Abstract
Background
The rate of disability and mortality associated with cerebral ischemia/reperfusion injury (CIRI) is high due to limited treatment options, making it a major challenge to clinical management. Calycosin is a biologically active compound hostile to inflammatory, neuroprotective, and tumor effects. Whether calycosin has an ischemia/reperfusion effect or mechanism is unclear.
Materials and Methods
For in vivo experiments, we randomly divided rats into five groups: blank control group, middle cerebral artery occlusion/reperfusion (MCAO/R) surgical group, calycosin + MCAO/R group (5 mg/kg), calycosin + MCAO/R group (10 mg/kg), and calycosin + MCAO/R group (20 mg/kg). Molding of the middle cerebral artery was performed. Calycosin’s neuroprotective effects were evaluated using the neurological deficit score, brain edema rate, and cerebral infarct volume. For in vitro experiments, we divided PC12 cells into five groups: blank control group, oxygen and glucose deprivation/reperfusion (OGD/R) group, calycosin + OGD/R group (1 × 10−6 mol/L), calycosin + OGD/R group (4 × 10−6 mol/L), and calycosin + OGD/R group (16 × 10−6 mol/L). The optimal concentration of calycosin on PC12 cells was determined using the cell counting kit-8 (CCK-8) cell activity assay. The expression of nuclear factor kappa-B (NF-κB)-related factors was detected using real-time quantitative polymerase chain reaction and Western blotting.
Results
In rats, the MCAO/R model resulted in elevated neurological deficit scores, increased brain infarct volumes, and increased brain edema rates. The OGD/R model decreased rat adrenal pheochromocytoma (PC12) cell activity, and calycosin had a significant cerebral protective effect on PC12 cells under OGD/R conditions. In addition, calycosin can inhibit the activation of the NF-κB pathway, and its neuroprotective effect may be related to the NF-κB pathway.
Conclusion
Calycosin can reduce focal CIRI, and the neuroprotective effect of calycosin may be related to the inhibition of the high mobility group protein 1/toll-like receptor 4 (TLR4)/NF-κB signaling pathways.
Introduction
Stroke is an acute, critical illness caused by ischemia and hypoxia in brain tissue due to blockage of blood vessels or bleeding from ruptured cerebral blood vessels. It puts a serious weight on families and society because of its high mortality and inability rate (Chen et al., 2020). The types of strokes include ischemic strokes and hemorrhagic strokes. Revascularization is an effective treatment for ischemic stroke, but when blood flow is revascularized, it may cause ischemic/reperfusion (I/R) injury (Green 2008; Khatri et al., 2012). The pathophysiological processes of I/R injury include free radical generation (Wei et al., 1996), excitatory glutamate toxicity (Mayor & Tymianski, 2018), oxidative stress (Eltzschig & Eckle., 2011), inflammatory response (Shi et al., 2019), and apoptosis (Cherubini & Miles., 2015; Zhang et al., 2015). Treatments are selected according to their pathogenesis to reduce stroke and cerebral ischemia/reperfusion injury (CIRI).
Calycosin is an isoflavonoid phytoestrogen isolated from Astragalus membranaceus, one of the most active components of which is calycosin (Efferth et al., 2016). Calycosin has many organic and drug effects, including hostility to inflammation (Wang et al., 2022), neuroprotective (Yang et al., 2019), hostility to the tumor (Chen et al., 2015), antiviral (Zhu et al., 2009), and so on. The neuroprotective effects of calycosin have already been reported in different models (Liu et al., 2020; Qian et al., 2012; Schreihofer & Redmond, 2009). However, its therapeutic roles in ischemic stroke have been less well studied. Therefore, in this study, we assessed the anti-ischemic effects of calycosin in in vivo and in vitro models.
