Abstract
Lipopolysaccharide (LPS) is widely used to establish various animal models, including models of acute lung injury, cardiomyocyte damage, and acute kidney injury. Currently, there is no consensus on the diagnosis and treatment of LPS-induced disease. We herein present a case series of four patients who developed dose-dependent multi-organ injury, including acute lung injury and acute kidney injury, after inhaling LPS gas in a sealed room. These patients exhibited varying degrees of multi-organ injury characterized by inflammatory cell infiltration and secretion of proinflammatory cytokines. One patient showed progressive symptoms even with active treatment, leading to mild pulmonary fibrosis. This study emphasizes the importance of early diagnosis and treatment of significant LPS exposure and suggests personalized treatment approaches for managing LPS poisoning.
Keywords
Introduction
Lipopolysaccharide (LPS), a component of gram-negative bacteria, is a potent inflammatory stimulant that primarily activates macrophages through the intracellular mitogen-activated protein kinase and nuclear factor-kappa B (NF-κB) pathways. The activated macrophages produce various chemokines and chemoattractant factors, which recruit new polymorphonuclear leukocytes and macrophages, resulting in a considerable increase in the production of proinflammatory factors. This process can cause tissue damage and lead to serious conditions such as endotoxic shock, sepsis, systemic inflammatory response syndrome, and even death.1–3 To date, research on LPS has been limited to animal tests and human volunteers who received LPS administration either parenterally or through inhalation.4–6 No serious adverse effects have been reported in healthy subjects. Typically, only mild flu-like symptoms or no symptoms at all were recorded, and all symptoms resolved within 24 hours. To the best of our knowledge, this is the first report on four patients who inadvertently inhaled LPS gas. Among these patients, three developed myocardial enzyme abnormalities, one developed abnormalities on routine urinalysis, and one developed mild pulmonary fibrosis despite receiving active treatment. This cases are being shared to remind clinicians of the importance of early diagnosis and treatment of LPS poisoning along with individualized treatment.
Case reports
Ethics and consent
This study was approved by the Ethical Committee of the Seventh People’s Hospital of Chongqing in China (Approval No. 2022120). All patients provided written informed consent for both treatment and publication. The reporting of this study conforms to the CARE guidelines. 7
Clinical course and management
Four previously healthy individuals, all of whom were nonsmokers and worked in the same laboratory, were simultaneously involved in LPS research within a closed room. Owing to negligence, all of them were exposed to LPS gas for approximately 1 hour without wearing protective masks. Within the next 24 hours, all four individuals presented to the emergency room (ER) with similar prodromal symptoms ranging from fever and chills to cough. Upon admission, all patients received oxygen therapy (2 L/minute nasal tube oxygen inhalation for at least 8 hours daily), intravenous corticosteroids (40 mg of methylprednisolone twice daily), oral N-acetyl cysteine (NAC) (0.6 g twice daily), and oxygen atomization inhalation of NAC (0.3 g twice daily). Remarkably, all patients showed symptom resolution within 1 to 4 days. Chest imaging results were normal for three of the patients, but one patient showed progression to mild pulmonary fibrosis despite receiving treatment (Figure 1). The clinical characteristics of these four patients are summarized in Table 1.
