Abstract
Background
Cervical cancer is the third most prevalent cancer globally, with high incidence and mortality rates. Current treatments often result in severe side effects and drug resistance. While Solanecio mannii (S. mannii) roots have been traditionally used in cancer treatment, there is a lack of scientific validation of their anticancer potential. Therefore, the present study sought to analyze the metabolomics profile of the extracts and evaluate its anticancer and pro-apoptotic potential.
Methods
S. mannii roots were collected, freeze-dried, and extracted using diethyl-ether: methanol (v:v) for total extraction, and hexane and ethyl acetate for fractionation. This study analyzed the metabolomic profiles of S. mannii root extracts using gas chromatography coupled to mass spectrometry and evaluated their selective cytotoxic effect on human cervical cancer cell lines, specifically Henrietta Lacks (HeLa) cell lines. Moreover, this study investigated the extracts’ potential to modulate autophagy and induce apoptosis. The pro-apoptotic effect of S. mannii was evaluated by analyzing chromatin morphological changes, the expression of Bcl-2 homologous antagonist/killer (BAK) and BCL2-Associated X (BAX) genes, and the activity of caspases.
Results
S. mannii exhibited selective anticancer and anti-proliferative activities against HeLa cells, with IC50 values of 113 ± 0.28 µg/mL for the total extract, 38.94 ± 0.87 µg/mL for the hexane fraction, and 12.16 ± 0.52 µg/mL for the ethyl acetate fraction. Additionally, S. mannii root extracts modulated autophagosome formation and autophagy-related genes. Furthermore, extracts exhibited an intrinsic pro-apoptotic potential by upregulating BAK and BAX and increasing the activity of caspase-9, −3, and −7 in a time- and dose-dependent manner. These effects are attributed to the bioactive phytocompounds identified in the extracts, including syringic acid, 4-coumaric acid, caffeic acid, vanillin, hydroquinone, oleic acid, beta-sitosterol, and stearic acid, among others.
Conclusion
This study showed the selective anticancer and pro-apoptotic activity of S. mannii root for the management of cervical cancer.
Introduction
Cervical cancer ranked as the third most common cancer (6.5%) and cancer-leading cause of death (7.7%) among women worldwide in 2020. 1 Singh et al (2023) highlight disparities along the geographic, socioeconomic, and temporal axes in cervical cancer incidence and mortality rates. 2 Within areas and between countries, there was a significant variation in the incidence and death rates, ranging from 40 to 50 times. Oceania, Asia, Latin America, and Sub-Saharan Africa had the highest within-region heterogeneity, with notably high rates of incidence and mortality. 2 While there has been a decline in the incidence of cervical cancer in many parts of the world over the past three decades, cervical cancer rates and burdens are still high globally, with 604,127 new cases and 341,831 fatalities recorded in 2020. 3 Twenty percent of worldwide cases and twenty-two percent of worldwide deaths occurred in Africa. Age-adjusted rates of 40, 36, 31, 23, and 6.2 cases per 100,000 women were recorded in Eastern, Southern, Central, Western, and Northern Africa, respectively. 2
Addressing the prevalence of cervical cancer goes beyond symptomatic management or singularly targeting specific signaling pathways and molecular mechanisms with targeted/immune therapy. Existing treatments for cervical cancer, including chemotherapy, targeted, and immunotherapy, result in severe side effects and the development of drug resistance in tumors, which is linked to genomic alterations, dysregulation of apoptosis and cellular homeostasis, efflux transporters, and autophagy.4–6 Therefore, there is an urgent need for new and improved therapeutic options with enhanced efficiency, where phytopharmaceutical drug discovery could expedite and economize the development of pharmaceuticals, potentially modulating major signaling pathways and enhancing the quality of life of cancer patients.
A potential approach to expedite and economize the development of pharmaceuticals involves the application of phytopharmaceutical drug discovery. 7 Research has demonstrated the anti-carcinogenic potential of several plant-based compounds, 8 including paclitaxel, vinblastine, camptothecin, curcumin, and resveratrol, 9 where major signaling pathways implicated in inducing apoptosis, suppressing cell proliferation, arresting the cell cycle, and inhibiting angiogenesis and invasiveness can be potentially modulated by single/multi-component phytopharmaceuticals. 10 Compounds and single/multi-component plant extracts were reported to enhance the quality of life of cancer patients. 11
Even though only 15% of approximately 250,000 species of higher plants that are known to exist underwent phytochemical screening and roughly 6% underwent biological property evaluation, there is undoubtedly a resurgence of scientific interest in natural plant-based pharmaceutical discoveries. 12 The initial, and most crucial, stage in the process of discovering drugs from plants is choosing plant candidates for the extraction of active principles and biological activity screening. 13 One of these approaches is to rely on ethnomedicine and ethnobotanical surveys. Solanecio mannii, a plant found in tropical and subtropical regions of Africa, has traditionally been utilized for its medicinal properties, as noted in ethnobotanical surveys. 14 Local communities have employed various parts of the plant for treating ailments such as wound healing, pain relief, and infections. Notably, ethnobotanical data suggested its use in traditional cancer treatment, attributing its efficacy to a rich uncharacterized phytochemical profile. 14 While ethnobotanical studies have shown promise regarding its anticancer effects, further rigorous research is essential to validate its efficacy and safety for modern medical applications. This study investigated the metabolomic profile of the roots at a high-throughput level, the attributed selective cytotoxic activity of the total extract and fractions against human cervical cancer cells (HeLa cell lines), and their potential to modulate autophagy and apoptosis, holding promise for developing novel anticancer therapies targeting cervical cancer.
Materials and Methods
Plant Collection, Identification, and Extraction
S. mannii root samples were collected from Kapseret, Kapsabet, Kenya (0°12′06.7″ N 35°05′58.3″ E). The plant samples were identified at the National Museum of Kenya. Dried and ground plant material was soaked in diethyl-ether/methanol (v/v) for total extraction. The resultant extract was filtered with Whatman filter paper number 1 and concentrated under a vacuum (55 °C). The total extract was subjected to sequential fractionation using organic solvents with increasing polarities. Using a separating funnel, the total extract was washed with 250 mL of n-hexane (20 min). The upper part (hexane phase) was collected, filtered, and concentrated at 69 °C, while the lower part was transferred for distilled water/ethyl acetate (v/v) partitioning (24 h). The ethyl acetate extract was recovered, filtered, and concentrated using a rotary evaporator (at 77 °C). Dried extracts were stored at −20 °C for further studies.
