Abstract
Chlorogenic acid has been proved to have cardiovascular protection, antibacterial, antiviral, hemostatic, and hypolipidemia effects. Modern scientific research on the bioactivity of chlorogenic acid has been extended to the fields of food, medicine, health care and daily-use chemical industry. The aim of this research was to optimize the extraction conditions for chlorogenic acid from Eucommia ulmoides (Eucommiaceae) leaves. The significant variables were screened and optimized by a combination of Plackett-Burman test and Box-Behnken design. Optimum extraction parameters with ethanol concentration of 50%, solvent pH value of 3, and particle size of 60 mesh were determined according to variance analysis and contour plots. Under these conditions, the yield of chlorogenic acid was up to 4.36 mg/g, which was basically consistent with the theoretical prediction value of 4.50 mg/g. This study also proved the potential antioxidant activity of E. ulmoides leaves. The optimal extract of E. ulmoides leaves rich in chlorogenic acid showed the highest antioxidant activity in the FRAP method, which was 219.8 μM Trolox equivalents (TE) per g extract weight (EW) (μM TE/g EW). The DPPH method gave a similar value (168 μM TE/g EW) to the ABTS method (152 μM TE/g EW). The established extraction process was efficient in the recovery of chlorogenic acid from E. ulmoides leaves, encouraging its valorization as a cheap and sustainable alternative for the isolation of chlorogenic acid.
Introduction
Eucommia ulmoides Oliver (E. ulmoides) is a distinctive medicinal material economic resource in China. It can be used as medicine, glue, as well as a health food and a source of other raw materials.1,2 E. ulmoides has a broad application prospect and has great potential in the field of medicine. Its dried bark is a valuable nourishing material in China, which belongs to the meridians of liver and kidney.
In traditional Chinese medicine, E. ulmoides bark is commonly used to treat rheumatoid arthritis, hypertension, sciatica and lumbago, and also has hypoglycemic and antiaging properties.3‐6 However, due to the cutting down of the trees and inefficient peeling of the bark, bark resources of E. ulmoides are scarce. In view of a large demand and wide use of E. ulmoides, research has been directed at the leaves of E. ulmoides. Modern scientific research shows that the pharmacological effects of the leaves of this species are similar to those of the bark,7,8 which indicates that the leaves have the prospect of being developed as substitutes for the bark. In the 2005 edition of Chinese Pharmacopoeia, E. ulmoides leaves were included as a new drug for the first time. Therefore, it is of practical significance to establish the optimum extraction process for the active components of E. ulmoides leaves.
Chlorogenc acid (CGA) is an unsaturated ester composed of quinic acid and caffeic acid (Figure 1), which has been commonly adopted as the quality control standard for E. ulmoides leaves. 9 CGA is an important bioactive substance and chemical reagent endowed with antibacterial and anti-inflammatory effects. Therefore, CGA is the subject of 170 kinds of patented Chinese medicines. It has been reported to contain a variety of pharmacological activities such as cardiovascular protection, antibacterial and antiviral effects, hemostatic and hypolipidemia properties, and increasing leucocytes. It has been widely used as a raw material in the fields of food and health care, and by the pharmaceutical industries.10‐12 Outside of China, CGA is mainly extracted from sunflower seeds and coffee beans, while in China, CGA is mainly extracted from sunflower seeds and honeysuckle.13‐15 The results showed that the CGA content in E. ulmoides leaves and honeysuckle was high and similar, but the price of E. ulmoides leaves was only one twentieth to one fortieth that of honeysuckle. In view of the great economic value of CGA, it would have better social and economic benefits if CGA was extracted from cheap E. ulmoides leaves. Although there are many procedures to extract CGA from plants, including ultrasonic, microwave assisted and enzyme methods, the traditional reflux extraction method is still the most widely used and most suitable for industrial production.16‐18 In addition, considering that CGA may be affected by high temperature and enzymic hydrolysis, isomerization usually occurs through hydrolysis and transfer of molecular lactone groups, causing structural instability. In this paper, reflux extraction and biphasic separation were combined to extract and purify CGA in order to obtain high purity CGA.
