Abstract
Background
The abnormal expression of metastasis-associated gene 1 (MTA1) is related to the proliferation of tumor cells. Curcumin lipid nanoemulsion can effectively inhibit the growth of cervical cancer cells.
Objectives
This study mainly explores whether curcumin lipid nanoemulsion can inhibit the disease process of cervical cancer by regulating MTA1.
Materials and Methods
20 µmol/L curcumin lipid nanoemulsion was used to intervene with Hela cells for 0, 6, 12, and 24 h, respectively. Observe cell proliferation and cycle after curcumin intervention at different concentrations and at different times, observe the protein expression of phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt)/mammalian target of rapamycin (mTOR) pathway factors, and verify that PI3K is the direct target of MTA1.
Results
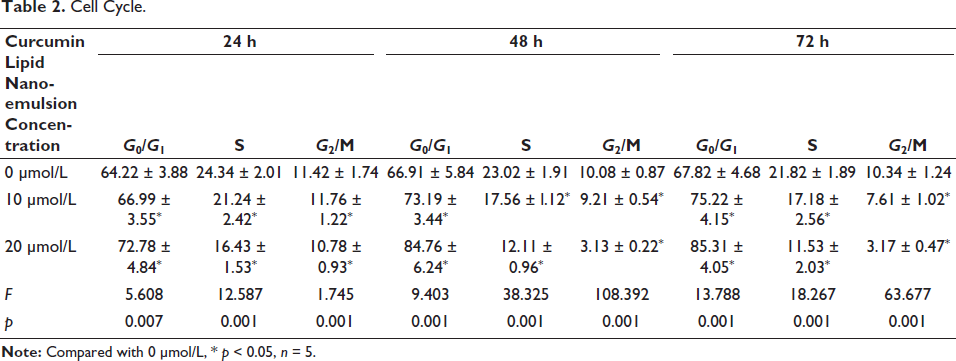
Curcumin lipid nanoemulsion dose-dependently inhibited cell proliferation with a more obvious at 10 µmol/L (p < 0.01). From 24 h onwards, cell number in the S phase in 20 µmol/L curcumin lipid nanoemulsion began to decrease significantly; cell number increased significantly in the G0/G1 phase and decreased in the G2/M phase (p < 0.05). The higher the concentration of curcumin lipid nanoemulsion, the higher the expression of the MTA1 inhibitor, showing a certain concentration and dose dependence. MTA1 can directly target PI3K. When the concentration of curcumin lipid nanoemulsion was 10 µmol/L, the expressions of PI3K, p-AKT, and p-mTOR decreased, and the decrease was more significant at 20 µmol/L (p < 0.01).
Conclusion
Curcumin lipid nanoemulsion can inhibit cervical cancer cell proliferation and control cell growth. Curcumin lipid nanoemulsion mainly acts through the MTA1/PI3K/Akt/mTOR axis. MTA1 negatively regulates the PI3K/Akt/mTOR pathway.
Keywords
Introduction
Curcumin is a common active compound (Yang, Han et al., 2021). Modern medical research shows that it mainly has anti-inflammatory and anti-hyperlipidemia functions. Curcumin drugs have anti-cancer effects (Yuan et al., 2022; Zheng et al., 2022), but the mechanism of their anti-cancer effects is still unclear. According to the study (Chang et al., 2021), curcumin mainly exerts anti-cancer effects through the following pathways, such as inhibiting deoxyribonucleic acid (DNA) methyltransferase; it can also modify histones; it can also regulate immune regulation, anti-oxidation, and interfering with hormone levels, and so on, pathways to exert anti-tumor effects. In addition, there are also studies that (Mahmoudi et al., 2022; Maleki Dizaj et al., 2022; Tiwari et al., 2014), curcumin drugs can regulate the expression of intracellular miRNA, control cell growth, and finally play an anti-cancer role.
Metastasis-associated gene 1 (MTA1) was identified in highly metastatic breast adenocarcinoma cells (Jia et al., 2019). Previous data have shown that MTA1 can act as a co-activator of the complex by interacting with histone deacetylases. Overexpression of MTA1 is related to the activities of some cancer cells (Guo et al., 2019), and it is found in many malignant tumor types. Among them, MTA1 overexpression was associated with higher tumor grade and poor prognosis (He et al., 2014). In cervical cancer cells, the MTA1 gene is abnormally expressed, and it is further found that MTA1 can act as a tumor suppressor gene, and its tumor suppressor mechanism is still unclear. Nicolson et al. (2003) found that the expression of MTA1 in cervical cancer was decreased. All the above studies indicated that the abnormal expression of MTA1 may be related to the progression of cervical cancer. However, the specific mechanism of action of MTA1 in cervical cancer remains unclear.
Phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt)/mammalian target of rapamycin (mTOR) is related to the progression of various tumor diseases and can participate in tumor cell proliferation. It shows that the PI3K/Akt/mTOR pathway may be linked to cervical cancer proliferation and cell cycle (Bossler et al., 2019; Sun et al., 2020; Zhang et al., 2021). MTA1 mediates the PI3K/Akt/mTOR pathway and regulates the biological behavior of tumor cells (Chen et al., 2022). Zhang et al. (2022) found that MTA1 can inhibit the activity of the PI3K/Akt/mTOR pathway and control the growth of cervical cancer cells. In our previous experiment (Wang et al., 2020), by constructing a mouse model of cervical cancer, the research results found that after curcumin drug intervention, MTA1 expression in cervical cancer tissue was increased. This preliminarily suggests that curcumin drugs may play an anti-cancer therapeutic role by promoting the expression of MTA1.
In addition, if curcumin drugs are directly used to intervene with cells, there may be instability, and the drug concentration will also be affected. Based on the advantages of improving drug stability and ensuring drug concentration, this study prepared curcumin lipid nanoemulsions, which are used to intervene in cervical cancer cells to ensure drug stability, and so on. This study analyzed the level of cell experiments to clarify further whether MTA1 can be used as a drug target for cervical cancer. At the same time, it is hoped that this study can further explain the potential mechanism of action of curcumin lipid nanoparticles in treating cervical cancer and provide a new direction for future research. The schematic diagram of the mechanism is shown in Figure 1.
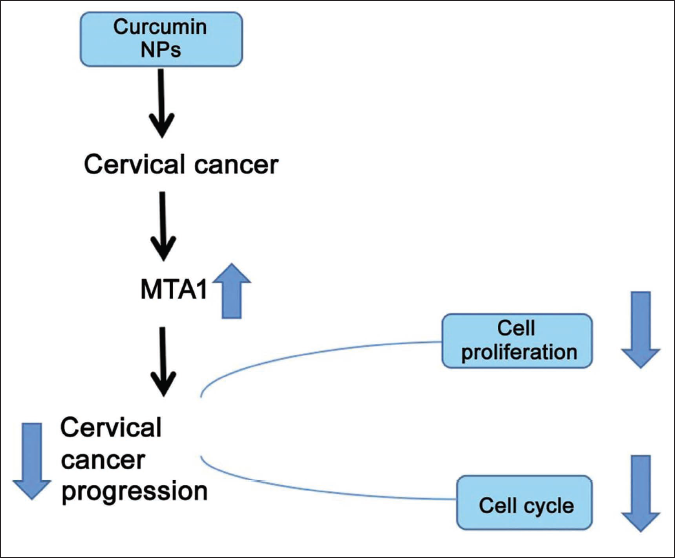
Materials and Methods
Experimental Reagents and Instruments
Cervical cancer cell Hela (Biometrics); primary anti-body (PI3K, AKT, p-AKT, mTOR, p-mTOR, 1:500; GAPDH, 1:1,000) (Abcam); secondary anti-body (Santa Cruz); Fetal bovine serum, penicillin, streptomycin (Costar); Lipofectamine™ 2000 kit, flow cytometer, electrophoresis tank (Merck); transmission electron microscope, particle analyzer (Zhuhai Omega).
