Abstract
Aging is the crucial co-morbidity that prevents the full cardioprotection against myocardial ischemia/reperfusion (I/R) injury. Combination therapy as a promising strategy may overcome this clinical problem. This study aimed to investigate the cardioprotective effects of Ginsenoside compound-Mc1 (GMc1) and Dendrobium Nobile Lindl (DNL) in myocardial I/R injury and explore the involvement of the TLR4/NF-κB signaling pathway in aged rats. In vivo I/R injury and myocardial infarction was established by temporary coronary ligation in 22–24 months’ old Sprague Dawley male rats. GMc1 (10 mg/kg) and DNL (80 mg/kg) were administered intraperitoneally for 4 weeks and orally for 14 days, respectively, before I/R injury. Infarct size was measured through triphenyl-tetrazolium-chloride staining. ELISA assay was conducted to quantify the levels of cardiotroponin, and myocardial content of TNF-α and glutathione. Western blotting was employed to detect the expression of TLR4/MyD88/NF-κB proteins. GMc1 and DNL significantly reduced the infarct size to a similar extent (p < 0.05) but their combined effect was greater than individual ones (p < 0.01). Combination therapy significantly restored the left ventricular end-diastolic and developed pressures at the end of reperfusion as compared with the untreated group (p < 0.01). Although the GMc1 and DNL reduced the levels of inflammatory cytokine TNF-α and increased the contents of antioxidant glutathione significantly, their individual effects on the reduction of protein expression of TLR4/MyD88/NF-κB pathway were not consistent. However, their combination could significantly reduce all parameters of this inflammatory pathway as compared to untreated I/R rats (p < 0.001). Therefore, the combined treatment with GMc1 and DNL increased the potency of each intervention in protecting the aged hearts against I/R injury. Reduction in the activity of the TLR4/MyD88/NF-κB signaling pathway and subsequent modulation of the activity of inflammatory cytokines and endogenous antioxidants play an important role in this cardioprotection.
Keywords
Introduction
Acute myocardial infarction (MI) is the main cardiovascular disease leading to death and disability worldwide. Reperfusion therapy is the essential treatment for reducing ischemic injury and succeeding infarct size in patients with MI. 1 However, in this situation, ischemia and subsequent reperfusion are two reasons for injuries inflicted on the myocardium. Myocardial ischemia-reperfusion (I/R) injury has numerous pathological mechanisms including intracellular pH alterations, intracellular calcium, and sodium overload, oxidative stress, inflammatory reactions, mitochondrial dysfunction, apoptosis, and microvascular endothelial cell injury, etc. 2 Despite achievements in cardioprotective interventions in clinical and preclinical studies, there is still no reliable approach to completely prevent the additional damages caused by these pathologies.
Moreover, the prognosis of myocardial I/R injury is poor when the patients have underlying co-morbidities like aging. Emerging data suggest that aging can impede the therapeutic effects of protective modalities on myocardial I/R injury. 3 One reason for this loss of cardioprotection is that aging-induced abnormalities and ischemic injuries share common underlying mechanisms, which can negatively influence the outcomes of cardioprotection. Among these shared mechanisms, the inflammatory response would be an important culprit to attenuate the potency of cardioprotection during aging. 4 A variety of damage-associated molecules are released from cardiac necrotic cells following myocardial I/R injury, where it has been shown to act on several recognition receptors such as Toll-like receptor-4 (TLR4) to activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB). Inhibition of TLR4 has been shown to reduce infarct size and restore cardiac function following I/R injury. 5 TLR4/NF-κB pathway serves to induce the release of several inflammatory mediators and cytokines including pro-interleukin-1β (IL-1β) and tumor necrosis factor-alpha (TNF-α) and play important roles in cell damage and relevant microcirculatory disorders.5,6 Such reactions intensify with advancing age. Moreover, the inflammatory responses continue to expand during aging, causing overproduction of free radicals and reduction of antioxidative power of the cardiomyocytes. 5 Accumulating evidence shows that natural anti-inflammatory and antioxidative agents can improve the outcomes of I/R injury.5,6 To reduce the burden of myocardial I/R injury during aging, finding the new and effective cardioprotective compound(s) while having anti-inflammatory effects is of great clinical importance.