Inflammation is one of the key factors contributing to the pathophysiology of stroke. Its secondary damage to brain tissue caused by I/R injury in the brain is closely associated with inflammation (Jiang et al., 2014; Qiu et al., 2008; Tao et al., 2015). Past investigations have shown that irritation is a significant part of the pathogenesis of ischemic stroke (O’Connell et al., 2017; Laird et al., 2014; Xu et al., 2018). When I/R occurs, High Mobility Group Protein 1 (HMGB1) secretion, reactive oxygen species (ROS) production, glutamate toxicity, and Ca2+ overload promote complement and platelets, activate inflammatory transcription factors, and release inflammatory signals (Laird et al., 2014). Among these, nuclear factor kappa-B (NF-κB) is significant in neurodegeneration and neuroinflammation (Wang et al., 2020). Late examinations have demonstrated that atorvastatin can balance the NF-κB flagging pathway during cerebral I/R with its potential NF-κB upstream factors like toll-like receptor 4 (TLR4) and HMGB1. HMGB1 concentrations in plasma are significantly elevated in patients with cerebral ischemia (Goldstein et al., 2006) and are positively correlated with cerebral infarct volume. Previous studies have concluded that the HMGB1/TLR4/NF-κB signaling pathway contributes to traumatic brain and spinal cord injuries (Sun et al., 2020; Wang et al., 2020).
B-cell lymphoma-2 (Bcl-2) and Bcl-2-associated X (Bax) are key factors in apoptosis; Bcl-2 protein exerts anti-apoptotic effects, while Bax protein exerts proapoptotic effects. Bax binds to Bcl-2 to form a heterodimer and inhibits the anti-apoptotic effect of Bcl-2 (Klemm et al., 2008). In past studies, methylcobalamin inhibits neuronal apoptosis. It regulates Bcl-2 and Bax expression in neurons during CIRI (Li et al., 2021). Liu et al. showed that electroacupuncture might prevent brain I/R damage by regulating Bax and Bcl-2 expression, significant pathways in apoptosis, and altering extracellular regulated protein kinases (ERK1/2), c-Jun N-terminal kinase (JNK), and p38 mitogen-activated protein kinases (p38MAPK) signaling pathways (Liu et al., 2018). Bcl-2 is activated after I/R injury and safeguards neurons against oxidative harm and fiery reactions (Chen et al., 2022; Wang et al., 2018). Calycosin applies its neuroprotective impacts by constricting the inflammatory reaction and neuronal apoptosis activated by mind I/R and rat adrenal pheochromocytoma (PC12) cells under oxygen and glucose deprivation/reperfusion (OGD/R) conditions. Furthermore, the defensive effect of calycosin in cerebrum CIRI included actuation of the HMGB1/TLR4/NF-κB flagging pathway. Consequently, according to our results, calycosin may be a promising therapeutic strategy for ischemic stroke and CIRI.
Materials and Methods
Chemicals and Materials
Calycosin was obtained from Priva Biotechnology Co., China. Anti-HMGB1, anti-TLR4, anti-NF-κB, and anti-IκB were obtained from Abcam, UK. p-NF-κB and p-IκB were obtained from Santa Cruz Biotechnology, USA. Anti-Bcl-2 and anti-Bax were obtained from Proteintch Group, China. Anti-β-actin was obtained from ZSGB-Bio, China. Dulbecco’s Modified Eagle Medium (DMEM) was obtained from Hyclone Laboratories, USA. The incubator was obtained from Billups-Rothenberg, USA. The cell counting kit-8 (CCK-8) assay kit was obtained from Dojindo, China.
Animals and Animal Experiments
Sprague–Dawley rodents (SD) (n = 50), by rejecting the dead creatures and ineffective models, including those without localized necrosis or localized necrosis with discharge (220–230 g), were bought from Hunan Silaikejingda, China. All trial creature conventions were supported by the Creature Trial and Error Morals Advisory group of Guilin Clinical College and were led by the Creature Care and Use Committee rules of Guilin Clinical College. Rodents were set in a steady temperature (20℃–25℃), consistent mugginess (40%–60%) day/night cycle of 12/12 h, and an unlimited eating routine and water for a brief multiweek time before the trial. Calycosin was disintegrated in dimethyl sulfoxide and weakened with sterile to the ideal focus. Day-to-day intraperitoneal infusions were given for 14 sequential days. A brief center middle cerebral artery occlusion/reperfusion (MCAO/R) was laid out by blocking the center cerebral course in rodents concerning our past exploratory method (Guo et al., 2021). The 50 rats were haphazardly allotted into the accompanying five gatherings (n = 10): the blank group, MCAO/R group, MCAO/R with 5 mg/kg calycosin, MCAO/R with 10 mg/kg calycosin, and MCAO/R with 20 mg/kg calycosin.