Gas Chromatography–Coupled Mass Spectrometry (GC-MS)-Based Metabolomic Profiling of S. mannii Root Extracts
Gas chromatography combined with mass spectrometry was used to analyze the metabolomics profile of the extracts. An Agilent Technologies 7890B gas chromatograph, equipped with a DB-5MS column (30 m × 0.25 mm internal diameter and 0.25 μm film thickness) and a 5977A mass spectrometer, was used for the GC-MS analysis. Hydrogen gas was used as the carrier gas, flowing at 1.0 mL/min, and the injection volume was 1 µL. The temperature was set at 60 °C for 1 min, rising to 320 °C (at 10 °C/min), and then held for 10 min. The injector and detector were held at 300 °C and 320 °C, respectively. Mass spectra were obtained by electron ionization at 70 eV using a spectral range of m/z 50–800 and a solvent delay of 6 min. The temperatures of the mass and quad were 230 °C and 150 °C, respectively. Identification of phytocompounds was achieved by comparing the spectrum fragmentation pattern with those stored in Wiley and NIST Mass Spectral Library data. All samples were subjected to derivatization before GC-MS analysis. In brief, dried samples were resuspended in 50 µL of bis(trimethylsilyl)trifluoroacetamide (BSTFA) supplemented with trimethylchlorosilane (TMCS) (99:1) silylation reagent and 50 µL of pyridine.
Extracts Preparation for In Vitro Studies
Briefly, 100 mg of dried extracts were dissolved in 1 mL of absolute Di-Methyl Sulf-Oxide (DMSO) to prepare a stock of 100,000 µg/mL. The stock solution was further diluted with complete growth media to a working solution of 200 µg/mL.
Cytotoxicity and Selectivity Analysis Using CCK8 Assay
In this study, kidney epithelial cells of African green monkey (Vero cells) and human cervical cancer cell line, specifically Henrietta Lacks (HeLa) cell lines were used. Both cell lines were cultured in 10% fetal bovine serum and 1% streptomycin/penicillin in Eagle's Minimum Essential Medium (EMEM). The growth media was supplemented with HEPES, sodium bicarbonate, and glutamine. The incubator environment was set at 37 °C, 5% CO2, and 95% humidity. Cells were inoculated into a 96-well plate at a density of 1 × 104 cells/well to attach overnight. Cells were treated with different extracts at a concentration of 200 µg/mL for 48 h (h) to select the active extracts. After the treatments, wells were washed with 100 µL of phosphate-buffered saline (PBS). Then, 90 µL of complete growth media and 10 µL of Cell Counting Kit 8 (CCK8, Beijing Solarbio Science and Technology Co., Ltd, Beijing, China, no CA1210) solution were added to each well and incubated for 3 h. Absorbances were measured at 450 and 650 nm using a microplate reader.
Positive control groups were incubated under identical conditions with the same concentration of rapamycin (Beijing Solarbio Science and Technology Co., Ltd, Beijing, China, no R8140), whereas the negative control group received only 0.2% DMSO-containing media. Corrected absorbances were used to calculate the cell viability.15,16
Furthermore, cells were treated with different concentrations (300, 150, 75, 37.5, 18.75, 9.375, and 4.6875 µg/mL) for 48 h to determine the half-maximal inhibitory concentrations (IC50) for cervical cancer cells (HeLa cell lines) and the half-maximal cytotoxicity concentrations (CC50) for normal cells (Vero cell lines). Dose–response inhibition curves were plotted using Splin/LOWESS fit in GraphPad Prism. To determine the selectivity index (SI), the following formula was used
17
:
Monodansylcadaverine (MDC) Staining
HeLa cells were seeded on membrane-treated slides, at a density of 2 × 105 cells/well, in 24-well plates and allowed to attach overnight, after which they were treated at ¼ IC50 and ½ IC50 for 18 h and 24 h, respectively. After treatment, cells were washed with PBS, followed by the wash buffer. Cells were stained with Monodansylcadaverine stain (MDC, Beijing Solarbio Science and Technology Co., Ltd, Beijing, China, no G0170) stain for 30 min (min), at room temperature, in the dark. Cells were washed with the wash buffer and observed under a fluorescent microscope.
4′,6-Diamidino-2-Phenylindole (DAPI) Staining
HeLa cells were seeded on membrane-treated slides, at a density of 2 × 105 cells/well, in 24-well plates and allowed to attach overnight, after which they were treated at ¼ IC50 and ½ IC50 for 18 h and 24 h, respectively. After treatment, slides were washed with PBS, fixed with 3.7% paraformaldehyde, and permeabilized with Triton-X (Beijing Solarbio Science and Technology Co., Ltd, Beijing, China, no T8200). Samples were stained with DAPI solution (Beijing Solarbio Science and Technology Co., Ltd, Beijing, China, no C0065) for 15 min at room temperature (RT). Finally, cells were rinsed with PBS twice and examined using a fluorescence microscope.
Hoechst Staining
HeLa cells were seeded on membrane-treated slides, at a density of 2 × 105 cells/well, in 24-well plates and allowed to attach overnight, after which they were treated at ¼ IC50 and ½ IC50 for 18 h and 24 h, respectively. After treatment, slides were washed with PBS and stained with Hoechst staining for 15 min (Beijing Solarbio Science and Technology Co., Ltd, Beijing, China, no C0030). Finally, slides were rinsed twice with PBS and observed under a fluorescent microscope.
Caspase-3, −8, and −9 Colorimetric Activity
Caspase-3, −8, and −9 activities were determined using a colorimetric assay kit (Beijing Solarbio Science and Technology Co., Ltd, Beijing, China, no BC3830, BC3880, BC3890). Briefly, cells were treated with S. mannii extract at ¼ and ½ IC50 for 18 h and 24 h, respectively. Following treatment, cells were harvested and lysed for 15 min. Lysate was centrifuged at 10,000 × g for 10 min at 4 °C to collect the supernatant. Protein concentrations were determined using the Bradford assay (Beijing Solarbio Science and Technology Co., Ltd, Beijing, China, Bradford Protein Assay Kit, no PC0010), and equal amounts of protein (100 μg) were incubated with caspase-specific substrate (Asp-Glu-Val-Asp-p-nitroanilide for caspase-3, N-acetyl-Ile-Glu-Thr-Asp-p-nitroanilide for caspase-8, and acetyl-Leu-Glu-His-Asp-p-nitroanilide for caspase-9 (at 37 °C for 2 h). Absorbances were measured at 405 nm using a microplate reader. Caspase activity was calculated as the fold change relative to the untreated control.
