Abstract
Background
Rheumatoid Arthritis (RA) is a persistent autoimmune disease. Triptolide (TPL) and celastrol (CEL) are both toxic components derived from traditional Chinese medicine Tripterygium wilfordii, which were proven to be potent candidates in the treatment of inflammatory and immunomodulatory.
Objectives
This study aims to explore the mechanism of action and efficacy of the combined application of TPL and CEL in the treatment of RA, as well as the advantages compared with their single use.
Materials and Methods
Network pharmacology predicted potential targets and pathways by analyzing interactions between RA and the 3D structures of TPL and CEL, and explored their combined effects. Cytotoxicity was assessed using Jurkat and RAW264.8 cell lines, and the dose ratio was validated. In vitro and in vivo studies further evaluated the synergistic therapeutic effects of TPL and CEL.
Results
Based on the topological importance of the “compound-target-pathway” network, inflammatory markers were identified as therapeutic targets of TPL and CEL, which play key roles in RA progression. TPL and CEL likely synergistically impact RA through these targets. Experimentally, their combination significantly inhibited inflammatory marker proliferation, mRNA/protein expression, and nuclear translocation in cells, outperforming individual treatments. The joint index confirmed their synergistic effect, aligning with network pharmacology predictions. In collagen-induced arthritis mice, TPL and CEL combined therapy markedly slowed RA progression, proving more effective together than alone.
Conclusion
The synergistic effect of TPL and CEL at a 1:160 ratio showed optimal efficacy for RA treatment with no significant toxicity. Both in vitro and in vivo studies confirmed its effectiveness over individual agents, attributed to nuclear factor kappa B and mitogen-activated protein kinase pathway inhibition.
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease causing synovial joint inflammation, cartilage damage, and bone erosion. In mainland China, RA prevalence is 0.42%, with a 61.3% disability rate over 15 years (Tian et al., 2021). Methotrexate (MTX), the primary RA treatment, has limited efficacy, with a success rate of only 15%–25% (Tang et al., 2020). RA mechanisms involve pathogenic cell migration and pro-inflammatory cytokine production (Huang et al., 2018). Current treatments like nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, and disease-modifying antirheumatic drugs (DMARDs) often cause significant side effects with long-term use (Bindu et al., 2020), highlighting the need for new therapies.
Traditional Chinese medicine (TCM), with over 5,000 years of history, offers bioactive compounds effective against inflammatory diseases (Yuan et al., 2019). Tripterygium wilfordii Hook F. (TwHF), a Celastraceae plant, has been used for centuries in RA treatment. Native to the northern hemisphere, TwHF is found in China, Korea, Japan, and the Americas (Shan et al., 2023). Its woody root is the primary medicinal part (Ru et al., 2020).
In TCM, TwHF is known for relieving stasis and internal warmth, treating joint pain and inflammation by stimulating blood flow, alleviating rheumatism, and reducing pain and swelling (Cheng et al., 2022). Modern research highlights its anti-inflammatory, cartilage-protective, and immunocyte-regulating properties in RA treatment (Spiering et al., 2019; Tang et al., 2020). However, its complex extraction and unclear mechanisms limit widespread use (Zhou et al., 2018). Key active components, triptolide (TPL) and celastrol (CEL), modulate immune and inflammatory cells, inhibit T-cell proliferation, and reduce inflammatory cytokines (Cui et al., 2022). Direct use of TPL and CEL could bypass complex extractions, and their potential synergistic effects warrant further exploration.
Network pharmacology, introduced by Hopkins, integrates biology, bioinformatics, pharmacology, and computational techniques to explore multi-component and multi-target drug mechanisms (Zhuang et al., 2020). Since 2010, it has been widely applied in TCM research (Wang et al., 2021). In this study, we used herb-compound-target (H-C-T) and protein–protein interaction (PPI) networks to identify key therapeutic targets through topological analysis. Using Cytoscape 3.7.1, we performed Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses to explore biological functions and compound–target interactions. We established a compound-target-pathway (C-T-P) network pharmacology dataset for TPL and CEL to investigate their synergistic anti-inflammatory effects, validated through in vitro, in vivo, and animal model experiments. These findings offer potential clinical therapy options for RA.