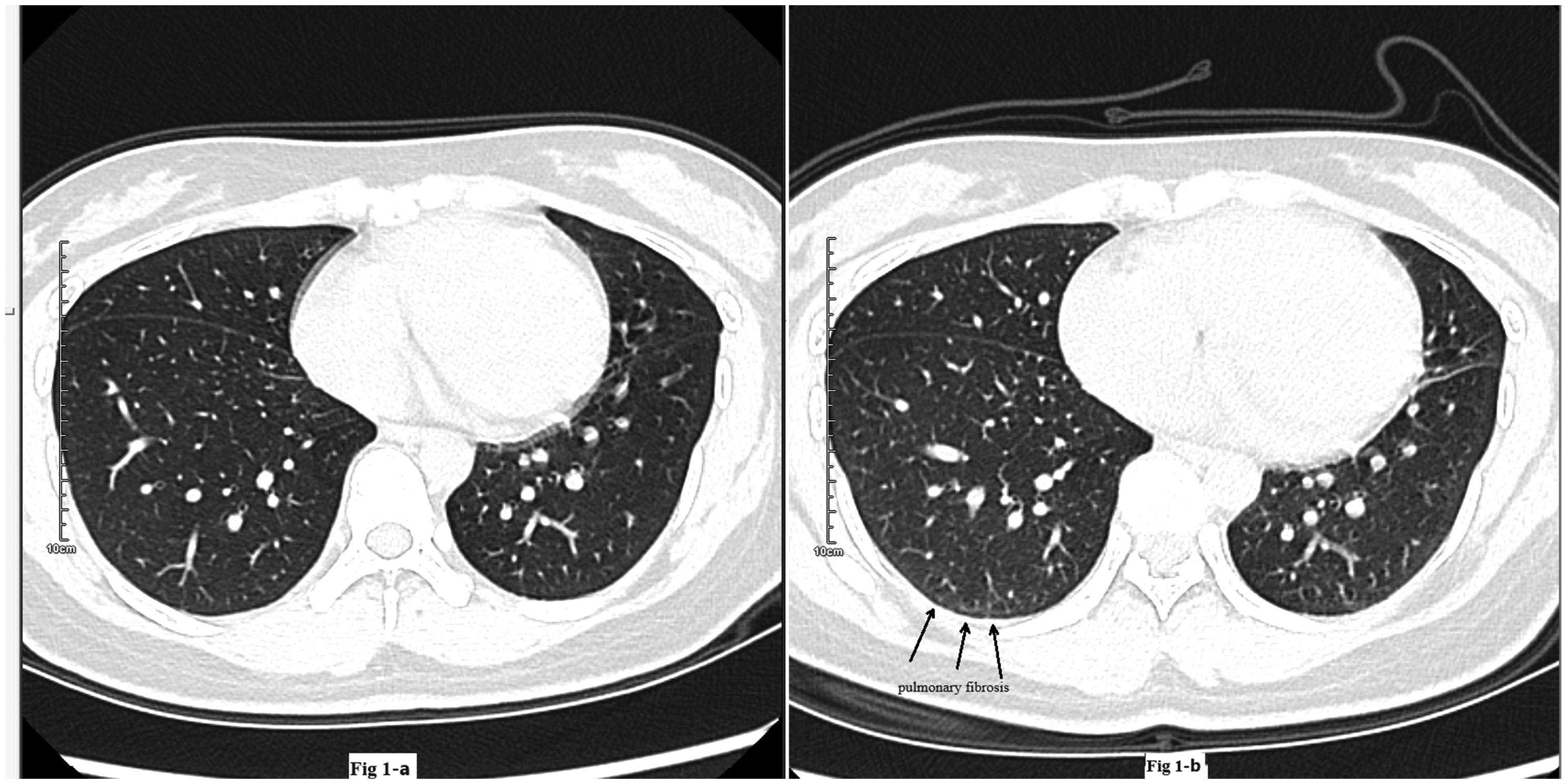
Axial chest computed tomography image of Patient 4 (a) before and (b) after therapy.
Clinical characteristics of the four study patients.