Analysis of the Relative Expression of Target Genes
RNA extraction was carried out using Pure Link RNA mini kit (Thermo Scientific™, USA, no 12183020) according to the manufacturer's protocol. In brief, HeLa cells were seeded in T75 flasks and allowed to reach 80% confluence. After treatments, cells were washed twice with PBS and lysed using a lysing buffer (supplemented with 1% 2-mercaptoethanol) for 5 min. The lysate was transferred into RNase-free Eppendorf tube and homogenized. An equivalent volume of absolute cold ethanol (100%) was added and vortexed. Using a spin cartridge, RNA was bound to the cartridge membrane after centrifugation. A total of 700 µL of wash buffer I was added to the spin cartridge and centrifuged at 12,000 rpm at room temperature for 15 s. A total of 500 µL of wash buffer II was added and centrifuged again at 12,000 rpm at room temperature for 15 s. The cartridges were spin-dried before the elution of RNA.
Using the SensiFAST ™ cDNA Synthesis Kit (Bioline, London, UK, no BIO-65053), cDNA was generated according to the manufacturer's instructions in a reaction volume of 20 μL containing 4 μL of RNA sample, 4 μL of buffer, 1 μL of reverse transcriptase, and 11 μL of nuclease-free water. The thermal cycling conditions began with a primer annealing step at 25 °C for 10 min, followed by reverse transcription at 42 °C for 15 min. Afterward, the reaction was inactivated by heating to 85 °C for 5 min and held at 4 °C. cDNA samples’ concentrations were quantified with a Thermo Scientific™ NanoDrop™ 2000/2000c Spectrophotometers, standardized to 900 ng/μL, and then diluted with nuclease-free water (1:9).
Quantitative real-time polymerase chain reaction (qRT-PCR) was performed with the SolisFAST® SolisGreen® qPCR Master Mix kit (Solis BioDyne, Estonia, no 28-41-00001) according to the manufacturer's protocol, using RT-PCR Quant Studio V system thermocycler (Thermo Scientific, USA), in a reaction volume of 10 μL (2 μL of the SolisFAST® SolisGreen® qPCR Mix (ROX, 5X), 0.3 μL of forward primer (10 μM), 0.3 μL of reverse primer (10 μM), 1 μL of cDNA template, and 6.4 μL of nuclease free water).
The thermal cycler was set as follows: Enzyme activation at 95 °C for 1 min, denaturation at 95 °C for 5 s, and the annealing/extension step was set at 57 °C for 20 s. Sequences of forward and reverse primers of target genes are shown in Table 1.
Forward and Reverse Primers’ Sequences of Target Genes.

Relative gene expression was quantified according to the 2−ΔΔCt method. β-tubulin was used as a housekeeping gene.
Statistical Analysis
All tests were replicated in at least three independent experiments. Two-way ANOVA was used to compare the quantitative data between the groups using GraphPad prism v.10; p < 0.05 was considered statistically significant.
Results
GC-MS-Based Metabolomic Profiles of S. mannii Root Extracts
The untargeted profiling yielded the identification of 61 sary metabolites in the total extract (RTT), 36 non-polar compounds in the hexane extract (RTH), and 15 compounds in the ethyl acetate extract (RTEA). Figure 1 shows the total number of ion chromatograms in the GC-MS-based untargeted metabolite profiling of the studied extracts.

GC-MS-based Total Ion Chromatograms of the Total
The analysis of S. mannii root phytocompounds in a high-throughput approach identified 9,12-octadecadienoic acid majorly constituting the total extract with 18.68% (Table 2). Hydroquinone was the major compound in the ethyl acetate extract, with an area sum of 59.57% (Table 3), whereas palmitic acid constituted 53.04% of the hexane extract (Table 4). Glycerol monostearate, 1-monopalmitin, 4-coumaric acid, caffeic acid, stearic acid, and azelaic acid were commonly found in the three studied extracts.
GC-MS-Based High-Throughput Secondary Metabolite Profiling of the S. mannii Root Total Extract.

GC-MS-Based High-Throughput Secondary Metabolites Profiling of S. mannii Root Hexane Extract.

GC-MS-Based High-Throughput Secondary Metabolites Profiling of S. mannii Root Ethyl Acetate Extract.

S. mannii Root Extracts Selectively Inhibited the Viability of HeLa Cells
Our study investigated the effect of S. mannii extracts on the viability of HeLa cells. Treatment with the extracts, over a period of 48 h, resulted in a significant inhibition of cell growth and a notable reduction in cell viability. These findings suggest that the total, hexane, and ethyl acetate extracts of S. mannii root possess cytotoxic properties against HeLa cells. To investigate the inhibitory concentrations of the active extracts, we performed a dose–response analysis. Our findings showed that the cell inhibition percentage increased in a concentration-dependent manner, with higher concentrations of the extract correlating with greater inhibition of HeLa cell viability (Figure 2). Specifically, the ethyl acetate extract showed the lowest IC50 of 12.16 ± 0.52 μg/mL, followed by the hexane and total extracts with IC50 values of 38.94 ± 0.87 μg/mL and 113 ± 0.28 μg/mL, respectively. Rapamycin, the positive control, exhibited an IC50 of 59.15 ± 2.44 μg/mL.
Our findings demonstrate selective activity of S. mannii root extracts against a human cervical cancer cell line. Table 5 shows the IC50 and CC50 depicted against HeLa cells and Vero cells, respectively, and the selectivity indexes of the extracts.

Effect of S. mannii Root Extracts on HeLa Cells.
Selectivity of S. mannii Root Extracts.

All data are presented as mean ± SD.
S. mannii Root Extracts Differentially Regulated Autophagy in HeLa Cells
This study investigated the potential of root extracts in autophagy induction in HeLa cells in a dose- and time-dependent manner using monodansylcadaverine (MDC) staining as a marker for autophagic vacuoles. RTH significantly modulated autophagy in HeLa cells. At ¼ IC50 for 18 h, RTH reduced autophagy, while at ½ IC50 for 24 h, it significantly enhanced autophagy compared to the untreated group. Treatment with RTEA at ¼ IC50 for 18 h decreased autophagic vacuole staining, but at ½ IC50 for 24 h, the staining returned to levels comparable to the untreated group. In contrast, RTT of S. mannii root did not alter autophagic vacuole staining at either dosage. Rapamycin caused no changes at lower dosages but significantly reduced MDC intensity at ½ IC50 for 24 h (Figure 3A, B).
