Abstract
Background
Chinese medicinal ointment was first recorded in the Shan Hai Jing (The Classic of Mountains and Seas). Different formulations of Chinese medicinal ointments have been made based on the traditional Chinese medicine (TCM) theory. Many TCMs possessed antibacterial properties that were used as microecological regulators to inhibit the growth of gram-negative bacteria, “Escherichia coli.”
Objectives
To develop and make an antibacterial ointment by Tripterygium wilfordii (TW) and evaluate its antibacterial function.
Materials and Methods
The methodologies included physical (cream selection), chemical [ultraviolet–visible (UV–vis) and high-performance liquid chromatography-diode array detector (HPLC-DAD)], and biological (agar assay) experiments for making TW ointment.
Results
An active ingredient, “celastrol,” was extracted from TW. Aqueous Cream BP was chosen as the base for ointment making since it did not have any smell and contained only natural substances. UV–vis and HPLC-DAD identified the active ingredient of “celastrol” from TW with an absorption peak at 230 nm and a symmetrical peak at a retention time of 19.855 min. The 1%, 2%, and 5% TW ointments with antibacterial properties and inhibitory effects on the colony formation units of E. coli were 109.33 ± 14.32, 85.67 ± 10.22, and 44.00 ± 6.66, respectively.
Conclusion
The Chinese medicinal ointment with antibacterial properties from TW was successfully developed using an Aqueous Cream BP and the Chinese medicinal plant “TW.” An active ingredient, “celastrol,” from “TW,” was determined by using UV–Vis and HPLC-DAD studies. The 1%, 2%, and 5% TW ointments were evaluated for its effectiveness for antibacterial properties. These Chinese medicinal ointments were the starting milestone for further studying the biological mechanisms or applications on human skin.
Introduction
Chinese medicinal ointment has a long history and was first recorded in the Shan Hai Jing (The Classic of Mountains and Seas). “Ointment” was widely used in the Tang Dynasty. Liangyao Yu Duyao (Good Medicine and Poisons) was the later version that clearly defined “ointment” and its pharmaceutical applications (Xu et al., 2019).
Over the past few years, many formulations have been made of Chinese medicinal ointments based on the traditional Chinese medicine (TCM) theory, which can impede signaling pathways and inhibit microbial growth, leading to a potent antibacterial effect and demonstrating remarkable efficacy against bacterial infections (Zou et al., 2023). Growing evidence has shown that TCMs with the advantage of low drug resistance because of a variety of active components, such as flavonoids, alkaloids, phenols, and quinones, that can inhibit the growth of drug-resistant bacteria and treat various drug-resistant bacterial infections (Li et al., 2022).
One of the most common gram-negative bacteria, “Escherichia coli,” can cause skin infections with the symptom of cellulitis. This was the type 1 necrotizing soft tissue infection (NSTI), where layers within the dermis, subcutaneous tissue, superficial fascia, or muscle become infected (Gallois et al., 2015). Many TCMs were microecological regulators to inhibit the growth of E. coli and restore the balance of the immune system (Wei et al., 2023).
Previous studied TCMs with antibacterial property include Cortex magnoliae officinalis, Cortex phellodendri, Flos caryophylli, Flos lonicerae japonicae, Fructus armeniaca mume, Fructus forsythiae suspensae, Herba cum radice viola yedoensis, Herba menthae haplocalycis, Pericarpium granati, Radix et rhizoma rhei, Radix gentianae, Ramulus cinnamomi cassia, and Rhizoma cimicifugae (Wong et al., 2010). The antibacterial mechanism involved altering membrane permeability, inhibiting protein and nucleic acid synthesis, suppressing enzyme activity in vivo, and managing the ability of pathogenic bacteria (Liu et al., 2022).
