Abstract
Background
Gliomas are the most aggressive and devastating brain tumors, with a poor prognosis due to their complex etiology, diverse clinical manifestations, and the limited efficacy of existing treatment methods.
Objectives
The present work was conducted to understand the anticancer activities of farnesol against glioma C6 cells.
Materials and Methods
The effects of farnesol on in vitro free radical scavenging were evaluated at various dosages (1–100 µM). A 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) test was used to assess the influence of farnesol on the growth of glioma C6 cells and nonmalignant Vero cells. The apoptotic levels in the cells were investigated using 4′,6-diamidino-2-phenylindole (DAPI) and dual staining assays. The levels of oxidative stress markers, inflammatory biomarkers, apoptotic protein levels, and phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT)/mammalian target of rapamycin (mTOR) pathway proteins were assessed using the respective test kits.
Results
The results of the free radical scavenging assays proved the antioxidant properties of farnesol. The MTT assay findings demonstrated a significant reduction in C6 cell viability following treatment with farnesol. The findings of the fluorescent assays proved the onset of apoptosis in the farnesol-treated C6 cells. Furthermore, the farnesol treatment effectively increased oxidative stress, increased apoptotic protein levels, and reduced inflammatory marker levels in the C6 glioma cells. The PI3K, AKT, and mTOR protein expression were also successfully downregulated in the C6 glioma cells by farnesol.
Conclusion
The current results demonstrate that farnesol effectively suppresses viability and triggers apoptosis in C6 cells. Thus, it has the capacity to be an effective anticancer agent to treat glioma. Nonetheless, additional work is still necessary in the future to determine the specific molecular mechanisms involved in farnesol-induced apoptosis in C6 cells.
Keywords
Introduction
Glioma, a type of brain tumor, has long been a subject of intense research and clinical concern due to its challenging nature and significant impact on patient outcomes. These tumors, which arise from the glial cells in the brain, are known for their aggressive behavior, complex biology, and significant morbidity and mortality in the affected individuals (Nguyen et al., 2021). Recently, considerable advancements have been made in our understanding of glioma, particularly in the areas of diagnosis, imaging, and treatment. Low-grade gliomas, in particular, present a unique challenge, as many patients may survive for a decade or more but face a higher risk of treatment-related complications. Malignant gliomas, such as glioblastoma, are the most widespread type of primary brain cancer in adults and account for between 2 and 3% of cancer deaths worldwide (Rubiano et al., 2023).
The difficulty in accurately differentiating between tumor and normal brain tissue during surgical resection is a major challenge in the management of gliomas. Unintentionally leaving behind tumor tissue can lead to tumor progression, while over-resection can cause permanent neurological deficits that significantly impact a patient’s quality of life (Yalamarty et al., 2023). Recent literature has emphasized the capacity of emerging technologies, such as hyperspectral imaging, to aid in intraoperative visualization and delineation of tumor boundaries (Pineda et al., 2023). Despite significant advancements in our understanding and management of gliomas, these tumors remain a formidable foe. Surgical resection, while an essential component of treatment, is not a standalone solution. Technological advancements and a deeper understanding of the underlying biology of gliomas are crucial for improving patient outcomes and developing more effective treatment strategies (Habashy et al., 2022).
Apoptosis is a highly controlled cell death mechanism that plays a crucial role in the progression of cancer. Cancer cells often exhibit deregulated apoptotic pathways, allowing them to escape apoptosis and multiply uncontrollably. Targeting these apoptotic mechanisms has appeared as a talented strategy in cancer treatment (Gousias et al., 2022). Mitochondrial apoptosis is a key process that inhibits oncogenesis at multiple stages from transformation to metastasis. However, cancer cells must disable specific mechanisms that would normally trigger apoptosis in order to survive and proliferate. This deregulation of apoptosis is a hallmark of most types of cancer. By disabling apoptosis, cancer cells can develop resistance to various anticancer treatments (Alves et al., 2021). In addition to directly triggering apoptosis, apoptosis-targeted therapies can also be used to enhance the effectiveness of existing cancer treatments such as chemotherapy, radiotherapy, and immunotherapy (Rodríguez-Camacho et al., 2022). These conventional therapies often rely on inducing apoptosis in cancer cells as their primary mechanism of action.