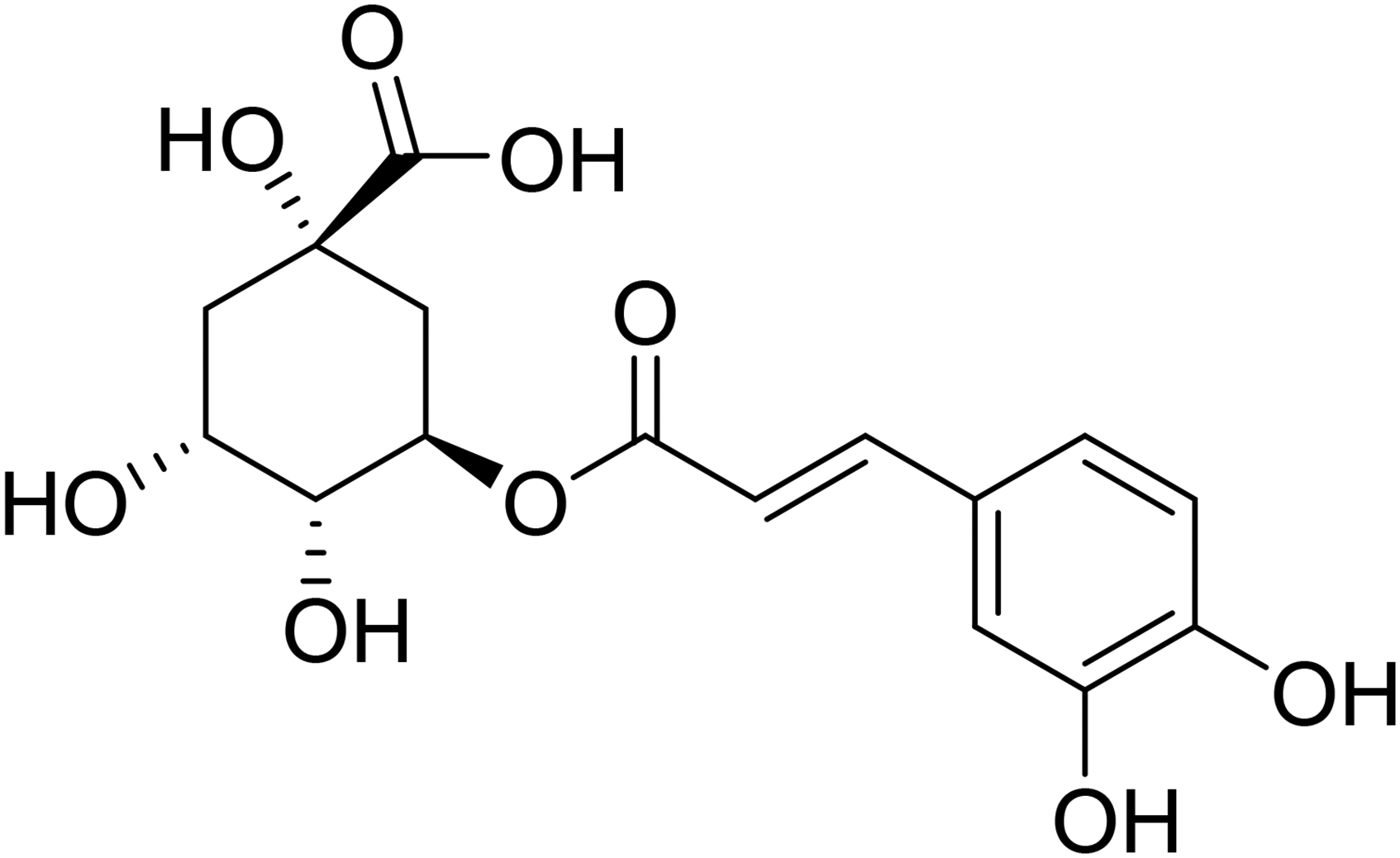
Structure of chlorogenic acid.