Chinese medicinal ointments are considered the simplest and most convenient medication dosage form with high compliance and flexibility (Kandhare et al., 2016). This was employed as the media for transdermal drug or cosmetic delivery with potential application prospects for skin diseases (Oberli et al., 2014). For example, Wang et al. (2021) developed a six-herb Chinese medicine compositions ointment for the treatment of hypertrophic scars, which included Salvia miltiorrhiza, Ligusticum wallichii, Sophora flavescens, Crocus sativus, Galla chinensis, and Brucea javanica. It was shown good immunohistochemical results with antibacterial and antihypertrophic scar properties that were inhibited hypertrophic scar formation (Wang et al., 2021). Ho et al. (2020) also identified that “Jinchuang ointment” was better used in external incised wounds, which accelerated wound closure significantly. The TCMs of this ointment consisted of Camphora officinarum, Boswellia sacra, Commiphora myrrha, Draconis sanguis, Senegalia catechu, and borneol (Ho et al., 2020). In 2022, Zhang et al. (2022) reported a dermatitis ointment that ameliorated inflammatory responses and dysregulation of itch-related molecules in atopic dermatitis. This ointment contained Coptidis Rhizoma, Phellodendri chinensis Cortex, Angelicae Sinensis Radix, Rehmanniae Radix, Curcumae Longae Rhizoma, and Sesamum indicum L. It effectively attenuated the pathological alterations of Th1/2 cytokines and itch-related mediators, as well as inhibited the phosphorylation of mitogen-activated protein kinase (MAPK) and nuclear factor-kappa B (NF-κB) (Zhang et al., 2022).
The above Chinese medicinal ointments are formulated with at least six herbals. They may have a chance to cause adverse reactions due to the potential of herbal–herbal interactions (Kam & Liew, 2002). The single herbal ointment is more stable and reliable compared to multiple herbal ointments. A herbal plant “Tripterygium wilfordii (TW)” whose root has antibacterial properties (Ru et al., 2019). Because it consists of an active ingredient, “celastrol,” that is natural and less toxic (Chen et al., 2018), this is pentacyclic nortriterpen quinone and belongs to the family of quinone methides (Law et al., 2020). Thus, the communication aims to develop and make ointments by TW, to determine the active ingredient, “celastrol,” from making TW ointment, and to evaluate the antibacterial properties of TW ointment.
Materials and Methods
TW was bought from a Chinese medicine shop in Hong Kong. Celastrol (98% HPLC, CAS No. 34157-83-0) was purchased from Shang Hai Shfeng Biological Technology Co., Ltd. Aqueous Cream BP was bought from a health and beauty chain store. Methanol (MeOH) and phosphoric acid (HPLC grade) were obtained from Anaqua Global International Inc., Ltd. Acetonitrile (HPLC grade) was purchased from RCI Labscan., Ltd. Difco™ Nutrient Agar BD was procured, and E. coli ATCC® 25922 was obtained from Thermo Fisher Scientific Inc., USA. All chemicals used were of analytical grade and were used as received without any further purification.
Preparation of the TW Powder
60 g of TW was grided to powder in the size of 80 mesh using a Gaoxin GX-08 blender (Figure 1) and placed in a sealed bag, which prevented raw material contamination and air oxidation for the growth of microbial, for example, mold.
Preparation of Tripterygium wilfordii (TW) Powder in 80 Meshes.

Physical Experiment
Cream Selection
A cream was chosen fragrance-free through a careful sniff. The cream selection should not have a scent and a greasy texture. Comparison information of cream selection will be reviewed in the result and discussion part.
Chemical Experiments
Determination of Ultraviolet–Visible (UV–Vis) Spectroscopy
Preparation of Standard Solution
A stock solution of 0.1% celastrol was prepared by accurately measuring 100 mg celastrol and dissolving it in 100 mL of ethanol. 1 mL of stock solution was pipetted and transferred to the UV cuvette for measurement.
Preparation of Sample Solution
A sample solution of 0.00255% TW extractant was prepared by accurately measuring 2.55 mg TW extractant and dissolved in 100 mL of ethanol. The sample solution was filtered through a 0.45 µm PTFE to remove the contaminants and impurities, then it was transferred to the UV cuvette for measurement.
Determination of High-Performance Liquid Chromatography-Diode Array Detector (HPLC-DAD)
Preparation of Standard Solutions
10 mg of celastrol was weighted accurately, which dissolved in 10 mL of MeOH to make a stock solution with a concentration of 1 mg/mL. The stock solution was diluted into 5 proportions, including 5, 10, 20, 40, and 80 mg/L, then sonicated for 10 min to ensure the celastrol standard was completely dissolved.