Materials and Methods
Construction of the H-C-T Database for TPL and CEL
The 3D structures of TPL and CEL were analyzed using SymMap and Swiss Target Prediction to identify target proteins (Wang et al., 2020). Cytoscape 3.7.1 was used to construct and analyze networks, focusing on topological characteristics. Node degree, indicating connection count, was calculated to assess importance, with higher-degree nodes being more significant (Wang et al., 2017).
PPI Network Construction and Module Analysis
Drug-disease crossover genes were identified to find shared targets between drugs and diseases, generating two target lists: drug-related and disease-related genes (Jing et al., 2019). Drug-correlation genes were sourced from SymMap and Swiss Target Prediction, while RA targets came from GeneCards (Zou et al., 2021). From 4,329 disease targets, the top 1,000 RA-related genes were analyzed. Bioinformatics & Evolutionary Genomics identified intersecting targets between TPL, CEL, and RA, visualized in a Venn diagram (Figure 1B.a). The STRING database was used to build a PPI network, filtering for high-confidence interactions (score > 0.4) (Yu et al., 2019). The PPI network was visualized using Cytoscape 3.7.1 (Wang et al., 2019).
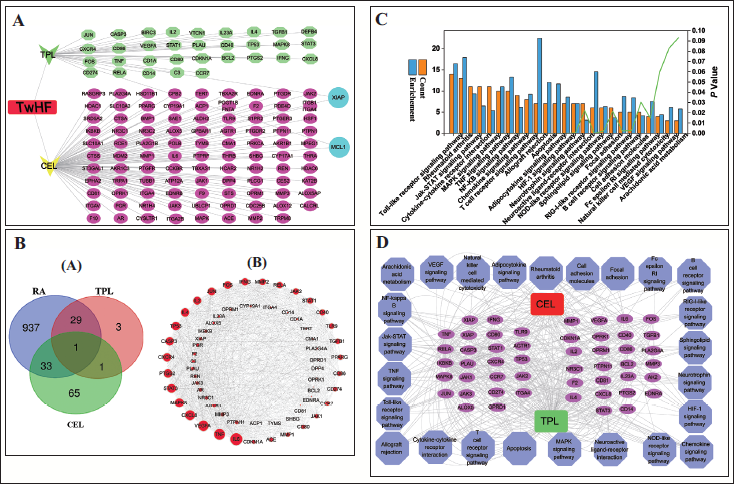
(A) Herb-Compounds-Targets (H-C-T) Network Diagram; (B.a) Venn Diagram of Rheumatoid Arthritis (RA) Correlation Targets and Drug-related Targets; (B.b) Protein–Protein Interaction (PPI) Networks of Triptolide (TPL) and Celastrol (CEL); (C) Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment Analysis; (D) Compound-Target-Pathway (C-T-P) Network Build.
KEGG Pathway Enrichment Analysis
Performed KEGG enrichment analysis on 63 target proteins to explore potential biological pathways influenced by the DAVID database. This analysis is highly credible for the localization of biological processes and molecular functions of proteins. The p value obtained from the bilateral hypergeometric test is less than .01 to determine the statistical significance of enrichment. This p value, to some extent, indicates the importance of enrichment pathways associated with TPL and CEL, which helps determine which biological and molecular functions are most relevant to these targets (Zhang et al., 2021).
Construction of the C-T-P Network for TPL and CEL
For KEGG predictions made by DAVID, a C-T-P network was constructed to link potential pathways with their associated targets (Jin et al., 2021). In the resulting diagrams, nodes show chemical compounds, targets, and pathways, while edges depict the interplay among these elements. This network helps visualize how compounds interact with targets and contribute to specific pathways.
Pharmacological Verification
Network pharmacology analysis predicted the synergistic effect and mechanism of TPL and CEL on RA. The network pharmacology revealed that toll-like receptor 4 (TLR4)-nuclear factor kappa B (NF-κΒ) signaling pathways and mitogen-activated protein kinase (MAPK) signaling pathways were the most relevant targets of TPL and CEL treating RA. Further, the synergistic effect was verified by cytological experiments, synergy analysis, reverse transcription-polymerase chain reaction (PCR), enzyme-linked immunosorbent assays (ELISAs), western blot, NF-κβ nuclear translocation, and animal experiments. The results suggested that TPL and CEL could synergistically inhibit inflammatory factors by the predicted signaling pathway (Supplementary Information).
