Abstract
Background
Acute lung injury (ALI) is an acute inflammatory condition defined by the excessive generation of inflammatory chemicals in lung tissue. Solasodine, a glycoalkaloid isolated from Solanum species, has been proven to exhibit anti-inflammatory and anti-spermatogenic activities.
Objectives
The present study intended to explore the protective potential of Solasodine on lipopolysaccharide (LPS)-stimulated ALI in rat models.
Materials and Methods
The animals were assigned to four groups: Normal control, LPS-induced at 4 mg/kg, Solasodine alone 50 mg/kg, LPS + Solasodine 50 mg/kg. The lung tissue was examined to assess the activities of the lung wet/dry weight ratios, lung myeloperoxidase (MPO), nitric oxide (NO) activity, oxidative stress parameters malondialdehyde (MDA), 8-hydroxy-2′-deoxyguanosine (8-OHdG), and superoxide dismutase (SOD) along with the histopathological analysis were evaluated. In addition, lactate dehydrogenase (LDH) activity in the bronchoalveolar lavage fluid (BALF), levels of inflammatory cytokines interleukin (IL)-1β, tumor necrosis factor-alpha (TNF-α), IL-6, and neutrophil gelatinase-associated lipocalin (LCN-2) in BALF were determined.
Results
The results revealed that Solasodine treatment substantially lowered the wet/dry weight ratio, improved the histopathologic modifications, increased the SOD, and downregulated the levels of LDH, MPO, nitric oxide (NO), MDA, and 8-OHdG. Moreover, Solasodine treatment decreased the neutrophil infiltration and cytokines and protein levels in the BALF.
Conclusion
The current investigation suggested that Solasodine improved LPS-stimulated ALI owing to its anti-inflammatory and antioxidative activity in rats. This study’s nursing interventions involve vigilant monitoring, ethical treatment, supportive care, and accurate data collection. Vigilant monitoring detects distress, and ethical treatment minimizes suffering, supportive care includes oxygen therapy, and accurate data collection evaluates Solasodine efficacy.
Introduction
Sepsis, or an uncontrollable host reaction to infection, causes life-threatening organ failure and is among the top causes of mortality in critical care units. The lung is among the most susceptible targeted tissue in sepsis, with acute lung injury (ALI) occurring in the initial stages and exhibiting a particularly high prevalence (Mowery et al., 2020). ALI is a life-threatening condition with a high death and morbidity rate resulting from sepsis, chest trauma, viral pneumonia, ischemia-reperfusion, as well as burns. Histopathological modifications include diffuse alveolar hemorrhage, edema of the lungs, apoptosis, along hyaline membrane development (Gao et al., 2018; Meng et al., 2018). Lipopolysaccharide (LPS), located in the gram-negative bacterial cell wall framework, is produced extracellularly and is regarded as the primary microbial stimulant of inflammation, triggering the immune system’s innate response at the outset of ALI. As a result, in experimental research, LPS is extensively employed as a promising approach for inducing the clinically relevant type of ALI in rodent models. Furthermore, earlier research has shown that anti-inflammatory and antioxidant substances have considerable protective benefits against LPS-stimulated ALI (Khan et al., 2019).
Inflammatory cell infiltration and inflammation, which is an indispensable part of the pathophysiology of ALI and is mainly accountable for tissue damage, are also linked to pro-inflammatory cytokine overproduction. LPS promotes the production of pro-inflammatory cytokines, including tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1β, and IL-6. These cytokines are primarily produced by inflammatory cells and are present in substantial concentrations in the lung. The cytokine cascade is then activated, accelerating tissue destruction (Liu et al., 2019). Furthermore, the inflammatory response increases the formation of reactive oxygen species (ROS), which serves a crucial role in LPS-induced ALI. Regardless of tremendous advancements concerning the pathogenesis of ALI, current therapeutic options have not dramatically reduced ALI-related mortality. Therefore, innovative solutions are still required to treat ALI successfully.