Preparation of Sample Solutions
0.4 g of TW powder was weighted accurately, which dissolved in 10 mL of MeOH to make a solution with a concentration of 0.04 g/mL. This was sonicated for 30 min and then centrifuged at 3,000 rpm for another 5 min. The test solution was finally filtered through a 0.45 µm PTFE filter to remove the contaminants and impurities.
HPLC Requirements and Conditions
The HPLC has a column of Eclipse Plus C18, 4.6 × 250 mm in length, with 5 µm diameter. Eluent A and B consisted of acetonitrile and 0.03% H3PO4/H2O, respectively. It was run in gradient elution, 50% of eluent A and B for 0 min. 100% of eluent A after 25 min. Column temperature was kept constant at 30°C. The HPLC was equipped with a DAD and measured at 250 nm for standard and sample solutions.
Extraction of Celastrol from TW
9.28 g of TW was weighed and dissolved in 50 mL of MeOH. It was sonicated for half an hour. The solution was then filtered through a 0.45 µm PTFE and transferred to a test tube. Different concentrations (1%, 2%, and 5%) of celastrol were determined by HPLC to make ointments.
Preparation of TW Ointments
The concentrations of TW extractants for each ointment were selected as 1%, 2%, and 5%. 5 mL of Aqueous Cream BP was mixed with the corresponding concentrations, 0.05 mL (1%), 0.1 mL (2%), and 0.25 mL (5%) of TW extractants. It was stirred gently and sonicated for 30 min, which was sealed with aluminum packaging boxes in a total volume of 10 g for each ointment, respectively. The final ointments were stored in the fridge for a night before use (Figure 2).
Preparation of 1%, 2%, and 5% Tripterygium wilfordii (TW) Ointments.

Biological Experiments
Preparation of Agar Plate
23 g of nutrient agar powder was weighed and dissolved in 1 L of deionized water. It was stirred, heated thoroughly, and boiled at 121°C for 15 min. The dissolved agar was autoclaved. After autoclaving, it was poured into the Petri dishes slowly and evenly to prevent any bubbles. The agar was cooled to room temperature and was further waited for 10 min for solidification, then closed with the lids. Agar plates were sealed tightly and stored in the fridge before use.
Serial Dilution of E. coli
9 mL of deionized water was pipetted into the four test tubes. 1 mL of E. coli solution with a concentration of 10–1 was pipetted into the first test tube and shaken thoroughly. It was then pipetted 1 mL of solution from the first test tube to the second test tube, shaken thoroughly again, with a concentration of 10–2, and so on. The dilution steps were repeated until the concentration of 10–4 was achieved.
Antibacterial Assay
100 µL of the prepared E. coli suspension (~10–4) was evenly spread on the Petri dishes. 50 µL of TW (1%, 2%, and 5%) with MeOH and (1%, 2%, and 5%) TW ointments were spread onto the Petri dishes evenly and correspondingly. It was incubated for 24 h at 37°C in a constant temperature biochemical incubator (Elbing & Brent, 2019). After 24 h, the production of colony forming units (CFU) was observed. This experiment was repeated three times.
Statistical Analysis
The statistical values were reported with the means of three separate experiments and standard deviations (±SD). Results were subjected to SPSS software.
Results
Cream Selection
Before starting the research, five creams were selected and compared from the market and their function, which was mainly to keep moisture and prevent dehydration on the skin. These creams consisted of different components, and most creams included extra essential oils, plant components, and aroma (Table 1). However, “Aqueous Cream BP” was the only cream that avoided the above-mentioned compounds. Since fragrances are common allergens that cause cutaneous side effects (Arribas et al., 2013), exposure to the skin must be limited; thus, the selected cream should not contain fragrance components. Therefore, “Aqueous Cream BP” was an additive-free cream that will be used in making our ointment.
List of Components for Different Creams.

Determination of UV–Vis Spectroscopy
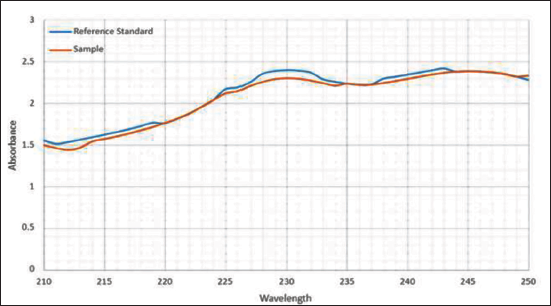
The UV–Vis absorption spectra were recorded from 210 to 250 nm. Results have shown that the reference standard (celastrol) and sample (TW extractant) displayed the absorption peak at 230 nm (Zhang et al., 2018), and the sample exhibited better absorption at 230 nm in a more concentrated solution. These results indicated that the sample contained the active ingredient “celastrol” (Figure 3). The other TW extracts, such as triptolide, triptophenolide, and demethylzeylasteral, with optimum absorptions were 218 nm (Ma et al., 2008), 430 nm, and 268 nm (Wang et al., 2016), respectively.