Assessment of Neurological Deficit
In the 24 h following reperfusion, the rats in the MCAO/R model had their neurological deficiencies evaluated using the Longa neurological shortfall scale, and the results were scored as follows. A score of 0 indicates that there are no neurological issues; 1 for failure to fully extend the left front paw and mildly limited neurological deficiencies; 2 for moderate neurological deficits as the rats turned to the left when walking; and 3 for severe neurological deficits as the rats A score of 3 shows that the rat leans to the left side when walking, with severe neurological deficits; a score of 4 shows that the rat cannot walk spontaneously or loses consciousness (Longa et al., 1989).
Infarct Volume Measurement
Toward the end of the neurological deficiency scoring, the rodents were euthanized with 2% sodium pentobarbital, executed, and the cerebrums were immediately eliminated. The brains were placed in molds for coronal sectioning and incubated with a 2% 2,3,5-triphenyl-2H-tetrazolium chloride (TTC) solution. A 2% TTC solution was obtained from Solarbio, China. Image analysis was performed using ImageJ. ImageJ was obtained from the National Institutes of Health (NIH), USA.
Brain Water Content
The degree of cerebral edema is determined by measuring the water content of the brain using wet and dry methods. A swiftly removed brain is weighed on an electronic analytical balance to determine its wet weight. After drying for 24 h at 100°C, the brain is weighed again to determine its dry weight.
Cells Culture and OGD/R Modeling
PC12 cells were bought from the Chinese community for type culture assortment. The cells were refined in DMEM at 37℃, 5% CO2, and 100% moisture with 10% fetal bovine serum and 100 units/mL penicillin/streptomycin. The OGD/R model was established by simulating in vitro CIRI. A 24-h pretreatment with calycosin was performed on PC12 cells, and then, cultured PC12 cells were in sugar-free Earle’s balanced salt solution (EBSS) to induce OGD and incubated in a hypoxic state at 37℃, 5% CO2, and 95% N2 incubator for 2 h. Control PC12 cells were cultured in EBSS containing glucose. After the OGD, PC12 cells were transferred to a basal medium for PC12 cells and incubated for 24 h at 37°C in a standard simulating reperfusion in a humidified incubator.
Analyses of Cell Viability
Cell viability was determined using the CCK-8 assay kit according to the instructions provided. PC12 cells were briefly purified in 96-well plates (1×104/well) for 24 h. Then, for the next 24 h, PC12 cells were exposed to three combinations of trichothecene (1, 4, and 16 M). After drug treatment, PC12 cells were refined in OGD/R conditions. CCK-8 was added to the appropriate wells and incubated at 37°C shading for 4 h, trailed by absorbance estimation at 450 nm utilizing a compound marker (Bio-Rad Model 680, Bio-Rad, CA, USA).
Real-Time Quantitative Polymerase Chain Reaction (RT-qPCR)
After sampling brain tissue or PC12 cells from each group of the experiment, samples were treated with Trizol lysis (Invitrogen, USA); ribonucleic acid (RNA) was extracted from the samples, and NanoDrop 2000 was used to determine its concentration and purity (Thermo Fisher Logical, USA). The ABI PRISM 7500 Sequence Detection System (Applied Biosystems, USA) was used for all RT-qPCR procedures. The appropriate gene expression was measured using the 2-∇∇CT technique, with actin acting as a normalization control. Table 1 displays the primer sequences for each qRT-PCR analysis.