Solasodine is a steroidal alkaloid derived from Solanum plants, including Solanum nigrum L. and Solanum lyratum Thunb. It is an aglycone of alkaloids, including solamargine and solasonine. It has been documented that Solasodine exerts diverse properties, which include antipyretic, antioxidant, anti-inflammatory, anti-spermatogenic, antifungal, antiepileptic, cardiotonic, and memory-enhancing effects in Alzheimer’s disease (Navabharath et al., 2022; Sharma et al., 2014; Xu et al., 2017). Also, Solasodine targets NF-κB signaling to overcome P-glycoprotein-mediated multidrug resistance in cancer (Bharathiraja et al., 2024). Owing to the antioxidant and anti-inflammatory properties of the compound, the study intends to assess if Solasodine could reduce pro-inflammatory cytokine expression and minimize lung edema and injury in rats with LPS-stimulated ALI.
The main aim of the current investigation is to elucidate the protective role of Solasodine against LPS-stimulated lung injury in rat models by examining the biochemical as well as histopathological parameters. The impact of Solasodine on the levels of myeloperoxidase (MPO), nitric oxide (NO), and lactate dehydrogenase (LDH) activities along with protein and cytokine levels in bronchoalveolar lavage fluid (BALF) in rat models has been examined.
The study investigates the protective role of Solasodine against LPS-induced ALI in rat models. Key nursing interventions include vigilant monitoring, ethical treatment, supportive care, and accurate data collection. Vigilant monitoring involves recording respiratory rate, heart rate, and body temperature to detect distress. Ethical treatment follows guidelines, minimizing suffering and providing a comfortable living environment. Supportive care includes oxygen therapy, fluid therapy, and nutritional support. Accurate data collection involves maintaining detailed records and conducting statistical analysis to evaluate the efficacy of Solasodine in mitigating ALI.
Materials and Methods
Chemicals and Reagents
Solasodine, LDH measurement kit, and LPSs were procured from Sigma–Aldrich (USA). Inflammatory cytokines enzyme-linked immunosorbent assay (ELISA) kits were procured from Thermo Fisher Scientific, USA. Before the start of the trial, all chemicals, reagents, and kits were purchased commercially and made available.
Animal
Albino male rats (220–250 g) were utilized for the experimental studies. The rats were housed in an animal enclosure with a regular temperature of 22±2°C, a photoperiod of 12:12 h of light and dark cycle, and relative humidity of 40–60%. The animals were fed a pellet-based diet and were provided access to unrestricted water. The Institutional Animal Ethical Committee (IAEC) approved the studies (YXLL-2023-306).
Experimental Protocol
Rats were divided into four groups, each with six animals. Group I received standard saline treatment and was considered the control, group II was induced with LPS (4 mg/kg), group III was subjected to Solasodine treatment alone (50 mg/kg), and group IV was simultaneously induced with LPS, and treated with Solasodine (50 mg/kg). Rats were sacrificed with a dose of pentobarbital sodium. Six animals from each group had their left lungs histopathologically examined, while the right lungs were tested for the wet/dry weight ratio. The remaining three animals in each group had their left lungs bronchoalveolar lavaged, while the right lungs were removed for biochemical measurements.
Estimation of Lung Wet/Dry Weight Ratio
For the assessment of lung edema, the wet/dry weight ratio of lung tissue was measured. The right lung was excised and its wet weight was reported. The lung’s dry weight was assessed after 24 h of incubation at 80°C, and wet/dry weight ratios were computed (Kao et al., 2015).
Estimation of Lactate Dehydrogenase (LDH) Activity in Bronchoalveolar Lavage Fluid (BALF)
The LDH activity in the BAL fluid was measured by employing a commercial LDH measurement kit. In summary, BALF supernatants were combined with Reagent A for 1 min before being added to Reagent B. Absorbance at 340 nm was measured every minute for 3 min, and LDH activity was assessed with the help of LDH standards (Chu et al., 2005).