Farnesol is a natural farnesane sesquiterpenoid compound extensively found in several plants, including citronella, cyclamen, balsam, musk, and several essential oils (Ishizaka et al., 2002). Previous studies have already reported that farnesol has numerous pharmacological properties such as anti-inflammatory (Jung et al., 2018), anticancer (Lee et al., 2015), cardioprotective (Szűcs et al., 2013), hepatoprotective (Vinholes et al., 2014), and antiparasitic (Sharma et al., 2023) properties. However, its anticancer activity against glioma has not been scientifically assessed yet. Hence, the present exploration was conducted to understand the anticancer effects of farnesol against glioma C6 cells.
Materials and Methods
Ferric-reducing Antioxidant Power (FRAP) Assay
The FRAP study was conducted using the previously established technique by Benzie and Strain (1996). Farnesol was added at diverse dosages (1, 5, 10, 25, 50, and 100 µM) together with the FRAP solution (1 mL), acetate buffer (300 mM), 2,4,6-tris(2-pyridyl)-s-triazine (TPTZ) reagent (10 mM), and ferric chloride solution (20 mM). The reaction solution (200 µL) was poured on a 96-well plate and left to incubate for 10 min. Subsequently, throughout the incubation period, the resultant product formed in the reaction solution was examined at 593 nm. Trolox, an analog of vitamin E, was utilized as a standard to compare the results.
2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Assay
The impact of farnesol on the removal of DPPH radicals was examined using the previously described procedure by Brand-Williams et al. (1995). The DPPH reagent (150 µL) was dissolved in ethanol (0.25 mM). It was then added with different concentrations of farnesol (1–100 µM) for 30 min at 37°C. Afterward, the absorbance was taken at 515 nm. Trolox, an analog of vitamin E, was utilized as a standard to compare the results.
Cell Culture Collection
The C6 cells (sourced from rat glioma) were purchased from ATCC, USA, and cultured in Dulbecco’s Modified Eagle Medium (DMEM) media with 10% fetal bovine serum (FBS) and 1% antimycotic mixture in a CO2 (5%) incubator. The mature cells were collected once they achieved 80% confluency and used for further treatment, fluorescence staining, and biochemical assays.
3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Assay
The viability of glioma C6 cells was examined using the MTT test (Mosmann, 1983), comparing control cells with farnesol-treated cells. The C6 cells were cultured for 24 h and subsequently treated with farnesol at various doses (1, 5, 10, 25, 50, and 100 µM) for an additional 24 h. After the treatment, a solution of MTT (20 µL) and DMEM (100 µL) was combined in the wells for 4 h. Once the formazan depositions had been dissolved using 100 µL of dimethyl sulfoxide (DMSO), the absorbance was taken at 570 nm.
Dual Staining
The dual staining test was performed on the control and farnesol-treated C6 cells to investigate apoptosis. The C6 cells were cultivated for 24 h and then exposed to 25 µM of farnesol and/or 2 µg of doxorubicin (DOX) for another 24 h. Subsequently, a solution containing 100 µg/mL acridine orange (AO)/ethidium bromide (EB) dyes was mixed into the wells for 5 min in the absence of light. Finally, the apoptotic cells were assessed in C6 cells using a fluorescence microscope.
4′,6-Diamidino-2-Phenylindole (DAPI) Staining
The DAPI staining method (Otto, 1990) was used to investigate the apoptotic cell nuclear morphology of control and treated C6 cells. Following the culturing of C6 cells into a 24-well plate, the cells were treated with farnesol at a 25 µM concentration for 24 h. After being fixed in paraformaldehyde (4%) for 30 min, the cells were stained with 200 µg/mL of DAPI for 20 min. Subsequently, the impact of farnesol on apoptosis in C6 cells was examined using a fluorescence microscope.