Results
H-C-T and PPI Network Construction and Analysis
The H-C-T networks of TPL and CEL were constructed by linking compounds to their potential targets, aiding in predicting pharmacological actions (Figure 1A). Network analysis identified key nodes, with 134 targets and 6,534 edges relevant to RA (Ashburner et al., 2000). Specifically, 34 targets for TPL and 100 for CEL were linked to RA treatment, with XIAP and MCL1 common to both. This suggests that TPL and CEL may have distinct and shared targets, potentially working synergistically in RA treatment.
By constructing the H-C-T network, we identified TPL and CEL targets, with a Venn diagram revealing 63 core RA-related targets (Figure 1B.a). In biological networks, interconnected genes or proteins form functional clusters, aiding cluster analysis and module development (Shi et al., 2020). PPI network analysis (Figure 1B.b) highlighted 63 core genes and 22,802 edges, with larger nodes indicating greater significance. The top 10 genes—IL-6, TNF, VEGFA, CXCL8, MAPK8, STAT3, PTGS2, CXCR4, CASP3, and TP53—were identified as central targets, suggesting TPL and CEL may synergistically treat RA by targeting these genes.
KEGG Enrichment Analysis
Similarly, KEGG enrichment analysis has revealed that TPL and CEL influence RA treatment through various pathways (Ashton, 2015). The pathways depicted in Figure 1C consist of the TLR signaling pathway, RA pathway, Jak-STAT signaling pathway, cytokine–cytokine receptor interaction, MAPK signaling pathway, TNF signaling pathway, NF-κB signaling pathway, chemokine signaling pathway, and T cell receptor signaling pathway. The results demonstrate that these pathways play a critical role in treating RA with TPL and CEL, and serve as potential targets for their potential action.
C-T-P Network Construction
According to a literature review, construct a C-T-P network as shown in Figure 1D, TPL and CEL were linked to 25 pathways and 50 targets, with key pathways including toll-like, RA, MAPK, JAK-STAT, and NF-κB. Top targets such as TNF, RELA, and IL-6 align with RA inflammation, providing a theoretical basis for TPL and CEL in RA treatment. Their potential synergy warrants further experimental validation (Yan et al., 2020).
Synergistic Effect of TPL and CEL on RA In Vitro
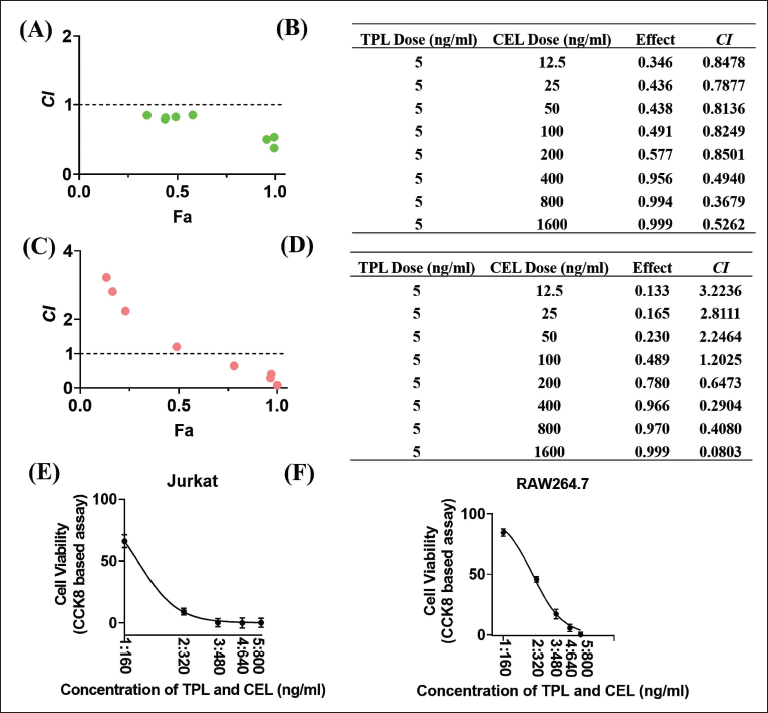
Synergistic Analysis and Verification by the CompuSyn Method and CCK8 Assay
To determine the interaction between TPL and CEL in Jurkat and RAW264.7 cells, CompuSyn software was used to calculate combination index (CI) values. A CI < 1 indicates synergy, CI = 1 additive effect, and CI > 1 antagonism (Zhang et al., 2020). In Jurkat cells (Figure 2A and 2B), the optimal synergistic ratio was 1:160 (TPL:CEL), while in RAW264.7 cells (Figure 2C and 2D), it was 1:320. The 1:160 ratio was chosen for further experiments due to its practicality and comparability. The IC50 of the 1:160 combination in RAW264.7 cells was 1.837 ng/mL (TPL) and 293.92 ng/mL (CEL), significantly lower than individual treatments (Figure 2E and 2F). These results support the synergistic effect of TPL and CEL, suggesting potential clinical benefits in efficacy, reduced side effects, and cost savings.