Assessment of Lung Myeloperoxidase (MPO) and Nitric Oxide (NO) Activity
Frozen tissue specimens from the right lower lung were homogenized using radioimmunoprecipitation assay (RIPA) buffer and sonicated (Li et al., 2007). The suspensions underwent centrifugation at 2,430 g at 4°C for 10 min. Colorimetric assay kits were employed according to the manufacturer’s directions to measure MPO activity and NO levels. Briefly, the MPO activity was measured by constantly recording the hydrogen peroxide (H2O2)-dependent Fast Blue B oxidation. In summary, the supernatant was combined with PBS comprising sodium dihydrogen phosphate and disodium monohydrogen phosphate, as well as sodium hydroxide, added with H2O2 and Fast Blue B. The absorbance at 460 nm was determined with a spectrometer.
Estimation of Oxidative Stress Parameters in Lung Tissue
The oxidative stress indicators were assessed to understand the antioxidant status in the tissues. The left lung tissue was obtained, homogenized, and subjected to centrifugation for 15 min at 4°C. The resulting supernatant was utilized for tests with commercially accessible kits. Malondialdehyde (MDA) levels were determined using the thiobarbituric acid reaction method. The activity of superoxide dismutase (SOD) was determined at 550 nm using the hydroxylamine method. The levels of 8-hydroxy-2′-deoxyguanosine (8-OHdG) were assessed with an ELISA kit as per the directions given by the manufacturer (Trocha et al., 2014).
Assessment of Differential Cell Counts and Neutrophil Count
Cell pellets from BAL fluid were reconstituted in PBS, and the total cell count was measured with a hemocytometer. BAL cytospin was prepared and stained using the Wright–Giemsa procedure. To examine differential cell counts, at least 500 cells were totaled to estimate the proportion of each cell type. The neutrophil count was estimated by multiplying the proportion of neutrophils by the total cell count in the BAL fluid (Ci et al., 2012).
Determination of Protein and Cytokines in BALF
The Bradford technique was employed to assess total protein concentration in BALF, with bovine serum albumin as a standard, as per the manufacturer’s guidelines. Cytokines such as TNF-α, IL-1β, neutrophil gelatinase-associated lipoprotein (LCN2), and IL-6 were quantified in the BALF employing ELISA kits as indicated by the manufacturer (R&D Systems, Minneapolis, MN, USA).
Histopathological Analysis of Rat Lung Tissues
Tissue samples collected from the middle lobe of the right lung were fixed with 10% formalin and later implanted in paraffin. Blocks of tissues were sliced at 5 mm thickness, immersed in lukewarm water, and positioned on glass slides. They were then stained with hematoxylin and eosin stain, dried, and the cover slipped. Light microscopy was employed for morphologic studies, which were then recorded using pictures (Wu et al., 2007).
Statistical Analysis
The experimental study’s values were presented as mean ± standard error of the mean (SEM). The statistical study was executed using GraphPad Prism, one-way analysis of variance (ANOVA), as well as Dunnett’s test. p values < 0.05 were deemed to be statistically significant.
Results
Impact of Solasodine on the Wet/Dry (W/D) Weight Ratio of Rat Lung Subjected to LPS Treatment
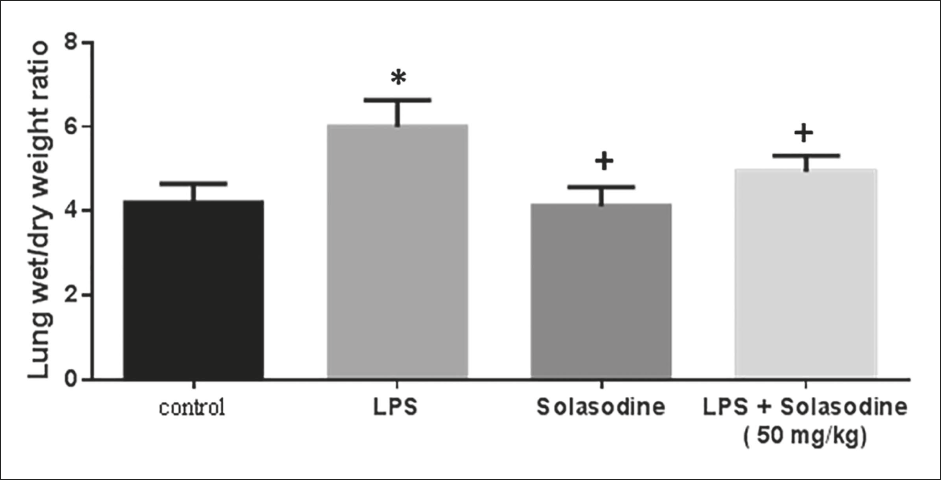
Figure 1 displays that the LPS group exhibited a considerably higher W/D ratio than the control group, indicating edema as well as inflammatory cell infiltration in the pulmonary region (p < 0.05). The LPS + Solasodine (50 mg/kg) group exhibited a significantly lower W/D ratio than the LPS group.
