Abstract
Background
Eucalyptol, a terpenoid oxide isolated from the essential oil of several Eucalyptus species, exhibits numerous pharmacological actions. Myocardial infarction is a major reason for death worldwide, although major advancements have occurred in its treatment.
Objectives
Existing study aims to reveal the cardioprotective action of Eucalyptol in myocardial infarction rat models and examines the toll-like receptor 4 (TLR4)/myeloid differentiation protein 88 (MyD88)/nuclear factor-kappa B (NF-κB) pathway as a possible mechanism of action.
Materials and Methods
Animals were distributed into five groups, which were treated by saline or Eucalyptol (100 or 200 mg/kg) for 3 weeks. Myocardial infarction was induced with isoproterenol on 20th and 21st days.
Results
Pretreatment with Eucalyptol revealed infarcted area-restricting effect, cardiac indicator enzymes diminishing, and electrocardiogram (ECG) adjustment, as all those parameters were challenged by isoproterenol-induced myocardial infarction insult. Furthermore, Eucalyptol reduced inflammatory mediator levels and apoptotic markers. Additionally, it suppressed messenger ribonucleic acid (mRNA) expression of TLR2, TLR4, MyD88, and TRIF, suggesting downregulation of the TLR pathway. The docking study of Eucalyptol with TLR4 demonstrates a binding affinity of –4.193 kcal/mol, indicating favorable interaction affinity.
Conclusion
Eucalyptol could act as a probable defending agent in myocardial infarction via diminishing inflammatory mediators and attenuating the apoptosis of myocardial cells through inhibition of TLR/NF-κB. These results might widen the clinical application of such a natural compound.
Introduction
Eucalyptol (synonym 1,8-cineole, 1,8-epoxy-p-menthane), Figure 1A, is a monoterpene epoxide identified in the essential oil of several Eucalyptus species such as Eucalyptus globulus Labill (Mączka et al., 2021). Eucalyptol has become a focus of attention due to its ability to inhibit and improve several chronic diseases (Hoch et al., 2023). Eucalyptol exhibited several pharmacological activities, including analgesic (Melo Júnior et al., 2017), anti-bacterial (Guzzo et al., 2020), anti-cancer action (Abdalla et al., 2020), anti-fungal (Li et al., 2022), and anti-inflammatory (Arooj et al., 2023). For cardiac actions, Eucalyptol lowered blood pressure via NO and lipid peroxidation regulation in a nicotine chronic exposure-induced hypertension model (Moon et al., 2014). Furthermore, Eucalyptol reduces heart rate as well as prompts hypotension by direct vasorelaxation (Lahlou et al., 2002; Soares et al., 2005).
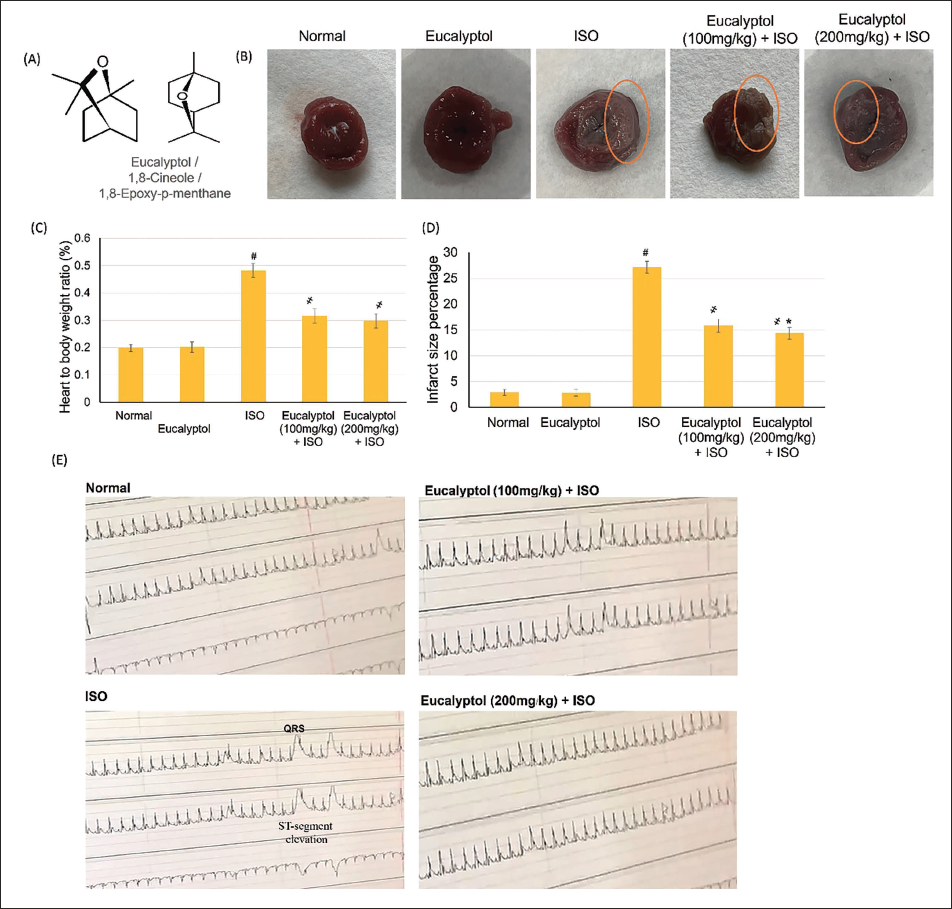
Eucalyptol Actions on Heart-to-body Relation and Infarct Size (%). (A) Eucalyptol Structure. (B) Representative Heart Sections Showing the Infarction. (C and D) Pretreatment with Eucalyptol (100 and 200 mg/kg) for 21 Days Actions on (C) Heart-to-body Weight Ratio and (D) Infarct Size (%) in Isoproterenol (ISO)-induced MI Animals, Respectively, and (E) Electrocardiogram (ECG) Demonstrative Images from Different Experimental Groups. Data Were Detailed as Mean ± Standard Deviation (SD). # Displays Statistically Significant from Normal Animals, ҂ Displays Statistically Significant from ISO Group When p < .05.
