0.01 against Normal Control (NC).
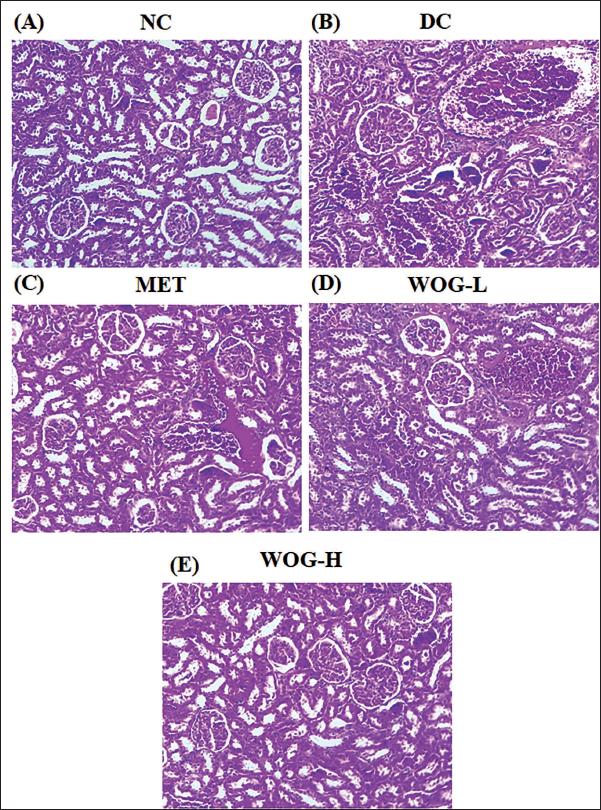
Analysis of Kidney Histopathology
DC rat’s histology of the kidney presented apparent glomerulus hypertrophy, the proliferation of mesangial cells, basement tissue thickening, and a few vacuolar tubular cells collapse with glycogen accumulation against NC. Conversely, the rats administered with MET and WOG were perceived as absent of palpable extension of the glomerulus, slight thickening of mesangial cells, and mild glycogen accumulation (Figure 8).
Influence of Wogonin (WOG) on Histopathology of Kidney in Control and Diabetic Control (DC) rats. (A) Normal Control (NC) Group Showing Normal Histology of the Kidney Cells. (B) DC Rats Display Apparent Glomerulus Hypertrophy, Proliferation of Mesangial Cells, Hardening of Basement Tissue, and Glycogen Deposition. (C, D, and E) The Rats Administered with Metformin (MET) and WOG (25 and 50 mg/kg) Noticed the Absence of Palpable Extension of the Glomerulus, Slight Thickening of Mesangial Cells, and Reduced Glycogen Level. Kidney Slices were Performed with Hematoxylin and Eosin (H&E) Staining.
Discussion
DM is a metabolic chronic syndrome described by hyperglycemia owing to insulin malfunction/deficiency, lipid dysfunction, extreme oxidative stress, and inflammation, which leads to further diabetic complications (Pareek et al., 2009; Paneni et al., 2014). In type 2 DM, hyperglycemia is the major problem; hence, averting and relieving hyperglycemia is a crucial task for human well-being. Several antidiabetic medicines have toxic side effects, thus herbal medicinal plant products are the hopeful substitute for DM treatment and its complications (Liu et al., 2020; Meng et al., 2018; Nasiry et al., 2019). In the research experiment, the HSHF food united with STZ-made DM is a classic investigational animal model (Reed et al., 2000; Zhang et al., 2008). In this work, we found that DC rats exhibited hyperglycemia, reduced levels of FINS, elevated HbA1c, HOMA-IR, oxidative stress, inflammation, and dysfunction of lipid metabolism. However, WOG demonstrated potential activity in glycemic switch, lipid management, oxidative stress relieving, antioxidant status, and renal defense during the experimental period. Here, DM rats of one group were also administered MET, the primary usage to manage type 2 DM. These findings exposed that either 25 or 50 mg/kg WOG administration is beneficial in dropping DM impediments through its antihyperlipidemic, antidiabetic, antioxidant, anti-inflammatory, and renoprotective activities.
STZ-prompted DM stimulates β-cells of islets of Langerhans damage owing to the reduction of insulin secretion, which further directs to the DM and associated complications, particularly DN (Wang et al., 2016). In our work, STZ-activated animals unveiled DM appearances and reduced BW together with elevated kidney weight. Loss of BW followed the administration of STZ caused by dehydration and fat/protein catabolism (Kumar et al., 2018). Supplementation of WOG displayed substantially higher BW, proposing a drop in dehydration and catabolism of fats/proteins. These findings specify that the administration of WOG may prevent kidney hypertrophy in DM rats. Similar results were proposed previously that WOG administration moderated RIPK-facilitated necroptosis after cisplatin-prompted acute kidney injury in rodent models. Recently, it was reported that WOG protects against STZ-induced diabetic cardiomyopathy, necrosis, apoptosis, and cardiac fibrosis through its action of antihyperglycemia, anti-insulin resistance, antioxidant, and anti-inflammation leading to improved cardiac function (Khan et al., 2016). Our current findings are consistent with this report.