Ginsenosides are the main constituents of ginseng extract which has been widely used in China as a traditional herbal medication. These agents have a variety of pharmacological effects against stroke, diabetes, hyperlipidemia, and cardiovascular disease.7,8 In contrast to glycosylated ginsenosides, Ginsenoside compound-Mc1 (GMc1) is a newly identified deglycosylated ginsenoside which has high bioavailability and greater ability to permeate across the cell membranes.8,9 Despite the antioxidative, anti-inflammatory, and protein kinase activating effects of GMc1, its cardioprotective effect, and the underlying mechanism remains unclear. Furthermore, dendrobium nobile Lindl. (DNL) is the alkaloid extract from a traditional Chinese herbal medicine Dendrobium, which is used to treat some inflammatory diseases. 10 Dendrobium extracts possess multiplex pharmacological features, such as neuroprotective, anti-oxidative, anti-inflammatory, and immune-modulatory functions. It has been well-documented that DNL has the potential to correct the molecular mechanisms of aging through the multi-target, multi-pathway, direct or indirect roles. 11
Recently it is believed that combinational therapies are a promising approach to protect the ischemic hearts in the presence of co-morbidities. 3 Interaction of aging with myocardial I/R injury is complex and multifactorial, so the administration of multi-targeted combination therapy can overcome the loss of cardioprotection during this co-morbidity. According to the previous reports, both GMc1 and DNL had their specific beneficial effects that, if used simultaneously, by activating different survival pathways and mediators, they can greatly correct the many pathophysiological changes caused by aging in the ischemic heart. Therefore, the cardioprotective potential of GMc1 and DNL within the context of myocardial infarction and I/R injury in aging has been investigated in the present study. Given that inflammatory responses have been shown to play an important role in myocardial I/R injury, this study hypothesized that this combination therapy may protect aged rat hearts from I/R injury by modulating the TLR4/NF-κB signaling pathway and subsequent inflammatory and oxidative mediators.
Materials and methods
Fifty Male Sprague-Dawley rats (at the age of 22–24 months) weighing between 310 ± 20 g were obtained from Shandong First Medical University of China and used in this study. All rats were maintained at 25 ± 2°C with 55% humidity on a 12-h light/12-h dark cycle. They had free access to food and water during the experiment. All animal experiments were conducted following the NIH Guide for the Care and Use of Laboratory Animals and approved by the Ethics Committee of the Shandong Provincial Hospital Affiliated to Shandong First Medical University, (Ethic Number: 202005-2801). GMc1 was obtained from Ambo Institute, Seoul, Korea. DNL was provided by the Natural Products Laboratory, Academia of China (Guiyang, Guizhou). Primary antibodies against TLR4, MyD88, NF-κB, β-actin, and the secondary antibody were purchased from Abcam (Cambridge, MA, UK). ELISA kits for TNF-α and Glutathione were obtained from MyBioscience, Inc., USA.
Experimental design
After 1 week of acclimation, the aged rats were randomly divided into 5 groups with 10 rats in each group:
Group Sham: administered normal saline by oral gavage (8 ml/kg) and intraperitoneal injection (2 ml/kg) for 4 weeks, then the MI surgery was performed and a thread was passed beneath the left anterior descending (LAD) coronary artery without LAD occlusion;
Group MI: administered normal saline as in the Sham group, then the MI surgery was performed and LAD coronary artery was ligated for 35 min followed by reperfusion for 2 h;
Group MI+GMc1: administrated GMc1 (10 mg/kg) by intraperitoneal injection for 4 weeks. 8 Then, MI surgery was performed and LAD coronary artery was ligated for 35 min followed by reperfusion for 2 h;
Group MI+DNL: administrated DNL (80 mg/kg) by oral gavage for 4 weeks. 12 Then, MI surgery was performed and LAD coronary artery was ligated for 35 min followed by reperfusion for 2 h;
Group MIR+GMc1+DNL: administrated GMc1 and DNL through their corresponding routes for 4 weeks. Then, MI surgery was performed and LAD coronary artery was ligated for 35 min followed by reperfusion for
Sample size calculation
Using PS power-sample size calculation software, sample size analysis for the primary endpoint infarct size revealed a required number of rats being five per group. The input for the sample size calculation was based on similar studies, with an expected difference in mean of infarct size of 20%, an expected difference in the standard deviation of 10%, a power of 80%, and a type I error of 0.05. Then, we considered 10 rats for each group (5 rats for infarct size detection and 5 rats for other measurements).