As an experimental design method combining statistics and mathematics, a response surface method (RSM) has been successfully used to optimize the extraction of effective substances from medicinal plants.19‐21 The greatest advantage of RSM is that fewer experiments are required to evaluate multiple polytomy variables and their mutual effects, saving labor and time compared with other methods.
Many assays have been used to evaluate antioxidants in food additives, foods, and natural products. These include the 2,2-diphenyl-1-picrylhydrazyl (DPPH•) free radical-scavenging method, 22 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS·+) procedure, 23 ferric reducing antioxidant power (FRAP) method, 24 oxygen radical absorbance capacity (ORAC) assay, 25 superoxide radical scavenging activity (SRSA) and metal chelating capacity (MCC) method. 26 These assays differ in measuring principles and test conditions. Due to the variety of mechanisms and reaction characteristics involved, no single method can accurately evaluate all antioxidant properties in a mixed or complex substance, therefore, various evaluation methods of antioxidant capacity are needed.
The aim of this study was to extract CGA from E. ulmoides leaves employing a response surface method (RSM). First, the Plackett-Burman method was used to screen out main process parameters affecting CGA content during extraction. The extraction parameters were then modeled and optimized by the three-level and three-variable Box-Behnken design (BBD) method. In addition, the antioxidant activity of the optimal extract was evaluated by DPPH, ABTS and FRAP assays in vitro.
Materials and Methods
Plant Source
Eucommia ulmoides leaves purchased from Wangping village, Henan Province, China, were identified by Prof. Ximing Lu, Medical College, Henan University of Science and Technology. Plant materials were air dried at room temperature and stored. A voucher specimen is maintained in the college Herbarium, certificate No. 817.
Chemicals and Standards
Chromatographic grade methanol and phosphoric acid were obtained from Thermo Fisher Scientific (Shanghai, China). Standard CGA was obtained from National Institutes for Food and Drug Control (Beijing, China). Analytical grade ethanol, ethyl acetate, cyclohexane, and hydrochloric acid were purchased from Sinopharm Chemical Reagent Co. Ltd (Shanghai, China).
2,2-Diphenyl-1-picrylhydrazyl (DPPH), 2,2′-azino-bis (3-ethylbenzothiazoline−6-sulfonic acid) diammonium salt (ABTS), 2,4,6-tris(2-pyridyl)-1,3,5-triazine (TPTZ) reagent and (±)-6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid (Trolox) standard were obtained from Shanghai yuanye Bio-Technology Co., Ltd
Extraction Procedure
Dried E. ulmoides leaves were crushed by a small grinder, then sifted, and 10 g put into a 250 mL flask. Solvent was then added to obtain a certain ratio of material to liquid. Specific extraction factor parameter conditions are shown in the experimental design. The acidic environment, heat and reflux at a constant temperature in a water bath were maintained, while a Brinell funnel was used to filter while hot. The filtrate was concentrated in a rotary evaporator, cooled and placed in a 25 mL volumetric flask, which was sealed and stored in a refrigerator.
Plackett-Burman Experimental Design for Main Factor Screening
As a two-level test design method, the Plackett-Burman test design has the greatest advantage of rapidly selecting the most significant parameters from many for further study. 27 This experimental design avoids wasting experimental resources in the later optimization experiment because there are either too many factors or some factors are not significant. In this experiment, 11 factors (seven actual factors and four virtual factors) were selected to screen the main influencing factors during extraction of CGA by the Plackett-Burman experimental design method, with 12 replications. The factors and the setting levels of the Plackett-Burman experiment are shown in Table 1.
Levels and Factors Adopted in Plackett-Burman Design Test.