Myocardial infarction (MI) is still a main cause of death globally (Altyar et al., 2023; Martin et al., 2024). Necrotic cardiac myocytes release an extensive range of endogenous damage-associated molecular pattern (DAMPs) molecules, for instance, heat shock protein (HSP) and others, which further prompt toll-like receptors (TLRs) (Cicchinelli et al., 2024). TLRs associate DAMPs to trigger downstream signaling as well as stimulate transcription factors, thus prompting the inflammation evolution (Rai et al., 2022). TLRs signal cascade is separated into two types: myeloid differentiation protein 88 (MyD88) and TRIF pathways (Ma et al., 2018). TLR4 binding to ligands eventually activates nuclear factor-kappa B (NF-κB) translocation with subsequent extensive inflammatory response as well as immune-associated gene expression. Therefore, TLR4/NF-κB signaling possesses an influential part in myocardial injury (Han et al., 2017). Hence, inhibiting TLR4/MyD88/NF-κB signaling may successfully recover cardiac performance and ameliorate myocardial inflammation (Luo et al., 2023).
Although Eucalyptol has demonstrated numerous cardiovascular benefits, its potential protective effects against MI remain unexplored. Given the critical role of TLR4/MyD88/NF-κB signaling in myocardial inflammation and injury, the current study hypothesizes that Eucalyptol exerts cardioprotective effects by inhibiting this pathway, thereby reducing inflammation and improving cardiac function post-MI.
Thus, the objective of this study was to investigate the protective role of Eucalyptol in myocardial cells following infarction insult. Additionally, it elucidates the underlying mechanisms of Eucalyptol’s action through in vivo and in silico analyses, focusing on its modulation of the TLR4/MyD88/NF-κB pathway.
Materials and Methods
Materials
Eucalyptol (W246506) and triphenyl tetrazolium chloride (TTC) (366019) were procured from Merck (Sigma–Aldrich). Tumor necrosis factor alpha (TNF-α) (ab100785), creatine phosphokinase (CPK) (ab187396), cleaved caspase-9 (ab119508), creatine kinase myocardial bound (CK-MB) (ab155901), cardiac troponin T (cTnT) (ab246529), interleukin-1 beta (IL-1β) (ab100768), cardiac troponin I (cTnI) (ab246529), nuclear factor kappa B (NF-κB) (ab46070), and cleaved caspase-3 (ab181418) kits were all obtained from Abcam (Massachusetts, USA). Eucalyptol was dissolved in normal saline with 2% Tween 80 as mentioned before (Lima et al., 2013).
Animals
The experiment protocols followed the Ethical Conduct for Use of Animals in Research Guidelines at King Faisal University (KFU-ETHICS2684).
Experimental Design
Male Wister rats (250–270 g) were disseminated into five groups randomly (n = 6). Normal and Eucalyptol groups: rats were given saline or Eucalyptol for 3 weeks and saline subcutaneous (s.c.) on 20th and 21st days. MI group: animals consumed saline orally for 3 weeks and were subsequently provoked with isoproterenol (ISO) (85 mg/kg, s.c.) as stated before (Bi et al., 2018; Hassan et al., 2016) on 20th and 21st days to induce MI. Eucalyptol pretreatment groups: rats were given Eucalyptol (100 or 200 mg/kg (Santos et al., 2022)) for 3 weeks by oral gavage, dissolved in corn oil as a vehicle, then confronted with ISO on 20th and 21st days. Rats were killed humanely 24 h following the 2nd of ISO.
ISO administration produces significant biochemical and histological changes that lead to myocardial injury (Rona, 1985). ISO injection can replicate several features of human heart disease (Brooks & Conrad, 2009), making it a useful tool for evaluating potential therapeutic strategies (Gupta et al., 2024). The selection of Eucalyptol doses (100 mg/kg and 200 mg/kg) was based on preliminary studies, as well as prior research (Arshad, 2024; Caldas et al., 2015; Lima et al., 2013).