Surgical procedure and induction of in vivo MI and I/R injury model
The induction of MI and myocardial I/R injury model was established as previously described. In brief, all rats were anesthetized with an intraperitoneal injection of sodium pentobarbital (40 mg/kg). A heating pad was used to maintain body temperature at 37°C. Tracheotomy and endotracheal intubation were done in all rats to perform ventilation with a rodent ventilator at a tidal volume of 1.2 ml/g of rats and respiratory rate of 65/min. Then the animal’s chest was opened by a middle thoracotomy. After removal of the pericardium, myocardial I/R injury was induced by the ligation of the LAD coronary artery for 35 min through a snare loop by a 4–0 silk, and reperfusion was done by releasing the thread for 2 h. To ligate the left anterior descending artery, the heart was exposed via a left lateral thoracotomy at the fourth intercostal space. After successful LAD occlusion, the area distal to the ligation site was changed to a visible blanched area.
Myocardial function measurement
Before the induction of ischemia, the right carotid artery of rats was cannulated with a Millar pressure micro-catheter attached to a bridge pressure amplifier (AD-Instruments, Australia). After fixation in the place, the catheter was then advanced to the left ventricle. Left ventricle end-diastolic pressure (LVEDP), and left ventricle developed pressure (LVDP) were recorded during the experiment.
Myocardial infarct size measurement
At the end of reperfusion, the LAD was ligated again and 0.25% Evans blue was injected via the femoral vein and then the hearts were isolated, weighed and stored at −20°C for 20 min and cut into five to six pieces from the base (below the ligature) toward the apex. Then, the slices were incubated in triphenyl tetrazolium chloride (TTC) staining for 15 min at 37°C. TTC staining causes healthy non-infarcted areas to remain red while the necrotic areas turn pale. An Image J software (1.46r version, NIH, USA) was used to measure the left ventricular volumes, areas at risk, and infarct sizes. Infarct sizes were calculated as (area at risk/ventricular volume) ×100.
Detection of myocardial enzyme levels in serum
During the reperfusion times, the peripheral blood was collected, and then the serum was separated to evaluate heart muscle damage indicator cardiotroponin-I (cTnI), using an ELISA kit (Thermo Fisher Scientific, USA), according to the provider’s instructions. The cTnI levels were reported U/L.
Detection of cytokine TNF-α and antioxidant glutathione in heart tissue
The changes of pro-inflammatory cytokines tumor necrosis factor-alpha (TNF-α) and glutathione in the left ventricular samples of the hearts were assessed via the specific ELISA kits, according to the instructions (MyBioscience, Inc., USA). The relative absorbance of parameters was read at 450 nm wavelength using a microplate ELISA reader and the final concentration was normalized to the protein content of each sample. The levels were reported as pg/mg of protein.
Western blotting for proteins expression
The expression of TLR4, MyD88, and NF-kB proteins was detected by the Western blotting technique. In brief, SDS-gels electrophoresed proteins of the samples (30 µg protein from left ventricular tissues) were transferred to a PVDF membrane and the membrane was blocked in 5% skim milk containing 0.1% Tween-20 for 1 h. Then, the blocked membranes were incubated overnight separately with a primary antibody for each target protein or β-actin (1:500, Cell Signaling) at 4°C. After washing steps with Tris buffer saline, the HRP-conjugated secondary antibody (1:3000, Cell Signaling) was added for an hour. Then, the membrane was washed and incubated with the enhanced chemiluminescence (Amersham) reagents in the darkroom and exposed to an X-ray film. The protein bands were visualized and the related intensities were quantified using Image J software (1.46r version, NIH, USA) and normalized to the intensity of β-actin in each sample.