Ultraviolet–Visible (UV–Vis) Spectrum of the Reference Standard (Celastrol, Blue Line) and Sample (Tripterygium wilfordii Extractant, Orange Line).
Determination of HPLC-DAD
Different Concentrations of Standard Solutions (Celastrol)
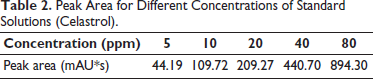
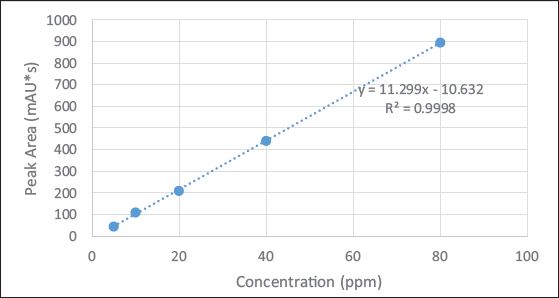
The calibration curves of different concentrations for standard solutions (celastrol) were established. It showed a linear relation within the range of 5–80 ppm. The slope or regression equation of the calibration curve was calculated as y = 11.299x – 10.632 with the correlation coefficient, R2 > 0.999, where y is the peak area and x is the concentration of celastrol. The calibration curve was the best-fit line, it demonstrated a good linearity between the peak height ratios versus concentrations (Table 2 and Figure 4).
Peak Area for Different Concentrations of Standard Solutions (Celastrol).
Calibration Curves at Different Concentrations for Standard Solutions (Celastrol) Between the Peak Height Ratios versus Concentrations.
Different Concentrations of Sample Solutions
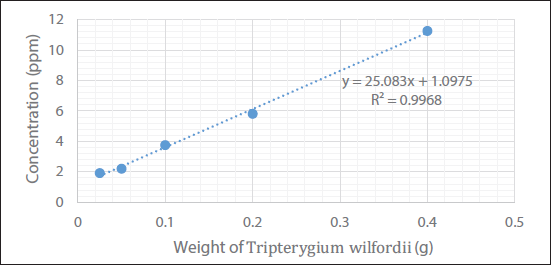
A calibration curve of the sample (TW extractant) showed a linear relation in the range of 0.025–0.4 g. The slope or regression equation of the calibration curve was calculated as y = 25.083x + 1.0975 with the correlation coefficient, R2 > 0.99, where y is the concentration of celastrol and x is the weight of TW. The calibration curve was the best-fit line, it demonstrated a good linearity between the concentration ratios versus weights (Table 3 and Figure 5).
Relationship for Different Weight of Tripterygium wilfordii (TW) with Different Concentrations.

Calibration Curves at Different Concentrations for Sample Solutions (Celastrol) between the Concentrations Ratios Versus Weights.
Specificity of the Reference Standard (Celastrol) and Sample (TW Extractant)
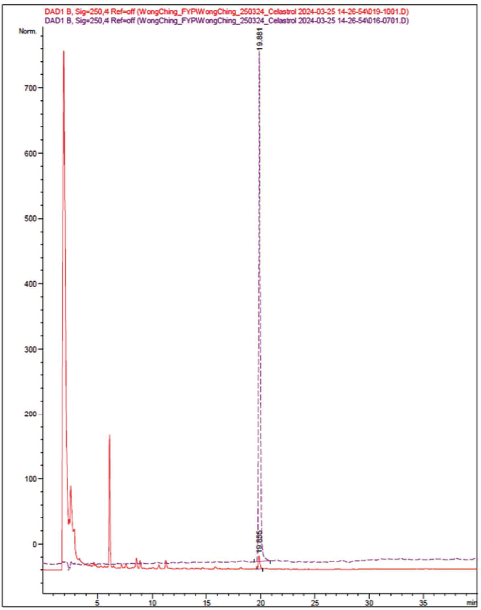
The HPLC-DAD chromatograms of the reference standard (celastrol) and sample (TW extractant) were overlapped, which showed a symmetrical peak at a retention time of 19.855 min with a detection wavelength of 250 nm (Figure 6).