Determination of the Cooperation Effect of the Therapy of Triptolide (TPL) with Celastrol (CEL) on Jurkat and RAW264.7 Cell; (A) Combination Index (CI) Plot; (B) CI Table Shows CI for the Selected Combination on Jurkat Cell; (C) CI Plot; (D) CI Table Shows CI for the Selected Combination on RAW264.7 Cell; (E) Cooperative Effect of TPL and CEL on Jurkat Cells; (F) Cooperative Effect of TPL and CEL on RAW264.7 Cell.
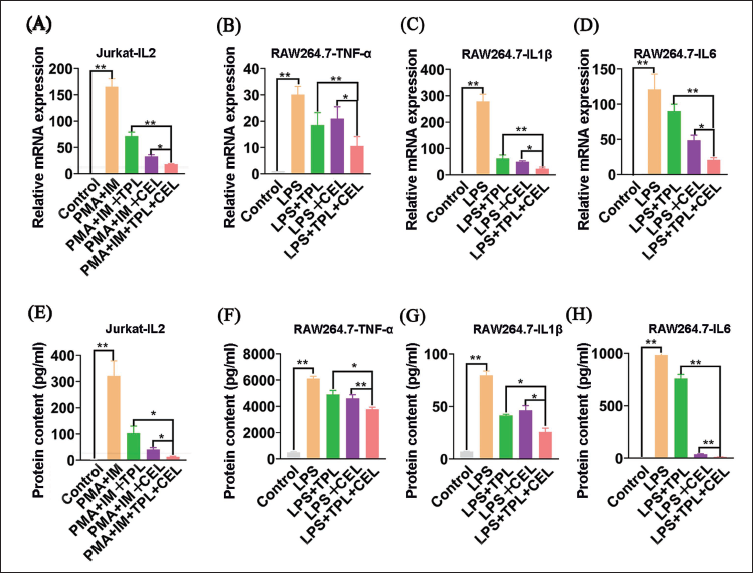
Regulation of mRNA and Pro-inflammatory Cytokines by TPL and CEL
Quantitative real-time PCR confirmed that TPL and CEL inhibit RA-related genes (TNF-α, IL-1β, IL-6). PMA and IM increased IL-2 levels in Jurkat cells, but TPL and CEL reduced IL-2 expression, with the TPL+CEL combination showing the most significant suppression (Figure 3A). Similarly, in RAW264.7 cells, the combination synergistically inhibited TNF-α, IL-1β, and IL-6 (p < .01) (Figure 3B–3D). The combined use of TPL and CEL was more effective than either compound in suppressing inflammatory mRNA expression, confirming their joint anti-inflammatory effect. ELISA detection of cytokines in Jurkat and RAW264.7 cell supernatants (Figure 3E–3H) showed that TPL+CEL most effectively reduced pro-inflammatory cytokine secretion, aligning with mRNA results (Ito et al., 2007). These findings demonstrate that TPL and CEL synergistically inhibit RA-related cell proliferation and cytokine release, consistent with network pharmacology analysis.