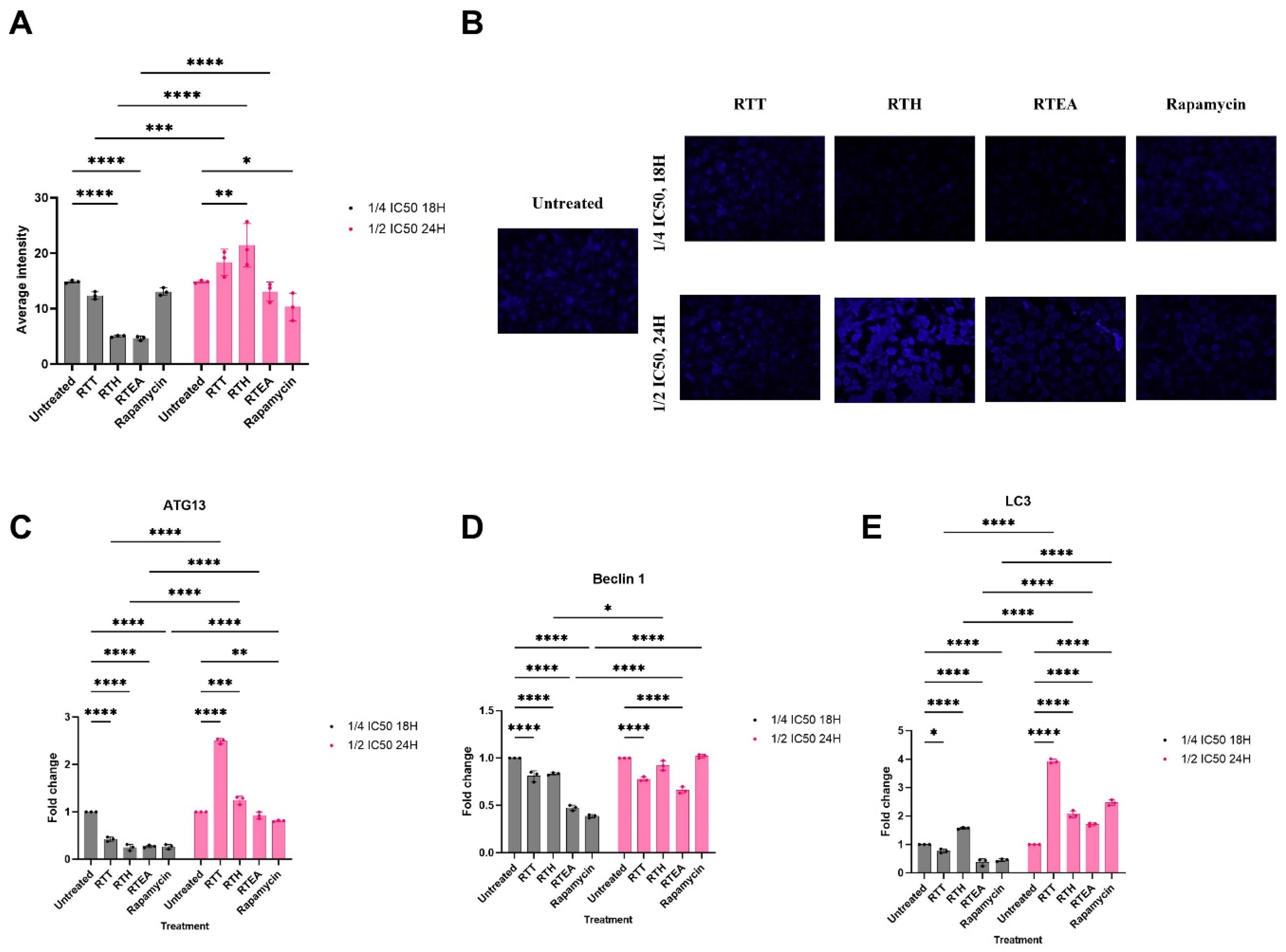
Effect of S. mannii Root Extracts on Autophagic Vacuoles and the Expression of Autophagy-related Key Proteins. (A). The Average Intensity of MDC Staining. (B). The MDC Staining of Autophagic Vacuoles Under Fluorescent Microscopy, 40×. (C–E). The Relative Ex-pression of Autophagy-related Key Proteins.
Transcriptomics analysis revealed decreased ATG13 expression across all treatments at ¼ IC50 for 18 h. At ½ IC50 for 24 h, RTH and RTT significantly increased ATG13 expression, while RTEA restored it to levels comparable to the untreated group. Rapamycin reduced ATG13 expression at both dosages (Figure 3C). Beclin 1 expression also decreased at ¼ IC50 for 18 h in all treatments. At ½ IC50 for 24 h, RTH and rapamycin restored Beclin 1 to untreated levels, while RTT and RTEA showed increases that remained significantly lower than untreated levels (Figure 3D). in addition, LC3 expression was similarly reduced by RTT, RTEA, and rapamycin at ¼ IC50 for 18 h, with significant increases at ½ IC50 for 24 h compared to untreated cells. Notably, RTH increased LC3 expression even at ¼ IC50 for 18 h, in a dose- and time-dependent manner (Figure 3E).
S. mannii Root Extracts Induced Intrinsic Apoptosis in Dose- and Time-Dependent Manner
This study investigated the impact of extracts on apoptosis induction in HeLa cells using DAPI and Hoechst staining. Irregular shape, reduced circularity (nuclear shape index), the condensation of the chromatin into sharply delineated masses, the shrinkage/swell of the nucleus, fragmentation, and the presence of lobules/herniation/invagination and ring-like structures were depicted upon treatment (Figure 4).

Effects of S. mannii Root Extracts on Nuclear Morphological Characteristics of Hela Cells Using DAPI and Hoechst Staining, 40×.