Statistical analyses
All data were shown as mean ± standard deviation (SD). One-way ANOVA and Tukey post hoc tests were used to compare the significance of differences among experimental groups. If p < 0.05, the data were statistically significant.
Data availability
The data associated with the paper are available from the corresponding author on a reasonable request.
Results
Effects of treatments on myocardial infarction
There was no significant difference in the volume of areas at risk in I/R groups, indicating the similar extent of injury imposed to all groups (Figure 1(a)). The results from TTC staining revealed that the infarct area in the MI group was increased considerably as compared to that in the sham group, confirming the successful establishment of the rat model of I/R injury (p < 0.01) (Figure 1(b)). Individual treatments with GMc1 or DNL significantly reversed the infarct size in rats subjected to I/R injury in a similar manner (p < 0.05). Besides, the combination of GMc1 and DNL reduced the infarct size more potently as compared with that of the MI group (p < 0.01).

(a, b) Effects of treatments on myocardial infarction in I/R rats (n = 5 in each group): (a) quantitative analysis of areas at risk (AAR) proportional to the volumes of the left ventricle (LV) in experimental groups and (b) quantitative analysis of infarct sizes proportional to the AAR in experimental groups.
Effects of treatments on myocardial function
In this study, the LVEDP and LVDP were recorded to evaluate the changes in myocardial function following treatment. The data of these hemodynamic parameters before ischemia and at the end of the reperfusion phase is seen in Table 1. Baseline values of LVEDP and LVDP were similar among groups, as there were no significant differences in these parameters before ischemia. However, induction of I/R injury significantly increased the LVEDP and significantly reduced the LVDP in the MI group as compared with those of the sham group (p < 0.01). None of the individual treatments could prevent I/R injury-induced increase in LVEDP. Moreover, only GMc1 individually restored the LVDP to some extent in comparison to the MI group (p < 0.05). On the other hand, the combination of GMc1 and DNL significantly reduced the LVEDP and improved the LVDP in the reperfusion phase as compared to the MI group (p < 0.01, table 1).
Effects of treatments on myocardial function in I/R rats.

LVEDP: left ventricular end-diastolic pressure; LVDP: left ventricular developed pressure (n = 5 in each group).
p < 0.01 compared with the Sham group; #p < 0.05 and ##p < 0.01 compared with the MI group.
Effects of treatments on myocardial enzyme marker, cTnI
The enzyme cTnI is the main indicator of myocardial injury. The levels of cTnI in the serum of the MI group was significantly increased as compared to that of the sham group (p < 0.01). Following single treatments of GMc1 and DNL in I/R rats, the levels of this enzyme were reduced but this effect was not statistically significant. However, combination treatment significantly reduced the I/R–induced increase in the levels of cTnI as compared to the MI group (p < 0.01) (Figure 2).
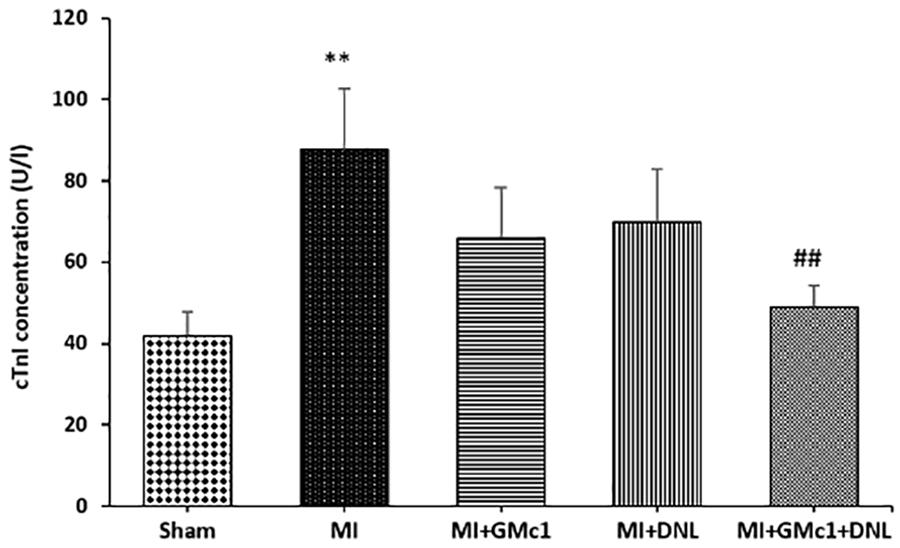
Effects of treatments on myocardial enzyme marker cTnI in I/R rats (n = 5 in each group).
