Abstract
Ischemic stroke is a leading cause of human death. The injury that is induced by oxygen-glucose deprivation/reperfusion in stroke remains unsolved. This study first investigated the effects of oroxylin A on oxygen-glucose deprivation/reperfusion-induced PC12 cells. This was performed by dividing the cells into a control group, an oxygen-glucose deprivation and reperfusion (OGD/R) group, a solvent control group, and experimental groups treated with different concentrations of oroxylin A. Cell viability was evaluated by Cell Counting Kit-8 assay. Relevant indicators of oxidant stress were detected by using the appropriate kits. Western blot was applied to detect the expressions of inflammatory cytokine and proteins of the signaling pathway. Oroxylin A pretreatment exerted anti-oxidative, anti-apoptotic, and anti-inflammatory effects in oxygen-glucose deprivation/reperfusion-induced PC12 cells, thus indicating it as a new avenue for stroke treatment and providing references for future studies.
Keywords
Ischemic stroke is a primary cause of death worldwide. 1 It is caused by blockage of the blood vessels in either the neck or brain. 2 Blood flow recovery via a thrombolytic drug is the conventional treatment, but the therapeutic efficacy is not ideal and is further restricted by a short therapeutic window and strict inclusion criteria. 3 In addition, the recovery situation of the patients is dependent on the ischemia duration, brain location, lesion size, and ischemia-evoked endogenous protection. It remains a challenge to improve therapeutic procedures and life quality of the patients with stroke. To find effective therapeutic targets, many studies have been conducted into the possible mechanism, as well as the discovery of the effective drugs in stroke. 4 -7
The mechanism for ischemic stroke is complex. Insufficient delivery of oxygen and glucose is unable to maintain homeostasis in ischemic stroke and leads to cell death by multiple processes including oxidative stress, inflammation, excitotoxicity, ionic imbalance, and apoptosis. 8 It is reported that cerebral oxygenation after perinatal arterial ischemic stroke is relevant to neurodevelopment. 9 Oxygen or sugar imbalances are vital factors for cerebral ischemic injury and new agents which improve oxygen supply/consumption balance are able to alleviate the injury. 10
Oxidative stress is the predominant cause of injury after ischemic stroke, which can be induced by insufficient oxygen and glucose. 11 ROS immediately generates after acute ischemic stroke. During the thrombolytic drug treatment for ischemic stroke, the rapid blood flow recovery elevates the tissue oxygenation level leading to a burst of ROS generation once more. Excessive ROS results in injuries of all cell components such as proteins, lipids, and DNA. 12,13 Inflammation initiated by stagnant blood flow is another pivotal event causing relevant damage in ischemic stroke. Increasing studies have focused on the inflammatory and oxidative stress responses to cerebral stroke. Currently, inflammatory research into ischemic stroke has mainly concentrated on inflammatory markers, relevant inflammatory pathways, and anti-inflammatory compounds. 14 -18 The development of anti-inflammatory and antioxidation agents is a promising avenue in the treatment of ischemic stroke.
Oroxylin A, a flavonoid extracted from scutellariae radix, is of high value with many pharmacological effects, including anti-inflammatory, antioxidant, neuroprotective, anti-coagulation, and anti-cancer. 19 Oroxylin A is low toxic. Orally 80 mg/kg oroxylin A has no effects on body weight, heart, liver, spleen, and kidney in mice. 20 Oroxylin A after intravenous administration was reported to conjugate rapidly and extensively with glucuronicacid in rat plasma and oroxylin A-7-O-glucuronide was the major metabolite of OA. 21
As reported, oroxylin A alleviates apoptosis and oxidative stress in PC12 cells induced by H2O2. 22 Oroxylin A has a protective effect on transient cerebral hypoperfusion-induced memory impairment. 23 The compound is also reported to suppress the inflammatory response induced by lipopolysaccharide. 24 As inflammatory and oxidative stress are the main causes of ischemia reperfusion injury in ischemic stroke, oroxylin A with anti-inflammatory and antioxidant properties is a promising agent. The effects of oroxylin A on PC12 cells induced by oxygen-glucose deprivation/reperfusion are reported in this study for the first time.