Impact of Solasodine on the LDH Activity in Bronchoalveolar Lavage Fluid (BALF)
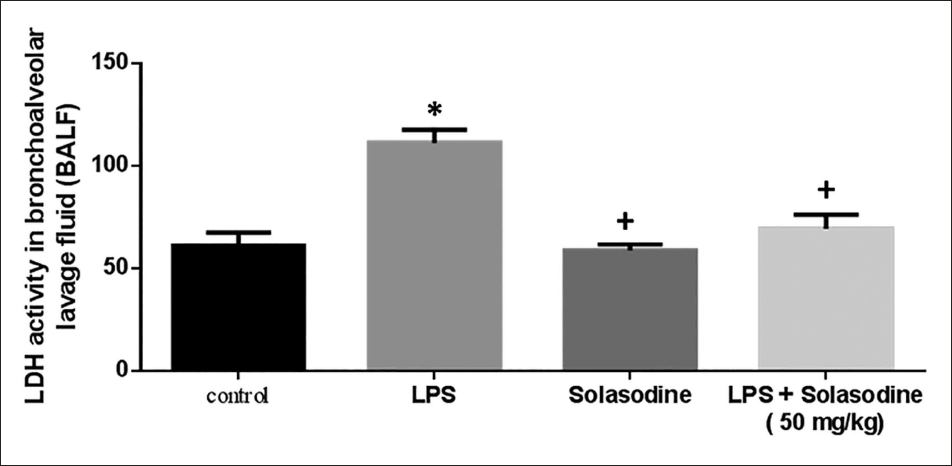
The effect of Solasodine on the LDH activity in BALF of rats is depicted in Figure 2. The LPS group displayed a substantially higher LDH level than the control group. The LPS + Solasodine (50 mg/kg) group exhibited a considerably reduced LDH level than the LPS group.
Impact of Solasodine on Lung Tissue Levels of MPO and NO
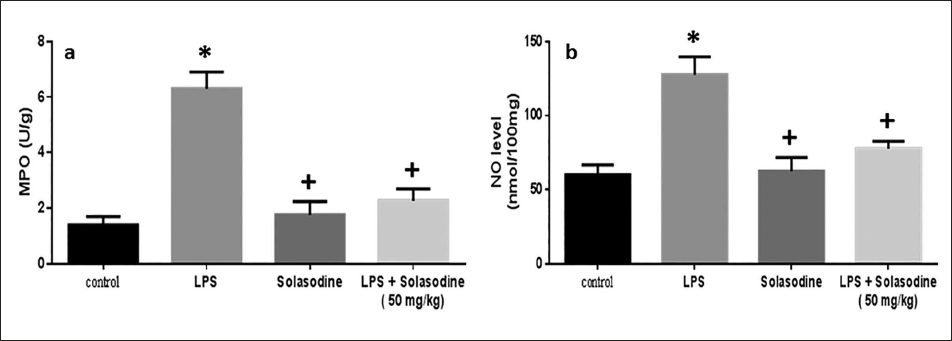
Lung MPO activity and NO levels were found to be substantially elevated in animals post-LPS exposure than in control models (Figure 3). Solasodine exposure considerably lowered the MPO and NO levels. Similarly, LPS + Solasodine treatment also reduced these levels more than that of the LPS group.