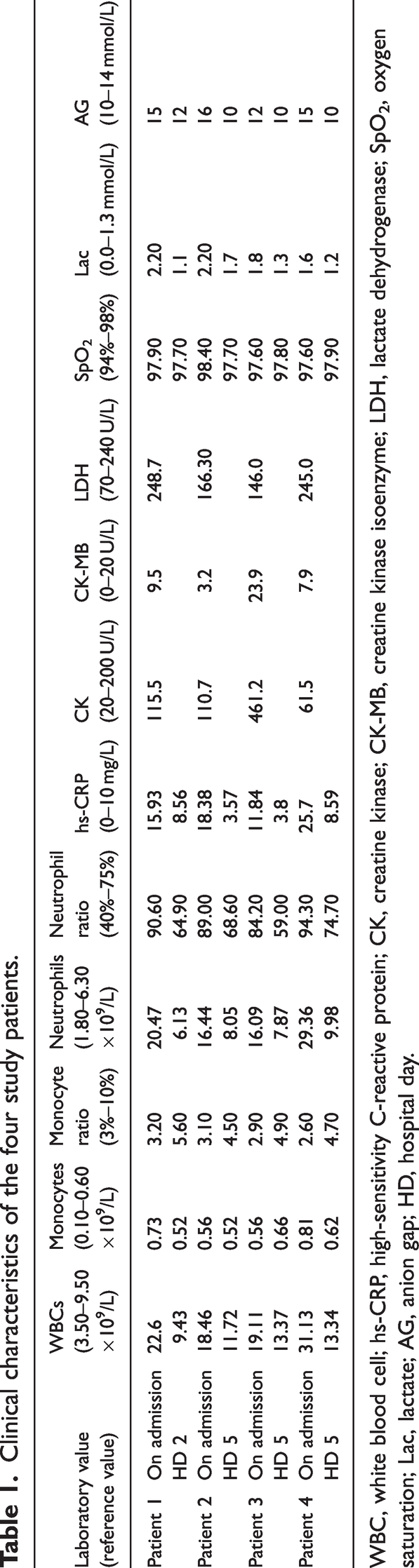
WBC, white blood cell; hs-CRP, high-sensitivity C-reactive protein; CK, creatine kinase; CK-MB, creatine kinase isoenzyme; LDH, lactate dehydrogenase; SpO2, oxygen saturation; Lac, lactate; AG, anion gap; HD, hospital day.
Case presentations
Patient 1 was a previously healthy 22-year-old man who presented to the ER with cough, chills, and fever (maximum temperature [Tmax], 39.4°C). At presentation, his vital signs were as follows: blood pressure (BP), 126/84 mmHg; heart rate (HR), 113 beats/minute; respiratory rate (RR), 21 breaths/minute; temperature (T), 37.4°C; and oxygen saturation (SpO2), 97.9% on room air. The patient did not experience any dyspnea. During the examination, clear breath sounds were heard bilaterally. Laboratory tests revealed obviously elevated levels of white blood cells (WBCs), neutrophils, monocytes, high-sensitivity C-reactive protein (hs-CRP), lactate dehydrogenase (LDH), anion gap (AG), and lactate (Lac). However, liver and kidney function, coagulation function, electrolytes, creatine kinase (CK), creatine kinase isoenzyme (CK-MB) were normal, and routine urine and stool examinations showed no obvious abnormalities. No significant electrocardiographic changes were observed. During hospitalization, the patient received oxygen, corticosteroids, and NAC. By hospital day (HD) 2, the patient’s fever had subsided, and follow-up laboratory findings showed significant improvement (Table 1). Initial and follow-up chest computed tomography (CT) conducted on HD 6 showed normal findings. The patient reported no discomfort and was subsequently discharged.
Patient 2 was a 25-year-old man who was admitted to the ER with cough, chills, and fever (Tmax, 38.2°C). On arrival, the patient’s vital signs were as follows: T, 37.2°C; HR, 84 beats/minute; RR, 21 breaths/minute; BP, 120/80 mmHg; and SpO2, 98.4%. Physical examination revealed no abnormalities. Laboratory studies showed high levels of WBCs, neutrophils, hs-CRP, Lac, and AG. After receiving the same treatment as given to Patient 1, his temperature returned to normal the next day. On HD 5, his laboratory indices had improved (Table 1). Chest CT before and after treatment revealed no abnormalities. The patient showed clinical improvement, and he was discharged on HD 5 and advised to continue oral NAC therapy at home.