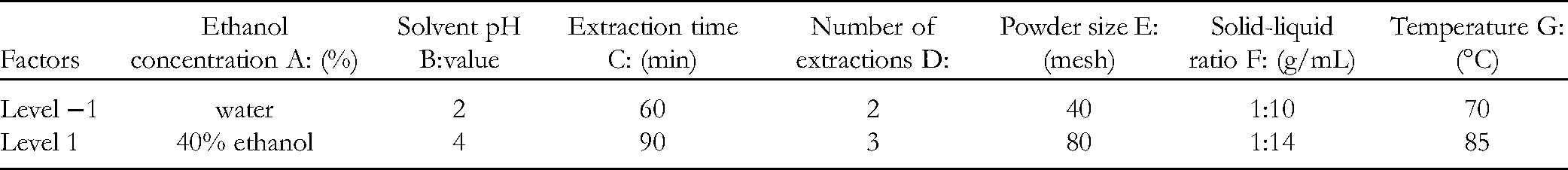
Box-Behnken Design Method to Optimize the Extraction Process
The extraction process parameters were screened by a Box-Behnken design with three levels and three variables. Based on the Plackett-Burman experimental design, ethanol concentration (A), solvent pH value (B) and powder size (C) were determined as the three most significant variables. Finally, seventeen experimental tests were performed. The extraction quantity of CGA (mg/g) was the response studied (Y). An empirical quadratic polynomial model is established through regression analysis, as shown in the following formula:
Two-Phase Separation
Two- Phase Separation Solvent System
CGA is a weak polar molecule. It can retain its molecular state under acidic conditions and dissolves easily in polar solvents (similar to the principle of miscibility). Therefore, we choose the polar ethyl acetate, which is insoluble in water, as the extraction agent. After extraction, n-hexane was used as a phase separation agent. When ethyl acetate and n-hexane are miscible to form a homogeneous phase, the CGA in the ethyl acetate is gradually released with very little water that does not dissolve in the extraction agent, and this process effectively filters out some impurities. High purity CGA samples were obtained. 28
Phase Separation Procedure
Ten mL extract was added to a separation funnel, an equal volume (10 mL) of ethyl acetate (adjusted pH = 3) was added, and the mixture shaken for 4 min. The upper phase was separated, and this extraction operation was repeated four times. The four extracts were mixed, and then extracted with n-hexane, which was equal to 40% of the total volume of the extraction phase (20 mL). After phase separation, the lower phase was poured into a flask and dissolved in methanol up to 10 mL for HPLC analysis and antioxidant activity determination.
Analysis and Identification of CGA by HPLC
The CGA content of extracts was determined using an HPLC system (Waters, USA), equipped with an ultraviolet detector, an automatic sampler and a high pressure pump. A Thermo Science Hypersil ODS C18 column (250 mm × 4.6 mm, 5 μm) was used to separate compounds. For determination, the extract was first filtered through a 0.45 μm microporous filter and then 10 μL of the filtered extracts were injected. The mobile phase consisted of 0.4% phosphoric acid water (A) and HPLC methanol (B), with a volume ratio of 13:87; the flow rate was 1 mL/min, and the detection wavelength was 327 nm. The standard curve showed a linear relationship for the CGA standard in the range of 0.16 μg ∼ 2.4 μg: Y = 357.71 X + 9.0902 (r = 0.9995, Y is the peak area for 10−4 times; X is the CGA content [μg]). Each sample was analyzed in triplicate to minimize the error.
Antioxidant Activity Determination
DPPH Free Radical-Scavenging Method
DPPH determination was based on the methods of Brand-Williams et al. 29 and Thaipong et al. 30 with some modifications. Two mL of properly diluted extract was added to 2 mL of 40.4 mg/L DPPH solution. After vortex mixing, the solution was placed in the dark at 25°C for 30 min, and the absorbance value was determined at 515 nm with an UV-Vis spectrophotometer. The analysis curve for Trolox methanol solution with a concentration range of 2∼24 μM L−1 showed a linear relationship: Y = 169.5X + 0.223, (r = 0.996; Y stands for clearance rate; X represents different concentrations of Trolox [μM L−1]). The results are presented as µM of Trolox equivalents (TE) per g of extract weight (μM TE/g EW).