Electrocardiogram (ECG) Recording
Urethane-anesthetized rats were prone-positioned to obtain uninterrupted ECG recordings using an automated ECG device by an independent blinded technician. ECG components were mechanically estimated.
Samples Collection
Subsequent to ECG determination, blood samples were composed, centrifuged (20 min/4,500 rpm and –4°C), and the obtained serums were frozen for further biochemical examinations.
Infarct Size Determination
Frozen hearts were fragmented into 4–5 transverse portions, incubated for 25 min at room temperature in 10% 2,3,5-TTC, and analyzed using the Image J® program. Cardiac tissue sections were coded before staining and examined by an independent blinded pathologist.
Histopathological Assays
Consuming light microscopy (Leica DM 300, NY, USA), the cardiac slices (5 µm-thick) were evaluated after being stained with hematoxylin and eosin (H&E). An independent blinded pathologist examined TTC staining and preformed ImageJ analysis who were unaware of the treatment groups to minimize bias in infarct area quantification.
Assessment of Biochemical Indicators
Cardiac tissues were homogenized in 10% phosphate-buffered saline (PBS) to quantify the biochemical parameters, including cardiac enzymes, apoptotic, and inflammatory markers. Using a microplate reader SpectraMax i3x, ELISA kits were consumed following to the protocols.
Polymerase Chain Reaction (PCR) Investigation of TLR Pathway
Real-time PCR was executed following the procedures defined before (Younis & Mohamed, 2019). Concisely, a Trizol reagent kit was used, then a reverse transcription PCR kit was utilized. Quantification analyses were completed via the Opticon-2 real-time PCR reactor. Step PE applied biosystems software was utilized to scrutinize RT-PCR outcomes. Gene expressions were correlated to β-actin. The following are the primer sequences used: Bcl-2-F: 5′-CCGGGAGA TCGTGATGAAGT-3′, Bcl-2-R: 5′-ATCCCAG CCTCCGT TATCCT-3′, F: 5′-GTGGTTGCCCTCTTCTACTTTG-3′, Bax-R: 5′-CACAAA GATGGTCACTGTCTGC-3′, TLR4-F: 5′-AGTGTATCGGTGGTCAGTGTGCT-3′, R: 5′-AAACT CCAGCCACACATTCC-3′, TLR2-F: 5′-AAACTGTGTTC GTGCTTTCTGA-3′, TLR2-R: 5′-CTTTCTTCTCAATGG GTTCCAG-3′, MyD88-F: 5′-GAGATCCGCGAGTTTGA GAC-3′, R: 5′-CTGTTTCTGCTGGTTGCGTA-3′, TRIF-F: 5′-TCAGCCATTCTCCGTCCTCTTC-3′, R: 5′-GGTCAGC AGAAGGATAAGGAA-3′, β-actin-F: 5′-TGACAGGATGC AGAAGGAGA-3′, and R: 5′-TAGAGCCACCAATCCAC ACA-3′.
Molecular Docking of Eucalyptol against TLR4
Preparation
The crystal structure of the TLR4 protein was recovered from the Protein Data Bank (PDB). In ChimeraX, the protein structure underwent preprocessing to prepare it for docking (Meng et al., 2023), which involved removing heteroatoms, water, and solvent molecules, and any bound ligands from the binding site. Additionally, any missing residues were completed, and protonation states were assigned based on physiological pH using ChimeraX’s structure-preparation tools.
Ligand Preparation
The 3D structure of Eucalyptol was attained from PubChem and prepared using SwissDock (
Molecular Docking using SwissDock
Both the prepared TLR4 protein and Eucalyptol structures were uploaded to SwissDock (
To validate the docking protocol, we performed re-docking of the co-crystallized ligand from the TLR4 structure and calculated the RMSD between the docked pose and the experimental pose. The RMSD was found to be less than 2 Å, indicating reliable docking accuracy. Additionally, resatorvid (TAK-242), a known TLR4 inhibitor, was docked as a reference compound to compare binding affinities and validate the docking methodology.
Post-docking Analysis in ChimeraX
The highest-ranked docked poses were acquired from SwissDock. The docked complexes were loaded into ChimeraX to inspect binding orientation, interaction distance, and score. The Contact Analysis tool in ChimeraX was utilized to recognize the critical interactions between Eucalyptol and TLR4. The docked pose with the highest binding affinity and favorable orientation was designated for comprehensive analysis.
Statistical Analysis
Data revealed as mean ± standard deviation (SD). The GraphPad Prism program was used for the analysis. One-way analysis of variance (ANOVA) followed by Tukey’s test was executed to determine statistical significant with p < .05.
Results
Heart-to-body Relation and Infarct Size (%)
Normal and Eucalyptol control groups exhibited a normal heart-to-body ratio and negligible infarcted areas, while the MI group revealed significant intensification in the heart-to-body ratio together with an upsurge in infarcted regions reaching 27.1%. Conversely, administering Eucalyptol (100 and 200 mg/kg) revealed significant infracted-restricting action compared to MI group; however, no substantial alteration between the two Eucalyptol doses, as illustrated in Figure 1.