Research Methods
Preparation of Curcumin Lipid Nanoemulsion
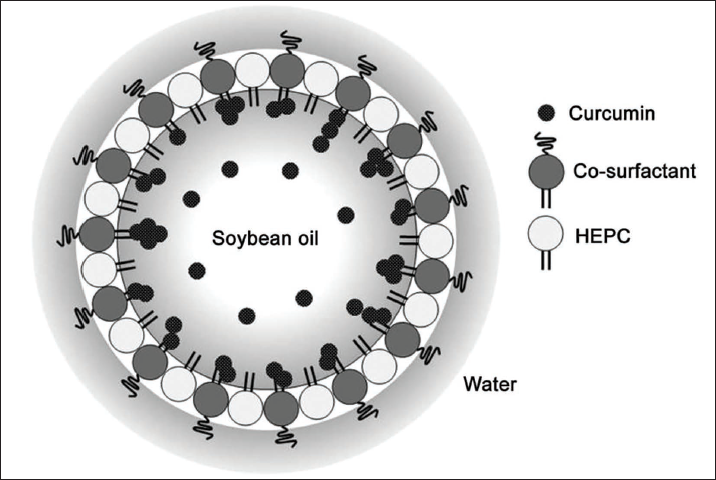
First, prepare the oil phase: mix palm oil, curcumin, and absolute ethanol. Add the oil to the water phase to form colostrum, homogenize for 10 min, and obtain curcumin lipid nanoemulsion. Take an appropriate amount of curcumin lipid nanoemulsion, deionized water, ultrasonic dispersion, take a proper amount of liquid sample dropwise onto the copper mesh, absorb the excess liquid with filter paper, dry it in the sun, put it into an electron microscope to observe the shape, and then use a particle analyzer to detect the size of granule. The synthetic diagram of the preparation is shown in Figure 2.
Schematic Diagram of Nanomaterial Synthesis.
Cell Intervention
Cell Culture
Cervical cancer cells were purchased from the Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences, and cultured in a Dulbecco’s Modified Eagle Medium (DMEM) medium.
Cell Transfection
According to the instructions of the kit, the negative control and the inhibitory tumor cells that down-regulate its expression were transfected, respectively. Cells were cultured and grouped as described above. Transient transfection was used. Liposomes and 20 nM negative control were mixed for transfection. After 48 h of action, a standard culture medium was used for culture.
Cell Intervention
Different concentrations (0, 10, and 20 µmol/L) of curcumin NPs were added to Hela cells (1 × 104/mL and 1 × 108/mL). Alternatively, 20 µmol/L curcumin lipid nanoemulsion was used to intervene in Hela cells, and the intervention time was 0, 6, 12, and 24 h, respectively. Or use 20 µmol/L curcumin lipid nanoemulsion to intervene Hela cells that have been transfected with MTA1 inhibitor.
Cell Biological Behavior Observation
5-Ethynyl-2′-Deoxyuridine (EDU) Staining
Take the above cells Hela, inoculate, add EDU reagent at the following time points (0, 5, 10, 15, 20 h), culture, wash with phosphate-buffered saline (PBS), fix, add Apollo solution, incubate, wash with PBS, stain with 4′,6-diamidino-2-phenylindole (DAPI), and count.
Cell Cycle
Take the above-mentioned HeLa cells after successful transfection, wash with PBS, centrifuge at 1,000 r/min for 5 min, make a single cell suspension, add Annenin V-PE, 7-AAD, incubate for 15 min, add 70% ethanol solution, dose 1 mL, and machine detects the cell cycle.
RT-PCR
Ribonucleic acid (RNA) was isolated for complementary deoxyribonucleic acid (cDNA) synthesis followed by reverse transcription polymerase chain reaction (RT-PCR) with conditions: 95°C for 5 min, 94°C for 30 s, 40 cycles, 72°C for 10 min, 35 cycles. The primer sequences are listed in Table 1.
Primer Sequences.

Western Blot
Isolated protein (50 µg) was separated by gel electrophoresis and transferred to polyvinylidene fluoride (PVDF) membrane for western blot. ECL chemiluminescent reagents detect signal expression.
Dual-Luciferase Reporter Gene Experiment
Transfect MTA1 and plasmids (wild-type plasmids, mutant plasmids) into MTA1 mimics or NC plasmids, dilute, blow off, centrifuge, add working solution, and detect.
Observation Indicators
Observe the proliferation and cell cycle of Hela cells, observe the protein expression of PI3K/Akt/mTOR pathway factors simultaneously, and verify that PI3K is the direct target gene of MTA1.
Statistical Methods
Data are presented as Mean ± SD and analyzed using SPSS21.0 software. The significant difference was confirmed by a one-way analysis of variance (LSD). p < 0.05 refers to a difference.
Results
Preparation of Curcumin Lipid Nanoemulsion
The synthesized curcumin lipid nanoemulsion is shown in Figure 3A. It was analyzed by transmission electron microscopy (TEM) (Figure 3A, 1:50 nm, B, 1:100 nm, and C 1:500 nm), showing that the curcumin lipid nanoemulsion sample consisted of an average diameter (SD) of 94.1 ± 11.6 nm spherical particle composition. As shown in Figure 3D.