Sonic hedgehog is widely expressed and plays a pivotal role in brain injury. It has been reported that sonic hedgehog is upregulated in multiple cell types after stroke and the function of the subacute phase is improved via sonic hedgehog agonist treatment. 25 The sonic hedgehog signal pathway as a regulator presents a vital function in self-renewal and functional recovery after brain injury, especially for ischemia. 25 -27
Based on the promising effects of oroxylin A, including anti-inflammatory, antioxidant, as well as neuroprotective, in this study, we first investigated the possible effects of oroxylin A on oxygen-glucose deprivation/reperfusion-induced PC12 cells, which are shown as the cell model of cerebral ischemia reperfusion, and also explored the possible relationship between oroxylin A and the sonic hedgehog signal pathway.
Cell viability was improved by oroxylin A pretreatment in OGD/R-induced PC12 cells. As shown in Figure 1, consistent with the previous study, cell viability in the OGD/R group was higher than that in the control group, confirming the successful establishment of OGD/R-induced PC12 cells. 28 The results for the solvent control and control groups were similar indicating that the solvent has no significant effect on the experiment results. Cell viability was increased with the concentration of oroxylin A. The groups that were treated with oroxylin A at different concentrations for 12 hours were significantly different from those treated for 6 hours. The following experiments were performed with oroxylin A treated for 12 hours. Cell viability was markedly higher as compared with the solvent control group, indicating that oroxylin A pretreatment before OGD/R improved cell viability significantly.
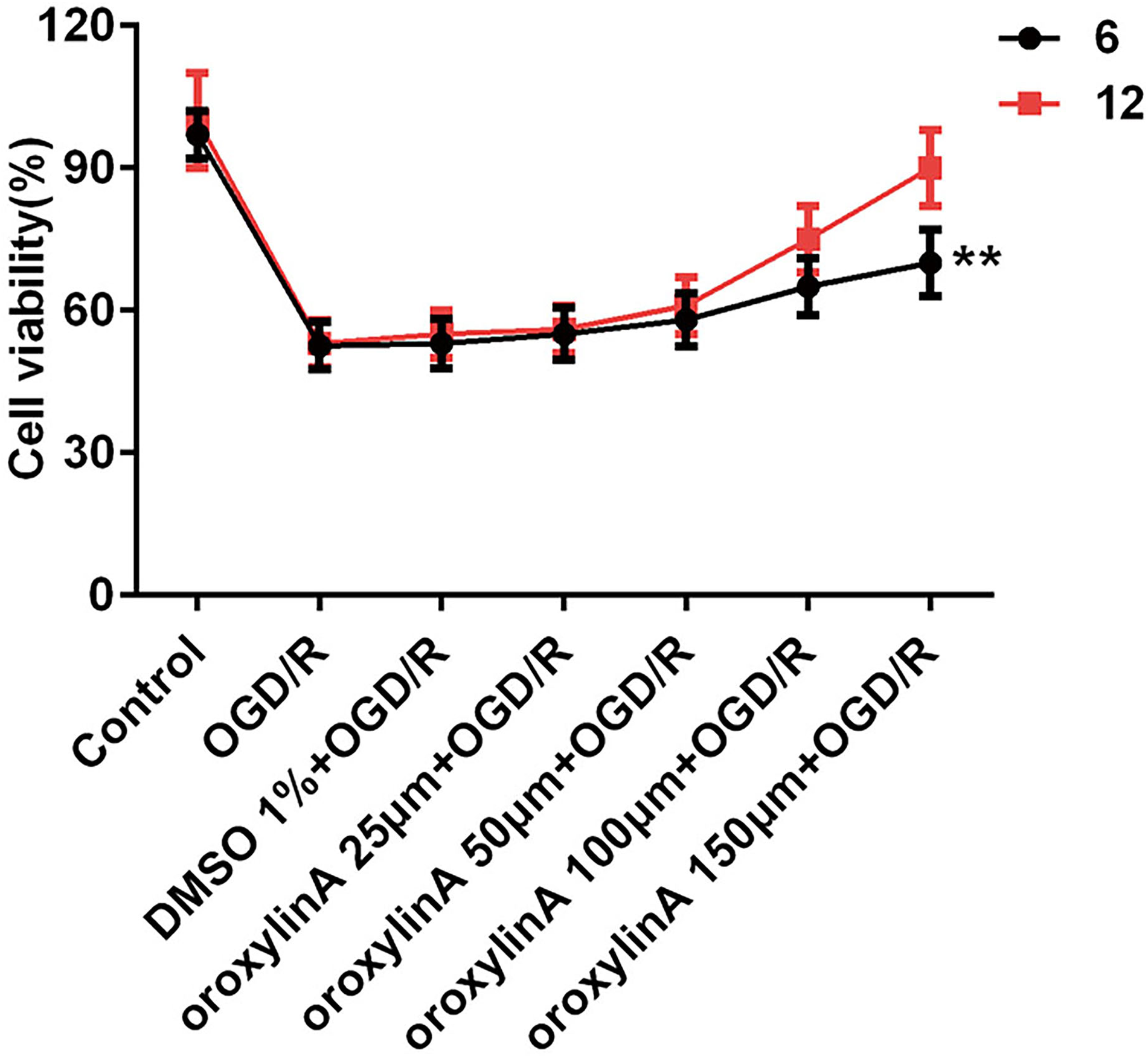
Cell viability in the control, solvent, and experimental groups treated with oroxylin A for 6 and 12 hours, respectively (** P < 0.01).