Electrocardiographic Traces
Normal as well as the Eucalyptol groups presented regular ECG, while ISO-prompted MI displayed frequent ECG alterations, including ST section and QT interval widening, shrinking P wave, QRS complex, and P-R and R-R intervals related to control groups. Alternatively, Eucalyptol (100 and 200 mg/kg) amended the ECG modifications, as illustrated in Table 1 and Figure 1D.
Pretreatment with Eucalyptol (100 and 200 mg/kg) for 21 Days Actions on Electrocardiogram (ECG) Components in Isoproterenol (ISO)-prompted Myocardial Infarction (MI).
Eucalyptol Effects on Cardiac Enzymes
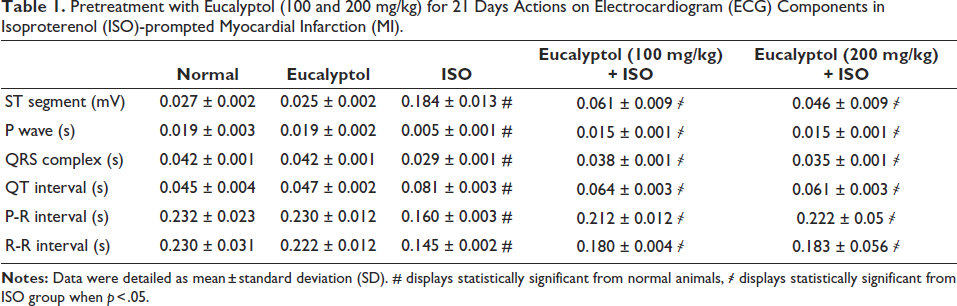
The ISO-induced MI group exposed an extensive escalation in heart enzymes, including CPK, CK-MB, low-density lipoprotein (LDL), cTnT, and cTnI, compared to normal group, whereas pretreatment with Eucalyptol (100 and 200 mg/kg) suggestively lessened these cardiac enzymes, as revealed in Figure 2.
Pretreatment with Eucalyptol (100 and 200 mg/kg) for 21 Days Actions on (A) Creatine Kinase Myocardial Bound (CK-MB), (B) Creatine Phosphokinase (CPK), (C) Low-density Lipoprotein (LDL), (D) Cardiac Troponin T (cTnT), and (E) Cardiac Troponin I (cTnI) in Isoproterenol (ISO)-prompted Myocardial Infarction (MI) Animals. Data Were Detailed as Mean ± Standard Deviation (SD). # Displays Statistically Significant from Normal Animals, ҂ Displays Statistically Significant from ISO Group When p < .05.
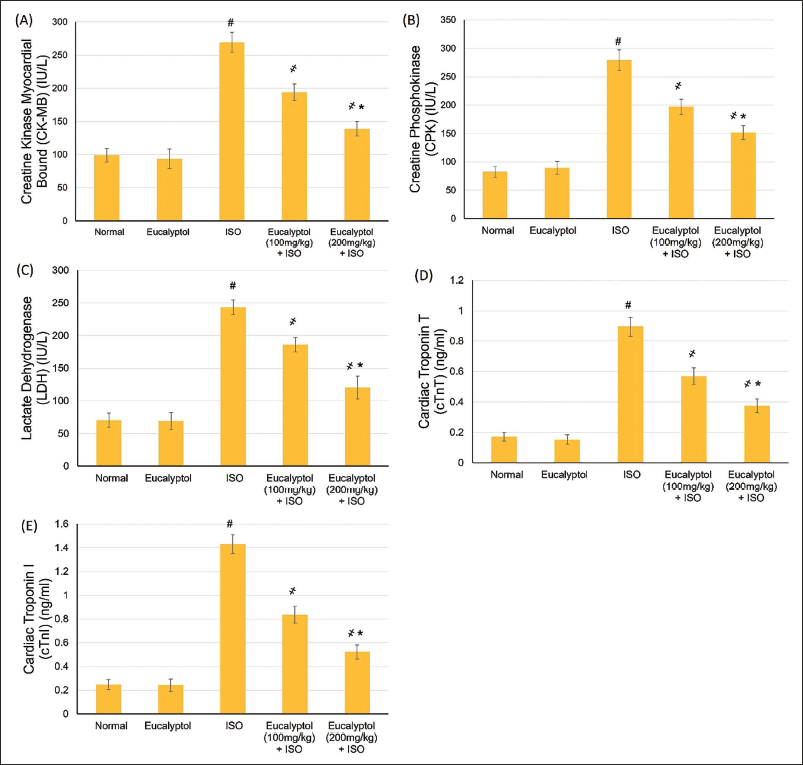
Eucalyptol Effects on the TLR Pathway mRNA Expression Levels
Gene expression outcomes demonstrated that animals suffering from MI caused by ISO exhibited a considerably increased expression of TLR2 and TLR4, as presented in Figure 3. However, pretreatment with Eucalyptol (100 and 200 mg/kg) noticeably declined TLR2, TLR4, MyD88, and TRIF relative expression levels when related to the ISO-induced MI control group (p < .05) (Figure 3).
