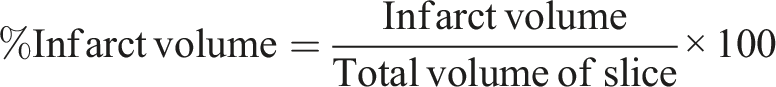Abstract
Objectives
This study aimed to evaluate the cardioprotective effects and mechanism of scutellarin (Scu) on heart ischaemia/reperfusion (I/R) injury by using an isolated Langendorff rat heart model.
Methods
Myocardial function was evaluated via measurements of cardiac haemodynamic parameters. Myocardial infarct size was macroscopically estimated by using TTC staining. The release of creatine kinase (CK) and lactate dehydrogenase (LDH) was used to evaluate cardiac injury. GSH/GSSG, SOD, and MDA were determined as being indicators of oxidative stress. CRP, IL-6, and TNF-α were also analysed to assess inflammation in the heart tissues. Cardiomyocyte apoptosis was quantified by using a TUNEL kit. PPARγ, Nrf2, and NF-κB were assayed via Western blotting.
Results
The results indicated that I/R induced significant cardiac dysfunction, myocardial infarct, and apoptosis, as well as decreasing the GSH/GSSG ratio, SOD activity, Nrf2 protein expression, and PPARγ protein expression, but increased levels of TNF-α, MDA, CRP, IL-6, and NF-κB.
Conclusion
These data suggest that Scu attenuates cardiac I/R.
Keywords
Introduction
Ischaemic heart disease is one of the main causes of disease mortality worldwide. 1 The restoration of blood supply through some surgical procedures, such as thrombolysis and coronary artery bypass surgery, is the mainstay of treatment for ischaemic heart disease. However, these procedures demonstrate the risk of myocardial ischaemia/reperfusion (I/R) injury. Myocardial I/R injury often occurs following surgery for ischaemic heart disease, which seriously endangers the life and health of patients. 2 It has been shown that the manipulation of the myocardial response to I/R can significantly impact the degree of I/R injury. Therefore, considerable efforts have been undertaken to elucidate the potential cardioprotective mechanisms. Some studies have shown that myocardial I/R injury can be attenuated by using drug pretreatment. 3 Therefore, it is feasible to identify a type of medicine that can reduce heart injury to improve the quality of life of ischaemic heart disease patients.
Heart I/R injury involved in pathological disorders includes oxidative damage, inflammatory damage, and myocardial cell apoptosis. 4 Excessive production of reactive oxygen species (ROS) is one of the important causes of heart I/R injury and the subsequent impairment of cardiac function. 5 A large number of ROS are produced during I/R, which leads to an imbalance between toxic free radicals and antioxidants, thereby increasing myocardial tissue damage. 6 Many studies have also shown that antioxidants can ameliorate heart I/R injury.7,8 Inflammation also plays an important role in heart I/R injury; a large number of inflammatory cytokines are released during I/R, and the reduction in the release of inflammatory factors can reduce I/R-induced heart damage.9,10
The flavonoid Scu is the primary active ingredient extracted from Erigeron breviscapus Hand-Mazz. 11 Previous studies have correlated the antioxidative and anti-inflammatory protective effects of Scu in cerebral I/R-induced brain injury concerning interactions with a wide variety of molecular targets. 12 However, the cardioprotective mechanism of Scu remains unclear. Therefore, the present study explored the antioxidative, anti-inflammatory, and cardioprotective effects of Scu on I/R-induced heart damage.
Materials and methods
Test compounds, chemicals, and reagents
Scutellarin (purity ≥98%) was purchased from Chengdu Must Bio-Technol Co., Ltd. (Chengdu, China). Oxidized glutathione, reduced glutathione, and 2,3,5-triphenyltetrazolium chloride (TTC) were purchased from Sigma Chemical Co. (Darmstadt, Germany). LDH detection assay kits, CK detection assay kits, MDA detection assay kits, SOD detection assay kits, and GSH/GSSG detection assay kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Additionally, TNF-α detection assay kits, IL-6 detection assay kits, and CRP detection assay kits were purchased from Shanghai Enzyme-linked Biotechnology Co., Ltd. (Shanghai, China). Anti-PPAR gamma antibody (ab178860), anti-Nrf2 antibody (ab92946), anti-NF-κB p65 antibody (ab32536), and anti-beta actin antibody (ab8226) were purchased from Abcam Plc. Moreover, HRP Anti-Rabbit IgG antibody (ab288151) was purchased from Abcam Plc. Finally, a protein extraction kit was purchased from Beijing Solarbio Science & Technology Co., Ltd. (Beijing, China).