High-Performance Liquid Chromatography-Diode Array Detector (HPLC-DAD) Chromatograms of the Reference Standard (Celastrol, Red Line) and Sample (Tripterygium wilfordii Extractant, Purple Line).
Antibacterial Assay
Nine sets of agar plate assay experiments were conducted for the determination of antibacterial activity (Figure 7), which included six control groups: (a) E. coli, (b) E. coli + MeOH, (c) E. coli + 1% TW in MeOH, (d) E. coli + 2% TW in MeOH, (e) E. coli + 5% TW in MeOH, and (f) E. coli + blank ointment. The remaining sample groups were (g) E. coli + 1% TW ointment, (h) E. coli + 2% TW ointment, and (i) E. coli + 5% TW ointment (Figure 7).

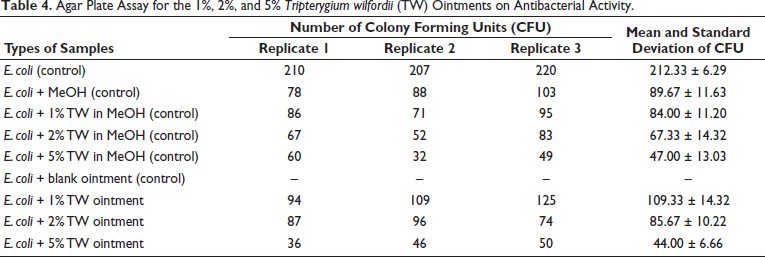
The results showed that control groups of (a) E. coli and (f) E. coli + blank ointment had no antibacterial effect. (b) E. coli + MeOH, (c) E. coli + 1% TW in MeOH, (d) E. coli + 2% TW in MeOH, and (e) E. coli + 5% TW in MeOH showed inhibition effects. Sample groups of (g) E. coli + 1% TW ointment, (h) E. coli + 2% TW ointment, and (i) E. coli + 5% TW ointment showed antibacterial effects. The greater the percentage of TW ointment, the stronger the inhibition effect for reducing the number of colony formation units. 1%, 2%, and 5% of TW ointments with inhibitory effects on the colony formation units of E. coli, which were 109.33 ± 14.32, 85.67 ± 10.22, and 44.00 ± 6.66, respectively (Table 4). It was indicated the colony formation units of E. coli proportionally decreased when the percentage of TW ointment increased.
Agar Plate Assay for the 1%, 2%, and 5% Tripterygium wilfordii (TW) Ointments on Antibacterial Activity.
Discussion
Natural ingredients and a fragrance-free cream base avoided harmful chemicals and were safe for all skin types, which provided gentle and effective skincare without causing any unwanted side effects. This cream base encapsulated and released its celastrol content at a higher rate in skin absorption (Darbre, 2023). Since celastrol was insoluble in water, an oily cream led celastrol to have more stability and efficacy.
E. coli was a gram-negative bacillus and acted as an indicator for the surveillance of antimicrobial resistance (AMR) in the environment. This tended to be more resistant to antimicrobial agents than the gram-positive since the extra protection afforded by the outer membrane. That was surrounded by two membranes, including the cytoplasmic cell membrane and the outer membrane. The monolayer of the outer membrane contained lipopolysaccharide (LPS) as the major lipid component (Epand et al., 2016). Because celastrol was a potent regulator of lipid metabolism, it exerted its lipid-regulating effects by modulating lipid synthesis, catabolism, absorption, transport, and peroxidation (Gu et al., 2023). Thus, celastrol was identified as a potential agent for lipid-related skin diseases, and E. coli was chosen for the research experiment to assess its antibacterial efficacy.
TW ointment inhibited the E. coli growth because of the terpenoids. The possible mechanism of TW ointment may be diminished mitochondrial content, resulting in altered levels of reactive oxygen species (ROS) and ATP generation. The inhibition of efflux pumps through the plasma membrane H+-ATPase supports the large transmembrane electrochemical proton gradient across the cell membrane and regulates intracellular pH, which was impaired by TW ointment, leading to inhibition of E. coli growth as well as the intracellular acidification and cell death (Xu et al., 2019).