Pretreatment with Eucalyptol (100 and 200 mg/kg) for 21 Days Actions on Messenger Ribonucleic Acid (mRNA) Expression of (A) Toll-like Receptor (TLR)2, (B) TLR4, (C) Myeloid Differentiation Protein 88 (MyD88), and (D) TRIF in Isoproterenol (ISO)-prompted Myocardial Infarction (MI). Data Were Detailed as Mean ± Standard Deviation (SD). # Displays Statistically Significant from Normal Animals, ҂ Displays Statistically Significant from ISO Group When p < .05.
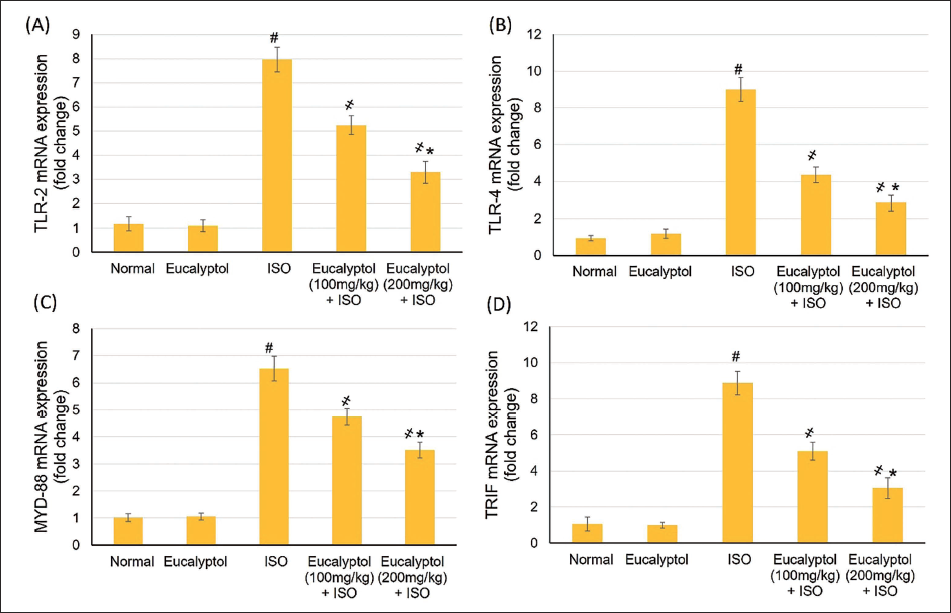
Pretreatment with Eucalyptol (100 and 200 mg/kg) for 21 Days Actions on (A) Interleukin (IL)-6, (B) Nuclear Factor Kappa B (NF-κB), (C) Tumor Necrosis Factor (TNF)-α, and (D) Heat Shock Protein (HSP)-60 in Isoproterenol (ISO)-prompted Myocardial Infarction (MI). Data Were Detailed as Mean ± Standard Deviation (SD). # Displays Statistically Significant from Normal Animals, ҂ Displays Statistically Significant from ISO Group When p < .05.
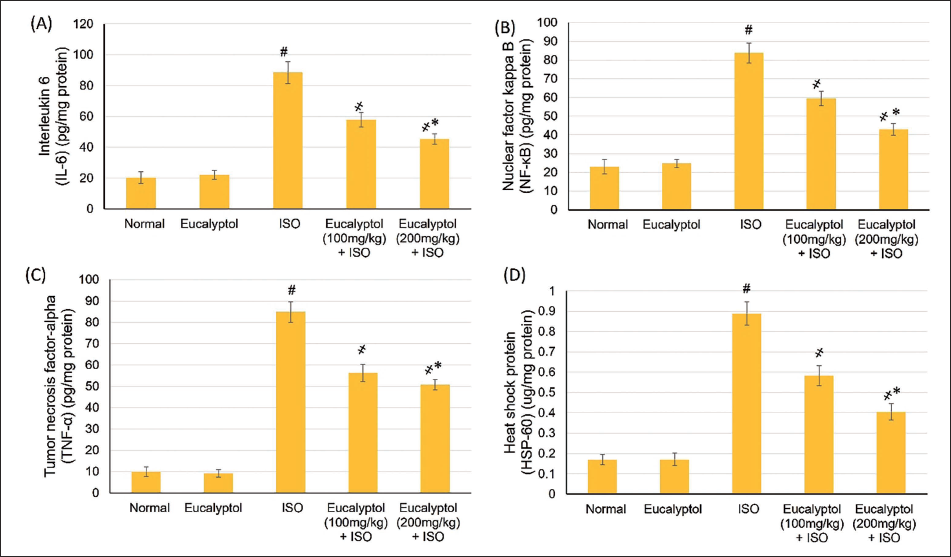
Eucalyptol Effects on HSP-60 and Inflammatory Mediators
Normal and Eucalyptol groups displayed no effect on HSP-60 and inflammatory indicators (TNF-α, IL-6, and NF-κB), while MI instigated a noteworthy (p < .05) elevation in HSP-60 and inflammatory markers levels. Pretreatment with Eucalyptol diminished the MI elevation in inflammatory mediators and HSP-60, as shown in Figure 5. Eucalyptol at a dose of 200 mg/kg caused a further noteworthy deteriorating in HSP-60, IL-6, and NF-κB than 100 mg/kg, except for TNF-α, in which a non-significant difference was found between both doses, as shown in Figure 4.