Analysis of Oxidative Stress Markers
The C6 cells were lysed using lysis buffer, and the lysate was prepared to assess the oxidative stress markers. The thiobarbituric acid reactive substances (TBARS), superoxide dismutase (SOD), and glutathione (GSH) levels were measured in the control and farnesol-exposed C6 cells. The assays were done using kits provided by the manufacturer (Abcam, USA), following the protocols given in the kit.
Analysis of Apoptotic Protein Expressions
To examine the levels of apoptotic biomarkers, the cell lysate was obtained from C6 cells using a cell lysis buffer. The B-cell lymphoma 2 (Bcl-2), Bcl-2-associated X protein (Bax), cyclin D1, caspase-3, and -9 levels were measured in the control and farnesol-exposed C6 cells. The assays were done using the commercial kits and the instructions given by the manufacturer (Elabscience, USA).
Analysis of Inflammatory Marker Levels
The tumor necrosis factor-alpha (TNF-α), nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), interleukin-6 (IL-6), and cyclooxygenase-2 (COX-2) levels were examined in the control and farnesol-exposed C6 cells using commercially available kits. The instructions provided by the manufacturer (Abcam, USA) were followed to conduct the experiments.
Measurement of Phosphoinositide 3-Kinase (PI3K)/Protein Kinase B (AKT)/Mammalian Target of Rapamycin (mTOR) Pathway Markers
The PI3K, AKT, and mTOR protein levels were assessed in the control and farnesol-exposed C6 cells using commercially available kits. The guidelines given by the kit’s manufacturer (Abcam, USA) were followed to perform the assays.
Statistical Analysis
The statistical assays were conducted using the statistical package for the social sciences (SPSS) software, and the results were given as the mean ± standard deviation (SD) of triplicates. The data were assessed using one-way analysis of variance (ANOVA) and Duncan’s multiple range test (DMRT), with a significance level of p < 0.05.
Results
Effect of Farnesol on the In Vitro Scavenging of Free Radicals
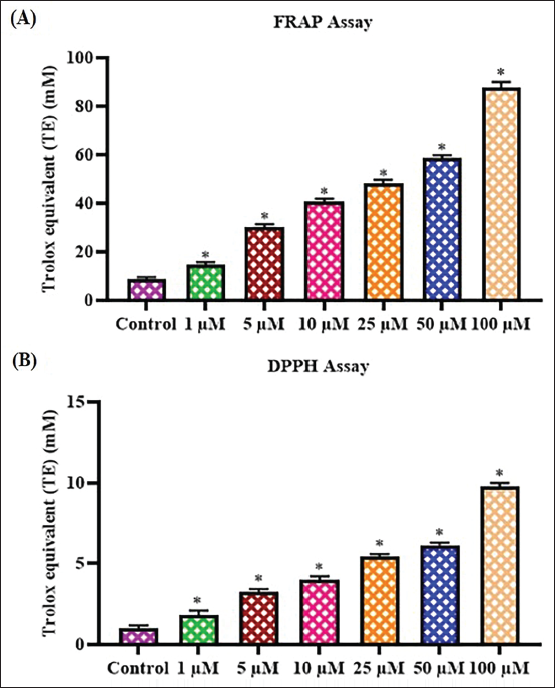
The in vitro antioxidant potentials of the farnesol were examined using FRAP and DPPH radical scavenging assays. The results of these assays are displayed in Figure 1. Farnesol treatment at different concentrations effectively decreased the levels of free radicals. Farnesol at 1–100 µM concentrations effectively suppressed the DPPH and showed ferric-reducing power. The in vitro antioxidant potentials of the farnesol were evidenced by the results of FRAP and DPPH scavenging assays (Figure 1).