Besides, TW ointments were against E. coli because of an active ingredient, “celastrol,” that affected the ribonucleic acid (RNA) and deoxyribonucleic acid (DNA) for protein synthesis. The cytoplasmic membrane disruption was caused by potassium leakage and the formation of mesosome-like structures. It inhibited oxygen consumption and disrupted the cellular structure of E. coli, leading to damage and death (Padilla-Montaño et al., 2021). As E. coli is a gram-negative entity, there were no simple rules for the physicochemical properties of the molecule energy via the outer membrane. Because celastrol influenced E. coli RNA and DNA synthesis, which indicated that it was effective in causing the cytoplasmic membrane disruption in E. coli (Silver, 2016). Hence, the number of colony formation units for E. coli proportionally decreased when slagged with TW ointments.
Conclusion
The Chinese medicinal ointment was developed successfully by using an Aqueous Cream BP and the Chinese medicinal plant, “TW.” Aqueous Cream BP was chosen for making ointments.
An active ingredient, “celastrol,” from TW was determined by using UV–vis and HPLC-DAD studies, with the absorption peak at 230 nm, and showed a symmetrical peak at a retention time of 19.855 min, respectively.
The 1%, 2%, and 5% TW ointments were evaluated its effectiveness for antibacterial properties, which decreased proportionally, and the number of colony formations against E. coli with 109.33 ± 14.32, 85.67 ± 10.22, and 44.00 ± 6.66. However, the biological mechanisms of TW ointments would be required for further investigation, such as cytotoxicity on human skin clinical studies.
Future Aspects
Based on the findings, the Chinese medicinal ointment from TW can be further developed and applied for skin diseases. The Chinese medicinal ointment possesses antibacterial properties, and the active ingredient “celastrol” inhibits the growth of E. coli, as E. coli can cause cellulitis, skin and soft tissue infection (SSTI). Besides, this Chinese medicinal ointment can also be studied for anti-inflammatory properties in the future. Growing evidence has shown that celastrol has an anti-inflammatory effect on skin diseases. For example, Lee et al. (2021) reported the topical application of celastrol, which alleviated atopic dermatitis symptoms by suppressing thymic stromal lymphopoietin production in keratinocyte cells through the activation of the NF-ĸB pathway. It reduced the levels of Th2 cytokines, including interleukin (IL)-4, IL-5, and IL-13, in atopic dermatitis skin lesions (Lee et al., 2021). Zeng et al. (2022) identified that celastrol may be a protective agent by inhibiting the LL37-activated Ca2+/CaMKII-mTOR-NF-κB pathway associated with skin inflammation disorders. It suppressed the expression of inflammatory cytokines and inhibited the Th17 immune response and cutaneous angiogenesis in human peptide LL37 (Zeng et al., 2022).
The developed Chinese medicinal ointment and proposed mechanisms demonstrated that celastrol has the potential to investigate anti-inflammatory and antibacterial properties on skin diseases, such as psoriasis and eczema. China has recommended Tripterygium extracts as adjuvant drugs for the treatment of eczema (Immunology Group of Chinese Society of Dermatology, 2011). If it is possible and suitable, celastrol can, with the help of nanotechnology, further improve on the stability of Chinese medicinal ointment. Therefore, the above information is just the preliminary results; it is a starting milestone for further studying the biological mechanisms or applications on human skin.
Footnotes
Abbreviations
E. coli: Escherichia coli; HPLC-DAD: High-performance liquid chromatography-diode array detector; MAPK: Mitogen-activated protein kinase; MeOH: Methanol; NF-κB: Nuclear factor kappa; SSTI: Skin and soft tissue infection; TCM: Traditional Chinese medicine; TW: Tripterygium wilfordii; UV–vis: Ultraviolet–visible.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This manuscript is part of the Ms. Ching Wong, 2024 BSc (Hons) Chinese Medicinal Pharmacy, Final Year Project (THEi).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Basic and Applied Basic Research Foundation of Guangdong Province (2022A1515111117) and Nanshan District Health System Major Science and Technology Project (NSZD2023031).
Ethical Approval and Informed Consent
Not applicable.