Effect of Solasodine on Lung Tissue Oxidative Stress Markers
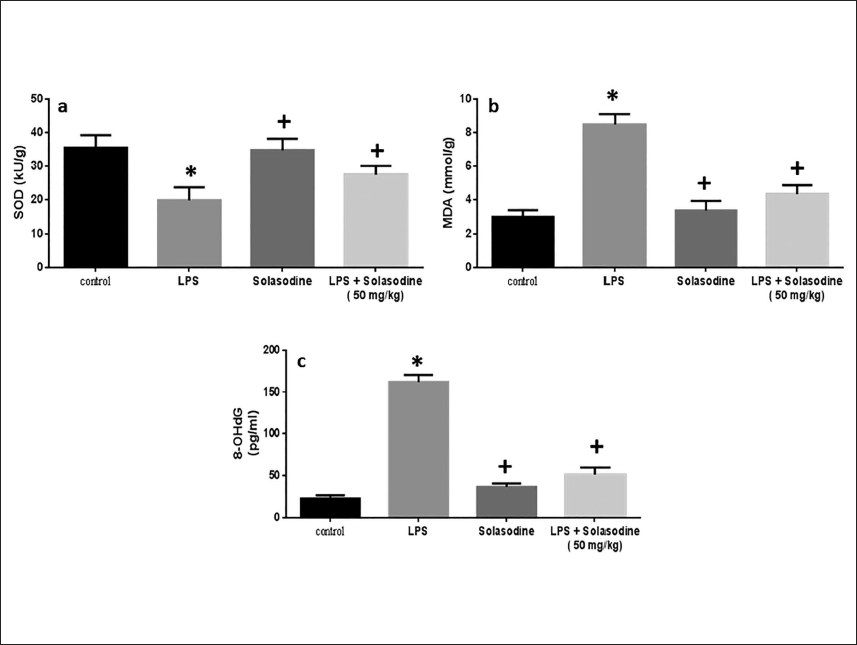
As depicted in Figure 4, the level of MDA was extensively elevated in the LPS-treated group relative to control animals. Solasodine diminished the MDA levels in comparison with the LPS group. The level of 8-OHdG also displayed a similar outcome. However, the SOD enzyme activity was significantly lowered in LPS-exposed animals than in control animals, which was again elevated upon treatment with Solasodine.
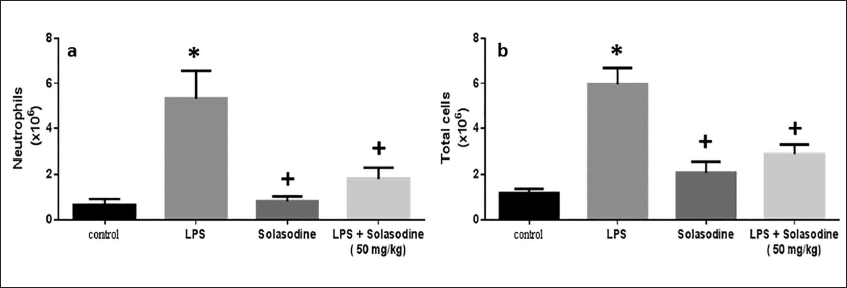
Effects of Solasodine on BAL and Differential Cell Counts
The neutrophil count (×106) and total cell count (×106) were estimated in the control as well as treated rat groups. Both the neutrophil and total cell count were increased in the LPS-induced group, whereas it decreased significantly with Solasodine exposure. Similarly, LPS + Solasodine treatment lowered the neutrophil and total cell count relative to the LPS group (Figure 5).