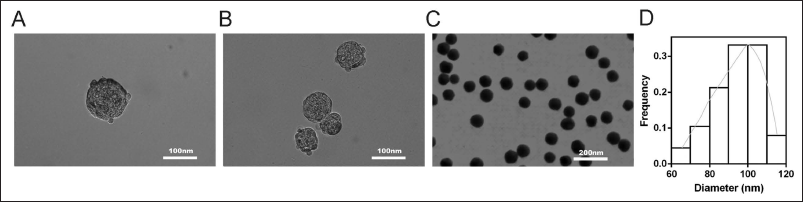
Curcumin Lipid Nanoemulsion Can Reverse the Cycle and Proliferation of Cervical Cancer Cells
Curcumin lipid nanoemulsion dose-dependently inhibited cell proliferation (p < 0.01). See Figure 4A. From 24 h onwards, cell number in the S phase in 20 µmol/L curcumin lipid nanoemulsion began to decline significantly with increased number in the G0/G1 phase and decreased number in the G2/M phase (p < 0.05) (Table 2). The results of flow cytometry showed that the cell proliferation in 15 µmol/L, and 20 µmol/L curcumin lipid nanoemulsion was obvious (Figure 4B and C).
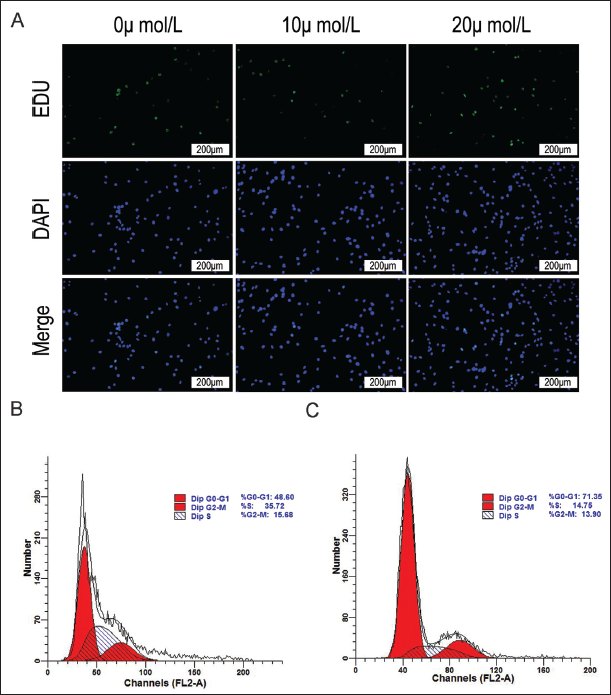
Cell Cycle.
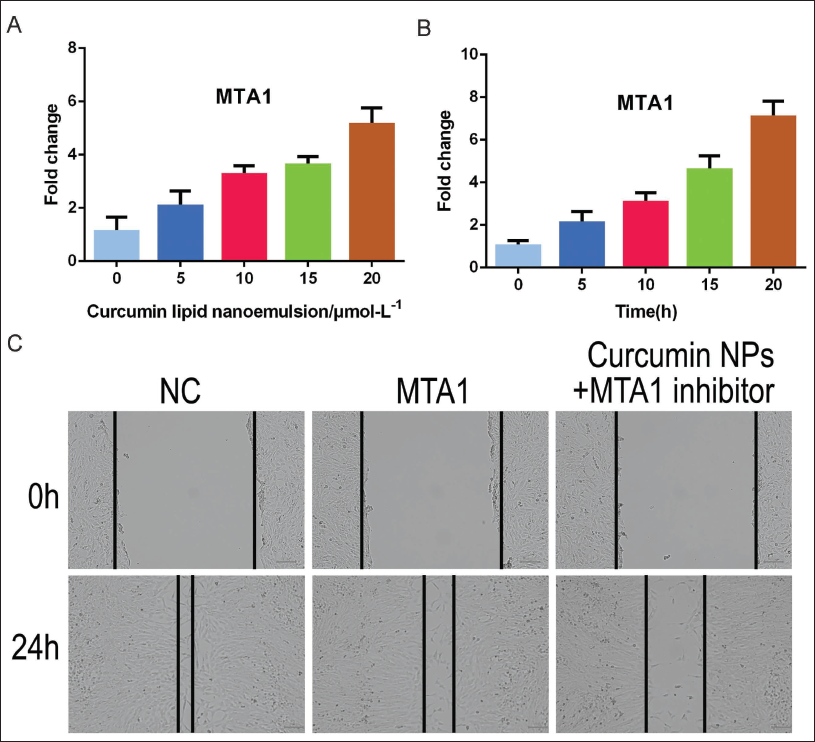
Curcumin Lipid Nanoemulsion Can Promote MTA1 Expression in Cervical Cancer Cells
The higher the concentration of curcumin lipid nanoemulsion, the higher the expression of MTA1 in cervical cancer cells, showing a certain concentration and dose dependence; the longer intervention time of 20 µmol/L curcumin lipid nanoemulsion, the higher MTA1 expression in cervical cancer cells. The higher it is, the more time-dependent it is (Figure 5A, B). Flavolipid nanoemulsions of 15 µmol/L and 20 µmol/L had consistent effects on cell proliferation and cycle, and 20 µmol/L was used for subsequent intervention. The results of cell scratching showed that under the intervention of 20 µmol/L concentration, the cell motility was weakened, as shown in Figure 5C.