Primer sequences for RT-PCR assays.

Western Blotting
Cerebrum tissue or PC12 cell test gatherings were homogenized in radioimmunoprecipitation assay (RIPA) buffer, and all proteins were separated (Beyotime Biotechnology, China). The protein content of the examples was then resolved to utilize a bicinchoninic acid assay (BCA) pack (Beyotime Biotechnology, China). Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) used equal amounts of protein samples (20 mg/sample), and the proteins were transferred onto 0.45 mm polyvinylidene fluoride (PVDF) membranes (Bio-Rad Laboratories, USA). The essential neutralizer was shut with 5% skim milk and broke down in tris-buffered saline with tween 20 (TBST) at 37°C (cradled salt arrangement containing 0.1% Tween-20) for 2 h, then for the time being at 4°C. anti-HMGB1 anti-TLR4, p-NF-κB, anti-NF-κB, p-IκB, anti-IκB, anti-Bcl-2, anti-Bax, and anti-β-actin. Antibody-related content is displayed. Wash three times the following day with the TBST solution for 5 min. The Super Signal West Pico Western blot detection reagent (Bridgen, China) was used to separate protein groups after 2 h of room-temperature incubation with secondary antibodies. The separated protein groups were then captured using Image Lab software (Bio-Rad, USA).
Statistical Analysis
Data were examined using SE and SPSS 23, USA. One-way analysis of variation and post hoc Dunnett’s tests were used to compare the groupings statistically. A statistically significant difference was defined as p < 0.05.
Results
Calycosin’s Neuroprotective Effect in Focal CIRI
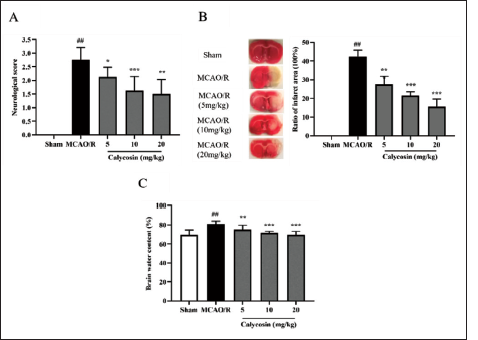
The neurological function scores of the rats in the MCAO/R model group were greater than those of the control group (p <0.05, Figure 1A). Rats in the MCAO/R model group demonstrated improved neurological function scores compared to the control group (p <0.05, Figure 1A). Calycosin gradually decreased with increasing drug concentration. Moreover, the rates of cerebral infarct volume (p <0.05, Figure 1B) and cerebral edema (p <0.05, Figure 1C) were essentially more excellent in the MCAO/R model group compared to the benchmark group. Brain infarct volume (p <0.05, Figure 1B) and cerebral edema rate were significantly higher in the MCAO/R model group in the calycosin administration group compared to the benchmark group. This suggests a role for calycosin in the rat brain I/R model (associated with neuroprotection).
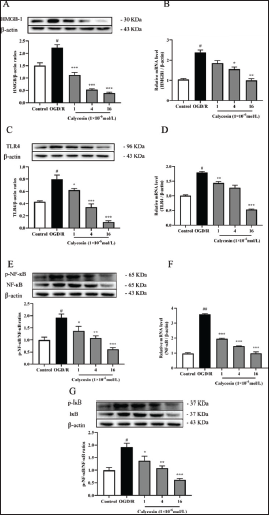
Calycosin Inhibits the HMGB1/TLR4/NF-kB Signaling Pathway to Attenuate the Inflammatory Response to CIRI
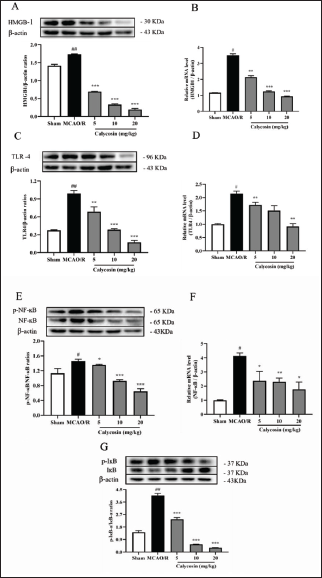
We further examined the expression levels of genes and proteins associated with inflammation since cerebral I/R damage can result in an inflammatory response (Franke et al., 2021). In the MCAO/R group compared with the sham operation group, the mRNA and protein levels of HMGB1, TLR4, p-NF-κB, NF-κB inhibitor kappa B alpha (IκB-α), and phosphorylation inhibitor kappa B alpha (p-IκB-α) were all significantly increased (p <0.05, Figure 2), and with increasing concentrations of calycosin administration HMGB1, the mRNA and protein levels of TLR4 and p-NF-κB decreased (p <0.05, Figure 2). The findings imply that calycosin lessens the inflammatory reaction to CIRI. It was postulated that calycosin could control the HMGB1/TLR4/NF-kB flagging pathway to reduce the inflammatory response of neuronal cells in CIRI.
