Abstract
Background
Myocardial ischemia-reperfusion (MI/R) injury is a leading cause of myocardial damage, characterized by apoptosis and impaired cardiac function. Dezocine, a mixed opioid receptor agonist/antagonist, has shown potential in mitigating MI/R injury, but its underlying mechanisms remain unclear.
Objectives
This study aimed to evaluate the effects of dezocine on myocardial cell apoptosis in rats with MI/R injury, focusing on the TLR4/NF-κB signaling pathway.
Materials and Methods
Thirty-six Sprague-Dawley rats were randomly divided into three groups: sham, model, and dezocine (n = 12 each). The sham group underwent thoracotomy without MI/R injury. The model group received normal saline prior to MI/R injury induction, while the dezocine group was administered dezocine. Toll-like receptor 4 (TLR4) and nuclear factor kappa B (NF-κB) p65 expression were analyzed using immunohistochemistry. Western blotting quantified Bax and Caspase-3 protein levels while quantitative polymerase chain reaction assessed their mRNA expression. Cell apoptosis was evaluated via terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay.
Results
TLR4 and NF-κB p65 expression were significantly elevated in the model and dezocine groups compared to the sham group but notably lower in the dezocine group versus the model group. Bax and Caspase-3 protein and mRNA levels followed a similar trend. TUNEL assay results demonstrated higher apoptosis rates in the model and dezocine groups compared to the sham group, with the dezocine group showing a marked reduction in apoptosis compared to the model group (p < 0.05).
Conclusion
Dezocine suppresses the TLR4/NF-κB pathway, reducing myocardial apoptosis in MI/R injury, suggesting its potential as a therapeutic agent in mitigating cardiac damage.
Introduction
Myocardial ischemia-reperfusion (MI/R) injury, often seen in MI patients, leads to damage to heart tissue, apoptosis, and even necrosis of myocardial cells, severely disrupting the myocardium’s normal structure and function (Tan et al., 2024; Yu et al., 2024). Evidence suggests that I/R injury to the myocardium significantly influences MI. In cases of severe injury, it can provoke massive fibrosis within the myocardial tissue, causing myocardial cell death (apoptosis), which intensifies the damage and impedes the healing process post-injury (Xu et al., 2024; Zhang et al., 2024). Current therapies, including pharmacological agents and mechanical interventions, have shown limited success in preventing myocardial cell death and ensuring full cardiac recovery. A major limitation is their inability to effectively address apoptotic mechanisms and inflammatory cascades, which play pivotal roles in exacerbating myocardial damage. Therefore, there is an urgent need to explore alternative therapeutic strategies targeting apoptosis and related pathways.
Recent studies highlighted the pivotal involvement of the TLR/NF-κB in modulating critical cellular processes like proliferation, apoptosis, and necrosis, especially in the context of MI/R injury (Ye et al., 2017; Zhang et al., 2017). In the context of MI/R injury, cytokines initiate the TLR4/NF-κB cascade, influencing the expression of apoptosis-related proteins such as Caspase-3 and Bax, which enhances cell death and aggravates the injury (Citalán-Madrid et al., 2017; Xu et al., 2017).
Dezocine, a mixed agonist/antagonist of opioid receptors, has been widely applied in clinics for its favorable analgesic effect (Li et al., 2017; Wang et al., 2017). Additionally, dezocine can improve MI/R injury well, but its mechanism of action remains elusive. This research aims to investigate the influence of dezocine on myocardial cell apoptosis in rats with MI/R injury, focusing on the TLR4/NF-κB signaling pathway to elucidate the mechanism involved.
Materials and Methods
Grouping and Treatment of Animals
A total of 36 specific pathogen-free (SPF) Sprague-Dawley (SD) rats, aged 1 month, were obtained from Shanghai SLAC Laboratory Animal Co., Ltd. Using a random number table, the rats were assigned into three groups: sham, model, and dezocine, with 12 rats in each group. They were housed in the animal facility under controlled conditions, receiving standard feed and sterilized filtered water, with a 12-h light/dark cycle and regulated temperature and humidity. The rats underwent a 7-day acclimation period before the start of the experiment.
In the sham group, the rats only underwent thoracotomy without MI/R injury. In the model group, the rats were first administered with normal saline by femoral vein drip at 5 min before the preparation of MI/R injury. Then, the model was established via MI for 30 min and reperfusion for 120 min. Finally, the materials were sampled. Besides, in the dezocine group, rats were given dezocine (0.5 mg/kg) via the femoral vein 30 min before inducing MI/R, followed by ischemia for 30 min and reperfusion for 120 min before tissue collection.