Oroxylin A pretreatment reduced the level of ROS in OGD/R-induced PC12 cells. ROS is a vital indicator in oxidative stress, as reported by previous study, and was evaluated herein. 29 As shown in Figure 2, ROS level in the control group was the lowest among all the groups (Figure 2). On the contrary, ROS content was higher in the OGD and solvent groups than that in any other group, suggesting that insufficient oxygen-glucose enhanced the ROS release, which is consistent with the previous studies. 30,31 ROS level was reduced with the increase in concentration of oroxylin A, indicating that oroxylin A pretreatment attenuated ROS generation in OGD/R-induced PC12 cells.

ROS levels in the different groups (# P < 0.05, ### P < 0.001 vs DMSO 1% + OGD/R; ***P < 0.001 vs control).
Oroxylin A pretreatment reversed the levels of MDA, SOD, and LDH induced by OGD/R in PC12 cells. MDA, SOD, and LDH as indicators of oxidant stress in OGD/R-induced PC12 cells were evaluated as well. As seen in Figure 3, the levels of MDA and LDH were higher in the OGD/R group as compared with the control group, suggesting that the release of MDA and LDH was enhanced by OGD/R, which was consistent with the previous report. 28 The levels of MDA and LDH were reduced in the oroxylin A pretreatment groups with the concentration of oroxylin A increasing. SOD level was reduced in the OGD/R group in comparison with the control group; the release of SOD was blocked by OGD/R, which was in accordance with the previous study, 28 whereas the release of SOD was reversed by oroxylin A pretreatment. As shown in Figure 3, SOD level in the oroxylin A pretreatment group was enhanced at high concentration of oroxylin A. All the results demonstrated that oroxylin A pretreatment attenuated the oxidant stress induced by OGD/R in PC12 cells.

The levels of MDA, SOD, and LDH in the different groups (# P < 0.05, ### P < 0.001 or ## P < 0.01 vs DMSO 1% + OGD/R; ***P < 0.001 vs control).
Oroxylin A pretreatment reduced the expressions of TNFα, MCP-1, and IL-1β in OGD/R-induced PC12 cells. TNFα, MCP-1, and IL-1β as indicators of inflammation were evaluated by western blot. As seen from Figure 4, consistent with the previous study, the levels of TNFα, MCP-1, and IL-1β were increased in the OGD/R groups in comparison with the control group, indicating that OGD/R enhanced the releases of TNFα, MCP-1, and IL-1β. 32 The expressions of TNFα, MCP-1, and IL-1β in the groups pretreated with oroxylin A were decreased significantly at high concentration of oroxylin A, confirming that oroxylin A pretreatment alleviated the expressions of TNFα, MCP-1, and IL-1β in OGD/R-induced PC12 cells.

Expressions of TNFα, MCP-1, and IL-1β in the different groups (### P < 0.001 vs DMSO 1% + OGD/R; ***P < 0.001 vs control).
Oroxylin A pretreatment activated the hedgehog signal pathway. The relevant proteins in the hedgehog signal pathway, including Shh, p-smo, and Gli2, were evaluated herein. As shown in Figure 5, the levels of Shh, p-smo, and Gli2 in the control group were higher than those in any other group, while the expressions of the relevant proteins in the OGD/R and OGD/R + solvent control groups were lower than in all the remaining groups. In the group pretreated with oroxylin A, the levels of Shh, p-smo, and Gli2 were increased with the concentration of oroxylin A pretreatment, demonstrating that the hedgehog signal pathway was activated by oroxylin A pretreatment in a dose-dependent manner.