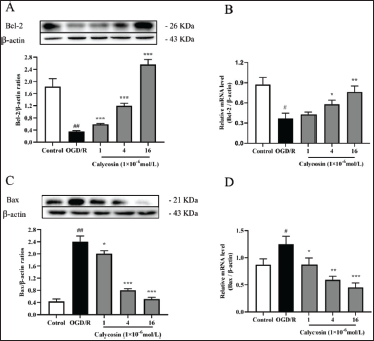
Calycosin Attenuates Neuronal Apoptosis After CIRI
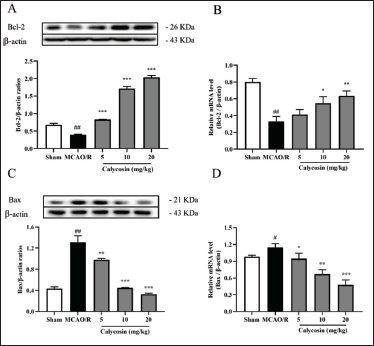
At 24 h post-MCAO reperfusion, we concentrated on the counter-apoptotic impact of calycosin. We looked at Bcl-2 and Bax’s mRNA and protein articulation. As displayed in Figure 3, compared to the sham group, rats in the MCAO/R model group had higher protein and mRNA levels of Bax and lower levels of Bcl-2 in their brain tissue. Still, this pattern was reversed after calycosin treatment (Figure 3). The effect of calycosin showed a dose-dependent trend, indicating that calycosin can reduce apoptosis of neuronal cells in CIRI.
Calycosin Enhances the Activity of PC12 Cells After OGD/R
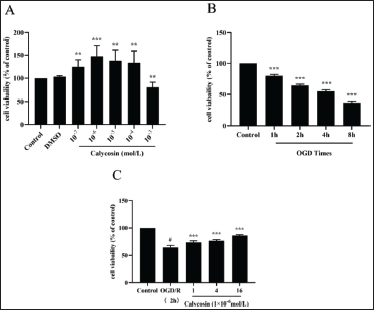
Next, we performed in vitro experiments with PC12 cells, which were treated with calycosin and OGD/R models after 24 h of culture, and CCK-8 measured cell viability. The results showed that calycosin promoted cell growth at 10−7 to 10−4 mol/L concentrations, with the most significant promotion at 10−6 mol/L (p <0.05, Figure 4A). Calycosin induced cell death when increased to 10−3 mol/L. Therefore, (1, 4, 16) × 10−6 mol/L was selected for subsequent experiments. By CCK-8 assay, PC12 cells were significantly damaged after 1–8 h under OGD conditions (Figure 4B). Then 2 h were chosen for additional research since there was only a minor degree of cell harm (64% viability) (Figure 4C). After 2 h of OGD, the cells were perfused for 24 h. The outcomes showed that calycosin might increase PC12 cell viability dose-dependently. Calycosin can thereby increase PC12 cell survival in OGD/R circumstances.