Effect of Farnesol on the Glioma C6 and Normal Vero Cell Viabilities
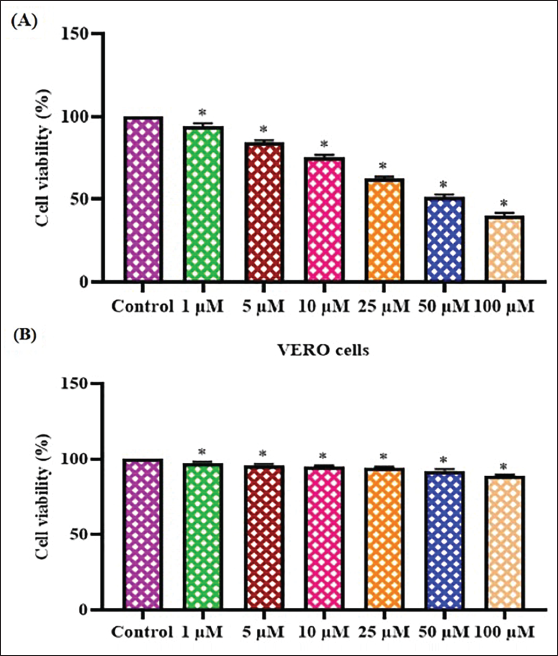
Figure 2 presents the findings of the MTT cytotoxicity assay, showing the influence of farnesol on the growth of glioma C6 and nonmalignant Vero cells. Farnesol treatment significantly reduced the viability of C6 cells over a range of concentrations (1–100 µM). In addition, the farnesol treatment at 1–100 µM did not have any impact on the viability of Vero cells. The increasing concentrations of farnesol had a slight inhibition on the growth of C6 cells, as seen in Figure 2. The half-maximal inhibitory concentration (IC50) of farnesol was noted at 25 µM against C6 cells, and the same dosage was chosen for subsequent experiments.
Effect of Farnesol on the Apoptosis in Glioma C6 Cells
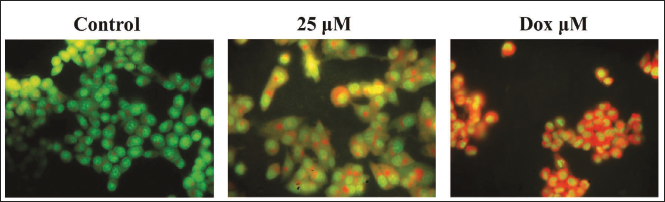
The dual staining technique was performed to study apoptosis in both the control and farnesol-treated C6 cells, and the outcomes are given in Figure 3. After being treated with 25 µM of farnesol, the C6 cells displayed higher levels of yellow and orange fluoresced cells, providing evidence of the occurrence of both early and late-stage apoptosis. The DOX treatment findings also demonstrated elevated cells demonstrating a vivid yellow/orange fluorescence, providing confirmation of the apoptosis onset in the farnesol-treated cells (Figure 3).
Effect of Farnesol on the Apoptosis in the C6 Cells
The apoptotic cell death level in the control and farnesol-exposed C6 cells was examined using DAPI staining, and the findings are shown in Figure 4. The C6 cells exposed to farnesol at a 25 µM concentration showed increased occurrences of apoptosis. The 25 µM of farnesol significantly enhanced apoptosis and resulted in a decrease in cell counts in the C6 cells, which was also evidenced by the results of the DOX treatment.

