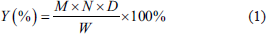Abstract
Background
Anemarrhenae asphodeloides polysaccharides (RAP) have demonstrated notable antioxidant activity, which is believed to be closely related to their monosaccharide composition and extraction conditions. However, systematic optimization of the RAP extraction process and a detailed understanding of its structure-activity relationship remain insufficiently explored.
Objectives
This study aimed to optimize the extraction process of RAP using the Box–Behnken design (BBD), analyze the monosaccharide composition of RAP from different origins using a PMP-UPLC-PDA method, and investigate the correlation between its antioxidant activity and monosaccharide constituents.
Materials and Methods
Extraction parameters including ethanol concentration, material-to-liquid ratio, extraction time, and extraction frequency were optimized using BBD based on RAP yield. The monosaccharide composition of six RAP samples from different regions was determined using 1-phenyl-3-methyl-5-pyrazolone (PMP) pre-column derivatization combined with ultra-performance liquid chromatography and photodiode array detection (UPLC-PDA). Antioxidant activities were evaluated via DPPH, hydroxyl, and superoxide anion radical scavenging assays. Correlation between monosaccharide composition and antioxidant capacity was analyzed using Pearson correlation and multivariate statistics.
Results
The optimal extraction conditions were identified as 77% ethanol concentration, a material-to-liquid ratio of 1:10 g/mL, extraction time of 124 minutes, and four extraction cycles, yielding RAP at 8.56% ± 2.74%. Antioxidant assays showed that higher RAP levels led to stronger free radical scavenging effects. Among all samples, the RAP from Yixian (a geo-authentic producing area) exhibited the highest antioxidant activity. Significant negative correlations were found between IC50 values and the proportions of Xyl, Fuc, Man, GalA, Ara, and GlcA, suggesting their contribution to antioxidant activity.
Conclusion
This study systematically optimized the RAP extraction process and revealed significant correlations between its monosaccharide composition and antioxidant activity. These findings provide new insights into the structure-function relationship and material basis of the antioxidant pharmacodynamics of A. asphodeloides.
Introduction
Anemarrhenae asphodeloides, first recorded in Shennong’s Classic of Materia Medica, is the dried rhizome of A. asphodeloides (Liliaceae) and is known for clearing heat-fire and nourishing Yin to moisten dryness (Bai et al., 2021). An increasing body of research in recent years has highlighted the remarkable effects of A. asphodeloides against pathogenic microbes, oxidative stress, and aging (Chen, Li, et al., 2023; Chen et al., 2024; Wang et al., 2019; Xie et al., 2023).
As a foundational component contributing to the medicinal properties of A. asphodeloides, polysaccharides exhibit notable effects on immune regulation, hypoglycemia, and anti-oxidation, with no observed severe side effects (Cao et al., 2021; Chae et al., 2011; Takahashi et al., 1985; Zhang et al., 2020). The complexity of polysaccharide structures presents significant challenges for researchers in terms of structural determination and synthesis. Monosaccharides, as essential components of polysaccharides, play a crucial role in determining key structural parameters, including molecular weight, constituent sugar profiles, and glycosidic bond configurations (Li et al., 2022). Therefore, its quality standards are not easy to formulate. Now, there are many reports about the saponin components of A. asphodeloides, but few about A. asphodeloides polysaccharide (RAP)’s process optimization and structure–activity relationship. Recently, ultrasound-assisted methods have been used to improve the extraction efficiency of A. asphodeloides (Prakash Maran et al., 2013). However, when extracting polysaccharides using ultrasound, it is necessary to examine whether the structure undergoes decomposition, if there are alterations in the pharmacological mechanism of action, and whether any unknown impacts on human metabolic processes occur (Henglein & Kormann, 1985; Jiang et al., 2020). Optimizing a preparation process with clear principles, high stability, and safety can lay a good foundation for studying the A. asphodeloides structure–activity relationship.
Recent studies have demonstrated that the majority of plant polysaccharides exert antioxidant and anti-inflammatory effects by eliminating free radicals and inhibiting lipid peroxidation (Ren et al., 2019). The efficient and low-toxicity antioxidants from natural products have attracted increasing attention from scholars (Carpentieri et al., 2021). Although the resources of A. asphodeloides are widely distributed in China, their popularity in health care and the prevention of elderly diseases is not enough. There are limited reports on the antioxidant activity of RAP, and its structure–activity relationship has not been explored. Thus, further research into the extraction and antioxidant activity of A. asphodeloides is needed.
Accordingly, the RAP preparation process was optimized using Box–Behnken design (BBD)-based response surface methodology (RSM), and its monosaccharide composition was determined via ultra-performance liquid chromatography with photodiode array detection (UPLC-PDA) following 1-phenyl-3-methyl-5-pyrazolone (PMP) pre-column derivatization. The antioxidant activity of RAP sourced from six different regions was investigated by hydroxyl and superoxide anion radical scavenging models. Building on these findings, a correlation analysis between monosaccharide composition and antioxidant activity was conducted to investigate the influence of varying monosaccharide ratios on antioxidant properties. These findings are anticipated to offer a theoretical foundation and methodological support for subsequent studies on RAP, particularly concerning the structure–activity relationship of polysaccharides.