Effects of treatments on the protein expression of TLR4, MyD88, and NF-κB
As the TLR4/NF-κB-mediated pro-inflammatory signaling pathway plays crucial roles in the outcomes of myocardial I/R injury, the involvement of this mechanism underlying the protective role of combination therapy in I/R injury during aging was then explored. Western blot analysis demonstrated that TLR4 (p < 0.05), its adaptor protein MyD88, and NF-κB (p < 0.01) were significantly increased in the MI group compared to the Sham group (Figure 3(a) to (c)). Notably, administration of GMc1 could reverse the expression of TLR4 (p < 0.05), and NF-κB (p < 0.01) with no effect on MyD88 expression, as compared with those of the MI group. Besides, alone administration of DLN reversed only the expression of NF-κB (p < 0.05) and had no effect on TLR4 and MyD88 expression levels. However, combining the treatments (GMc1 plus DNL) significantly and more potently downregulated the expression of all signaling mediators TLR4, MyD88, and NF-κB in comparison to the MI group (p < 0.01 to 0.001).

(a–c) Effects of treatments on the TLR4/MyD88/NF-κB signaling pathway as indicated by Western blot results (n = 5 in each group).
Effects of treatments on myocardial inflammatory cytokine TNF-α
Inflammatory cytokines are the key mediators of myocardial I/R injury as well as the aging process. As shown in Figure 4, Induction of MI and I/R injury in aged rats significantly increased the level of TNF-α in comparison to the Sham group (p < 0.001). Conversely, pretreatment with GMc1 and DLN individually (p < 0.01) or in combination with each other (p < 0.001) significantly reduced I/R-induced increase in the production of inflammatory TNF-α as compared to the MI group (Figure 4).

Effects of treatments on myocardial inflammatory cytokine TNF-α level in I/R rats (n = 5 in each group).
Effects of treatments on the myocardial glutathione level
Finally, we investigated the changes in the levels of glutathione in the myocardium to clarify whether the alteration of the NF-κB pathway by the treatments can modulate the myocardial oxidative stress. In comparison to the Sham group, I/R injury (MI group) significantly down-regulated the levels of glutathione as a main endogenous antioxidant in the myocardium (p < 0.001; Figure 5). Nevertheless, individual administration of GMc1 and DLN in their corresponding groups (p < 0.01) as well as their combination significantly increased the levels of this antioxidative marker as compared to the MI group (Figure 5). Again, the effect of combination therapy on this marker was also greater than those of individual treatments (p < 0.001).

Effects of treatments on glutathione levels in I/R rats (n = 5 in each group).
Discussion
In this study, the effect of herbal medicine-based combination therapy on cardioprotection during aging and the role of the TLR4/NF-κB inflammatory pathway was investigated in an in vivo I/R model in rats. The results showed that although DNL and GMc1 had some positive effects on myocardial infarction and underlying mechanisms when administered separately, these effects were not consistent in all parameters. However, their combination significantly reduced the infarct size and cTnI in aged I/R rats, restored the myocardial function as indicated by reduced LVEDP and improved LVDP, and weakened the activity of the TLR4/MyD88/NF-κB inflammatory signaling pathway compared to the untreated aged I/R rats. Also, following these effects, the level of inflammatory cytokine TNF-α decreased and the level of myocardial endogenous antioxidant glutathione increased after combination therapy.