Effect of Farnesol on the Oxidative Stress Marker Levels in the C6 Cells
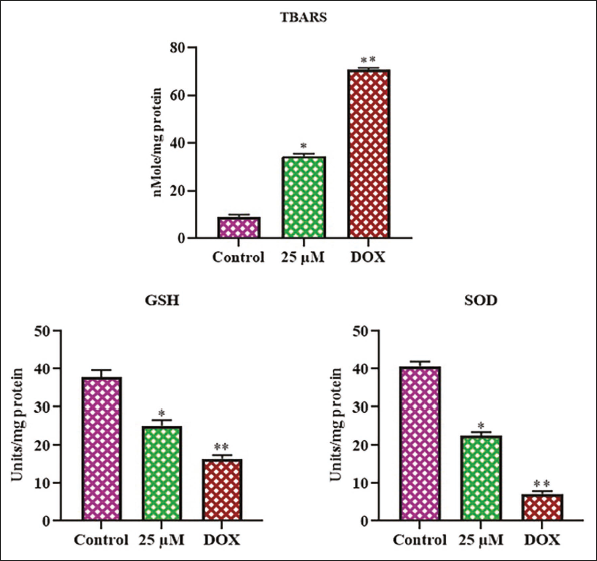
Figure 5 displays the TBARS, GSH, and SOD contents in the control and farnesol-exposed C6 cells. The control cells exhibited elevated levels of SOD and GSH, while there was a decrease in TBARS levels. Meanwhile, when C6 cells were treated with 25 µM of farnesol, there was a considerable elevation in TABRS levels and a reduction in antioxidant levels compared with the control. The treatment with DOX led to an elevation in the TBARS and a reduction in the GSH and SOD in the C6 cells. These outcomes support the hypothesis that farnesol causes oxidative stress in C6 glioma cells.
Effect of Farnesol on the Apoptotic Protein Expressions in the Glioma C6 Cells
Apoptotic protein expressions in the control and farnesol-exposed C6 cells were studied, and the outcomes are presented in Figure 6. In the control cells, the expressions of Bax, caspase-3, and -9 were decreased, whereas the cyclin D1 and Bcl-2 expressions were increased. Nevertheless, the treatment of glioma C6 cells with 25 µM farnesol resulted in a substantial enhancement of Bax, caspase-3, and -9 activities, accompanied by a reduction in cyclin D1 and Bcl-2 expressions. The outcomes of the DOX treatment confirmed these findings since it also enhanced the production of apoptotic proteins in the C6 cells. Thus, it was clear that farnesol induces apoptosis in the C6 glioma cells.
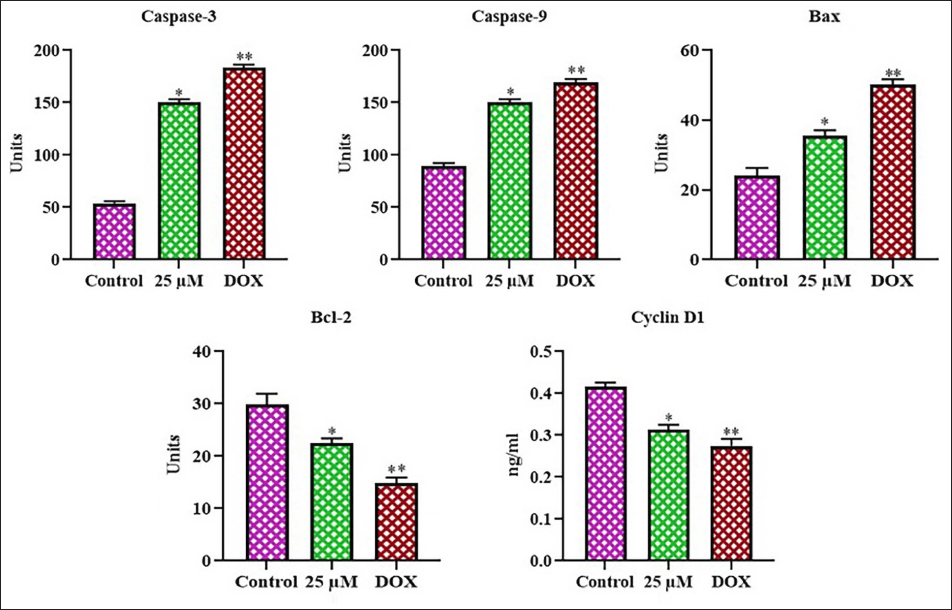
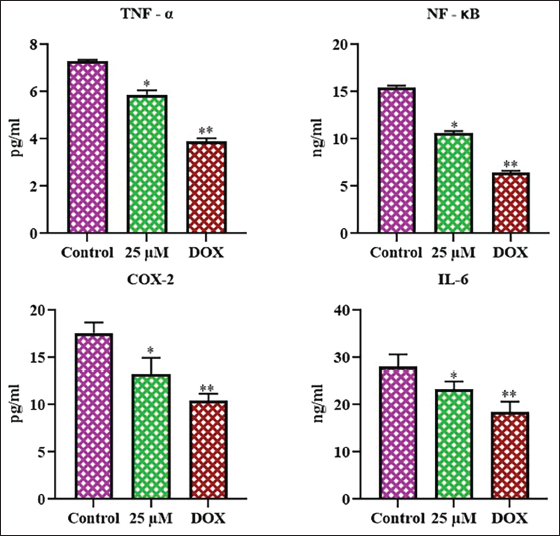
Effect of Farnesol on the Inflammatory Protein Levels in the C6 Cells
Figure 7 displays the findings of the analysis of inflammatory marker levels in control and treated C6 cells. Treatment with 25 µM of farnesol considerably decreased the TNF-α, NF-κB, IL-6, and COX-2 levels in C6 cells when compared with control. Therefore, it was evident that farnesol may trigger apoptosis in C6 cells by downregulating inflammatory protein expression.
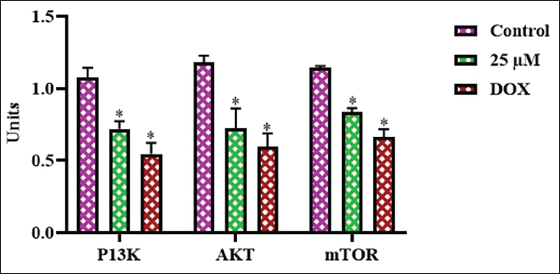
Effect of Farnesol on PI3K/AKT/mTOR Pathway Protein Levels in the Glioma C6 Cells