Materials and Methods
Plant Materials and Reagents
Plant Materials
The dried rhizome of A. asphodeloides Bge. from three different origins was purchased from Guangzhou Caizhilin Co., Ltd. (RAP1: Hanzhong, Shanxi; RAP2: Xinzhou, Shanxi; RAP3: Yulin, Shanxi; RAP4: Yixian, Hebei; RAP5: Anguo, Hebei; RAP6: Bozhou, Anhui Province) in 2020 and was identified by Professor Jizhu Liu of Guangdong Pharmaceutical University (GDPU). A voucher specimen has been deposited at the Guangdong Engineering Technology Research Center for Standardized Processing of Chinese Materia Medica, GDPU, Guangzhou, China.
Reagents
Man, Rha, Glc, Gal, Xyl, Fuc, Rib, Ara, GalA, and GlcA were purchased from Sigma-Aldrich (St. Louis, MO, USA). All other reagents were of analytical grade unless specified otherwise. 2,2-diphenyl-1-picrylhydrazyl (DPPH) was obtained from Shanghai McLean Biochemical Technology Co., Ltd. Vitamin C APIs (VC, batch number 28288902, purity >99.7%) were purchased from Tianjin Tianyin Fine Chemical Development Center. Hydrogen peroxide (H2O2) was purchased from Guangdong Hengjian Pharmaceutical Co., Ltd. All other reagents were of analytical grade unless otherwise specified.
Extraction of RAP
The prepared pieces of A. asphodeloides were extracted continuously in the following order: the prepared pieces (500 g) were relaxed with 10 volumes of 80% ethanol three times, 2 h each time. The residue was dried for later use, and then an appropriate amount of decoction pieces (20 g) was taken, extracted by water and alcohol precipitation, washed alternately with absolute ethanol and acetone, deproteinized by savage, and then dried (Mei et al., 2020).
Determination of Polysaccharide Content
Polysaccharide content was determined by the phenol–sulfuric acid assay (Liu & Huang, 2019). First, 50 mg of Glc (anhydrous) reference was precisely weighed to obtain a 1.0 mg/mL reference solution. Second, the reference solution of 0.2, 0.4, 0.6, 0.8, 1.0, 1.2, and 1.4 mL was sucked into a 10 mL test tube with a plug, supplemented with ultrapure water to 2 mL, and shaken well. Finally, 2 mL of ultrapure water was sucked in as a blank control, and 1 mL of 6% phenol solution and 5 mL of concentrated sulfuric acid solution were added in turn and shaken. After incubation in a boiling water bath for 20 min and cooling to room temperature, the absorbance was measured at 490 nm using a spectrophotometer. The standard curve was calculated by the absorbance value as the Y-axis and the mass concentration of Glc in the reference solution as X-axis: Y = 3.487X + 0.066 (0.2–1.4 (mg/mL), R2 = 0.9993, n = 6).
Polysaccharide content was calculated using the following Eq. (1):
Y represents the polysaccharide yield; M is the volume of the crude polysaccharide solution; N is the measured glucose concentration (mg/mL); D is the dilution factor, applied if the extraction solution exceeds the quantifiable range; W is the dry weight of A. asphodeloides.
Effect of Material-to-liquid Ratio on the Extraction Yield of RAP
Weighing 20 g of alcohol-extracted A. asphodeloides, with an extraction time of 90 min, alcohol content of 70%, extraction one time, and a solid–liquid ratio of 1:6, 1:8, 1:10, 1:12, and 1:14, was used to extract the samples, respectively. Each experiment was conducted in triplicate to ensure reproducibility. The optimal material-to-liquid ratio was established by comparing the RAP content from each extraction.
Effect of Extraction Time on Yield of RAP
Weighing 20 g of alcohol-extracted A. asphodeloides, with a solid–liquid ratio of 1:10, alcohol content of 70%, extraction one time, and extraction times of 60, 90, 120, 150, and 180 min, was used to extract the samples, respectively. All experiments were performed in triplicate. The optimal solid-to-liquid ratio was identified by evaluating the RAP yield from different extraction conditions.
Effect of Extraction Times on Yield of RAP
Weighing 20 g of alcohol-extracted A. asphodeloides, with a solid–liquid ratio of 1:10, alcohol content of 70%, extraction time of 120 min, and extraction 1, 2, 3, 4, and 5 times, was used to extract the samples, respectively. All experiments were performed in triplicate. The optimal solid–liquid ratio was determined by comparing the amount of RAP content obtained from each extraction.
Effect of Ethanol Concentration on Yield of RAP
Weighing 20 g of alcohol-extracted A. asphodeloides, with a material-to-liquid ratio of 1:10, extraction time of 120 min, extraction one time, and alcohol content of 60%, 65%, 70%, 75%, and 80%, was used to extract the samples, respectively. All experiments were performed in triplicate. The optimal solid–liquid ratio was determined by comparing the amount of RAP content obtained from each extraction.