Our research further established that unrestrained chronic hyperglycemia instigated kidney disorder as designated by high levels of Scr, urea, BUN, and reduced levels of serum protein along with a momentous high volume of urine. Microproteinuria is the typical DN feature and it may suggest the progression of DN. Sustained hyperglycemia makes DN a known severe DM-allied microvascular complication (Papadopoulou-Marketou et al., 2017) and supports our present outcomes. Diverse portions of the kidney’s nephrons are inflated by DN. The basement tissues of glomeruli and mesangial get hardened because of the extreme gathering of ECM (Kolset et al., 2012). This is obvious from the relative kidney weight enhancement in the DC rats in these current trials. Administration of WOG alleviated the level of BUN, Scr, urine volume, and protein excretion in urine. These findings put forward the renoprotection of WOG, which was further supported by the reduction of histopathological variation of kidney and kidney weight by WOG administration confirming its nephroprotective effects on STZ-activated DM rats.
It is renowned that hyperlipidemia has a profound action on DM pathology and insulin resistance (Alkhateeb & Qnais, 2017). We found that the DC rats altered the normal lipid profile, including elevated TG, LDL-C, and TC, and a decline in HDL-C in serum-specified momentous dyslipidemia in DC rats. This result agrees with the preceding documents (Kaur & Meena, 2012). These lipid profile variations may be ascribed to augmented cholesterol biosynthesis and intestinal absorption (Palumbo, 1998). Treatment with WOG attenuated TG, LDL-C, TC, and enhanced HDL-C in serum. These findings could be accredited to constraining the cholesterol synthesis pathway, diminished intestinal lipids absorption, improved glycemic control, and elevate the removal of LDL by triggering its receptors in liver cells (Subash Babu et al., 2007). The treatment of WOG (25 and 50 mg/kg) and MET to DC rats prominently upgraded hypolipidemic action by either enhanced secretion of insulin and/or improved insulin sensitivity.
Hyperglycemia may directly or indirectly develop diabetes-allied inflammation and oxidative stress, leading to renal and cardiac function impairment (Akarshini & Aruna, 2014; Toth-Manikowski & Atta, 2015). ROS-triggered oxidative stress is regarded as a usual pathophysiology of DM-facilitated complications (Varghese & Jialal, 2023). In this study, the oxidative status markers, namely MDA levels were pointedly augmented with a lessening of the T-SOD, CAT, and GSH-Px activities in DC rats. Our results also found that STZ treatment expressively elevated FBG, HbA1c, DM-activated insulin resistance, pro-inflammatory cytokines stimulation, and repressed antioxidants status. Interestingly, WOG administration could reverse STZ’s effects on DC rats. These findings are reliable with the earlier reports established that WOG alleviates hyperglycemia and dyslipidemia in mice model and restrains HG-prompted vascular inflammation and adipocyte osteopontin levels in both in vitro and in vivo models (Bak et al., 2014; Ku & Bae, 2015). Furthermore, WOG has been exposed to have anti-inflammatory and antioxidative capabilities in several cell forms, including vascular smooth muscles, endothelial cells, and adipocytes (Lee et al., 2015; Wang et al., 2015). In the present study, WOG was found to prevent IL-1β, IL-6, and TNF-α cytokines, diminish oxidative stress, and restore antioxidant status, suggesting that WOG could subdue DC-induced inflammation and oxidative stress. Related to MET, WOG also plays an eminent antidiabetic, anti-lipidemic, anti-inflammatory, antioxidant, and nephroprotective actions. It would be essential to further explore the WOG’s latent mechanisms on DM and its complications.
Similarly, recent studies, such as those explained about diabetes by Kaliaperumal et al. (2024), revealed that Tribulus terrestris (L) (TT) and Curcuma amada (R) (CA) extract-treated diabetic groups significantly lowered the mean blood glucose levels followed by increased glycogen and insulin levels. Urea, creatinine, and HbA1c levels were considerably reduced in TT and CA-treated diabetic animals as compared to that of antidiabetic drug glibenclamide-treated groups. TT and CA-treated diabetic animals showed considerable net BW gain at the end of the experimental day. A concluding remark of the study shows that Saeed et al. (2021) suggested that shikonin could be an effective hypoglycemic drug that inhibits PTP1B, presumably in the catalytic domain. The ligands’ binding affinities to the target PTP1B active site were determined using docking and molecular dynamics simulations. The combined in silico experiments revealed structural changes that could aid in the development of more effective PTP1B inhibitors via strategic molecular architecture design and optimization. The shikonin scaffold and its antidiabetic actions as PTP1B inhibitors are thoroughly explored in this article, opening new avenues for the creation of novel medications for the treatment of type 2 diabetes.
Conclusion
In summary, this study provides compelling evidence that WOG exerts renoprotective effects in experimental DN through multi-targeted mechanisms including antihyperglycemic, lipid-lowering, anti-inflammatory, and antioxidant actions. WOG-treated DC rats also attenuate kidney dysfunction, microproteinuria, and oxidative injury to the kidneys. Further research is warranted to elucidate the specific molecular pathways involved. Clinical studies would also be valuable to determine if WOG may have therapeutic potential for treating or preventing kidney disease in human diabetes. Therefore, it could be established that WOG demonstrated a nephroprotective action in DC rats. Further research is desired to explore WOG in clinical use.
Abbreviations
DM: Diabetes mellitus; CVD: Cardiovascular disease; DN: Diabetic nephropathy; ECM: Extracellular matrix; WOG: Wogonin; STZ: Streptozotocin; FBG: Fasting blood glucose; DC: Diabetic control; NC: Normal control; BW: Body weight; H&E: Hematoxylin and eosin; ANOVA: Analysis of variance.
Footnotes
Acknowledgments
None.
Data Availability
All data generated or analyzed during this study were included in this manuscript. Further inquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