Expressions of Shh, p-smo, and Gli2 in the different groups (# P < 0.05, ### P < 0.001 and ## P < 0.01 vs DMSO 1% + OGD/R; ***P < 0.001 vs control).
Stroke is the leading cause of long-term disability and the second cause of death worldwide. 33 Increasing studies have proved that oxidant stress and inflammation play pivotal roles in stroke. 34 -36 To diminish the injury induced by oxidant stress and inflammation in stroke is a promising strategy for therapy. As ischemic stroke is accompanied by insufficient oxygen-glucose, PC12 cells induced by oxygen-glucose deprivation/reperfusion, which has frequently served as the cell model of cerebral ischemia reperfusion, was used as the cell model in this study. 37 -39
Oroxylin A, a flavonoid, possesses low toxicity in vivo and in vitro and is a promising compound. It possesses many functions, including anti-inflammation, neuroprotective, and oxidation resistance. The effect of oroxylin A on oxygen-glucose deprivation/reperfusion-induced PC12 cells was investigated herein. As is known, there are two stages to control injury from ischemic stroke. The first stage is the ischemic attack, and the second is the reperfusion injury from the excessive ROS in the blood. Therefore, oroxylin A was administrated by pretreatment before OGD/R in this study.
It has been reported that OGD/R resulted in cell apoptosis. 39 Consistent with that study, our data showed that cell viability was decreased by OGD/R compared with the control group, whilst, cell apoptosis decreased by OGD/R was reversed by oroxylin A pretreatment in a dose-dependent manner, which confirmed that oroxylin A protected the cells from the injury induced by insufficient oxygen-glucose.
Oroxylin A presented a suppression effect on H2O2-induced oxidative stress in PC12 cells. 40 In the current study, the levels of indicators of oxidative stress, including ROS, MDA, SOD, and LDH induced by OGD/R, were reversed by oroxylin A pretreatment in a dose-dependent manner, confirming that oroxylin A pretreatment exerted protective effects on OGD-induced PC12 cells via attenuation of the oxidant stress.
Accumulative evidence has shown that oroxylin A has anti-inflammatory effects. Oroxylin was reported to inhibit pro-inflammatory cytokines in microglial BV-2 cells and downregulate expressions of inflammatory genes. 41,42 In the current research, levels of TNFα, MCP-1, and IL-1β were upregulated by OGD/R. The expressions of the above-described inflammatory cytokines were reduced, evidently by oroxylin A pretreatment at a high dose. All the results discussed above showed that oroxylin A, with the effects of oxidation resistance and anti-inflammation, was a promising agent for alleviating the injury from OGD/R.
The possible signal pathway mechanism was also explored herein. The hedgehog signal pathway upregulated in multiple cell types was reported to have an effect on the outcome of stroke. 25 Sonic hedgehog with anti-oxidative, anti-apoptotic, and pro-angiogenic effects is reported to improve the function after focal cerebral ischemia. 43 Therefore, we speculated that the hedgehog signal pathway is possibly activated by oroxylin A. The relevant proteins of the hedgehog signal pathway were investigated herein. The levels of shh, p-smo, and Gli2 were upregulated in the oroxylin A pretreatment group compared with the OGD and solvent control groups. The results confirmed our speculation that the hedgehog signal pathway is activated by oroxylin A.
All the results in this study demonstrate that oroxylin A, with promising anti-oxidative, anti-apoptotic, and anti-inflammatory effects, alleviated the injury from oxygen-glucose insufficiency by activating the hedgehog signal pathway (Figure 6). The findings in this study indicate a new, promising therapeutic avenue for treating ischemia stroke, but the effects of oroxylin A on the injury caused from oxygen-glucose insufficiency in vivo should be further investigated in vivo.

Illustrations of this article. The red arrows indicate the effect of OGD/R. The blue arrows indicate the effects of oroxylin A. The “ ” indicates the inhibition effect (Ptch was the receptor of shh).
” indicates the inhibition effect (Ptch was the receptor of shh).