The expressions of PI3K, AKT, and mTOR proteins were analyzed in the control and farnesol-exposed C6 cells (Figure 8). In the control cells, PI3K, AKT, and mTOR expressions were increased. However, the administration of farnesol at a 25 µM concentration to the C6 cells led to a considerable decrease in the PI3K, AKT, and mTOR expressions in comparison to the control group. The results of the DOX treatment also supported these findings, since it also reduced the PI3K/AKT/mTOR expressions. Hence, it was clear that farnesol treatment blocks cell growth and promotes apoptosis in glioma cells.
Discussion
Antioxidants are compounds that can neutralize free radicals and reactive oxygen species (ROS), thereby preventing or delaying oxidative damage to cells and tissues (Marino et al., 2023). The assessment of the antioxidant properties of bioactive compounds is crucial for understanding their potential health benefits and applications in the food and pharmaceutical industries. Two widely used assays for assessing in vitro antioxidant capacity are the FRAP assay and the DPPH scavenging assay. The FRAP analysis determines the efficacy of samples to reduce Fe3+ ions to Fe2+ ions, while the DPPH study evaluates the radical scavenging capacity of samples (Marques et al., 2014; Valko et al., 2007). These assays provide complementary information about the antioxidant properties of bioactive compounds as they measure different mechanisms of action. The FRAP assay is particularly useful for assessing the antioxidant activity of bioactive compounds. The DPPH assay, on the other hand, determines the efficacy of bioactive compounds to scavenge the stable DPPH radical. This assay provides information on the free radical scavenging capacity of bioactive compounds, which is another important mechanism of antioxidant action (Aini et al., 2019). Combining the results of FRAP and DPPH assays can provide a more comprehensive understanding of the antioxidant properties of bioactive compounds. The present findings prove that farnesol demonstrated effective antioxidant properties.