Pretreatment with Eucalyptol (100 and 200 mg/kg) for 21 Days Actions on (A) Bcl-2 and (B) Bax Messenger Ribonucleic Acid (mRNA) Expressions, (C) Caspase-3, and (D) Caspase-9 Levels in Isoproterenol (ISO)-prompted Myocardial Infarction (MI). Data Were Detailed as Mean ± Standard Deviation (SD). # Displays Statistically Significant from Normal Animals, ҂ Displays Statistically Significant from ISO Group When p < .05.
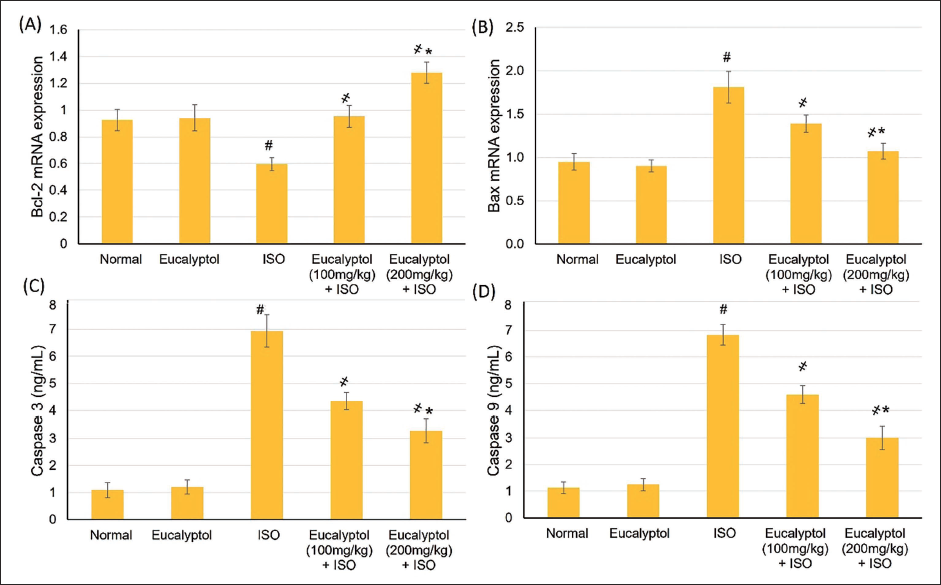
Eucalyptol Effects on Apoptotic Status within the Myocardium
Bax mRNA expression level and caspase-3 and caspase-9 activities were suggestively augmented, whereas Bcl-2 gene expression was lesser in the MI group, demonstrating an apoptotic condition ascending in MI-experiencing rats, as shown in Figure 5. However, in Eucalyptol and ISO, Bax gene expression and caspase-3 and caspase-9 activities were obviously declined, while Bcl-2 was increased, indicating that Eucalyptol limited MI-associated myocardial apoptosis, as shown in Figure 5.
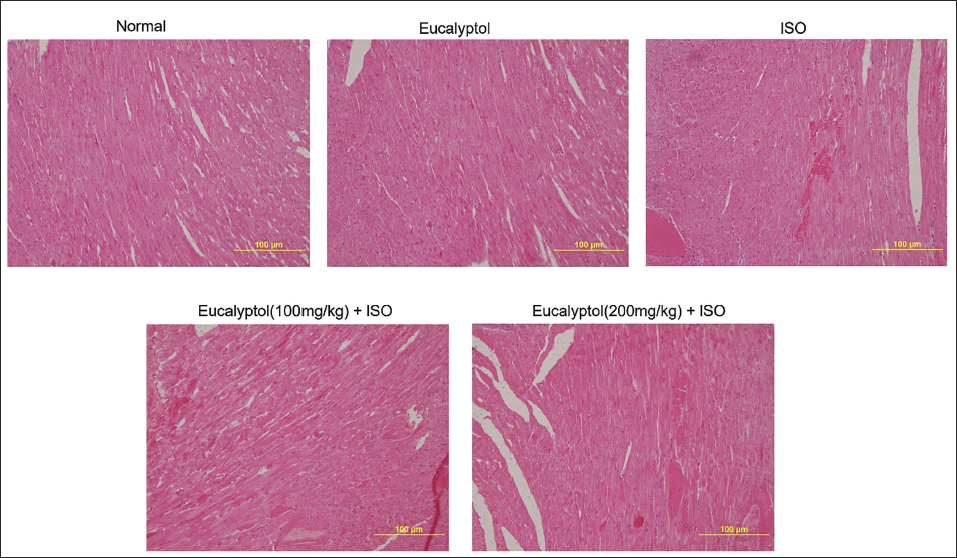
Eucalyptol Effects on Histopathological Examination within the Myocardium
In the control and Eucalyptol, myocardial sections appeared normal, with intact myocardial fibers with no signs of edema, necrosis, or inflammation. Alternatively, the ISO group showed disrupted myocardial fibers associated with edema and widespread infiltration of the immune cells. Groups that were pretreated with Eucalyptol exhibited preserved myocardial fibers, lessened myocardial necrosis, and deterred edema and immune cells infiltration (Figure 6).