Multiple intracellular changes during aging including increased inflammatory and oxidative responses, decreased mitochondrial function, and diminished protein kinase activity predispose cardiomyocytes to damages induced by various stressors. 13 Therefore, the occurrence of cardiac I/R damage during aging can have more severe consequences, and the aforementioned negative changes prevent the full effectiveness of cardioprotective interventions. 14 In this regard, it has been documented that the protective effects of interventions that significantly reduce the infarct size during no risk factor conditions are reduced or eliminated in the presence of aging as one of the main comorbidities. 14 As a result, it is concluded that in the case of comorbidities, we must somehow increase the power of individual interventions. In the present study, we used a combination therapy strategy to try to restore the therapeutic weakness of the individual interventions by increasing their potency. The combined administration of GMc1 and DNL in aged rats had a stronger effect on reducing infarct size and enzymatic index cTnI than their single administration. Moreover, this cardioprotective effect in combination therapy was associated with a significant decrease in the TLR4/NF-κB pathway activity.
In ischemic conditions, TLR4 mediates the inducible inflammatory response in cardiomyocytes and activates the NF-κB by activating its adapter protein MyD88. 15 In the present study, GMc1 alone was able to reduce TLR4 and MyD88 expression levels compared to the untreated MI group, but DNL did not have a significant effect on these parameters. On the other hand, both compounds alone had almost the same significant effects on NF-κB expression. If these two compounds are combined, the expression levels of all three proteins of the signaling pathway are reduced. These findings suggest that the reduction in NF-κB expression following the alone administration of interventions is not necessarily achieved by the reduction in TLR4 pathway activity, and there is probably another pathway to reduce NF-κB activity in this regard. However, if both therapeutics are used simultaneously in a combination manner, changes in NF-κB expression are associated with similar changes in the expression of its upstream pathway, TLR4/MyD88. However, this association does not rule out the existence of other pathways such as PI3K/Akt and JAK/STAT leading to the reduction in NF-κB activity. 8 Following the increase in intracellular NF-κB activity, the expression and production of inflammatory cytokines will be augmented, triggering the cellular inflammatory reactions. 16 Inter-and intra-cellular swelling, infiltration of inflammatory cells, and necrosis are the outcomes of these reactions, which eventually lead to the exacerbation of I/R pathophysiology, apoptosis, and cell death.6,17
Studies have also shown that, besides exacerbating inflammatory responses, NF-κB leads to increased oxidative stress and thereby reduction in the levels of tissue endogenous antioxidants. 18 To test this hypothesis, we assessed glutathione levels as the strongest endogenous antioxidant, along with TNF-α, and observed a significant decrease in the level of this antioxidant following MI injury compared to the sham group. The administration of both interventions alone or in combination was able to increase the intracellular level of this antioxidant, and here, the combination therapy effect was still stronger than the individual effects. This finding also confirms that in old age, in addition to inflammatory responses, oxidative stress can also be involved in reducing the response of cardioprotective interventions, and therefore targeting main players in this phenomenon can restore the loss of cardioprotection by interventions in the aged heart. 19 In this regard, the endoplasmic reticulum stress and mitochondrial oxidative burden and the subsequent production of apoptotic factors are important mediators 20 which the extent of their involvement in the effect of combined treatment of DNL and GMc1 in the protection of the aged heart requires additional studies.
Conclusion and limitation
To sum up, the antioxidative and anti-inflammatory combination therapy with GMc1 and DNL increased the potency of each other in protecting the aged hearts against I/R injury and reduced the myocardial infarction, and improved myocardial function. Reduction in the activity of the pro-inflammatory TLR4/MyD88/NF-κB signaling pathway and subsequent decrease in the production of inflammatory cytokine and an increase in the production of myocardial endogenous antioxidants play an important role in this cardioprotection. Due to the inability to maintain more rats for longer periods, we were unable to investigate the long-term effects of this combination therapy, and verifying the long-term effects of this combination would be clinically significant. The contribution of mitochondrial mechanisms to cardioprotection resulting from this treatment in chronic conditions is suggested for future studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by a grant from Shandong Provincial Hospital affiliated to Shandong First Medical University, China.
Ethical approval
Ethical approval for this study was obtained from Ethics Committee of the Shandong Provincial Hospital Affiliated to Shandong First Medical University, (Ethic Number: 202005-2801).
Animal welfare
The present study followed international guidelines for humane animal treatment and complied with relevant legislation.