Additionally, treatment with S. mannii root extracts revealed the upregulation of BAK and BAX in a time- and dose-dependent manner (Figure 5). Notably, the expression level of BAX and BAK was significantly elevated the most when treated with RTT at ½ IC50 for 24 h. Treatment with the hexane extract significantly increased the expression of BAK and BAX compared to the untreated group, but not change in a dose-dependent manner. Additionally, treatment with RTEA increased the expression of BAK and BAX in a dose-dependent manner.

Effects of S. mannii Root Extracts on the Expression Level of BAK and BAX. All Data are Presented as Mean ± SD.
Moreover, we analyzed the fold-increase activity of caspase-8, −9 and −3. S. mannii root extracts induced a dose-dependent increase in caspase-9 activity by 2.032 ± 0.144 and 3.223 ± 0.371 folds when treated with RTT at ¼ IC50 for 18 h, and ½ IC50 for 24 h, respectively. Additionally, RTH increased the activity of caspase-9 by 2.029 ± 0.093 and 2.5 ± 0.233 folds when treated at ¼ IC50 for 18 h, and ½ IC50 for 24 h, respectively. An increase in the caspase activity was shown upon treatment with RTEA in a dose- and time-dependent manner by 2.936 ± 0.233 and 4.132 ± 0.551 folds, compared to the untreated control (Figure 6). The significant increase in caspase-8 activity was only detected in rapamycin-treated group in a dose- and time-dependent manner. Additionally, there was a significant increase in caspase 3 activity upon treatments, most notably at higher dosages.

Effects of S. mannii Root Extracts in the Activity of Caspase-3, Caspase −8 and Caspase 9. All Data are Presented as Mean ± SD.
Discussion
Cervical cancer is a major global public health issue, particularly in low- and middle-income countries, where reductions in prevalence and mortality have been sluggish despite prevention strategies such as vaccination against human papillomavirus (HPV) and screening. 3 Cervical cancer is currently managed primarily through surgical resection, chemotherapy, immunotherapy, local targeted therapy, and radiotherapy. While conventional treatments demonstrate effectiveness in managing cervical cancer in its early stages, their effectiveness diminishes when applied to locally staged and metastatic forms due to the presence of severe adverse effects, drug resistance, multiple recurrences, and metastasis. 18 As a result, the development of drugs with improved safety profiles and enhanced efficacy for the treatment of cervical cancer is imperative. Rich in bioactive chemicals that have molded contemporary medicine, medicinal plants are priceless tools for the search and development of new medications. Additionally, the development of herbal medicine depends on the techniques of analysing and extracting plant materials, which help to refine and standardize herbal compositions thereby guaranteeing their safety, effectiveness, and quality.
Phytotherapy, a combination of ethnobotanical knowledge and modern drug discovery techniques, has shown promise in treating cancer. Traditional plant medicinal properties have led to the identification of bioactive compounds with anticancer properties, including paclitaxel, vinblastine, camptothecin, curcumin, and resveratrol. 9 This paradigm shift has led to the exploration of herbal-based extracts and formulations, which can modulate multiple targets and pathways simultaneously. This approach enhances treatment efficacy, overcomes drug resistance, and mitigates cancer cell heterogeneity.19,20 Ethnobotanical studies have demonstrated that the roots of S. mannii exhibit therapeutic properties. 14 Traditionally, it has been utilized to treat wounds, infections, and cancer, crediting its effectiveness to its abundant phytochemical content. Despite its ethnobotanical significance, S. mannii has not been extensively investigated for its anticancer properties, particularly in the context of cervical cancer. Additionally, there exists a substantial deficiency of evidence concerning its metabolomics profile. In this study, we have demonstrated the selective cytotoxic potential of S. mannii against HeLa cells, and its potential in regulating autophagy and inducing apoptosis.
Our pioneering study demonstrates, for the first time, the selective cytotoxic activity of S. mannii on cancerous cells, in accordance with previous ethnobotanical surveys. Selectivity index (SI) values >1 indicate desirable selectivity against cancerous cells. 21 In the present study, the ethyl acetate fraction exhibited the most potent anticancer activity, having the lowest IC50, followed by the hexane fraction (P < 0.0001), with SI of 1.18 and 1.09, respectively. The solvent used in the plant extraction and partitioning of the total extract can influence the extraction efficiency and the yielded extracts’ components. This might, therefore, affect the extract's biological properties, particularly its cytotoxic potential. The cytotoxic activity of RTEA against HeLa cells with an IC50 of 12.16 ± 0.52 μg/mL demonstrated a comparable effect to that of paclitaxel, a widely used chemotherapeutic agent for cervical cancer treatment, typically reported in the range of 5–10 nM. 22 This similarity in potency suggested that the ethyl acetate extract of S. mannii has comparable efficacy in inhibiting the proliferation of HeLa cells, highlighting its potential as a therapeutic agent for cervical cancer.
The cytotoxic activities are not only attributed to the solvent but also to the complexity and diversity of the bioactive components of plant extracts. According to the concept of “like dissolves like” solubility, polar compounds can only dissolve in polar solvents, while semipolar and non-polar compounds can only dissolve in semipolar and non-polar solvents.23,24
In our study, the total extract exhibited a significantly higher IC50 compared to RTH, RTEA, and rapamycin (P < 0.0001). In regards to the guidelines of the National Cancer Institute, RTT exhibited a weakly cytotoxic effect (IC50 > 50 μg/mL). 25 Despite this classification, the total extract exhibited the most selective effect against HeLa cells (SI = 1.47). Lower IC50 indicates a higher potency and effectiveness, which was depicted in the ethyl acetate and hexane extracts of S. mannii root extracts. Human cervical cancer cell lines (HeLa cells) are sensitive to S. mannii extracts and could be a potential anticancer candidate for the management of cervical cancer, without drug resistance, compared to common treatment approaches used in the management of cervical cancer.
The phytochemical profiles of the three extracts revealed distinct chemical compositions that correlate with the polarity of the extraction solvents. The metabolomics profile of the RTT extract highlighted a broad range of compounds, including fatty acids (palmitic acid, oleic acid, and stearic acid), organic acids (azelaic acid and sebacic acid), sugars (β-D-fructofuranose and β-D-tagatopyranose), and glycerides (glycerol monostearate), reflecting both polar and nonpolar compounds. In comparison, the RTH extract exhibited a marked enrichment of nonpolar constituents, predominated by lipophilic compounds, including palmitic acid, monopalmitin, and beta-sitosterol, aligning with hexane's low polarity. Additionally, the RTH extract showed a significant relative abundance of arachidic acid and stearic acid, both long-chain fatty acids are hydrophobic metabolites. On the other hand, the RTEA extract showed a distinct profile dominated by hydroquinone, caffeic acid, and syringic acid. Therefore, the ethyl acetate extracted was enriched with moderately polar compounds, including phenols and certain acids, including azelaic acid and 4-coumaric acid. This underscores the solvent's role in constituent segregation and fractionation. The demonstrated anticancer activity of S. mannii is attributed to the identified phytocompounds. The GC-MS analysis of S. mannii extracts reveals a rich profile of bioactive compounds.