Apoptosis plays a significant role in the regulation of cellular homeostasis and is considered a potential target for cancer therapies (Thang et al., 2023). In this study, to investigate apoptotic cell death in glioma C6 cells, we have employed DAPI and dual staining assays. DAPI is a fluorescent stain that binds to deoxyribonucleic acid (DNA), enabling the visualization of nuclear morphological changes associated with apoptosis, such as chromatin condensation and fragmentation. Dual staining assays, on the other hand, utilize a combination of fluorescent dyes to differentiate between apoptotic and necrotic cell populations (Moldovan et al., 2023). The present results from both DAPI and dual staining assays indicate that farnesol successfully triggered apoptosis in the glioma C6 cells.
Understanding the underlying mechanisms contributing to glioma progression and treatment resistance is crucial to developing more therapeutic interventions (Awuah et al., 2022). Oxidative stress, an imbalance between ROS accumulation and the cellular antioxidant defense mechanism, has been implicated in the pathogenesis of glioma. TBARS, SOD, and GSH are key markers of oxidative stress, and their levels can provide insight into the cellular redox status and its impact on apoptosis (Acevedo-León et al., 2023). The present research work aims to investigate the TBARS, SOD, and GSH levels and to elucidate the role of oxidative stress in the induction of apoptosis in glioma C6 cells. Increased levels of TBARS, a marker of lipid peroxidation, have been observed in glioma tissues, indicating enhanced oxidative damage to cellular membranes. Additionally, altered activities of antioxidant enzymes, such as SOD, have been reported in glioma cells, suggesting a dysregulation of the cellular defense system against ROS (Acevedo-León et al., 2022). GSH, a crucial antioxidant, plays a pivotal role in regulating the cellular redox balance. Depletion of GSH has been linked to the stimulation of apoptotic signaling, such as the activation of proapoptotic factors and the inhibition of antiapoptotic proteins (Chiang et al., 2023). The outcomes from this work show the mechanisms by which farnesol-mediated oxidative stress modulates glioma cell apoptosis. The farnesol treatment effectively promoted oxidative stress, thereby facilitating apoptosis in glioma C6 cells.
Apoptosis is a pivotal cell death mechanism that maintains cellular homeostasis and eradicates damaged or abnormal cells. Understanding the intricate interplay between key apoptotic regulators is paramount for developing effective therapeutic strategies for glioma (Rana et al., 2022). The Bcl-2 family of proteins, both prosurvival (e.g., Bcl-2) and proapoptotic (e.g., Bax) members, plays a central role in the mitochondrial pathway of apoptosis. Bcl-2, an antiapoptotic gene, is often overexpressed in glioma cells, conferring a survival advantage (Vera et al., 2022). In contrast, the proapoptotic protein Bax is a crucial regulator of membrane permeabilization in mitochondria, which promotes cytochrome c release and the activation of caspase cascades (Merati et al., 2023). The cyclin-dependent kinase inhibitor, cyclin D1, has also been implicated in the regulation of apoptosis in glioma cells. Cyclin D1 overexpression can inhibit the proapoptotic function of Bax, thereby promoting cell survival and proliferation (Jeffreys et al., 2022). Additionally, the caspase family of proteases, which act as the executioners of apoptosis, plays an imperative role in the demise of glioma cells. The activation of caspases, such as caspase-2, can lead to the silencing of the antiapoptotic protein survivin, further tipping the balance towards apoptosis (Eskandari & Eaves, 2022). The delicate interplay between the Bcl-2 family members, cyclin D1, and the caspase cascade is crucial in determining the fate of glioma C6 cells. Understanding these complex regulatory mechanisms can offer more insights to develop effective therapies focused on reestablishing the balance between pro and antiapoptotic signals, ultimately leading to the effective elimination of glioma cells. The present outcomes revealed that the farnesol substantially enhanced the Bax, caspase-3, and -9, and a subsequent reduction in Bcl-2 and cyclin D1 expressions in the C6 cells. Hence, it was clear that farnesol successfully induced apoptosis in the C6 glioma cells.