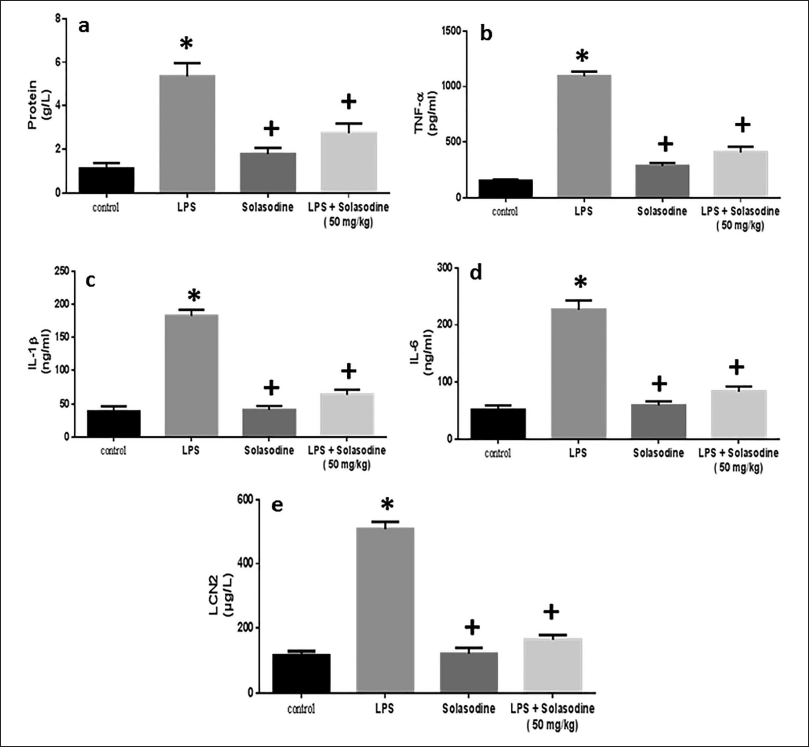
Effects of Solasodine on Analysis of BALF on Protein and Cytokine Expression
Figure 6 depicts inflammatory cytokine expression data. LPS significantly elevated pulmonary TNF-a and IL-1β levels as compared to the control group. Furthermore, LPS stimulation dramatically elevated protein content, pro-inflammatory IL-6, and LCN2 expression. TNF-a, IL-1β, LCN2, IL-6, and protein levels were all considerably lower in the LPS + Solasodine group than in the LPS group after treatment.
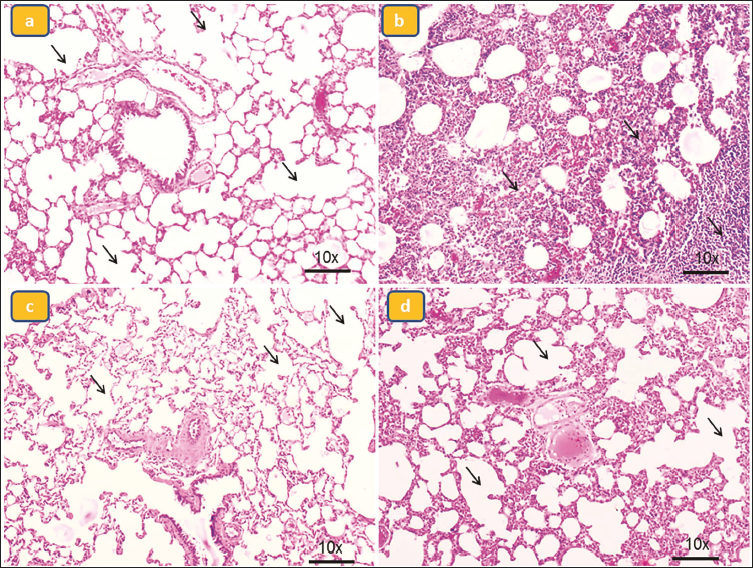
Histopathological Examination of Lung Tissue in Rats
As demonstrated in Figure 7a, the control group rats demonstrated normal lung architecture, with a thinner alveolar wall and zero visible infiltration. In contrast, LPS-treated mice that did not receive Solasodine showed severe lung injury, as seen by substantial infiltration of inflammatory cells into the interstitium and alveolar gaps of the lungs, as well as thickening of the alveolar wall (Figure 7b). Notably, Solasodine treatment drastically decreased these lung histological alterations (Figures 7c and 7d).