Caffeic acid is well-documented for its pro-apoptotic and anti-proliferative properties, particularly in colon, breast, and lung cancer models. 26 Caffeic acid promoted apoptotic morphological changes, increased ROS levels, and altered matrix metalloproteinases in ME-180, HT-1080, and HeLa cell lines.26,27 In breast cancer, caffeic acid inhibited the proliferation of MCF-7 cell lines. 28 Additionally, in colon cancer, caffeic acid demonstrated an anti-tumor property by activating AMPK signaling pathway. 29 Caffeic acid triggered cell death by blocking Bcl-2 activity, which released and activated caspase-3 in B cell lymphoma, squamous cell carcinoma, and cervical cancer.26,30–32 Caffeic acid reduced cell viability in human colorectal adenocarcinoma HT-29 cell lines, regulated cell cycle, inhibited their clonogenic potential, and upregulated caspase expression.29,33,34 According to Li et al (2015), caffeic acid reduced the viability of cancer stem cells by inhibiting TGFβ-SMAD2 signaling. 35 According to research conducted by Kabała-Dzik et al (2017) and Kleczka et al (2020), caffeic acid reduced the migration of MCF-7 and induced apoptosis and cell cycle arrest in MDA-MB-231 cells.36,37
Similarly, 4-coumaric acid showed mitochondrial-mediated apoptosis in colorectal cancer cell lines by inhibiting Bcl-2 and Bcl-xL, Bcl-2 protein family members, and activating BAK and BAX.38–41 Sharma et al revealed that coumaric acid has induced mitochondrial membrane depolarization, promoted cytochrome-c release, and promoted caspase-dependent intrinsic apoptosis in human colon cancer cell lines (HT-29 and SW480). 39 Hu et al (2020) demonstrated the potential of coumaric acid to inhibit melanoma cell proliferation and to induce cell cycle arrest in the G0/G1 phase by downregulating Cyclin E and CDK2. In addition, coumaric acid induced intrinsic apoptosis in A375 and B16 cells by upregulating Apaf1, and Bax, and increasing the activity of caspase-3 and caspase-9. 42
Azelaic acid exhibits cytolytic and cytotoxic effects on acute myeloid leukemia cell lines and inhibits their viability. According to Dongdong et al (2019), azelaic acid promoted NK and T cell proliferation and activation, and the secretion of cytolysis-related cytokines in vitro. 43 Additionally, azelaic acid reduced oxidative stress and intracellular ROS Levels and increased the total antioxidant capacity in acute myeloid leukemia by increasing SOD and GSH levels. It induced cell cycle arrest in G0/G1 and apoptosis. 44
Linoleic acid, for instance, has demonstrated anticancer properties against endometrial cancer cell lines (HEC-1A and KLE) and in transgenic mice with endometrial cancer. Linoleic acid was shown to induce cell cycle arrest in G1 and to promote apoptosis via a dose-dependent increase in the activity of caspase3, −8, and −9. 45 Additionally, linoleic acid altered cancer cells' energy metabolism, induced cell quiescence, and decreased colorectal cancer growth in mice injected with CT26 cell line. 46
Other pharmacologically active phytocompounds included beta-sitosterol, stearic acid, hydroquinone, syringic acid, and vanillin. Mounting evidence demonstrated the anti-cancer and pro-apoptotic activities of these molecules in several cancer types, including human non-small cell lung cancer (NCI-H460 and NSCLC cell lines), breast cancer (MCF-7 and MDA-MB-231 cell lines), and cervical cancer (Caski, HeLa, and SiHa cell lines).47–51
Umereweneza et al (2023) showed the presence of umutagarananol, umutagarinine A and B, umutagarinine B, senaetnine, 2-angeloyloxy-5,8-dihydroxypresilphiperfolane, caffeic acid, methyl caffeate, and lupeol in the CH2Cl2-MeOH (1:1) extracts of root and stem bark of S. mannii. 52 These compounds showed anticancer activities of 378.2, 35.6, 21.7, 12.5, 486.2, > 1110.1, 844.1, and 164.9, respectively, against MCF7 breast cancer cell lines. Notably, umutagarananol and umutagarinine A and B demonstrate potent cytotoxic effects. These effects may be achieved by activating caspase pathways and modulating pro- and anti-apoptotic proteins, which potentially induce apoptosis. Additionally, caffeic acid and methyl caffeate, which are known for their antioxidant properties, contribute to the anticancer effect by inducing oxidative stress and subsequent DNA damage in cancer cells, thereby promoting apoptosis.53,54 Similarly, lupeol, a triterpenoid, is recognized for its ability to impede the proliferation of cancer cells by inhibiting NF-κB signaling.55–58 These compounds enhance apoptotic pathway signaling and reduce cancer cell survival. 59 Similarly, Ndom et al (2007) identified styrylpirones in the aerial parts of S. mannii, including 4-methoxy-6-(11-O-ß-D-glucopyranosylstyryl)-αpyrone, 4-methoxy-6-(11-hydroxystyryl)-α-pyrone, 4-methoxy-6-(11-O-α-L- rhamnopyranosylstyryl)-αpyrone, L-rhamnopyranose, and α-amyrin. 60 The alkaloid extract of Solanecio mannii contained 7-O-senecioylplatynecine and 7-O-tigloylplatynecine (81.9% and 17.1% of the total alkaloid content). 61 Furthermore, Dou et al (2017) have studied the antitumoral effects of crude polysaccharides, extracted from Solanecio scandens (S. scancdens) (dried whole plant, using ethanol), against human lung cancer, leukemia, sarcoma, and hepatocellular carcinoma cell lines (A549, HL60, S180, and H22, respectively). S. scandens crude polysaccharides showed anti-proliferative activity, mostly against H22 cell lines, with an IC50 of 42.4 μg/mL, followed by S180 (60.3 μg/mL), A549 (102.8 μg/mL), and HL60 (117.9 μg/mL). 62
Altogether, our findings offer a new perspective for research on the pharmacological activities of S. mannii, mainly with regard to its anticancer activity.