Animals
Adult male Sprague Dawley (SD) rats (250 g to 300 g) were housed in the animal facility of the Yantai Yuhuangding Hospital Experimental Animal Center, in accordance with institutional guidelines. Animal housing conditions were maintained between 22°C and 25°C, with a relative humidity of 50–60% and a 12 h light/12 h dark cycle. All of the experimental procedures were approved by the Ethics Committee of Yantai Yuhuangding Hospital.
Experimental groups
The rats were randomly divided into five groups: (1) control group, in which the rat hearts were subjected to 0 min of zero-flow global ischaemia and 95 min stabilization; (2) I/R group, in which the rat hearts were subjected to 20 min of zero-flow global ischaemia, followed by a 45 min reperfusion; (3) Scu treatment groups, in which the rat hearts were stabilized for 20 min, followed by treatment with Krebs–Henseleit (KH) buffer solution containing different doses of Scu (1, 5, and 25 µg/ml) for 10 min and 20 min of zero-flow global ischaemia, followed by a 45 min reperfusion.
I/R model establishment
The heart I/R injury model was induced in the rats via Langendorff, according to a previously described method. The experiments were performed as previously described. 7
Measurement of heart haemodynamic parameters
Haemodynamic parameters were continuously monitored by using a computer-based data acquisition system. The left ventricular developed pressure (LVDP) and maximum rise/down velocity of the left intraventricular pressure (±dp/dt max ) were continuously monitored by using a 4S AD Instruments biology polygraph (Powerlab, Australia). The coronary flow (CF) was measured by using a flowmeter with an in-line probe.
Measurement of myocardial cell membrane damage
The markers of myocardial injury (LDH and CK) were measured to evaluate myocardial cell damage by using spectrophotometric cytotoxicity detection assay kits for LDH and CK, according to the manufacturer’s protocols.
Determination of myocardial infarct size
We used TTC staining to measure the myocardial infarct size. The experiments were performed as previously described.
7
The heart slices were incubated in 1% TTC in pH 7.4 buffer for 15 min, fixed in 10% formaldehyde solution, and photographed with a digital camera to distinguish the red-stained viable tissues and the white-unstained necrotic tissues. Areas stained red and white were measured by using Image-Pro Plus 7.0 (Media Cybernetics, Wyoming, USA). The infarction size percentage was calculated by using the following equation:
Assay of oxidative stress and inflammation
After 45 min of perfusion, the hearts were frozen and homogenized. The supernatant was obtained by using cryogenic centrifugation for testing.
The malondialdehyde (MDA) level and SOD activity were determined by using spectrophotometric assay kits (Nanjing Jiancheng Bioengineering Institute, China). We used a kit to detect the contents of glutathione (GSH) and glutathione disulfide (GSSG) and calculated the ratio of GSH/GSSG. The tumour necrosis factor-α (TNF-α), C-reactive protein (CRP), and interleukin-6 (IL-6) levels were analysed by using ELISA, according to the manufacturers’ instructions.
Detection of cardiomyocyte apoptosis
We used an In Situ Cell Death Detection Kit, POD (Roche, Basel, Switzerland) to evaluate myocardial cardiomyocyte apoptosis. The experiments were performed as previously described. 7
Western blot analysis
Protein levels of PPARγ, Nrf2, and NF-κB were measured via Western blotting. The experiments were performed as previously described. 7 The same part of the rat heart was cut and collected from each sample, and protein samples were extracted by using a protein extraction kit. The proteins were separated via electrophoresis on a 12% SDS polyacrylamide gel, transferred to nylon membranes via an electrophoretic transfer system, and then serially incubated with rabbit anti-rat PPAR-γ, rabbit anti-rat NF-κB, rabbit anti-rat Nrf2, and rabbit anti-rat β-actin polyclonal antibodies at 4°C overnight. The membranes were then washed with TBST buffer and incubated with HRP anti-rabbit IgG antibody. Finally, the bands were visualized by using the ECL-plus reagent, and the Bio–Rad Gel Doc 2000 imaging system and software were used to calculate the integrated absorbance (IA) of the bands.