Establishment of MI/R Injury Model
An intraperitoneal injection of 5 mL/kg of 7% chloral hydrate was administered to the rats to induce anesthesia. Following this, the chest area was depilated to expose the skin and was then disinfected. The rats were restrained, and the chest was opened with scissors to reveal the heart, locate the left anterior descending artery, and perform ligation at the middle section. The electrocardiogram revealed signs of MI, including a pale left anterior wall of the heart, faint heartbeats, ST-segment elevation, and sharp T-waves. The blood supply to the left anterior descending artery was reinstated after 45 min of ischemia once the blockage was cleared. The rats’ health was carefully tracked, and once they were stable, the wound was sutured in multiple layers. Finally, the rats were housed individually in separate cages.
Sampling
Following the induction of anesthesia and blood collection from the abdominal aorta, cardiac tissues were removed from six rats per group, washed with saline, and frozen at –80°C in Eppendorf tubes for Western blot and quantitative polymerase chain reaction (qPCR) evaluations. Perfusion fixation was performed on the remaining six rats from each group for sampling purposes. The rats underwent a thoracotomy to expose the heart, followed by the administration of 400 mL of 4% paraformaldehyde to the left atrial appendage. The heart tissue was removed, placed in a 4% paraformaldehyde solution, and fixed for immunohistochemistry and terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assays.
Immunohistochemistry
Tissue samples embedded in paraffin were sliced into 5 µm sections and placed in water heated to 42°C for mounting. The sections were deparaffinized by soaking in xylene and graded ethanol. Sections underwent antigen retrieval by heating in citrate buffer in a microwave for three 3-min cycles, followed by a 5-min incubation. Endogenous peroxidase activity was blocked with a blocking agent for 10 min, followed by washing. After incubating the sections with primary antibodies against toll-like receptor 4 (TLR4) (1:200) and nuclear factor kappa B (NF-κB) p65 (1:200) at 4°C overnight, they were washed and then incubated with the secondary antibody for 10 min. The sections were further treated with streptavidin-peroxidase for 10 min, and finally, 3,3′-diaminobenzidine (DAB) was applied for the chromogenic reaction. Hematoxylin was used for counterstaining, and the slides were sealed and observed under a microscope.
Western Blotting
Protein lysates were prepared from heart tissues stored at ultra-low temperatures, incubated on ice for 1 h, and then centrifuged at 14,000 g for 10 min. The concentration of the protein was measured using the bicinchoninic acid (BCA) method. After denaturation, proteins were separated using sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and subsequently transferred onto polyvinylidene fluoride (PVDF) membranes, which were then blocked for 1.5 h using a blocking buffer before being incubated with primary antibodies (anti-Bax and anti-Caspase3, both at 1:1,000) and a secondary antibody (1:1,000). For Western blotting, β-actin was used as a housekeeping protein to normalize protein levels, ensuring the reproducibility and accuracy of the results. After thorough washing, chemiluminescent reagents were used to develop the signal in the dark for 1 min.
qPCR
RNA was isolated from heart tissue samples, and cDNA was synthesized using a reverse transcription kit. qPCR was performed in a reaction volume of 20 µL. The PCR program included an initial step of 5 min at 53°C, followed by 10 min at 95°C, 10 s at 95°C, and 30 s at 62°C, with a total of 35 cycles. Gene expression levels were determined with the ∇Ct method. Primer sequences are provided in Table 1.
List of Primer Sequences.

TUNEL Apoptosis Assay
Paraffin-embedded tissue was sectioned and deparaffinized as described above. TdT enzyme reaction buffer was applied for 1 h in the dark, followed by 15 min of deionized water incubation to terminate the reaction. To block endogenous peroxidase activity, the sections were treated with the working solution for 60 min. DAB chromogenic solution was added, followed by rinsing and mounting for observation.
Statistical Analysis
SPSS version 20.0 was adopted for statistical analyses, and the data were expressed as mean ± standard deviation. In cases of normally distributed data with equal variance, a t-test was carried out; for data with unequal variances, an adjusted t-test procedure was used. The analysis of data that was not normally distributed was carried out using non-parametric tests. Rank-sum tests were applied to ordinal data, and chi-square tests were used for categorical data.
Results
Immunohistochemistry Detection Results
The sham group exhibited fewer positive expressions of TLR4 and NF-κB p65 compared to the model and dezocine groups, where more tissues showed positive staining. Statistical analysis (Figure 1) indicated that the mean optical density values for TLR4 and NF-κB p65 expressions were significantly higher in the model and dezocine groups than in the sham group (p < 0.001). Moreover, the optical density in the dezocine group was notably lower than that in the model group (p < 0.001).