ABTS Free Radical-Scavenging Method
ABTS cation radical scavenging activity was measured using the method of Re et al, 31 with some modifications. At room temperature and in darkness, 7 mM L−1 potassium persulfate and 5.2 mM L−1 ABTS were used to prepare ABTS·+ stock solution. The working solution of ABTS·+ was diluted with ethanol and the absorbance at 734 nm was 0.70 ± 0.05. Properly diluted solution (500 µL) was introduced into 4.5 mL diluted ABTS·+ solution. After vortexing and mixing, the solution was left in the dark for 30 min. The absorbance value was determined at 734 nm. Within the range of 100∼2000 μM L–1, the standard curve Y = 50.509X + 2.5927 (r = 0.998; Y stands for clearance rate; X represents the concentration of Trolox solution at different concentrations [μM L−1]) showed a linear relationship with Trolox (water-soluble vitamin E). The measurement results are indicated as µM of Trolox equivalents (TE) per g of extract weight (μM TE/g EW).
FRAP Ferric Reducing Antioxidant Power Method
FRAP analysis was used as described by Lahbib et al. 32 The stock solution included 0.3 M L−1 acetic acid buffer solution (pH = 3.6), 10 mM L−1 TPTZ solution (prepared with 40 mM L−1 hydrochloric acid) and 20 mM L−1 FeCl3•6H2O solution. The above three solutions were mixed at 10:1:1 volume ratio (considering the current use, 25 mL acetic acid buffer, 2.5 mL TPTZ solution, as well as 2.5 mL FeCl3•6H2O solution were selected, respectively) to prepare the working solution. The diluted extracts (200 μL) were reacted with 4 mL FRAP solution, and placed in the dark condition for 30 min after vortex mixing. The absorbance was determined at 593 nm. In the range of 20 ∼ 240 μM L–1 concentration, the analysis curve was drawn with Trolox ethanol solution (Y = 0.0019X + 0.4635, r = 0.997; Y is absorbance; X is the concentration of Trolox solutions at different concentrations [μM L−1]). The results are indicated as µM of Trolox equivalents (TE) per g of extract weight (μM TE/g EW).
Results and Discussion
Main Factors Screening
Design Expert 8.0.6 software was adopted to screen the main influence factors, and the results of Plackett-Burman test were analyzed. The CGA content of the extract and the influence degree of various factors on the content of the extract are shown in Table 2.
Results of Plackett-Burman Design Test.

Abbreviations: A presents ethanol concentration (%); B presents solvent pH; C presents extraction time (min); D presents number of extractions; E presents powder size (mesh); F presents solid-liquid ratio (g/ml); G presents temperature (°C);and H、I、J、K stand for four virtual factors. Y presents chlorogenic acid yield (mg/g); X presents the effects of various factors on the extraction of chlorogenic acid (%).
Among the seven factors, the results showed that ethanol concentration, solvent pH value and powder size had great influence on the extraction rate of CGA, and the influence of ethanol concentration on the CGA yield was 84.98%. The effect rate on the results of solvent pH on the extraction of CGA was 5.74%, and that of powder size was 5.47%. The influence rate of the other four practical factors was less than 2%, indicating that solid-liquid ratio, extraction time, number of extractions and extraction temperature had little influence on the actual results. Therefore, the Box-Bohnken experimental design was optimized by selecting ethanol concentration, solvent pH value and powder size as three factors.
Box-Bohnken Optimization of Extraction Parameters
Model Fitting
According to the principle of Box-Behnken, the three main factors of ethanol concentration, solvent pH value and powder size selected from the experimental design of Plackett-Burman were optimized at three levels, and the other factors were optimized from the point of view of energy saving. The low level in the two-level experimental design was selected to carry out the experiment, and the results are indicated in Table 3. The variance analysis of the experimental results is indicated in Table 4. The determination coefficient (R2) of the model is 0.9874, indicating that 98% of the response variability can be explained by the response surface model developed. A higher F value indicates that a certain factor has a higher significant influence on the corresponding response. The model F value was acquired from the formula of F = MSF/MSE, where, MSE and MSF are mean square of error and mean square of factor, respectively. The P value was employed to check the significance of each variable, which is necessary to understand the pattern of interactions between factors. The smaller the P value, the more significant the corresponding coefficient. An independent variable or interaction between variables with a P value lower than 0.05 indicated a significant effect on CGA extraction yield. In this case, A, B, C, AB, A2, B2 and C2 had significant effects on the response studied, namely CGA extraction yield. Referring to the results of high R2 and significance of P value, ethanol concentration was the main contributor to CGA yield among the three variables.