Statistical analysis
Data are presented as the mean ± standard deviation from at least six independent experiments. Statistical differences were determined by using a Student’s t test, where p < .05 was considered to be statistically significant. The analyses were performed by using the Statistical Program for Social Sciences Software (International Business Machines Corporation, New York, USA).
Results
The effect of Scu on cardiac function
Scu improves cardiac dysfunction (mean ± SD, n = 8).

Note: Con: Control group; L-Scu: 1 µg/ml group; M-Scu: 5 µg/ml group; H-Scu: 25 µg/ml group. ## p < .01 versus control; * p < .05 and ** p < .01 versus I/R.
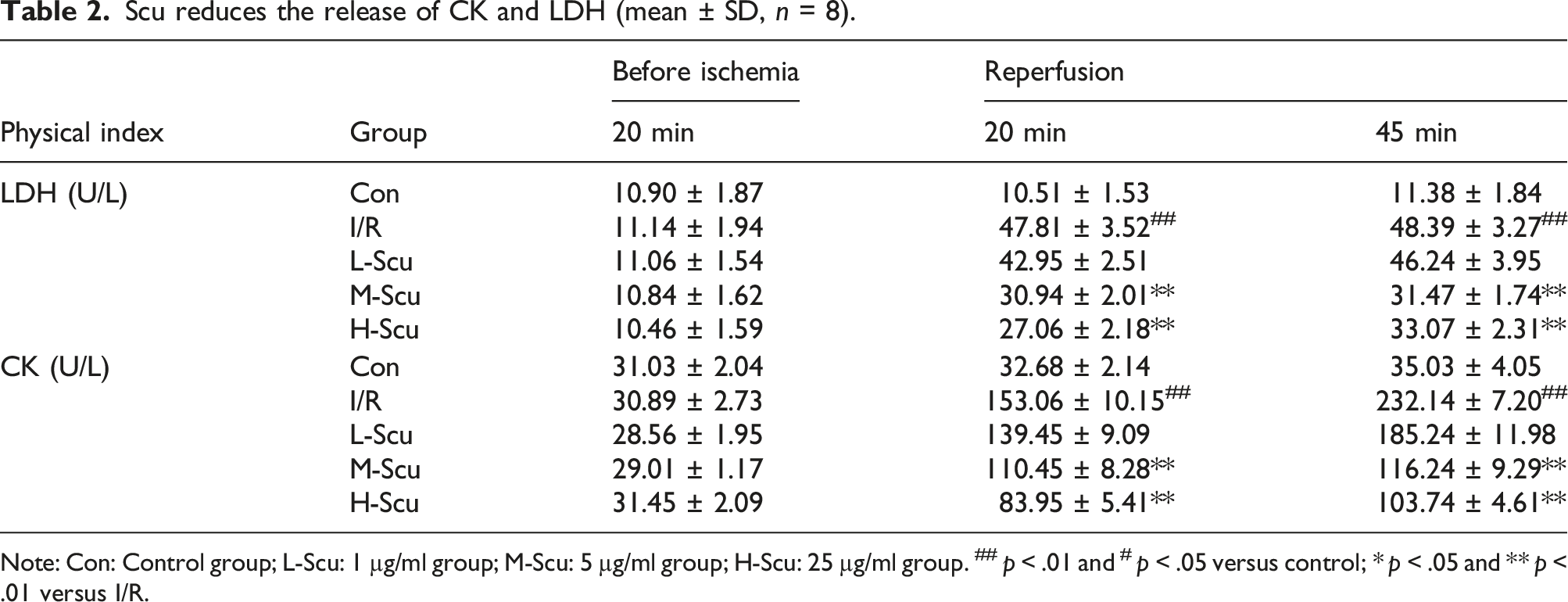
The effect of Scu on myocardial injury
As shown in Figure 1, hearts obtained from the I/R group exhibited a remarkable increase in infarct area (45.24% ± 3.56%). In contrast, Scu preconditioning relatively reduced the heart infarct size to 30.29% ± 1.54% and 22.49% ± 1.98%, respectively. Measurements of LDH and CK release were used to quantify necrotic cell death in these experiments. As shown in Table 2, I/R significantly increased the content of LDH and CK in coronary effluent, whereas pretreatment with Scu significantly decreased the content of LDH and CK in the coronary effluent. Scu alleviates heart infarction (mean ± SD, n = 8). Con: control group; L-Scu: 1 µg/ml group; M-Scu: 5 µg/ml group; H-Scu: 25 µg/ml group. ## p < .01 versus control; *p < .05 and **p < .01 versus I/R. Scu reduces the release of CK and LDH (mean ± SD, n = 8). Note: Con: Control group; L-Scu: 1 µg/ml group; M-Scu: 5 µg/ml group; H-Scu: 25 µg/ml group.