Histopathological Changes in Lung Tissue. Group I is Normal Control (a), Group II is Lipopolysaccharide (LPS)-induced (b), Group III is Solasodine Alone, and Group IV is Treatment LPS + Solasodine 50 mg/kg (c, d).
Discussion
Diffuse alveolar destruction, edema of the lungs, and widespread infiltration of inflammatory cells are histological features of ALI (Meyer et al., 2021). The etiology of ALI is multifactorial and poorly understood. In this investigation, an LPS-stimulated ALI mouse model was carefully established, and the protective effect of Solasodine against lung injury was evaluated through the analysis of both biochemical and histological markers. The effects of Solasodine on protein and cytokine levels, MPO, NO, and LDH activities in BALF in rat models have been examined. Over the past decade, an increasing array of study findings have demonstrated that phytochemicals with minimal toxicity and fewer adverse effects can be employed in diverse disease models for medicinal benefits (Polat et al., 2018).
LPS, a constituent of the bacterial outermost membrane, is a potent trigger of acute sepsis as well as chronic inflammation and is often referred to as an endotoxin. LPS-stimulated ALI is distinguished by major edema of the lungs and inflammatory lung cell infiltration (Kaya et al., 2020). In this investigation, we discovered that LPS treatment induced a substantial increase in the W/D ratio, suggesting the presence of pulmonary edema and inflammatory cell infiltration, which is under prior findings (Li et al., 2016). Solasodine treatment significantly lowered the W/D ratio in the LPS + Solasodine group as compared to the LPS group, demonstrating that Solasodine alleviated pulmonary edema.
The influx of neutrophils into the lung is a characteristic of ALI. It is commonly acknowledged that neutrophils are a major reservoir of pro-inflammatory cytokines that lead to inflammation-triggered lung injury. The MPO enzyme, which detects neutrophil buildup in tissues, is mostly found in neutrophil granules. MPO activity implies neutrophil adherence and margination in lung tissues; therefore, MPO stimulation is regarded to be crucial in the progression of ALI (Huang et al., 2015). LDH activity in the BAL fluid, an indication of cell damage along with death, increases and persists after a respiratory infection (Zhao et al., 2017). Our results demonstrated that Solasodine administration drastically lowered the total count of neutrophils and LDH activity in BALF and MPO activity in the lung. Altogether, our findings suggest that Solasodine exhibits anti-inflammatory properties against LPS-stimulated ALI.
A vast amount of data indicates the involvement of oxidants as well as oxidative damage in the pathophysiology of ALI (Ge et al., 2017). Sepsis generates large amounts of reactive oxygen and nitrogen species, which induce cell damage by attacking DNA, peroxiding lipids, forming vasoactive molecules, and oxidizing proteins (Huang et al., 2013). Lipid peroxidation disrupts the membranes of cells as well as mitochondria and leads, ultimately, to necrosis and apoptosis. Modifications in the SOD levels, which is necessary for eliminating ROS, and in MDA levels, which is a result of lipid peroxidation, generally represent the body’s capacity to neutralize ROS and avoid oxidative damage to lipids, whereas 8-OHdG levels are an indication of DNA peroxidation (Prauchner, 2017). Our findings further revealed that LPS-stimulated neutrophil recruitment and stimulation in the lung was associated with the emergence of pulmonary oxidative stress, as proven by higher MDA levels and reduced antioxidant enzymes, including SOD, MDA, and 8-OHdG.
It is now commonly acknowledged that lung tissue is directly damaged by sepsis as it progressively intensifies a series of proinflammatory cytokines, including TNF-α, IL-1β, and IL-6 (Gil et al., 2016). In granulocytes, LCN2/neutrophil gelatinase-associated lipoprotein (NGAL) is constantly expressed. In inflammatory conditions, LCN2 expression is increased, driven by epithelial cells. There is a substantial correlation between neutrophil exudation and LCN2 expression levels. LCN2 protein levels serve as another indicator of oxidative stress. Significantly elevated LCN2 expression was found in damaged lung tissue in a rat model of inflammation-related lung injury. In addition, clinical research has indicated that NGAL levels in sepsis patients predict a variety of organ problems. These experimental findings demonstrate that LCN2 might have a significant role in the progression of sepsis-induced lung damage (Liu et al., 2022).