mRNA and Protein Demonstration of RA-linked Genes on Cells. (*p < .05. **p < .01.) (A) Demonstration of IL-2 mRNA in Jurkat Cells; (B) Demonstration of TNF-α mRNA in RAW264.7 Cells; (C) Demonstration of IL-1β mRNA in RAW264.7 Cell; (D) Demonstration of IL-6 mRNA in RAW264.7 Cell; (E) Demonstration of IL-2 Protein in Jurkat Cells; (F) Demonstration of TNF-α Protein in RAW264.7 Cells; (G) Demonstration of IL-1β Protein in RAW264.7 Cells; (H) Demonstration of IL-6 Protein in RAW264.7 Cells.
Inhibition of the NF-κΒ and MAPK Signaling Pathways by TPL and CEL
KEGG pathway analysis revealed that TPL and CEL primarily target RA’s NF-κB and MAPK signaling pathways. Western blotting confirmed elevated levels of P65, p-P65, ERK1/2, and p-ERK1/2 in PMA- and IM-activated Jurkat cells, indicating T cell activation of these pathways, which may promote pro-inflammatory cytokine release and RA progression (Figure 4A, 4B, 4E, and 4F). TPL and CEL treatment reduced these protein levels, with combined use showing enhanced inhibition, underscoring their therapeutic synergy in RA.
Western Blotting (WB) was Used to Check the Protein Expression of Rheumatoid Arthritis (RA)-linked Genes on Cells; (A) Demonstration of P65 and p-P65 Proteins in Jurkat Cells; (B) Demonstration of PERK1/2 and p-PERK1/2 Protein in Jurkat Cell; (C) Demonstration of TLR4, P65, and p-P65 Protein in RAW264.7 Cell; (D) Demonstration of PERK1/2 and p-PERK1/2 Protein in RAW264.7 Cell.
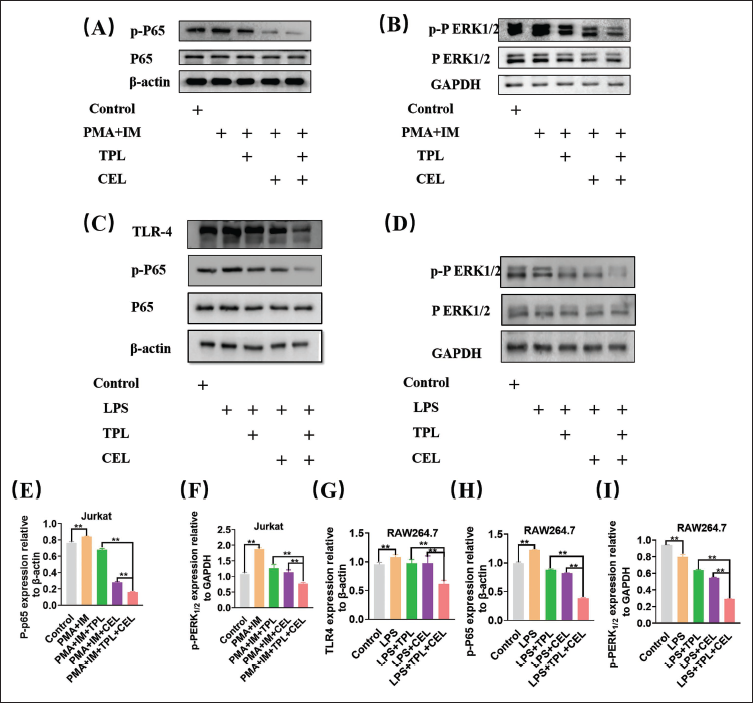
Additionally, macrophages, crucial in RA, were examined for TLR4, P65, p-P65, PERK1/2, and p-PERK1/2 expression in RAW264.7 cells. The TPL and CEL combination significantly decreased these protein levels, suggesting a synergistic suppression of NF-κB and MAPK pathways, thereby mitigating inflammatory and immune responses in RA (Figure 4C, 4D, 4G, 4H, and 4I). The results of the nuclear translocation experiment of NF-κB (P65) detected by laser confocal microscopy further confirmed that TPL and CEL synergistically inhibit the nuclear translocation of P65, thereby blocking the activation of the NF-κB signaling pathway (Supporting Information).