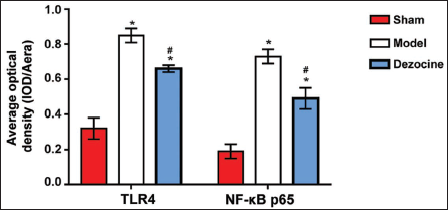
Relative Protein Expression via Western Blotting
Bax and Caspase-3 protein levels were lower in the sham group, whereas the model and dezocine groups demonstrated an increase in their expressions. Statistical analysis revealed that the relative protein levels of Bax and Caspase-3 were significantly elevated in the model and dezocine groups compared to the sham group (p < 0.001). However, these protein levels were significantly reduced in the dezocine group compared to the model group (p < 0.001) (Figure 2).
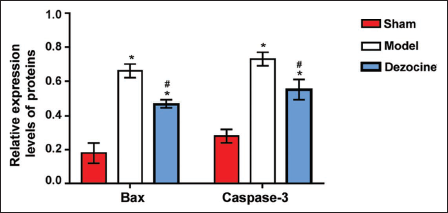
qPCR Results
Bax and Caspase-3 mRNA expression levels were higher in the model and dezocine groups compared to the sham group (p < 0.001). However, the dezocine group showed markedly lower expression levels than the model group (p < 0.001) (Figure 3).
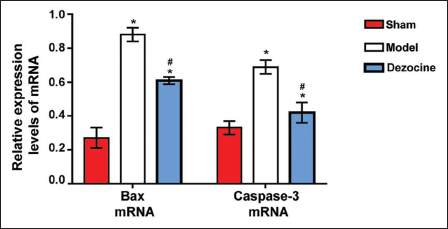
Apoptosis Rate via TUNEL Assay
The sham group exhibited a significantly lower number of apoptotic cells compared with the model and dezocine groups, which showed a higher incidence of apoptosis. Statistical analysis indicated that the cell apoptosis rates in the model and dezocine groups were significantly elevated relative to the sham group (p < 0.001). Furthermore, the dezocine group exhibited a significantly lower apoptosis rate compared with the model group (p < 0.001) (Figure 4).
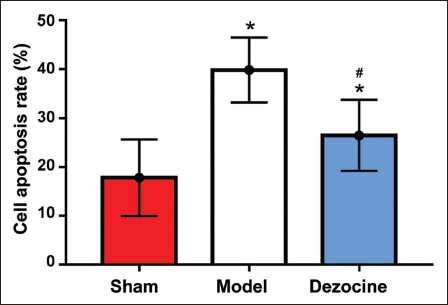
Discussion
I/R injury, as a pivotal pathological process in MI, has vital influences on the physiological and pathological states of myocardial cells, especially a crucial regulatory effect on myocardial cell apoptosis (Yang & Zhang, 2023; Yanpiset et al., 2023). Massive myocardial cell apoptosis can be regarded as one of the major pathological injuries in I/R injury, and effectively reducing myocardial cell apoptosis can facilitate the repair of myocardial tissues with I/R injury after MI. Recent studies (Ferdinandy et al., 2023; Wang et al., 2023) have shown that after MI, I/R injury triggers the release of numerous damage-associated factors in myocardial tissues, which in turn activate various downstream signaling cascades, such as the TLR4/NF-κB pathway. This pathway plays a key role in regulating apoptosis and has significant physiological and pathological implications in I/R injury following MI, contributing to the apoptosis of cells induced by I/R damage. As a crucial inflammation-associated portal protein in human bodies, TLR4 can modulate the downstream target proteins, such as NF-κB, thereby regulating multiple biological effects. Studies have manifested that (Alatorre et al., 2018; Shah et al., 2018) active TLR4 can be abnormally highly expressed to mediate inflammation and upregulate the expression and release of various inflammatory factors. Besides, the aberrantly high expression of TLR4 can promote NF-κB nuclear translocation to modulate the further release of the downstream inflammatory factors so that a vicious circle is formed to promote the transcription and translation of the downstream apoptosis-associated target genes and proteins and regulate the pathological reactions in apoptosis. As a positive regulator of apoptosis, Bax, when overexpressed, can cause a shift in the equilibrium between pro-apoptotic and anti-apoptotic proteins, triggering a rise in Caspase-3 activity and accelerating apoptosis (Yip & Malaspina, 2012; Zhang et al., 2013). The results indicated a marked alteration in TLR4 and NF-κB expression, alongside an increase in Bax and Caspase-3 in myocardial cells in rat heart tissue, suggesting that MI/R injury triggers the TLR4/NF-κB signaling pathway, which plays a crucial role in promoting myocardial cell apoptosis.