Autophagy is a physiological intracellular degradation/catabolic pathway mediated by autophagosomes that deliver degraded damaged proteins and cytoplasmic components to the lysosome to be recycled for further cellular metabolism. 63 In this study, we employed sub-inhibitory concentrations to ensure the most selective anticancer activity and modulation of cellular processes while minimizing non-specific cytotoxic effects. Our findings demonstrated the potential of S. mannii root extracts to regulate autophagy in HeLa cells. At a lower dosage, S. mannii regulates the autophagy process on the nucleation and the maturation level, demonstrated by the reduced average intensity of autophagosomes, via the downregulation of ATG13, Beclin 1, and LC3. Kumari et al (2023) demonstrated that the inhibition of autophagy in HeLa cells by lichen extract induced ROS generation, membrane depolarization, and lipid peroxidation, thereby promoting cell cycle arrest and apoptosis. 64
At higher dosages (½ IC50 for 24 h), an increase in the MDC staining was depicted in HeLa cells, together with an increase in the expression of ATG13 and LC3, demonstrating an increase in the number of autophagic vacuoles. Nevertheless, the relative expression of Beclin 1 is persistently significantly reduced, demonstrating a stronger regulatory effect of the extracts on the fusion step of autophagosomes with lysosomes, where HeLa cells are unable to overcome the suppression of autophagosome fusion with lysosomes (incomplete/defective autophagy). In this context, targeting the fusion step between autophagosomes and lysosomes represents an anticancer strategy of S. mannii root, where the degradation of autophagic cargo within lysosomes is hindered, leading to the accumulation of damaged organelles and toxic proteins within the cell. This buildup can trigger apoptotic pathways or induce cellular senescence, ultimately inhibiting cancer cell proliferation and promoting their demise. Chloroquine was found to inhibit autophagy by decreasing autophagosome-lysosome fusion and autophagosome degradation rate, thereby blocking autophagic flux and enhancing the pro-apoptotic effect of treatments. 65 Oblongifolin C, extracted from Garcinia yunnanensis Hu was found to induce apoptosis in human cholangiocarcinoma cells (QBC939) via inhibiting the autophagic flux and mitochondrial dysfunction. 66
According to McKnight et al (2014), Beclin 1 is essential for the trafficking of vacuoles and their fusion with endosomes/lysosomes, where it plays a major role in the localization of PI(3)P. 67 Thoresen et al (2010) demonstrated that depletion of one of PI3K-III subunits (VPS15, VPS34, Beclin 1, UVRAG or BIF-1) in HeLa cells im-paired the degradation of epidermal growth factor receptor (EGFR). 68 In addition, defective PI3K-III complexes (including the loss of Beclin 1) impaired degradative endocytic receptor trafficking. In the same study, the depletion of Beclin 1 or any member of the PIK-III (but not ATG14L) increased the number of midbodies, resulting in impaired cytokinesis in HeLa cells and, therefore, human cervical cancer cell proliferation. 68 Further, Beclin 1 has been demonstrated to be critical for maintaining the proliferation and the tumorigenicity of breast cancer stem cells. 69 MCF-7 mammospheres reduced in size when Beclin 1 was downregulated. McKnight et al (2014) reported an endosome maturation defect-based impaired autophagy when Beclin 1 was depleted due to the loss of Rab5 GTPase and the UVRAG-VPS34 complex and associated activity. 67 Overall, when Beclin 1, the stability of members of the autophagy and late endosomal trafficking and fusion is compromised. This supports, in turn, our hypothesis, as when HeLa cells were treated with higher dosage of total and ethyl acetate S. mannii root extracts, we found an increased autophagosome generation (high MDC stain, high expression of LC3 and ATG13) in response to the defective late stage of autophagy due to decreased expression of Beclin 1; however, due to financial constraints, the autophagic defect in a post-autophagosomes production context was not validated in this study.
The enhanced autophagosome formation was depicted upon treatment with RTH at ½ IC50 for 24 h, with high intensity of MDC staining, upregulated LC3 and ATG13, and no significant change in the Beclin 1 expression level, compared to the untreated group. Autophagy has been described as a tumor suppressor mechanism in other contexts. The loss of autophagy through Beclin 1 and ATG 5 loss increased DNA damage and genomic instability (gene amplification, centrosome abnormalities, and aneuploidy), promoting tumorigenesis and apoptosis deficiency in immortalized and transformed primary epithelial cells. 70 Autophagy inhibits cell malignancy by preventing gene and chromosome mutations and reducing reactive oxygen species production, primarily by clearing dysfunctional mitochondria and cheesy proteins during redox reactions. 71 In cervical cancer, the increase in autophagy promoted apoptosis in c-ray-irradiated HeLa and CaSki. 72 The differential regulation of autophagy is likely attributed to the identified phytocompounds. Palmitic identified in the hexane extracts, is known to promote apoptosis by inducing excessive autophagy. 73 Tu et al (2014) demonstrated that palmitic acid promoted autophagy via JNK2 activation. 74 Conversely, in a study conducted by Hernández-Cáceres et al (2020), palmitic acid was found to regulate the autophagic flux by impairing autophagosome-lysosome fusion and endolysosome trafficking. 75 Previous research has demonstrated the anticancer activity of beta-sitosterol by activating autophagy in hepatocellular carcinoma and gastric cancer. 76 Further research should be conducted to further investigate the effect of RTH on autophagy. Our findings highlighted a stable ATG13 expression level and MDC staining when treated with a lower dosage of rapamycin, compared to the untreated group. Previous studies have shown that HeLa cells exhibit reduced sensitivity to rapamycin at suboptimal concentrations, due to compensatory signaling mechanisms, involving mTORC2 activity, and/or alterations in upstream regulators. 77
Our findings show how ATG13, Beclin 1, and LC3 affect autophagic processes at various dosages of S. mannii root extracts; the control of autophagy is closely linked to the expression of key autophagy-related genes. Lower dosages (¼ IC50 for 18 h) suggest decreased autophagic vacuole generation since the downregulation of ATG13, Beclin 1, and LC3 corresponds with a reduction in the intensity of MDC staining. Particularly, ATG13 is a vital component of the autophagy initiation complex and its downregulation indicates early-stage autophagy suppression. Analogous to this, the reduction in Beclin 1 expression points to reduced autophagosome generation and trafficking, which is crucial for the maturation and nucleation phases of autophagy. Since LC3 is involved in autophagosome membrane elongation, the decreased expression of LC3 supports even more a lower autophagic flow. This joint downregulation in gene expression helps to limit the cells’ capacity to remove damaged organelles and proteins, therefore triggering either death or other stress-related processes, or incomplete or inhibited autophagy. Increased MDC staining at higher dosages (½ IC50 for 24 h) together with ATG13 and LC3 overexpression hint to improved autophagosome generation. However, the ongoing downregulation of Beclin 1 points to a deficit in the fusion of autophagosomes with lysosomes even as autophagy is triggered and autophagosomes are generated. This flaw in the fusion step causes an accumulation of autophagosomes, a characteristic of faulty autophagy, where autophagic cargo is not efficiently broken down inside the lysosome. Under these conditions, the increased expression of ATG13 and LC3 suggests that, despite the activation of the autophagic process, cargo breakdown and poor fusion keep it incomplete. This disturbance of late-stage autophagy might gather broken proteins and organelles inside the cell, therefore causing cellular damage. Furthermore, the expression of LC3 indicates maturity as well as the generation of autophagosomes. The higher expression of LC3 at higher dosages corresponds with our finding of more autophagic vacuoles, therefore supporting the hypothesis that S. mannii extracts can induce autophagosome synthesis with compromised fusing phase, pointing to a continuous deficiency in autophagosome-lysosome fusion.