Response Surface Analysis for Optimization of Extraction Condition for RAP
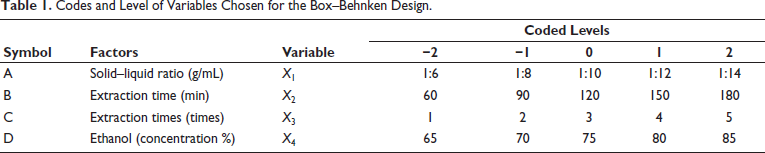
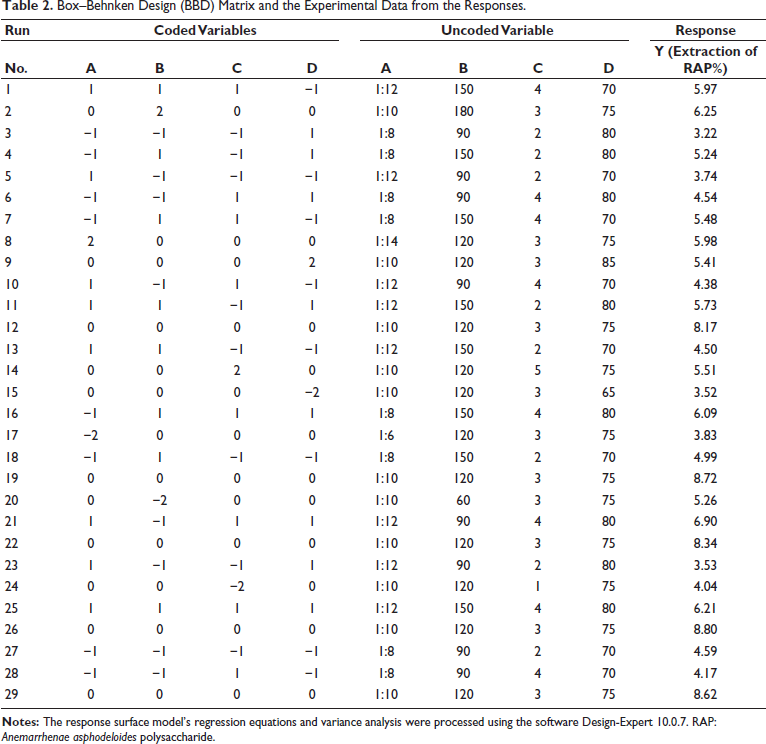
Single-factor experiments were initially conducted, and according to the results, four independent variables (A, solid–liquid ratio; B, extraction time; C, extraction times; and D, ethanol concentration) at five levels were employed for the optimization of extraction parameters for polysaccharides from A. asphodeloides to obtain the best yield of RAP by BBD-RSM (Zhu et al., 2023). Table 1 presents the ranges of the independent variables, along with their corresponding response values. Table 2 shows the range and levels of the independent variables, along with the results from the 29 test points conducted in random order. The response surface model’s regression equations and variance analysis were processed using the software Design-Expert 10.0.7.
Codes and Level of Variables Chosen for the Box–Behnken Design.
Box–Behnken Design (BBD) Matrix and the Experimental Data from the Responses.
Analysis of Monosaccharides of RAP
The monosaccharide composition of A. asphodeloides from six different origins was analyzed using the PMP pre-column derivatization technique (Liao et al., 2018). The sample was dissolved in ultrapure water to a concentration of 1.0 mg/mL, combined with 2 mL of 2 mol/L trifluoroacetic acid (TFA) solution, sealed with nitrogen gas, subjected to hydrolysis at 120°C for 160 min, and subsequently cooled to ambient temperature. The samples were centrifuged for 8 min (1,500 r/min); the supernatant was concentrated under reduced pressure to eliminate the remaining TFA. Methanol was added during this period, and after repeated distillation 3–4 times, ultrapure water was added to dilute it to 2.0 mL and was set aside.
An appropriate amount of 10 monosaccharide standards was accurately weighed. The concentration of the standard solution was 1.0 mol/L with ultrapure water, and the mixed standard solution of 1.0 mol/L was prepared according to the above method. The standard solution and the hydrolyzate of anemones were taken separately and placed in a 5.0 mL centrifuge tube. Then, 500 µL of 0.5 mol/L PMP methanol solution and 500 µL of 0.3 mol/L NaOH solution were added. After mixing, the mixture was placed in a 70°C water bath and heated for 30 min. After removal, the mixture was maintained at room temperature for 10 min, then neutralized by adding 500 µL of 0.3 mol/L hydrochloric acid (HCl). The extraction was carried out three times using chloroform added in equal amounts. After shaking, the sample was centrifuged at 1,500 rpm, the supernatant was separated, and filtered using a 0.45 µm microporous membrane.
Monosaccharide analysis of RAP was performed on a Waters Acquity UPLC system using an ACE C18 column (1.7 µm, 2.1 × 100 mm), with photodiode array detection at 254 nm. The mobile phase was acetonitrile (15%–18%–3%–10%–20%–15%) and 0.05% glacial acetic acid solution containing 0.02 mol/L ammonium acetate, the flow rate was 0.4 mL/min, and the sample injection volume was 0.8 µL.
DPPH Radical Scavenging Activity of RAP
A previously established method was used to evaluate DPPH radical scavenging activity (Shimada et al., 1992), with some modifications. Briefly, a 0.5 mM solution of DPPH in ethanol was prepared, and 1.0 mL of this solution was mixed with 1.0 mL of the polysaccharide solutions at concentrations of 1.0, 2.0, 4.0, 6.0, and 8.0 mg/mL in water. The sample was thoroughly mixed and incubated at 25°C in darkness for 30 min, after which the absorbance was determined at 517 nm against a blank. The scavenging percentage was calculated by the following Eq. (2):
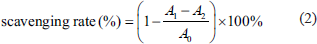
A0 represents the control absorbance (with water replacing the RAP solution), A1 is the sample absorbance, and A2 is the sample absorbance measured under the same conditions as A1 but with water replacing the DPPH solution.