##
p < .01 and
#
p < .05 versus control; * p < .05 and ** p < .01 versus I/R.
The effect of Scu on oxidative stress in myocardial tissues
Compared to the control group, I/R significantly increased MDA, as well as SOD and the GSH/GSSG. Additionally, pretreatment with Scu significantly reduced MDA but increased SOD and the GSH/GSSG (see Figure 2). Scu alleviated oxidative stress in heart I/R (mean ± SD, n = 8). (a) SOD activity. (b) MDA content. (c) GSH/GSSG ratio. Con: control group; L-Scu: 1 µg/ml group; M-Scu: 5 µg/ml group; H-Scu: 25 µg/ml group.
##
p < .01 versus control; * p < .05 and ** p < .01 versus I/R.
The effect of Scu on inflammation
Compared to control group, I/R significantly increased TNF-α, CRP, and IL-6. Additionally, pretreatment with Scu significantly reduced the levels of these inflammatory cytokines (see Figure 3). Scu attenuated the inflammation in heart I/R (mean ± SD, n = 8). (a) TNF-α concentration. (b) IL-6 concentration. (c) CRP concentration. Con: control group; L-Scu: 1 µg/ml group; M-Scu: 5 µg/ml group; H-Scu: 25 µg/ml group.
##
p < .01 versus control; * p < .05 and ** p < .01 versus I/R.
The effect of Scu on cardiomyocyte apoptosis
As shown in Figure 4(a), I/R significantly increased cardiomyocyte apoptosis, whereas pretreatment with 5 µg/mL and 25 µg/mL Scu significantly decreased cardiomyocyte apoptosis (p < .01). The percentage of apoptotic cells is shown in Figure 4(b). Scu reduced cardiomyocyte apoptosis in I/R injury (×200). Blue nuclear staining indicates normal cardiomyocyte nuclei, and brown nuclear staining indicates apoptotic cardiomyocyte nuclei (mean ± SD, n = 8). (a) Cardiomyocyte apoptosis was quantified by using a TUNEL kit. (b) Apoptosis percentage. Con: control group; L-Scu: 1 µg/ml group; M-Scu: 5 µg/ml group; H-Scu: 25 µg/ml group.
##
p < .01 versus control; * p < .05 and ** p < .01 versus I/R.
The effect of Scu on PPARγ, Nrf2, and NF-κB
Compared to control group, NF-κB protein was significantly increased, and the Nrf2 protein was significantly decreased in the I/R group. The expression level of PPARγ decreased slightly in the I/R group, and there was no difference between the I/R group and the control group. Compared to I/R group, the expression levels of PPARγ and Nrf2 were markedly increased, and the NF-κB expression level was markedly decreased in the H-Scu group (see Figure 5). Effects of Scu on PPARγ, Nrf2, and NF-κB protein (mean ± SD, n = 3). Con: control group; H-Scu: 25 µg/ml group.
##
p < .01 versus control; * p < .05 and ** p < .01 versus I/R.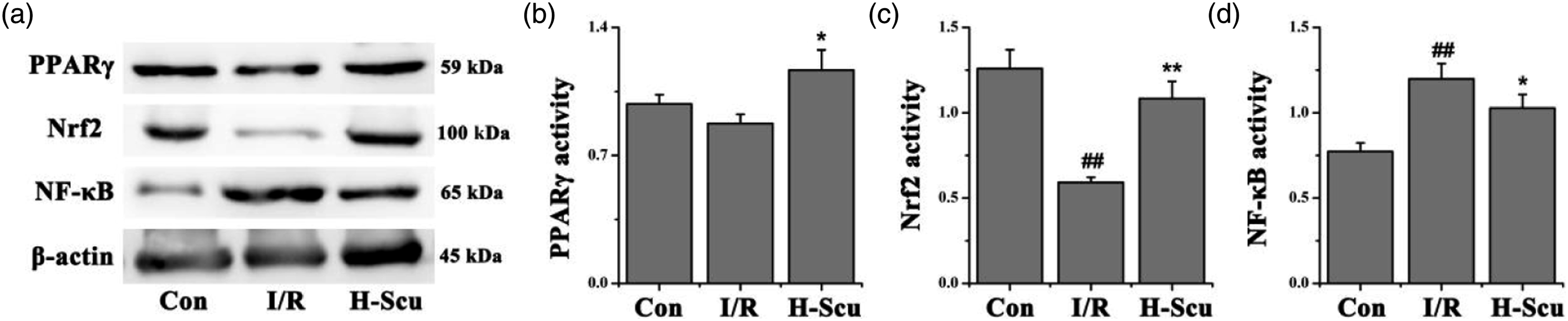
Discussion
Oxidative damage is a critical factor that causes heart I/R injury.13,14 Excessive ROS can cause oxidative damage, and some antioxidant enzymes can resist oxidative damage (e.g., SOD and GSH). 15 Cardiomyocytes will generate a large amount of ROS during the I/R process, reducing SOD activity and GSH activity, resulting in myocardial oxidative damage. 16 Flavonoids have unique antioxidant properties. 17 The present study demonstrated that the flavonoid Scu protects against heart I/R injury, enhances both the GSH/GSSG ratio and SOD activity. Therefore, one mechanism for the cardioprotection of Scu may be associated with its antioxidant effects.