Pretreatment with Eucalyptol (100 and 200 mg/kg) for 21 Days Actions on Histopathological Examination in Isoproterenol (ISO)-provoked Myocardial Infarction (MI) in Animals.
The Docking Study of Eucalyptol with TLR4
The docking study of Eucalyptol with TLR4 demonstrated a binding affinity of –4.193 kcal/mol, indicating a favorable interaction, though with moderate binding compared to resatorvid (TAK-242/CLI-095), a well-known inhibitor of TLR4 (Matsunaga et al., 2011), which scored –5.387 kcal/mol (Figure 7). While the observed binding affinity of –4.193 kcal/mol is moderate and does not imply strong inhibition, it does suggest that Eucalyptol is capable of interacting with the TLR4 receptor. Analysis of the binding mode revealed key interactions, including hydrophobic interactions with HIS 458, MET 437, LYS 435, and ALA 462, GLN 436, and ARG 460 contributed significantly to the stability of the complex. Although the presence of an oxygen atom in Eucalyptol, no hydrogen bonding affinity was detected, which could be attributed to the hydrophobic nature of the TLR4 binding site. Collectively, these findings provide insight into how Eucalyptol could modulate TLR4 signaling pathways, underscoring its potential therapeutic applications in all diseases that could involve the modulation of TLR4.
Docking Results of Eucalyptol with TLR4 Protein. The Docking Process Was Implemented by AutoDock Vina Via SwissDock (http://www.swissdock.ch ), and Then Photos Were Generated Through the Manipulation of the Docking File using ChimeraX. Different Hydrophobic Interactions Are Shown in Green Dotted Lines.
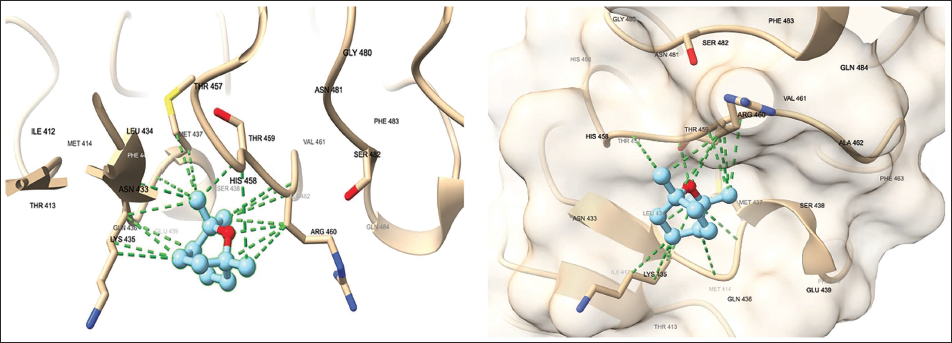
Compared to known TLR4 inhibitors, such as resatorvid (–5.3 kcal/mol), Eucalyptol exhibited a moderate binding affinity (–4.193 kcal/mol). This suggests that while Eucalyptol interacts with TLR4, its binding strength is lower than established inhibitors, indicating potential differences in its mechanism of action or bioavailability.
Discussion
The preclinical research has revealed the role of diet, rich in certain natural compounds, in the prevention of various cardiovascular diseases (Grewal et al., 2021; Ibrahim et al., 2020; Kabir et al., 2022; Tagde et al., 2021). Well-organized myocardial inflammation is indispensable for defense against injury and tissue repair. Nevertheless, extreme or continuing inflammation may provoke severe myocardium injury (Wu et al., 2023).
ISO-prompted MI group exposed an upsurge in heart-to-body ratio, expanded infarcted region, escalation in numerous cardiac enzymes indicators, as well as numerous ECG alterations, suggesting infarcted myocardium manifestation. Several previous reports mentioned similar changes occurring with ISO administration (Xing et al., 2022; Yang et al., 2022). On the other hand, pretreatment with Eucalyptol revealed infarct-limiting effect, cardiac indicator enzymes diminishing, and ECG adjustment revealing a cardioprotective effect of Eucalyptol. Earlier cardiovascular studies showed that Eucalyptol prompted hypotension in anesthetized or conscious normotensive animals (Lahlou et al., 2002). Further, Eucalyptol’s cardiovascular action includes a negative inotropic effect (Soares et al., 2005). Another report proved that Eucalyptol induced hypotension via NO and lipid peroxidation regulation (Moon et al., 2014).