Hydroxyl Radical Scavenging Activity of RAP
The superoxide radical scavenging activity was evaluated following a previously reported method with slight modifications (Chen et al., 2012). In brief, a range of concentrations of RAP were prepared 1.0, 2.0, 4.0, 6.0, and 8.0 mg/mL, and then incubated with 6.0 mM FeSO4 (2.0 mL), 6.0 mM H2O2 (2.0 mL), and 2.0 mL of 6.0 mM salicylic acid-ethanol solution for 30 min at 30°C. Hydroxyl radicals were detected by measuring the absorbance at 517 nm. The hydroxyl radical scavenging rate was calculated using the following Eq. (3):
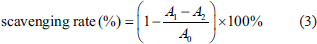
A0 is the control absorbance in the hydroxyl radical generation system (with water replacing the RAP solution), A1 is the test group absorbance, and A2 is the sample absorbance (with water replacing the H2O2 solution).
Superoxide Anion Radical Scavenging Activity of RAP
The superoxide radical scavenging effect was evaluated based on a previous method with slight modifications (Marklund & Marklund, 1974). Briefly, 4 mL of 50 mM Tris–HCl buffer (pH 8.0) was mixed with 1.0 mL of sample solutions at concentrations of 1.0, 2.0, 4.0, 6.0, and 8.0 mg/mL, and incubated at 25°C for 20 min. Then, 1.0 mL of 25 mM pyrogallol (pre-equilibrated at the same temperature) was added, and the reaction proceeded at 25°C for 4 min. The reaction was then terminated by adding HCl, and the absorbance was measured at 325 nm against a blank. The superoxide radical scavenging percentage was calculated using the following Eq. (4):
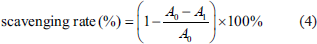
where A0 is the absorbance of the control group in the superoxide radical generation system (water was used instead of RAP solution), and A1 is the change speed of the absorbance of the test sample.
Statistical Analysis
Experimental data were analyzed using GraphPad Prism 9.0. One-way analysis of variance (ANOVA) was applied for multiple-group comparisons, while a t-test was used for pairwise comparisons. p < .05 was regarded as statistically significant. Through the R language visualization function, Pearson correlation analysis was performed on the monosaccharide composition ratio and antioxidant activity index parameters of RAP by RStudio 2022.02.0+443 software, and a correlation coefficient visualization diagram was made.
Results
Results from Single-factor Experiments
Figure 1 presents the single-factor test results, showing a significant increase in RAP yield from 3.20% to 5.07% as the solvent-to-solid ratio increased from 6 to 14 g/mL (Figure 1A). The maximum yield was reached at 1: 10. As the volume of the solvent increases, the polysaccharide yield increases. When it reaches its maximum dissolution rate, the yield no longer increases (Zhu et al., 2022). Consequently, a solid-to-liquid ratio of 1:10 was determined to be optimal.
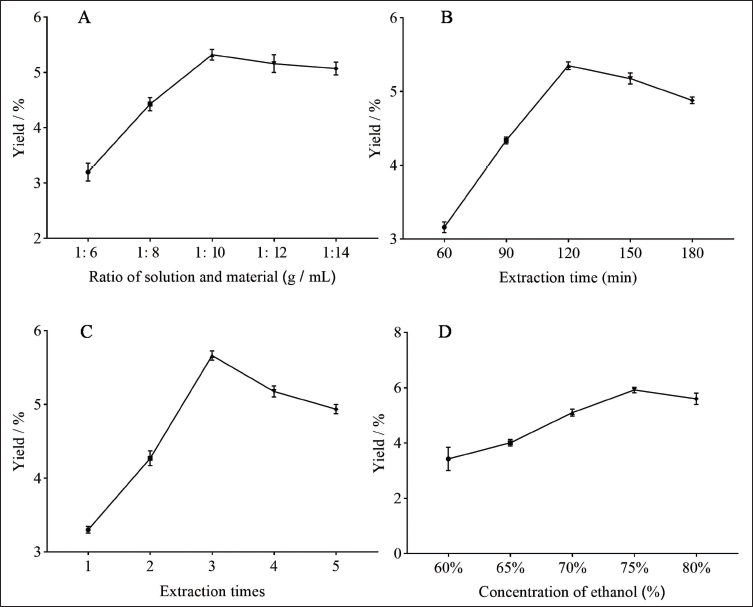
As the extraction time increased from 60 to 120 min (Figure 1B), the yield of RAP increased from 3.16% to 5.35%. As shown in the figure, the highest yield of the polysaccharide was obtained at 120 min. The decrease in polysaccharide yield may be attributed to prolonged exposure to high-temperature boiling environments, which can cause the destruction and degradation of the polysaccharide structure (Zhao et al., 2013). Therefore, 120 min was chosen as the best value.