Experimental
Cell Culture and Treatment
PC12 cells, purchased from American Type Culture Collection (ATCC), were cultured in Dulbecco’s modified Eagle medium (DMEM) containing penicillin (100 U/mL), streptomycin (100 µg/mL), 5% FBS, and 10% horse serum at a density of 1 × 104 cells/well at 37℃. The cells were cultured in 96-well culture plates in a humidified atmosphere of 95% air and 5% CO2 and were passaged every 3 days. Oroxylin A (Glpbio) was dissolved in 1% DMSO. The cells were treated with oroxylin A at concentrations of 25, 50, 100, and 150 µm for 6 and 12 hours, respectively, before oxygen-glucose deprivation/reperfusion.
Oxygen-Glucose Deprivation and Reperfusion
The cells, after treatment as described above, were exposed to an atmosphere of 95% (v/v) N2 and 5% (v/v) CO2 and cultured in a glucose-free Earle’s balanced salt solution for 2 hours. The cells were then cultured in a normal medium in an incubator with 5% CO2 and 95% air for 12 hours. The cells without oroxylin A treatment were treated as described above and served as the OGD/R group. The cells without OGD/R and oroxylin A treatment were taken as the control group. The cells treated with 1% DMSO were the solvent control group.
Cell Viability Assay
The cells (1 × 104) were cultured as describe above and were pretreated with oroxylin A at concentrations of 25, 50, 100, and 150 µm for 6 and 12 hours, respectively. Then the cells were washed with PBS, 3 times. Oxygen-glucose deprivation and reperfusion were performed as described above. Cell viability in the different groups was detected by using Cell Counting Kit-8 (CCK8). Each well of the cells in different groups was incubated with 10 µL CCK-8 kit at 37 ℃ in an incubator with 5% CO2 for 2 hours. The absorbance at 450 nm was obtained by using a microplate reader (RNE90002, REAGEN).
ROS Detection
ROS was detected using a ROS Activity Assay Kit (Abnova, China) according to the manufacturer’s instruction. Briefly, the cells (1 × 104) were cultured as described above and were pretreated with oroxylin A at concentrations of 25, 50, 100, and 150 µm for 6 and 12 hours, respectively. Then the OGD was performed, as described above. The cells of the different groups were incubated with the redox‐sensitive fluoroprobe 2′,7′‐dichlorofluorescein diacetate (carboxy‐H2DCFDA; Invitrogen) under dark conditions at 37℃ for 30 minutes. The ROS level was determined by a FACSCalibur™ flow cytometer (BD Biosciences) at an excitation wavelength of 490 nm and an emission wavelength of 525 nm.
Measurement of MDA, LDH, GSH-Px, and SOD Levels
MDA, as a naturally occurring product of lipid peroxidation and one of the oxidative stress parameters, was detected using an MDA assay kit. LDH, GSH-Px, and SOD served as indicators of oxidative stress and were determined by the corresponding assay kit according to the instructions of the manufacturer. The cells were pretreated with oroxylin A at concentrations of 25, 50, 100, and 150 µm for 6 and 12 hours, respectively. Then OGD was performed as described above before the cells of the mentioned group were lysed and centrifuged. The supernatants of the corresponding group obtained were removed to measure the levels of MDA, LDH, GSH-Px, and SOD according to the instructions of manufacturers.
Western Blot Analysis
Sonic hedgehog (Shh), p-smo, Gli2, and smo as the relevant proteins of the sonic hedgehog signal pathway were evaluated by western blot. The cells of the different groups were lysed to acquire the total proteins for detection. The target proteins, including Shh, p-smo, Gli2, and smo, in the different groups were separated on a polyacrylamide gel and then transferred electrophoretically to a polyvinylidene difluoride membrane. To the Tris‐buffered saline-Tween-20 solution 5% fat-free milk was added to block the membranes. Then they were reacted with the primary anti-sonic hedgehog antibody (1:2000 dilution), anti-p-smo antibody (1:2000 dilution), anti-Gli2 antibody (1 µg/mL), and anti-smo antibody (1:2000 dilution). The amounts of primary antibodies were based on the polyvinylidene difluoride membrane area (0.1 mL/m2) and secondary antibody overnight in succession. The anti-GADPH monoclonal antibody served as the internal control. The images were obtained by ImageQuant_LAS500 (GE Healthcare).
Statistical Analysis
SPSS software 10.0 and GraphPad Prism 5.1 were applied to analyze the data. All are presented as mean ± SD; the significance was evaluated by either one-way or two-way analysis of variance test.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