Emerging evidence connect TLRs, particularly TLR2 and TLR4, to ischemic injury (Parizadeh et al., 2018; Yang et al., 2016). Within the initial few minutes, NF-κB translocates to the nucleus to prompt pro-inflammatory and proapoptotic gene expression, triggering cell dysfunction and death (Parizadeh et al., 2018). In the existing study, the MI group established a substantial increase in mRNA expression of TLR2, TLR4, MyD88, and TRIF, as well as elevation in inflammatory and apoptotic markers, demonstrating the inflammatory and apoptotic consequences following MI propagation. Previous reports demonstrated that ISO elevated TLR4 mRNA, MyD88, TNF-α, IL-6 (Kadry & Abdel-Megeed, 2018; Yang et al., 2016), and apoptotic markers (Zhang et al., 2017).
Results from the current experiment displayed that Eucalyptol diminished MI elevation in inflammatory mediators and apoptotic markers. Earlier studies documented that several monoterpenes, including Eucalyptol, were able to constrain the NF-κB pathway (Akcakavak et al., 2024). Eucalyptol inhibited NF-κB expression and decreased various inflammatory mediator levels in bleomycin-induced pulmonary fibrosis (Rui et al., 2022), monosodium urate (MSU)-induced gout arthritis (Yin et al., 2020), and acetaminophen-induced liver injury (Jiang et al., 2019).
Several mechanisms were proposed to illuminate Eucalyptol’s anti-inflammatory actions. For instance, Eucalyptol inhibited cytokine expression via deterring the phosphorylation of p38 mitogen-activated protein kinase (MAPK) and TLR4 expression in HBECs (Lee et al., 2016). Another study suggested that Eucalyptol prohibited the LPS-provoked IκBα degradation and NF-κB p65 translocation (Linghu et al., 2019). Additional mechanism was introduced by verifying that Eucalyptol caused an increase in IκBα protein level in an IKK-independent mode in U373 cells, in human peripheral blood mononuclear cells (Khan et al., 2014). Similarly, the anti-inflammatory activity of Eucalyptol may be modulated via selective downregulation of the pattern-recognition receptors (PRR) pathways (Yadav & Chandra, 2017). Furthermore, Eucalyptol diminished inflammation and oxidative stress via mitigating NF-κB (Kennedy-Feitosa et al., 2016).
The results showed that Eucalyptol diminished mRNA expressions of TLR2, TLR4, MyD88, and TRIF, indicating minimized post-MI TLR pathway activity with subsequent lessening in MI inflammatory mediators and apoptotic markers. DAMP activates TLR, leading to MyD88 recruitment, IKKs phosphorylation, IκBs phosphorylation and degradation, NF-κB translocation, and, finally, downstream genes become triggered. Some studies demonstrated that Eucalyptol may inhibit TLR4 expression and subsequent pathway. For instance, Eucalyptol inhibited the LPS-induced acute inflammation via downregulating TLR and NF-κB expression in mouse lungs (Zhao et al., 2014) and deterred the Der p-associated cytokine expression via deterring the intracellular TLR4 expression in HBECs (Lee et al., 2016).
Combining all data together, Eucalyptol demonstrated cardioprotective effects in our study by modulating the TLR4/MyD88/NF-κB pathway, diminishing inflammatory mediators as well as attenuating the apoptosis of myocardial cells. This protective effect suggests potential clinical applications in reducing post-MI inflammation, preventing adverse cardiac remodeling, and enhancing vascular function as an adjunct therapy alongside standard treatments. However, several translational hurdles must be addressed before clinical application. Bioavailability and pharmacokinetics remain key challenges, requiring further studies. Another critical barrier is the need for mechanistic validation in human cardiomyocytes and endothelial cells. Addressing these challenges through pharmacokinetic studies and human trials is essential for integrating Eucalyptol into MI management.
A limitation of the current study is the absence of protein-level confirmation via Western blotting or immunohistochemistry, which would provide additional mechanistic depth.
Conclusion
Eucalyptol may exhibit a cardio-protective action from the insult of MI in preclinical experiments. Eucalyptol acts through the TLR cascade, leading to the diminishing of the MI-accompanied inflammation and apoptosis. Eucalyptol demonstrates promising cardioprotective effects in an experimental MI animal model. However, further research is needed to address certain limitations, such as lack of chronic model validation, absence of functional outcome assessments, as well as clinical studies.
Footnotes
Acknowledgments
The authors would like to thank DSR and COCP at the University of King Faisal for continuous encouragement and support. Molecular graphics and analyses were performed with UCSF ChimeraX, developed by the Resource for Biocomputing, Visualization, and Informatics at the University of California, San Francisco, with support from the National Institutes of Health R01-GM129325 and the Office of Cyber Infrastructure and Computational Biology, National Institute of Allergy and Infectious Diseases.
Author Contributions
Conceptualization, investigation, methodology, formal analysis, software, writing—original draft, NSY, BA, and MEM; investigation, methodology, formal analysis, validation, resources, NSY and MEM; resources, data curation, validation, writing—review and editing, visualization, NSY, BA, and MEM; funding acquisition, BA. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The ethical approval has been granted from the institutional Ethics Committee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was sponsored by the Deanship of Scientific Research, King Faisal University, through the project number KFU241938.
Informed Consent
The informed consent was obtained for the study.