Dezocine has a favorably potent analgesic effect and an effective protective effect on tissues, thereby improving MI/R injury (An et al., 2017; Feng et al., 2017). The present investigation further validates that dezocine can significantly lower the expression of apoptosis effector molecules, Bax and Caspase-3, after MI/R, consequently reducing myocardial cell apoptosis. Moreover, TLR4 and NF-κB, the key molecules in the TLR4 and NF-κB signaling pathway closely related to apoptosis, were detected to investigate the relevant mechanism of action of dezocine. The use of dezocine was found to significantly decrease the expression of TLR4 and NF-κB in the myocardium after I/R injury, pointing to its role in inhibiting these signaling pathways. This action may be responsible for the attenuation of myocardial cell apoptosis observed following I/R injury.
Despite the promising results, certain limitations of this study warrant consideration. First, the experimental model employed in this study involves acute MI/R injury in rats, which may not fully capture the complexity of human myocardial infarction. Additionally, while the focus on the TLR4/NF-κB pathway provides valuable mechanistic insights, other signaling pathways contributing to apoptosis in I/R injury were not investigated. For instance, the PI3K/Akt and JNK pathways are known to play critical roles in myocardial apoptosis and may interact with TLR4/NF-κB signaling. Future research should aim to elucidate these interactions to provide a more comprehensive understanding of the mechanisms underlying I/R injury and dezocine’s protective effects.
Furthermore, the dose and timing of dezocine administration were optimized for this study based on prior literature. However, a detailed pharmacokinetic and pharmacodynamic analysis is necessary to establish the optimal therapeutic regimen for clinical applications. Clinical trials would also be essential to confirm the translational potential of dezocine in mitigating I/R injury in MI patients.
The potential clinical implications of these findings are significant. Given that apoptosis and inflammation are key contributors to myocardial damage, dezocine’s ability to target both processes positions it as a promising candidate for adjunctive therapy in MI/R management. Importantly, dezocine’s dual action as an opioid receptor agonist/antagonist offers additional benefits in managing perioperative pain, which is a common concern in cardiac patients.
However, translating these preclinical results into clinical settings requires further investigation. Specifically, clinical trials are necessary to evaluate the safety, efficacy, and optimal dosing of dezocine in humans. Understanding its long-term effects, particularly in preventing post-MI complications, is crucial to integrating it into existing treatment protocols.
There are several limitations to consider when interpreting these findings. First, this study was conducted in a rat model, which may not fully replicate human pathophysiology. Second, the duration of treatment was limited to the acute phase of MI/R injury. Future studies should explore the chronic effects of dezocine and its potential interactions with other therapeutic agents.
Unlike traditional therapies like beta-blockers and calcium channel blockers, which primarily target hemodynamic regulation, dezocine’s mechanism of action uniquely focuses on apoptotic pathways and inflammation through TLR4/NF-κB modulation. Unlike conventional opioids, dezocine demonstrates additional anti-inflammatory properties, making it a potentially superior option for comprehensive myocardial protection.
In addition, it is important to investigate whether dezocine affects other signaling pathways involved in myocardial protection, such as the PI3K/Akt and JNK pathways. This will provide a more comprehensive understanding of its mechanism of action and potential off-target effects.
Future research focused on clinical trials to determine the efficacy of dezocine in humans is required. Dose–response relationships, long-term outcomes, and potential combination therapies should be explored. Such studies could guide the development of optimized therapeutic protocols and potentially broaden the clinical applications of dezocine beyond cardiac protection.
Conclusion
Our study highlights the therapeutic potential of dezocine in reducing myocardial apoptosis following I/R injury by targeting the TLR4/NF-κB signaling pathway. These findings not only provide a foundation for further exploration of dezocine in cardiac protection but also underscore the importance of addressing inflammation and apoptosis in the management of I/R injury. Future investigations should aim to expand on these findings by exploring additional pathways, conducting long-term studies, and evaluating dezocine’s efficacy in clinical settings.
Footnotes
Abbreviations
MI/R: Myocardial ischemia-reperfusion; NF-κB: Nuclear factor kappa B; SD: Sprague-Dawley; SPF: Specific pathogen-free; TLR: Toll-like receptor; TUNEL: Terminal deoxynucleotidyl transferase dUTP nick end labeling.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the Animal Ethics Committee of The First Affiliated Hospital of Hunan Normal University Animal Center (Approval No. 2025-003).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