The yield of RAP increased in the range of 1–3 extraction times and then decreased slightly. At the extraction of three, the maximum value of 5.66% is reached (Figure 1C). Considering the increasing number of extractions, the consumption of solvent, energy, and time also increases. Therefore, three times were chosen as the best extraction time.
The yield of RAP increased in the range of 60%–75% ethanol and then plateaued. A maximum value of 5.92% was reached at a concentration of 75% (Figure 1D). There was no significant change in the yield of polysaccharides with increasing the concentration of alcohol precipitation. Therefore, 75% was chosen as the optimal concentration of alcohol sedimentation.
Response Surface Analysis for Optimization of Extraction Condition for RAP
The design plan and results obtained using Design-Expert 10.0.7 are presented in Table 2. Based on these results, multiple linear regression analysis was performed, yielding the following regression Eq. (5):
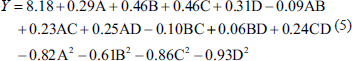
The regression equation results are shown in Table 3. The correlation coefficient was found to be R2 = 0.8993. For the regression model, p < .0001, indicating a highly significant result. The lack of fit term was 0.6983, with p > .05, suggesting that the model selection was reasonable and suitable for analyzing and predicting the extraction yield of RAP. In the variance analysis of the regression equation, p values for factors B and C were both <.01, indicating that these two factors had highly significant effects on the extraction yield of RAP. Additionally, by comparing the F-values, the influence of each factor on the extraction yield of RAP was determined to follow this order: B > C > D > A.
The Analysis of Variance Results for the Response Surface Quadratic Models for the Yield of Anemarrhenae asphodeloides Polysaccharide (RAP).
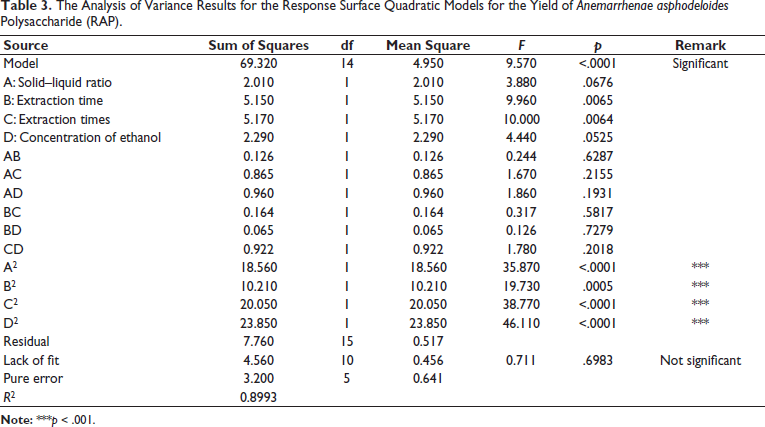
A 3D response surface map, generated in the “Model Graphs” under the “Analysis” module, is shown in Figure 2. The 3D plot provides a clearer view of the interactions among various factors. The surfaces of factors B and C are relatively steep, while the surfaces for factors A and D are more gradual, indicating that B and C have a greater impact on the extraction yield of RAP.
Four factors were analyzed using Design-Expert 10.0.7, and the optimal extraction conditions were determined to be as follows: the ratio of solid to liquid was 1:10.38 g/mL, the extraction time was 124.11 min, the times of extractions was 3.53, and the concentration of alcohol was 76.77%. Under these optimal conditions, the predicted RAP yield was 8.337%. Considering the feasibility of operation, the final extraction condition was determined as follows: solid–liquid ratio of 1:10 g/mL, extraction time of 124 min, extraction frequency of four times, and alcohol precipitation with a content of 77%. Under these modified conditions, each experiment was conducted in triplicate, resulting in an RAP extraction yield of 8.56% (RSD = 2.47%). The measured value of the optimized extraction closely matched the predicted value of the model, suggesting that both the optimized method and the established model were reliable and predictable.
Monosaccharide Composition Analysis of RAP
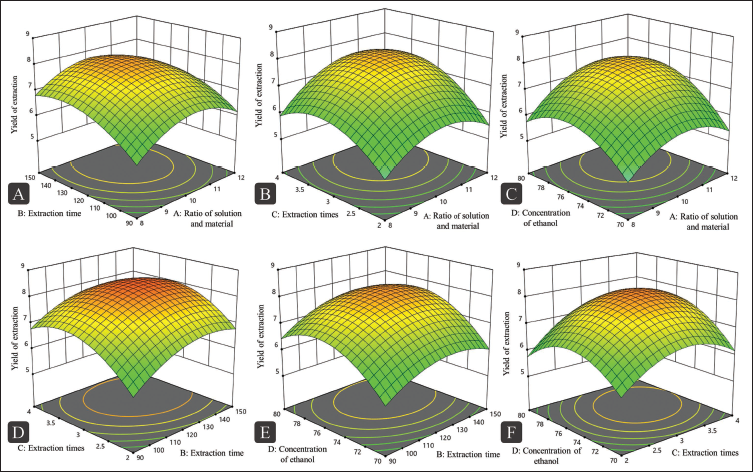
Polysaccharide bioactivity is largely influenced by the types and proportions of constituent monosaccharides (Zhang et al., 2017). In this study, the individual monosaccharide contents in RAP were quantitatively analyzed using UPLC-PDA. The high-performance liquid chromatography with photodiode array detection (HPLC-PDA) chromatogram of PMP-labeled mixed monosaccharide standards (Figure 3A) illustrates the efficient resolution of 10 representative sugars, including Man, Rha, Glc, Gal, Xyl, Fuc, Rib, Ara, GalA, and GlcA. The linear regression equation is shown in Table S1 (Supplementary Material). Figure 3B presents the sample of RAP. Composition and molar ratio of monosaccharide of RAP1-6 are shown in Table 4. The results suggested that RAP was a heteropolysaccharide. According to the findings, RAP primarily consisted of Man and GalA. These results are consistent with those of previous studies on A. asphodeloides (Chen, Lan, et al., 2023; Chen, Li, et al., 2023). The results showed that the extraction methods influenced the monosaccharide composition of the RAP samples, though the types of monosaccharides remained unchanged. The UPLC chromatograms of RAP2~6 samples are shown in Figure S1 (Supplementary Material).