Recent advances in understanding the molecular mechanisms underlying glioma cell survival and resistance to apoptosis have revealed the crucial roles of several key signaling pathways and inflammatory mediators. Among these, the cytokines TNF-α and IL-6, the transcription factor NF-κB, and COX-2 have been widely assessed for their involvement in glioma pathogenesis and their potential as therapeutic targets (Aguilar-Cazares et al., 2022). TNF-α is a potent mediator of inflammation in the brain, and its signaling through the NF-κB pathway is known to drive the expression of numerous prosurvival and antiapoptotic proteins in glioma cells (Borovcanin et al., 2023). Activation of NF-κB by TNF-α leads to the upregulation of genes like Bcl-2, which counteracts the proapoptotic signals and promotes glioma cell survival (Jarmuzek et al., 2023). In addition, TNF-α can also induce the production of IL-6, another important inflammatory cytokine that has been implicated in glioma progression. IL-6 signaling has been shown to activate parallel prosurvival pathways, such as the Janus kinase (JAK)/signal transducer and activator of transcription 3 (STAT3) cascade, further enhancing the resistance of glioma cells to apoptosis (Singh et al., 2022). Likewise, the enzyme COX-2, which is commonly overexpressed in gliomas, can contribute to the inflammatory microenvironment and inhibit apoptosis through the production of prostaglandin E2 and other lipid mediators. Targeting the COX-2 pathway has appeared as a hopeful therapeutic target to sensitize glioma cells to apoptosis (Lombardi et al., 2022). The interplay between these key inflammatory mediators, TNF-α, NF-κB, IL-6, and COX-2, plays a pivotal role in sustaining the survival and proliferation of glioma cells, as well as conferring resistance to apoptosis. The present study proved that the farnesol treatment significantly reduced the levels of these markers in the glioma cells, thereby promoting apoptosis.
The PI3K/AKT/mTOR pathway is an imperative regulator of numerous cellular mechanisms, such as cell growth, survival, and metabolism. The PI3K/AKT/mTOR pathway is dysregulated in many types of cancer, including glioma. Activation of this pathway can result in the inhibition of apoptosis, permitting tumor cells to evade programmed cell death (Stanciu et al., 2022). Specifically, the PI3K/AKT cascade inhibits proapoptotic genes, like BAD, while promoting the activity of antiapoptotic genes, like Bcl-2. This shift in the balance of apoptotic regulators ultimately favors cell survival and contributes to the aggressive nature of gliomas (Glaviano et al., 2023). It has been highlighted the efficacy of targeting the PI3K/AKT/mTOR signaling as a therapeutic strategy for glioma (Su et al., 2022). Inhibition of this pathway, either through pharmacological interventions or genetic approaches, has been shown to induce apoptosis and suppress tumor growth in glioma models (Mohamed et al., 2022). In this work, the outcomes showed that the farnesol treatment substantially reduced the PI3K/AKT/mTOR expression in the glioma cells.
Conclusion
The current results demonstrate that farnesol effectively suppresses viability and triggers apoptosis in glioma cells. Farnesol treatment triggered oxidative stress and apoptosis by increasing the levels of Bax and caspases while decreasing the antiapoptotic protein Bcl-2 expression in C6 cells. Furthermore, the farnesol also blocked the PI3K/AKT/mTOR pathway in the glioma cells. Thus, it has the potential to be an effective anticancer agent to treat glioma. Nonetheless, additional work is still necessary in the future to determine the specific molecular mechanisms involved in farnesol-induced apoptosis in C6 cells.
Footnotes
Abbreviations
AKT: Protein kinase B; Bax: Bcl-2-associated X protein; Bcl-2: B-cell lymphoma 2; COX-2: Cyclooxygenase-2; DAPI: 4′,6-Diamidino-2-phenylindole; DPPH: 2,2-Diphenyl-1-picrylhydrazyl; FRAP: Ferric-reducing antioxidant power; GSH: Glutathione; IL-6: Interleukin-6; mTOR: Mammalian target of rapamycin; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; PI3K: Phosphoinositide 3-kinase; SOD: Superoxide dismutase; TBARS: Thiobarbituric acid reactive substances; TNF-α: Tumor necrosis factor-alpha.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