Calycosin Inhibits the HMGB1/TLR4/NF-kB Signaling Pathway, Thereby Attenuating the Inflammatory Response of PC12 Cells Under OGD Conditions
We conducted in vitro tests to get PC12 cells to examine the effects of calycosin on PC12 cells under OGD/R conditions to expand on those mentioned above. We found significantly increased protein levels of HMGB1, TLR4, p-IκB-α, and p-NF-κB after OGD/R treatment compared to controls (p <0.05, Figure 5), whereas HMGB1, TLR4, p-IκB-α, and p-NF-κB were dose-dependently reduced after calycosin treatment (Figure 5). RT-PCR results likewise showed that mRNA and protein articulation of these provocative elements were diminished by calycosin treatment, as was predictable from the in vivo results.
Following OGD/R Therapy, Calycosin Lowers Apoptosis in PC12 Cells
To thoroughly examine calycosin’s effect on apoptosis in PC12 cells under OGD/R conditions, we looked at the changes in Bcl-2 and Bax. Bax levels rose compared to the baseline group, and Bcl-2 levels fell in PC12 cells gathered for the OGD/R model. However, following calycosin treatment, the trend, as mentioned above, was reversed (Figure 6), and the above results were consistent with the in vivo experiments.
Discussion
Epidemiological findings recommend estrogens, and estrogen-like compounds are powerful agents to protect neurons from neuropathological and neurotoxic challenges and promote their survival (Engler-Chiurazzi et al., 2017; Ma et al., 2016; Simpkins et al., 2004). Calycosin, an isoflavone phytoestrogen isolated from Astragalus membranaceus, is found to possess various potential pharmacological activities. Calycosin exerted anti-tumorigenesis (Chen et al., 2015), anti-oxidation (Zhao et al., 2016), anti-virus (Wang & Zhao, 2016), and apoptosis-modulation effects (Zhu et al., 2009). In this study, we have analyzed the role of calycosin in cerebral I/R-induced SD.
As of late, many examinations have shown that ischemic stroke remains one of the main sources of death worldwide, with the primary drivers being cerebral ischemia and reperfusion injury (Tuttolomondo et al., 2011; Xie et al., 2018). The inability of the brain to supply blood adequately can lead to structural damage to brain tissue and a range of dysfunctions. Complete or severe ischemia for an extended period might result in tissue necrosis and neuronal death (Khoshnam et al., 2017). Blood–brain barrier breakdown and brain tissue edema frequently occur in the I/R region after brain I/R injury (Chomova & Zitnanova, 2016). Proinflammatory substances are also more likely to penetrate the tissues and worsen the injury because the blood–brain barrier is compromised (Durukan & Tatlisumak, 2007). Guo et al. (2012) showed that calycosin reduced neurobehavioral scores and brain infarct volume in rats after cerebral I/R. Our results showed calycosin can minimize I/R injury in animal models (Sato et al., 2011; Watanabe et al., 2007). Neuroinflammation and increased neuronal deficit scores were reported in the untreated MCAO-injured rats.
The most life-threatening complication of cerebral ischemia is cerebral edema, which causes 5% mortality in stroke patients (Thorén et al., 2017). Cerebral edema occurs due to blood–brain barrier disruption, causing infiltration and leading to increased intracranial pressure (Brogan & Manno, 2015; Jüttler et al., 2007). Therefore, treating cerebral edema is a critical step in preventing the complications induced by ischemic conditions. Calycosin treatment significantly decreased the percentage of water content in the brain of MCAO-injured rats, thereby decreasing the cerebral infarct percentage and neuronal deficiency in rats.