Cardiac dysfunction, myocardial injury and myocardial apoptosis are the main features of cardiac I/R injury.18,19 The current study observed remarkable myocardial dysfunction, including increased the release of enzymes, disturbance of haemodynamic parameters, and induced heart infarct, as well as significant cardiomyocyte apoptosis, following heart I/R. Scu significantly improved the haemodynamic parameters, decreased enzyme release, attenuated apoptosis, and reduced heart infarct size.
Inflammation has previously been implicated in I/R injury, with roles for proinflammatory cytokines.20,21 To explore whether a relationship exists between the cardioprotective and anti-inflammatory effects of Scu, the current study assessed whether Scu can modulate the proinflammatory responses triggered by I/R injury. Although I/R injury increased proinflammatory cytokines (such as IL-6 CRP, and TNF-α) production, Scu treatment reduced the levels of these cytokines in the coronary effluent.
Many signalling pathways are involved in the development of heart I/R processes, e.g., PPARγ, Nrf2, and NF-κB.7,22,23 Studies have suggested that I/R promotes the release of a large number of inflammatory factors from cardiomyocytes by activating the PPARγ and NF-κB signalling pathways, ultimately causing severe inflammatory damage. 7 Nrf2 has been shown to attenuate heart I/R injury through a number of mechanisms, including reductions in inflammatory responses and oxidative stress. 24 Some studies have also found that Scu can exert antioxidant and anti-inflammatory effects by activating these three pathways. 22 We found that Scu significantly increased the I/R-induced decrease in PPARγ and Nrf2 protein expression and decreased the I/R-induced increase in NF-κB protein expression. Therefore, we thought that the cardioprotective effects of Scu may be related to the PPARγ, Nrf2, and NF-κB signalling pathways.
However, there are still some limitations in this study, such as no power calculation for the selected sample size. This study is only a preliminary study of the cardioprotective effect of Scu, and its mechanism of action has not been studied in depth. This study found that PPARγ, Nrf2 and NF-κB may be involved in the process of Scu protecting the heart, but no related inhibitors were used for further research, which is a limitation of this study. This study also has some flaws in the design of the experiment, and the storage of experimental data is not standardized, resulting in the loss of some original data. In future research, we will correct these errors.
Conclusion
In summary, Scu has significant cardioprotective effects against I/R injury, including decreases in myocardial enzyme release, infarct size, oxidative stress, cytokine levels, and cell apoptosis. The cardioprotective effects of Scu may be closely associated with the PPARγ, Nrf2, and NF-κB signalling pathways.
Supplemental Material
Supplemental Material - Effects of scutellarin on acute myocardial ischemia/reperfusion injury in isolated rat heart
Supplemental Material for Effects of scutellarin on acute myocardial ischemia/reperfusion injury in isolated rat heart by Daoxu Qu, Peiwen Feng, Xinjie Zhang and Mingjie Zhou in European Journal of Inflammation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Yantai Yuhuangding Hospital Scientific Research Development Fund (NO. 2021-43).
Ethics approval
Ethical approval for this study was obtained from National Standard for Laboratory Animals (APPROVAL NUMBER/ID: GB14925-2010).
Animal welfare
The present study followed Guidelines for Ethical Review of Laboratory Animal Welfare (GB/T35892-2018).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.