Moreover, S. mannii extract showed a pro-apoptotic potential. Based on the morphological changes depicted with DAPI and Hoechst staining, S. mannii root extracts appear to induce late apoptosis, characterized by extensive nuclear alterations. These changes are indicative of advanced stages of cell death, where the cell is committed to the apoptotic process and irreversibly destined for death/destruction. 78 The observed increase in apoptotic cells suggests that S. mannii roots possess pro-apoptotic properties, prompting the activation of programed cell death pathways in HeLa cells. This study outlined evidence of the pro-apoptotic potential of S. mannii root, specifically the mitochondrial intrinsic pathway. The mitochondrial intrinsic apoptotic pathway is a tightly regulated process, initiated by internal cellular stress signals, including DNA damage, oxidative stress, or cytokine deprivation. Key players in this pathway are the pro-apoptotic proteins BAK (Bcl-2 homologous antagonist/killer) and BAX (Bcl-2-associated X protein), which belong to the Bcl-2 family. Upon activation, BAK and BAX oligomerize and form pores in the mitochondrial membrane, disrupting its integrity, which leads to the release of cytochrome C from the mitochondria into the cytosol. Cytochrome C binds to apoptotic protease activating factor-1 (Apaf-1), forming the apoptosome, which recruits and activates procaspase-9. Active caspase-9 subsequently triggers a caspase cascade, activating executioner caspases-3 and −7. 79
In the present study, S. mannii increased the activity of caspase-9, a hallmark of the intrinsic apoptotic pathway, together with an increase in effector caspases (3 and 7). In accordance with Ulloth et al (2003), palmitic and stearic acids induced caspase-dependent apoptosis in PC12 cell lines. These fatty acids induced apoptosis-related morphological changes of the chromatin, including cell shrinkage, membrane blebbing, condensation, and fragmentation. 80 Additionally, beta-sitosterol was found to induce mitochondrial-mediated apoptosis via ROS generation and loss of mitochondrial membrane potential, together with an increase in caspase-9, −3, and −7.81–83 Furthermore, Numerous studies have demonstrated the potential of oleic acid to inhibit cell proliferation in different tumor cell lines by increasing intracellular ROS levels and caspase-3 activity.84–88 According to Cortez et al (2024) and Nasiri et al (2018), caffeic acid upregulated the expression of BAK and BAX and induced mitochondrial-mediated apoptosis.89,90 Rapamycin, the positive control, increased the activity of both caspase-8 and −9, demonstrating its potential to induce both intrinsic and extrinsic apoptotic pathways. Our findings are in accordance with previous studies.91,92 Notably, this study speculated the lack of dose dependency in the pro-apoptotic potential of RTT and RTH. This may be due to complex regulatory mechanisms underlying BAK expression or the presence of bioactive compounds in the extract with opposing effects at different concentrations. Further investigations are required to elucidate the potential interactions with other apoptotic regulators. Our findings align with previous studies on related species within the Solanecio genus. The antitumor effect of Solanecio scandens against hepatocellular carcinoma was pronounced through the regulation of the expression level of Bcl-2 (down) and the upregulation of Bax, caspase-3, and caspase-9, depicting a mitochondria-mediated apoptosis pathway, in vivo. 62
To the best of our knowledge, this study is the first to report the anticancer activity of S. mannii root total extract and fractions, demonstrating its potential for cervical cancer management. Our investigation revealed that S. mannii root extracts selectively induced cytotoxic effects on cervical cancer cells, thereby corroborating the plant's ethnobotanical application in the treatment of cancer. The study highlighted the pro-apoptotic effects and regulation of autophagy by S. mannii root, in accordance with ethnomedicine. Nevertheless, further research is needed to identify the specific bioactive compounds responsible and to evaluate the effects of S. mannii root extracts in animal models and clinical settings.
Conclusion
The findings of this study suggest that S. mannii root extracts have a potential selective cytotoxic effect on human cervical cancer cells, attributed to their metabolomic profiles. In addition, S. mannii root extracts differentially regulated autophagy and triggered intrinsic mitochondrial-mediated apoptotic cell death in a dose- and time-dependent manner. Nevertheless, there is a need for further research to fully elucidate the molecular mechanism of S. mannii root against cervical cancer and the selective anti-cancer activity reported herein. Finally, the present study suggests that S. mannii root extracts may be further investigated in cervical cancer models, in vivo, and in clinical settings as a potential candidate for the treatment and management of cervical cancer.
Footnotes
Acknowledgments
This research was partially supported by KEMRI, under grant number KEMRI/COV/SPEC/003, and by the Africa-AI-Japan Project.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions/CRediT
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported with funding from the Pan African University under the African Union Commission scholarship.
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data used and analyzed during the current study are incorporated in this paper.