Ultra-performance Liquid Chromatography (UPLC) Analysis of 1-Phenyl-3-methyl-5-pyrazolone (PMP) Pre-column Derivatives of (A) Nine Standard Monosaccharides (1. Man; 2. Rha; 3. Rib; 4. GlcA; 5. GalA; 6. Glc; 7. Gal; 8. Ara; 9. Xyl; 10. Fuc) and (B) RAP1 Hydrolysate.
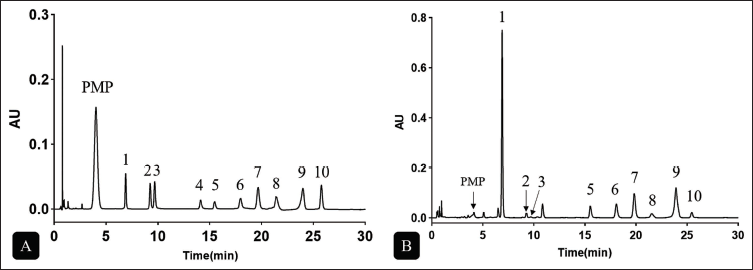
Composition and Molar Ratio of Monosaccharide of RAP1-6.
DPPH Radical Scavenging Activity of RAPs
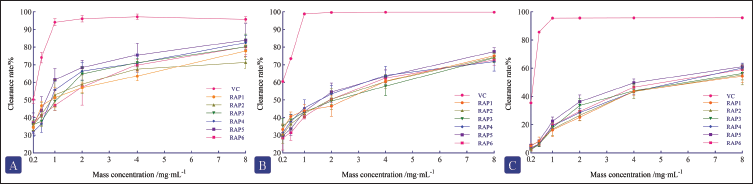
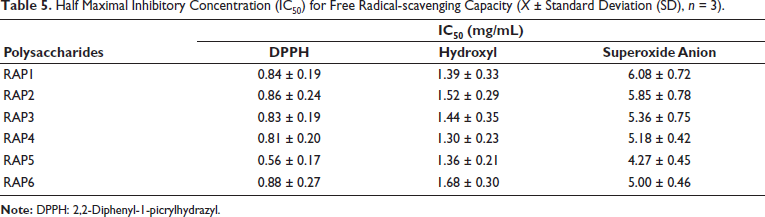
RAPs scavenge DPPH radicals by transferring electrons or protons to DPPH radicals, thereby forming a stable molecular state of DPPH. The DPPH radical has a characteristic absorption at 517 nm. When radical scavengers are present, the absorption gradually diminishes. Therefore, this method can be used to quantitatively evaluate the radical scavenging activity of RAPs (Hossain et al., 2013). Figure 4A displays the DPPH radical scavenging activity of the RAP samples. All RAP samples exhibited DPPH scavenging ability, with a concentration-dependent response across the range of 0.2–8.0 mg/mL. However, their scavenging capacity was still lower than that of the positive control group. The half scavenging rate (IC50) of DPPH free radical of RAPs is shown in Table 5. It is known that the smaller the IC50, the stronger the antioxidant activity. Among them, RAP5 showed stronger DPPH radical scavenging activity than other samples.
Hydroxyl Radical Scavenging Activity of RAPs
Excessive hydroxyl radicals can induce oxidative stress, leading to the impairment of intracellular biomolecules and aberrant cellular functions. The generation of hydroxyl radicals through the Fenton reaction, which involves H2O2 and Fe2+, has been extensively reported. Salicylic acid has been shown to efficiently scavenge these hydroxyl radicals and react with them to form the colored substance 2,3-dihydroxybenzoic acid (characteristic absorption at 510 nm). When a sample with antioxidant properties is introduced, the absorption peak at 510 nm tends to decrease (Zhao, 2023). Figure 4B shows the hydroxyl radical scavenging activity of the RAP samples. All RAP samples were able to scavenge hydroxyl radicals, with a concentration-dependent effect observed across concentrations ranging from 0.2 to 8.0 mg/mL. Nevertheless, their radical scavenging ability was still inferior to that of the positive control. The half maximal inhibitory concentration (IC50) of hydroxyl free radical of RAPs is shown in Table 5. It is known that the smaller the IC50, the stronger the antioxidant activity. Notably, RAP4 and RAP5 showed enhanced DPPH radical scavenging effects in comparison with other groups.