As an endogenous damage-associated molecular pattern, HMGB1 contributes to inflammatory and innate immune responses in various neurological disorders (Huang et al., 2020; Paudel et al., 2019). In inflammatory states, HMGB1 can be released passively from necrotic cells (Wang et al., 2020). TLR4 is a pattern recognition receptor that recognizes its endogenous ligand, HMGB1, to promote neuroinflammation. When CIRI occurs, HMGB1 binds to TLR4 to activate myeloid differentiation factor, leading to phosphorylation of NF-κB (IκB). NF-κB immediately dissociates from the complex and translocates into the nucleus. In the nucleus, NF-κB binds with specific DNA sequences and initiates gene transcription and expression of inflammatory response proteins, thereby promoting I/R injury (Tornatore et al., 2012). Several recent studies have reported that activation of the HMGB1/TLR4/NF-κB signaling pathway is responsible for the worsening of CIRI, and inhibition of this signaling pathway attenuates the injury (Zhang et al., 2020; Zhu et al., 2018). Our results confirmed increased TLR 4 protein expression, enhanced NF-κB and IκBα phosphorylation, and increased production of HMGB1 in MCAO/R brain tissues and in OGD/R PC12 cells, demonstrating activation of the HMGB1/TLR4/NF-κB signaling pathway.
A large number of studies have confirmed that apoptosis plays an important role in cerebral reperfusion injury. Apoptosis is the most critical molecular process in reperfusion injury. In fact, apoptosis is a key pathological mechanism in I/R-induced brain injury and neuronal cell loss (Thorburn, 2008). Sustained I/R accelerates irreversible apoptosis. Cell survival is substantially impacted by the interaction of the anti-apoptotic Bcl-2 family protein and the proapoptotic Bax protein on the outer mitochondrial membrane (Vela et al., 2013). Bcl-2 is an anti-apoptotic protein, a crucial protein in the Bcl-2 family that regulates apoptosis. It plays a significant role in advancing cell endurance and restraining the activity of supportive apoptotic proteins. Transgenic mice overexpressing Bcl-2 are tolerant of cerebral ischemia (Chen et al., 2000). Moreover, calycosin strongly protected rats with CIRI by increasing Bcl-2 expression and reducing apoptosis (Wang et al., 2014). The outcomes showed that calycosin diminished Bax’s outflow and expanded the expression of Bcl-2 in the cerebral cortex of OGD/R-actuated PC12 cells and rodents.
Conclusion
Overall, the results of this review indicate that calycosin has significant neuroprotective effects in cerebral ischemia and reperfusion injury. It may do this by directly or indirectly inhibiting HMGB1 emission and NF-κB nuclear movement in the brain tissue of MCAO/R rodents and OGD/R-activated PC12 cells. Hence, we propose that calycosin be regarded as an efficient pretreatment strategy to prevent ischemic stroke, characterized by delayed damage following acute and severe neuroinflammation. This is because calycosin modulates the interaction between HMGB1, TLR4, and NF-κB. Then, further research is required to fully understand calycosin’s mode of action before it is used to treat people experiencing ischemic attacks.
Footnotes
Abbreviations
MCAO/R: Middle cerebral artery occlusion/reperfusion; CIRI: Cerebral ischemia/reperfusion injury; RT-qPCR: Real-time quantitative; PC12: Rat adrenal pheochromocytoma; OGD/R: Oxygen and glucose deprivation/reperfusion; NF-κB: Nuclear factor kappa-B; HMGB1: High mobility group protein 1; TLR4: Toll-like receptor 4; ROS: Reactive oxygen species; Bcl-2: Bax B-cell lymphoma-2; BCL2: Associated X; ERK1/2: Extracellular regulated protein kinases; JNK: c-Jun N-terminal kinase; p38MAPK: p38 mitogen-activated protein kinases.
Acknowledgments
This work was supported by Guilin Medical University, China and Hainan University, Hainan Province. This work was supported by Guilin Medical University, China and Hainan University, Hainan Province. Yong Wang and Shifeng Wang are Co-first author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation (81860231), the Open Project Program of Guangxi Key Laboratory of Brain and Cognitive Neuroscience, Guilin Medical University (GKLBCN-20200108-02), and Guangxi University Young And Middle-aged Teachers Basic Ability Improvement Project (2021KY0515).
Statement of Informed Consent and Ethical Approval
All the animal experiments were conducted according to the animal ethical guidelines.