Solasodine’s ability to increase SOD (antioxidant defense) and reduce MDA (lipid peroxidation) and 8-OHdG (DNA oxidative damage) correlates strongly with its reduction of inflammatory cytokines like TNF-α, IL-1β, and IL-6. This suggests that its protective effects against inflammation are at least partly due to its role in reducing oxidative stress, thereby dampening oxidative damage-driven inflammatory responses. By lowering oxidative stress markers (SOD, MDA, and 8-OHdG), Solasodine breaks the harmful cycle between ROS and inflammation. This leads to reduced activation of inflammatory cytokines like TNF-α, IL-1β, and IL-6, suggesting that Solasodine’s anti-inflammatory effects are closely linked to its antioxidative properties. A previous study on the effects of curcumin on asthma in mice found that its aerosol form showed better absorption at 1 h (Singh et al., 2023). Curcumin, an anti-inflammatory and antifibrotic molecule, can effectively treat silica dust-induced occupational lung disease, reducing oxidative stress, inflammation, and granuloma nodules (Kumari, Singh, Dash, et al., 2024). The previous study also found increased extracellular matrix deposition, ROS, inflammatory cell recruitment, and aberrant mitochondria, suggesting fibrosis severity is linked to repeated silica exposures (Kumari, Singh, & Singh, 2024).
Administration with Solasodine reduced the level of BALF protein and the lung’s wet/dry weight ratio, suggesting a decrease in lung hyperpermeability and edema. Solasodine suppressed LPS-induced TNF-α and IL-6 release as well as neutrophil cell influx in BALF, together with decreased lung MPO activity. Additionally, the MDA level decreased and the activities of CAT, SOD, and GPx increased as a result of the Solasodine treatment. Collectively, our findings demonstrate that Solasodine at least partly protects ALI caused by LPS by lowering oxidative stress along with inflammation. The pathophysiology of ALI is significantly influenced by the secretion of several cytokines and chemokines, which increases the pulmonary penetration of the alveolar-capillary membrane and impairs arterial oxygenation as a result. Therefore, one of the primary objectives of the pharmacologic therapy of ALI patients has been the suppression of lung inflammation.
Conclusion
In conclusion, Solasodine prevents LPS-stimulated ALI by substantially lowering inflammatory cell infiltration, and lung edema, as well as TNF-alpha, IL-1 beta, and IL-6 levels. Our findings also indicate that Solasodine treatment exerts a preventative impact on LPS-induced ALI, which is primarily mediated by increased antioxidant enzymes and decreased oxidative stress, LDH, MPO, and NO levels. The results obtained suggest that Solasodine could be a valuable medication for treating or improving the prognosis of LPS-stimulated ALI. More pharmacological studies should be done to warrant its use as a Solasodine LPS-stimulated ALI agent in the future. Further, these outcomes necessitate additional investigation into the clinical usefulness of Solasodine in both preventing and treating ALI.
Footnotes
Abbreviations
8-OHdG: 8-hydroxy-2′-deoxyguanosine; ALI: Acute lung injury; BALF: Bronchoalveolar lavage fluid; ELISA: Enzyme-linked immunosorbent assay; H2O2: Hydrogen peroxide; IL-1: Interleukin-1; IL-6: Interleukin-6; LCN2: Neutrophil gelatinase-associated lipoprotein; LDH: Lactate dehydrogenase; LPS: Lipopolysaccharide; MDA: Malondialdehyde; MPO: Myeloperoxidase; NO: Nitric oxide; ROS: Reactive oxygen species; SOD: Superoxide dismutase; TNF-α: Tumor necrosis factor-alpha.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The study was approved by the Ethics Committee of Shanxi Bethune Hospital, Taiyuan, China (YXLL-2023-306).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