Synergistic Effect of TPL and CEL on RA In Vivo
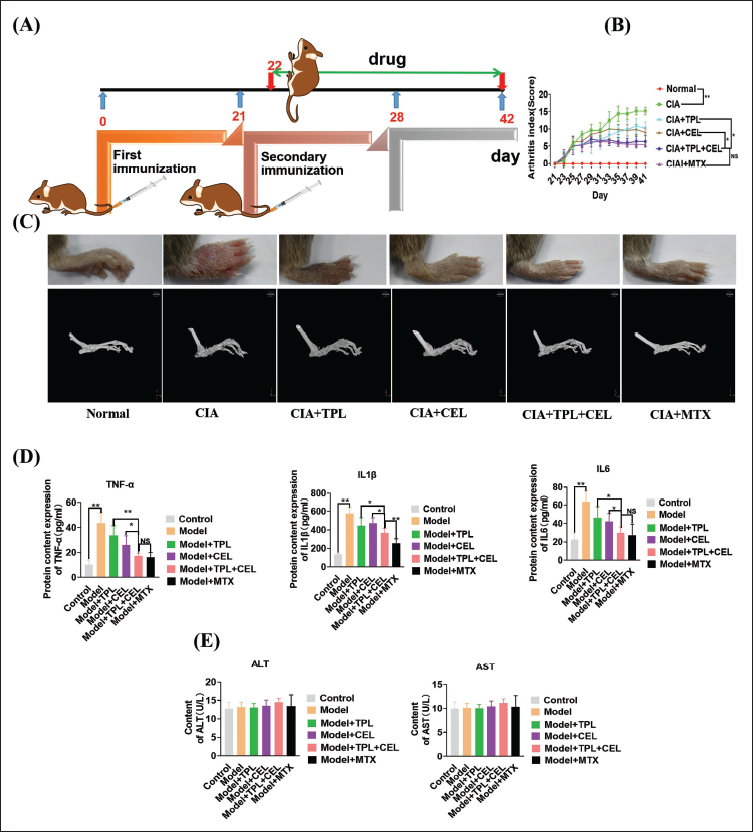
The therapeutic efficacy of TPL+CEL was evaluated in collagen-induced arthritis (CIA) mice (Figure 5A). Arthritis scores in the CIA group averaged 15.2 ± 1.10, significantly higher than those of healthy controls. Post-treatment, TPL, CEL, TPL+CEL, and MTX groups showed arthritis indices of 10.2 ± 1.91, 9.00 ± 1.00, 6.40 ± 1.67, and 5.40 ± 0.55, respectively (Figure 5B). TPL+CEL significantly suppressed RA progression, with effects comparable to MTX. Micro-CT imaging confirmed TPL+CEL’s efficacy in reducing cartilage damage and bone erosion (Figure 5C). Cytokine levels (Figure 5D) decreased post-treatment, with TPL+CEL showing stronger suppression of pro-inflammatory cytokines than single treatments, matching MTX’s effectiveness. Safety assessments (Figure 5E) indicated no significant liver function impact, as alanine aminotransferase (ALT) and aminotransferase (AST) levels remained stable. Histopathological and immunohistochemical studies also demonstrated that TPL and CEL exhibit significant synergistic effects in the treatment of RA (Supporting Information).
Medicinal Outcome of Triptolide (TPL) and Celastrol (CEL) on Collagen-induced Arthritis (CIA) Mice. (A) Experimental Arrangement; (B) The Arthritis Index of Each Group Changes Over Time; (C) At the End of Each Treatment Group, Paw Images and Micro-CT Scans were Taken; (D) Changes in Pro-inflammatory Cytokines in Mouse Serum After Every Treatment; (E) Changes in Alanine Aminotransferase (ALT) and Aminotransferase (AST) Levels in Mouse Serum After Every treatment. Data were Presented as Mean ± SD, n = 5, *p < .05. **p < .01.