Box-Behnken Design Matrix and Their Corresponding Chlorogenic Acid Extraction Yields From Eucommia Ulmoides Leaves.

ANOVA Results of Box-Behnken Design Quadratic Model.

Abbreviations: A presents ethanol concentration (%); B presents solvent pH; C presents powder size (mesh).
The quadratic model designed by Box-Behnken was employed to obtain the response surface image of the main mutual effects between the three independent variables. Through regression analysis, the corresponding second-order mathematical model for predicting the available extraction yield is given as follows:
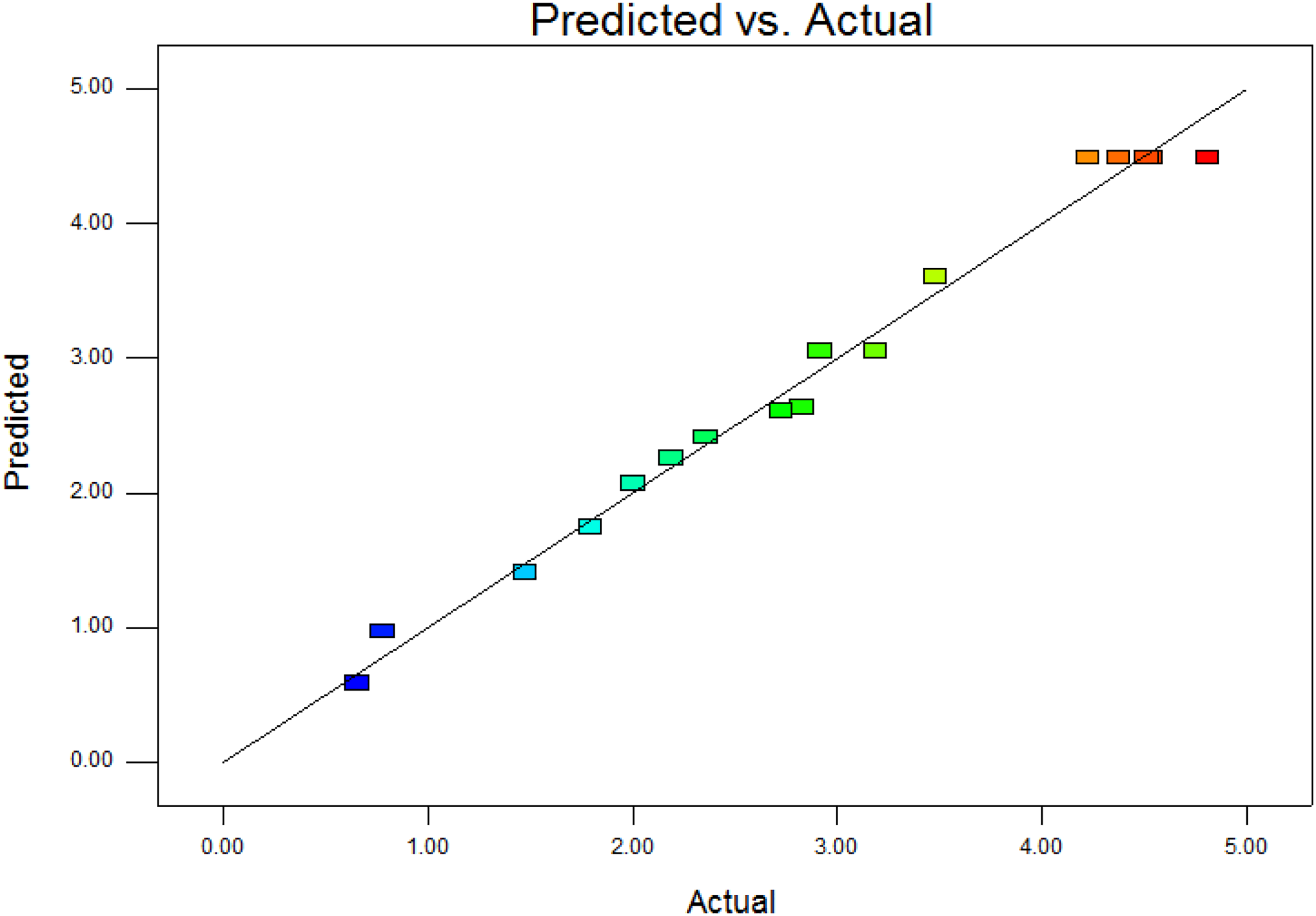
Correlation graph between the actual and predicted yield.