Half Maximal Inhibitory Concentration (IC50) for Free Radical-scavenging Capacity (X ± Standard Deviation (SD), n = 3).
Superoxide Anion Scavenging Activity of RAPs
The superoxide anion is produced through the nicotinamide adenine dinucleotide-phenazine methosulfate-nitroblue tetrazolium (NADH-PMS-NBT) system, where a superoxide anion scavenger can diminish the blue color of NBT. The antioxidant capacity of the active ingredients within the system can be assessed by measuring the absorbance value at 560 nm (Kozuleva & Ivanov, 2023). It can be seen that the scavenging rate of RAPs to superoxide anion is not high within the experimental concentration range; the maximum is not more than 60% (Figure 4C). In the concentration range of 0.2–8.0 mg/mL, all RAP samples demonstrated the ability to scavenge superoxide anion. The IC50 of the superoxide anion of RAPs is shown in Table 5. Among them, RAP5 showed stronger superoxide anion scavenging activity than other samples.
Correlation Analysis Between Monosaccharide Composition and Antioxidant Activity
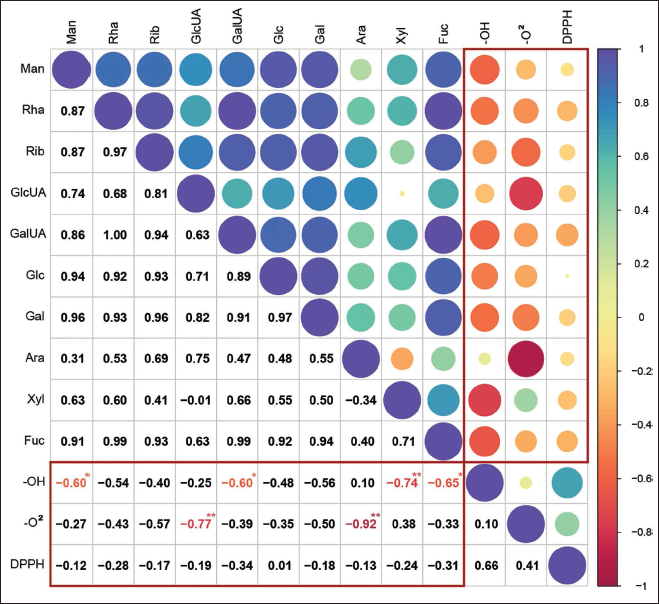
In R language, the IC50 values of DPPH radicals, hydroxyl radicals, and superoxide radicals were analyzed using Pearson correlation. The relationship between the monosaccharide composition and antioxidant activity of polysaccharides is illustrated in Figure 5. In Figure 5, the circle size corresponds to the Pearson correlation coefficient, where blue to purple hues signify positive correlations, and red to yellow hues indicate negative correlations. The correlation coefficient is displayed in the bottom-left corner of the figure (*p < .05, **p < .01). As the IC50 values of most monosaccharides are negatively correlated with DPPH, hydroxyl, and superoxide radicals, it is evident from the figure that a higher concentration of monosaccharides corresponds to lower IC50 values, indicating an enhanced ability to scavenge radicals. Figure 5 illustrates that the contents of Man, GalA, Xyl, and Fuc are significantly negatively correlated with the IC50 value of the hydroxyl radical. This suggests a positive correlation between hydroxyl radical scavenging activity and the relative proportions of Man, GalA, Xyl, and Fuc, with the strength of correlation ranked as Xyl > Fuc > Man = GalA. Among them, the IC50 values of GlcA and Ara are negatively correlated with the superoxide anion radical. These findings suggest a positive correlation between RAP’s scavenging activity and the relative contents of GlcA and Ara, with Ara exhibiting a stronger correlation than GlcA.
Discussion
A. asphodeloides has garnered attention for its potential applications in health and medicine for over two millennia. It is currently a focal point of research in modern medicine and the healthcare industry for its potential to provide highly efficient and low-toxicity antioxidants capable of combating free radicals in the body (Lee et al., 2023; Piwowar et al., 2020). Therefore, the author optimized the extraction process and studied the antioxidant activity of RAP and its active ingredients to lay the foundation for future development and application.
In clinical practice of traditional Chinese medicine (TCM), A. asphodeloides is frequently used to treat various ailments such as hyperthermia, polydipsia, bone steaming, hot flashes, dryness of the intestine, and constipation. TCM theory posits that inflammation is a “hot disease” primarily caused by external stimuli and internal damage. Modern pharmacological research has demonstrated that A. asphodeloides can inhibit the release of inflammatory substances from astrocytes and macrophages when stimulated (Bhatia, 2018; Peng et al., 2022). However, the body’s response to oxidative stress and inflammation is closely linked, as an imbalance between free radicals such as DPPH, hydroxyl, and superoxide anion and the body’s antioxidant defense system can result in oxidative stress, subsequently contributing to disease (Devasagayam et al., 2004; Gill et al., 2010).