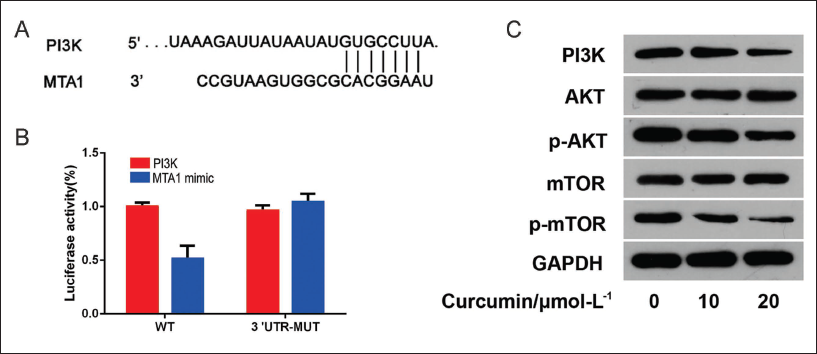
PI3K is the Direct Target Gene of MTA1
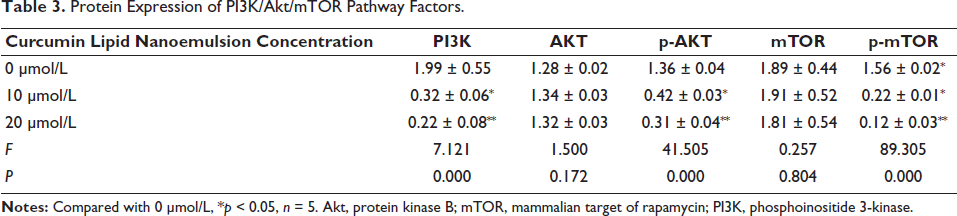
After analysis by Targetscan software, there is a specific binding region between the MTA1 sequence and the 3′-UTR 545-565 sequence of the PI3K gene, and PI3K is the target gene of MTA1 (Figure 6A). The fluorescence intensity of the mutant plasmid was higher than the wild-type plasmid (p < 0.05) (Figure 6B). When the concentration of curcumin lipid nanoemulsion was 10 µmol/L, the protein expressions of PI3K, p-AKT, and p-mTOR all decreased significantly, and the decrease was more significant at 20 µmol/L (p < 0.01). In addition, when comparing different concentrations of AKT and mTOR, p > 0.05 (Table 3 and Figure 6C).
Protein Expression of PI3K/Akt/mTOR Pathway Factors.
Discussion
To improve the stability of the curcumin drug and its concentration without interference, this study successfully prepared curcumin lipid nanoemulsion to interfere with cervical cancer cells. First, observe the proliferation and cell cycle changes of cervical cancer cells. Our results found that when the concentration of curcumin lipid nanoemulsion is 10 µmol/L, cells number in the S phase and G2/M phase is significantly reduced. The results show that curcumin lipid nanoemulsion, can interfere with the cell cycle, thereby inhibiting the cell proliferation process and finally controlling the abnormal growth of cervical cancer cells, thereby exerting an anti-cancer therapeutic effect. The results of the study found that curcumin lipid nanoemulsion drugs can promote apoptosis of cervical cancer cells and play a therapeutic role, both of which indicate that curcumin lipid nanoemulsion drugs can exert an anti-cancer effect on cervical cancer, consistent with our results (Park et al., 2011; Rao et al., 2011; Wei et al., 2021).