Influence of Factors
Figure 3(a) to (c) show the 3D response surface diagrams of the extraction yield corresponding to the interaction effects. These graphs were obtained by fixing each factor in turn at its central level and changing the other two to better understand the interaction between the variables and to determine the optimal factor value for the maximum extraction yield.

3D Response surface diagrams demonstrating the effect of the interaction between two independent variables on the CGA yield. (a) 3D response surface plots of solvent concentration and solvent pH value (A × B), (b) 3D response surface plots of solvent concentration and powder size (A × C), (c) 3D response surface plots of solvent pH value and powder size (B × C).
Figure 3(a) shows the mutual effect between ethanol concentration and solvent pH on CGA yield when powder size is fixed at the 60 destination center level. Thus, the mutual effect of ethanol concentration and solvent pH was significant. The CGA yield continued to increase as the ethanol concentration increased from 40% to 50%, but when the concentration increased from 50% to 60%, the yield decreased. The effect of ethanol concentration can be illustrated by the principle of a similar polar solvent dissolving a similar polar solute. CGA is an ester formed by caffeic acid and quinic acid. Its molecular structure includes an ester bond, and unsaturated double bond and is a polyphenol. In the process of extraction from plants, isomerization usually occurs through hydrolysis and migration of molecular lactone groups. Since CGA can be in a molecular form under acidic conditions, it has good stability at pH = 3, and the yield is higher when the polarity of the extraction solvent matches that of the compound. The experimental results showed that the solubility of CGA increased within a certain concentration range of ethanol and water, thus increasing the extraction yield.
Figure 3(b) demonstrates the mutual effect between ethanol concentration and powder size on CGA yield when the solvent pH was fixed at the central level. It can be seen that compared with the concentration of ethanol, the particle size has little influence on the extraction rate. When the particle size increased from 40 mesh to 60 mesh, the yield increased continuously, but when it continued to rise from 60 to 80 mesh, the yield decreased slowly. The effect of powder size on the yield could come from two aspects. On the one hand, the finer the powder size, the better the contact with the extraction solvent and the higher the yield of CGA. With the increase of particle size, this effect will become gradually insignificant. On the other hand, the further reduction of particle size will also lead to local boiling during the extraction process, resulting in the loss of raw materials. Although the ethanol concentration and particle size had significant influence on CGA yield, the interaction between them was not significant.
The influence of pH value of solvent and powder size with the ethanol concentration fixed at its central level is represented in Figure 3(c). It was shown that the yield of CGA is sensitive to changes in powder size and solvent pH value. However, the interaction between the two factors was not significant. The yield was highest when pH = 3 and powder size was 60 mesh.
Model Validation
The optimum extraction conditions of CGA were as follows: ethanol volume fraction 53.17%, solvent pH value 3.11, and powder size 63.81 mesh. Under optimized conditions, the maximum predicted yield of CGA was 4.50 mg/g. Considering the operability in actual large scale production, the optimization conditions were modified to: ethanol concentration 50%, solvent pH value 3, and particle size 60 mesh. Under this condition, the average experimental value was 4.36 ± 0.23 mg/g (n = 3), and the predicted value had a good correlation with the experimental value, indicating that the RSM model could better reflect the expected optimization effect.