The complex structure of polysaccharides, natural polymers found in plants, determines their properties. Previous research suggests that the connection method between neighboring monosaccharide groups in the basic structure of polysaccharides may influence the potency of their antioxidant activity. Additionally, the hemiacetal hydroxyl group of polysaccharides serves as a reducing end that can scavenge free radicals (Bai et al., 2022; Ji et al., 2022). BBD is a technique used to optimize complex extraction processes, which can continuously analyze all levels of test factors compared to the single-factor method. Experimental studies have found that RAP has good antioxidant activity. In order to maximize the yield of RAP, this study used BBD to optimize the extraction process of RAP based on the single-factor experimental results of solid–liquid ratio, extraction time, extraction times, and alcohol precipitation concentration, and determined the best extraction process of RAP. In addition, the Pearson correlation analysis method was used to calculate the correlation coefficient between the monosaccharide content and antioxidant activity of A. asphodeloides, examining the connection between antioxidant activity and polysaccharide structure.
The IC50 values of six aliquots of RAP for scavenging free radicals were determined using DPPH, hydroxyl, and superoxide anion radical as experimental subjects. The correlation between the IC50 value and the composition ratio of 10 monosaccharides was then examined. The results indicated that RAP exhibited better free radical scavenging effects at higher concentrations within a specific mass concentration range. Specifically, RAP5 showed the best DPPH radical and superoxide anion scavenging activity, while RAP4 exhibited the best hydroxyl scavenging activity. Correlation analysis revealed a significant positive correlation between RAPs hydroxyl radical scavenging activity and the composition ratios of Man, GalA, Xyl, and Fuc, as well as a significant positive correlation between RAPs superoxide anion radical scavenging activity and the composition ratios of GlcA and Ara (Zhang et al., 2021). Therefore, it was concluded that the higher the proportion of Man, GalA, Xyl, Fuc, GlcA, and Ara in RAP, the greater the capacity to scavenge free radicals, demonstrating enhanced antioxidant activity. Among these monosaccharides, Man, Fuc, GlcA, Ara, and Xyl have known physiological actions that include immune-system regulation and anti-inflammatory responses (Cheudjeu, 2020; Liu et al., 2020; Michiels et al., 2023). Previous studies have also found a favorable correlation between different monosaccharides and the in vitro antioxidant activity of polysaccharide samples, with a strong overall correlation between monosaccharide composition and activity. The experimental results showed that the samples containing GlcA and Xyl showed significantly higher antioxidant activity than those without these monosaccharides, aligning with previously reported findings (Li et al., 2015; Ni et al., 2014). The study of polysaccharides from nine herbs shows that those containing GlcA and Xyl have significantly higher antioxidant activity. However, the specific degree of their impact requires further study through experiments with a more representative range of samples. In the radical scavenging experiment, Man, Fuc, and GlcA were identified as key influencing factors based on the association between polysaccharide composition and anti-oxidative properties of Brasenia schreberi, consistent with the findings of this study (Li et al., 2020). While numerous studies have examined the antioxidant properties of different monosaccharides, polysaccharides are not simply the sum of individual monosaccharides (Caicedo et al., 2019; Ito et al., 2021; Wang et al., 2024). Thus, they cannot be solely evaluated based on the cumulative antioxidant properties of their monosaccharide components. This study performed correlation analysis between the monosaccharide composition and antioxidant activity of RAP, offering initial insights into the compositional patterns underlying antioxidant polysaccharides. Further research is needed to fully elucidate the structure–activity relationship of the antioxidant activity of plant polysaccharides, particularly regarding the fundamental structure of polysaccharides and its antioxidant activity.
Conclusion
In this study, the BBD response surface method was utilized to optimize the preparation process of RAP, yielding the following optimal conditions: a solid–liquid ratio of 1:10 g/mL, an extraction time of 124 min, an extraction frequency of 4 times, and alcohol precipitation with a content of 77%. The relative proportions of 10 monosaccharides in six polysaccharide samples were further quantified using PMP derivatization combined with UPLC-PDA analysis. The polysaccharides’ antioxidant capacities were assessed through their ability to scavenge DPPH, hydroxyl, and superoxide anion radicals, and the relationship between monosaccharide composition and antioxidant activity was subsequently examined. The study provides an initial exploration of the relationship between monosaccharide composition and the antioxidant activity of RAP. It was observed that Xyl, Fuc, Man, GalA, Ara, and GlcA had significant effects on antioxidant activity in vitro. A higher proportion of these monosaccharides corresponded to stronger free radical scavenging abilities, indicating enhanced antioxidant activity.
However, the study of polysaccharides in this study only stayed at the monosaccharide composition, and the detailed structure of polysaccharides was not systematically studied. In future research, GC-MS and NMR techniques may be utilized to elucidate the linkage patterns of monosaccharides, further clarifying their connection to antioxidant activity.
Footnotes
Abbreviations
AA: Anemarrhenae asphodeloides; Ara:
Acknowledgments
The authors thank the TCM Processing Platform of Guangdong Pharmaceutical University for providing facilities to carry out this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
As this study did not involve human participants or animals, ethical approval and informed consent were not required.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Famous Old Traditional Chinese Medicine Experts Inheritance Studio Construction Program of National Administration of TCM [Grant Number: (2022) No. 75]. The Seventh Batch of National Famous Old Traditional Chinese Medicine Experts Experience Heritage Construction Program of National Administration of TCM [Grant Number: (2022) No. 76]. Guangdong Key Field Research and Development Program Project (No.2020B111112-0002). Basic and Applied Basic Research Project of Guangzhou Basic Research Program (SL2022A04J01286).
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.