Previous studies have found that curcumin lipid nanoemulsion has an anti-cancer effect on cervical cancer and abnormal changes in the expression of MTA1 (Park et al., 2011; Rao et al., 2011; Wei et al., 2021). The effect of different intervention concentrations and time on MTA1 expression in cervical cancer cells was analyzed. Our results found that with the increase of the concentration of curcumin lipid nanoemulsion, MTA1 expression also increased. In addition, it was also found that 20 µmol/L Curcumin lipid nanoemulsion for intervention. We further indicate that MTA1 expression in cervical cancer cells has a dose-dependent and time-dependent relationship with the concentration of curcumin lipid nanoemulsion and the intervention time.
Compared with the cells that interfered with curcumin lipid nanoemulsion, the research results found that after transfection with curcumin, the number of cell proliferation was also significantly reduced, but after adding MTA1 inhibitor on this basis, the cell proliferation and cell cycle changes were similar to those of On the contrary, the results of this study suggest that curcumin lipid nanoemulsion mainly inhibits cells in S phase and G2/M phase by promoting the expression of MTA1, but the MTA1 inhibitor easily releases this, that is, after the MTA1 inhibitor is added to the cells, the curcumin lipid The nanoemulsion could not play a therapeutic role, which further indicated that curcumin lipid nanoemulsion played a role in promoting the expression of MTA1. Previous studies have shown that after MTA1 mimics interfered with cervical cancer cells (Xiao et al., 2021), cell proliferation was inhibited, but after MTA1 inhibitor was added, cell proliferation was promoted, and both groups of cells were under the premise of curcumin lipid nanoemulsion drug intervention. The MTA1 transfection operation suggested that curcumin lipid nanoemulsion may inhibit cell proliferation by promoting MTA1 expression.
To reveal the therapeutic mechanism of curcumin lipid nanoemulsion drug on MTA1. First confirmed the targeting relationship between MTA1 and PI3K (Xiao et al., 2021; Yang, Wang et al., 2021). It can induce apoptosis, and some studies have also shown that (Yin et al., 2021) Inhibiting PI3K/Akt/mTOR axis can inhibit cervical cancer cells proliferation. This study further observed the changes of PI3K/Akt/mTOR pathway factors after different concentrations of curcumin lipid nanoemulsion interfered with cervical cancer cells. With the higher concentration of curcumin lipid nanoemulsion, the PI3K, p-Akt, and p-mTOR protein decreased significantly. After the expression of PI3K decreases, cells number in the S and G2/M phases decreases, and inhibit the proliferation of cervical cancer cells (Borah et al., 2020; Zhan et al., 2021; Zhang et al., 2016). We further indicate that MTA1 can be used as a drug target of curcumin lipid nanoemulsion to inhibit cervical cancer cells by inhibiting the activity of the PI3K/Akt/mTOR pathway.
Conclusion
Curcumin lipid nanoemulsion inhibits cell number in the S phase and G2/M phase, causing G0/G1 arrest, inhibiting cervical cancer cells from entering the next cell cycle, and then controlling cell growth. Curcumin lipid, the mechanism of the therapeutic effect of nanoemulsion on cervical cancer, is related to MTA1. Curcumin mainly acts through the MTA1/PI3K/Akt/mTOR axis, and MTA1 targets PI3K to regulate cell proliferation. However, this study also has limitations. The involvement of the PI3K/Akt/mTOR pathway in cervical cancer cells needs further research. In addition to PI3K, the target genes of MTA1 include ZHX1, mTOR, and so on. Whether MTA1 affects cervical cancer cells through other target genes requires further study.
Footnotes
Abbreviations
AKT: protein kinase B; MTA1: metastasis-associated gene 1; mTOR: mammalian target of rapamycin; PI3K: phosphoinositide 3-kinase.
Acknowledgments
The authors gratefully acknowledge the Ganzhou People’s Hospital Laboratory for providing the necessary equipment for this study.
The author Yanhua Yu is also affiliated with Department of Clinical Laboratory, Huangshi Love & Health Hospital, Huangshi, Hubei, China.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the ethics committee of Ganzhou People’s Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