Antioxidant Activity of Chlorogenic Acid-Rich Extracts
DPPH, ABTS and FRAP assays were adopted to evaluate the antioxidant activity of extracts, as shown in Table 5. The extract of E. ulmoides leaves rich in CGA showed the highest antioxidant activity in the FRAP method, which was 219.8 μM TE/g EW. The DPPH method gave a similar value (168 μM TE/g EW) to the ABTS method (152 μM TE/g EW).
Antioxidant Activity of Chlorogenic Acid-Rich Extracts From Eucommia ulmoides Leaves Determined by the DPPH, ABTS, and FRAP Assays.

Abbreviations: TE/g EW presents Trolox equivalents per gram of extract weight.
The difference in results is due to the different principles of the various methods. DPPH and ABTS were used to determine the free radical scavenging capacity. For DPPH, if an antioxidant is present, it will pair with the free single electron of DPPH•, reducing its absorption peak and changing the color of the solution from dark purple to light purple or light yellow. The colorimetric change is positively correlated with the antioxidant activity of the solution. In the ABTS method, a stable blue-green cationic radical ABTS·+ is generated in the reaction system. Also, the absorbance of ABTS·+ decreased after the reaction of antioxidants with the free radicals that allows the assessment of the antioxidant capacity of chlorogenic acid-rich extracts. The FRAP analysis is considered as the dynamic reaction between the metal iron ion in the oxidation state and reduction state. The principle of the FRAP method to determine the total antioxidant capacity is that, under acidic conditions, the tripyridine-ferric complex (Fe3+-TPTZ) is reduced into blue-purple tripyridine-ferrous (Fe2+-TPTZ). The absorbance is measured at 593 nm, which can be considered as an indicator of the total antioxidant capacity of the sample. The higher the absorbance, the better the antioxidant activity. However, not all reductants that reduce Fe3+ are antioxidants. 33
Ultrasonic extraction of CGA from E. ulmoides leaves has been reported, and Soxhlet, reflux heating and ultrasound-assisted extraction were compared for the yield of CGA from E. ulmoides leaves. In this study, the content of CGA obtained by the response surface optimization extraction process combined with two-phase separation technology was 10 times higher than that reported in the literature. 34 Three different in vitro assays were used to express the antioxidant activity of the obtained chlorogenic acid-rich extract, using Trolox as the equivalent control.
Conclusion
So far, reports on antioxidant activity of E. ulmoides leaves mostly focused on the mixture of polysaccharides, flavonoids and phenolic components,35‐37 while studies on CGA in plants and its antioxidant activity are mostly found in species such as honeysuckle and topinambur.38,39 In this paper, we employed Plackett-Burman combined with Box-Behnken experimental designs to optimize the enrichment process of CGA in E. ulmoides leaves. Then the antioxidant activity of the optimal extract (ethanol concentration 50%, solvent pH value 3, and particle size 60 mesh) obtained from E. ulmoides leaves was proved by in vitro assays, namely ABTS, DPPH and FRAP methods. This study provided a scientific basis for the extraction of CGA with strong antioxidant activity from E. ulmoides leaves.
Footnotes
Acknowledgments
This research is supported by a grant from the Natural Science Foundation of Henan Province of China (No.162300410104), Science and Technology Planning Project of Henan Province of China (No.182102311125) and Doctoral Research Foundation Project (No.13480058).
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Hua Jiang: Methodology, Software, Writing - Original Draft, Supervision, Writing - Review & Editing; Jun Li: Conceptualization, Methodology, Resources, Project administration, Writing - Review & Editing, Funding acquisition; Ning Zhang: Validation, Investigation, Data Curation; Hai-Yang He: Validation, Investigation, Data Curation; Jia-Min An: Validation, Investigation; Ya-Ning Dou: Validation, Investigation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Doctoral Research Foundation Project (grant number No.13480058, No.182102311125, No.162300410104).
Trial Registration
Not applicable, because this article does not contain any clinical trials.