Discussion
A multidisciplinary approach is commonly used in RA treatment, with MTX and dexamethasone (Dex) combination therapy being widely adopted for their anti-inflammatory and immunomodulatory effects (Sales et al., 2020). TwHF has also shown promise in RA treatment due to its immune-suppressing and anti-inflammatory properties, though its side effects and quality control remain challenges. Analyzing its active components, such as TPL and CEL, can improve quality control and reduce toxicity. Network pharmacology, integrating systems biology and bioinformatics, offers a systematic approach to explore the synergistic effects and mechanisms of TPL and CEL. This study employs network pharmacology methods, including target identification and pathway analysis, to predict the molecular mechanisms of TPL and CEL, providing insights for their safe and effective use in RA treatment.
In cytotoxicity assays, a TPL:CEL ratio of 1:160 showed the best synergistic effect, with CIs of 0.3679 and 0.408 in Jurkat and RAW264.7 cells, indicating synergistic growth inhibition. This combination also effectively suppressed IL-2 expression in T cells and pro-inflammatory cytokines (IL-1β, IL-6, TNF-α) in macrophages, highlighting its potential in RA treatment. Mechanistically, TPL and CEL inhibited inflammatory factor release by targeting the NF-κB and MAPK signaling pathways. In Jurkat cells, IL-2 expression was suppressed via these pathways, while in RAW264.7 cells, TLR4-NF-κB and MAPK pathways were primarily involved. These pathways play key roles in inflammation and cell regulation, suggesting TPL+CEL could modulate RA progression.
In vitro experiments demonstrated that TPL+CEL had a strong synergistic effect, reducing foot swelling, cartilage damage, and bone erosion in RA models, comparable to MTX. Safety evaluations of AST and ALT levels further supported the combination’s security. Overall, TPL+CEL shows promising therapeutic potential for RA, offering a novel approach to modulate key inflammatory pathways.
Conclusion
This study predicted the joint effect of TPL and CEL through network pharmacology methods. The prediction was further verified in several experiments, indicating that the combination of TPL with CEL showed a synergistic effect on RA-related cells and in CIA mice. Although the precise mechanisms underlying these effects were not completely determined in this research, we found that the combined anti-inflammatory action of TPL and CEL was mediated by intercepting the activation of the NF-κΒ and MAPK signal path. These findings offer valuable insights into the pharmacological role of TPL and CEL, and they suggest that TPL and CEL could be promising future treatment options for patients with RA.
Footnotes
Abbreviations
ALT: Alanine aminotransferase; AST: Aminotransferase; C-T-P: Compound-target-pathway; CEL: Celastrol; CI: Combination index; CIA: Collagen-induced arthritis; GO: Gene Ontology; H-C-T: Herb-compound-target; KEGG: Kyoto Encyclopedia of Genes and Genomes; MTX: Methotrexate; PPI: Protein–protein interaction; RA: Rheumatoid arthritis; TCM: Traditional Chinese medicine; TPL: Triptolide.
Acknowledgments
The authors thank the Shanghai University of Traditional Chinese Medicine for providing a good research platform.
Authors’ Contributions
Conception and design: JGZ and RXL; Collection and assembly of data: QHZ and ZFW; Data analysis and interpretation: GZC and RXL; Manuscript writing: All authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of the Shanghai University of Traditional Chinese Medicine (No. PZSHUTCM200807020).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the National Natural Science Foundation of China (81903809), the Shanghai Municipal Education Commission (SMEC 2019-01-07-00-10-E00072), the Science and Technology Commission of Shanghai Municipality (STCSM 18401933500), the State Administration of Traditional Chinese Medicine of People’s Republic of China (GZYYGJ2019059), the Expenditure Budget Program of Shanghai University of Traditional Chinese Medicine (Grant No. 23KFL001), the Shanghai Pujiang Programme (23PJD085), the Shanghai Frontiers Science Center for Traditional Chinese Medicine Chemical Biology, Shanghai Science and Technology Development Fund from Central Leading Local Government (YDZX20223100001004), the Traditional Chinese and Western Medicine Collaborative Guidance Project of Integrated Hospital from Shanghai Municipal Health Commission/Shanghai Municipal Administration of Traditional Chinese Medicine (ZXXT-202303).
Informed Consent
Not applicable.
Supplemental material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