Patient 3 was a previously healthy 24-year-old man who presented to the ER approximately 11 hours after the onset of cough, chills, and fever (Tmax, 38°C). His initial vital signs were as follows: T, 37.3°C; BP, 144/66 mmHg; HR, 101 beats/minute; RR, 22 breaths/minute; and SpO2, 97.6% on room air. His respiratory and cardiac examination findings were normal. Notably, certain laboratory indices, including CK and CK-MB, were found to be significantly abnormal (Table 1). However, under the same treatment plan, the patient’s fever did not persist after HD 2. Chest CT performed 5 days after starting the therapy showed no significant abnormalities. By the following day, all his symptoms had resolved. On HD 5, the patient remained stable without any further complications and was subsequently discharged.
Patient 4 was a 23-year-old woman who had been positioned closest to the laboratory table and was admitted to the ER with cough, chills, and fever (Tmax, 38°C). Her vital signs were as follows: T, 37.1°C; HR, 112 beats/minute; RR, 21 breaths/minute; BP, 106/78 mmHg; and SpO2, 97.6%. Biochemical examination revealed significantly high levels of WBCs, neutrophils, hs-CRP, and LDH (Table 1). In addition, routine urine examination showed abnormalities, including 22 pcs/µL of urine-derived epithelial cells and 10 pcs/µL of casts. Despite receiving active treatment, the patient’s condition progressed to mild pulmonary fibrosis (Figure 1).
Discussion
LPS is the major structural component of the outer wall of gram-negative bacteria. In scientific research, LPS is widely used to establish various animal models that induce infiltration of inflammatory cells and trigger the production of various inflammatory cytokines in vivo. These models are employed to study conditions such as acute lung injury (ALI), cardiomyocyte damage, acute kidney injury, and endotoxin-mediated sepsis.8–11,22 This report has presented a case series of four patients who inadvertently inhaled LPS gas owing to operational errors, resulting in a clear exposure history. All four patients were hospitalized for fever but experienced steady recovery and were ultimately discharged after receiving anti-inflammatory and antioxidant treatment.
ALI is characterized by a disrupted alveolar–capillary barrier, resulting in increased lung permeability and the infiltration of activated neutrophils into the lungs. ALI may even progress to acute respiratory distress syndrome with significant morbidity and mortality. 12 Studies involving the examination of endothelial cells after LPS administration in vivo have shown that ALI can be induced by LPS. 13 Increasing evidence suggests that the signal pathways of the LPS receptor complex, which is composed of CD14, Toll-like receptor 4 (TLR4), and myeloid differentiation protein 2, play a crucial role in the activation of macrophages by LPS. The receptor complex is involved in LPS-induced release of proinflammatory cytokines, such as tumor necrosis factor-α, interleukin (IL)-6, and IL-8, by activated monocytes and alveolar macrophages; this release of cytokines leads to an inflammatory response, oxidative stress, and the development of ALI.14,15 The characteristics of LPS-induced ALI include lung infiltration with inflammatory cells and secretion of proinflammatory cytokines. 16
In this case series, we described four patients with significantly elevated levels of neutrophils, leukocytes, and hs-CRP, which is consistent with findings from animal studies. The antioxidant NAC afforded protective effects against LPS-induced ALI by inhibiting inflammation via the TLR4/NF-κB signaling pathway.17–19 The levels of neutrophils, leukocytes, and hs-CRP in all four patients significantly decreased after NAC and hormone treatment. We also found that Patient 4, who was located closest to the laboratory table and likely inhaled a higher concentration of LPS gas, exhibited higher levels of inflammatory cells and cytokines. Despite receiving active treatment, this patient’s condition progressed to mild pulmonary fibrosis. This outcome highlights the importance of maintaining an appropriate balance of inflammatory cytokines: an adequate response can protect against pathogenic microorganisms and stress, whereas excessive production of cytokines in response to LPS can be detrimental to health.
Myocardial dysfunction or sepsis-induced cardiomyopathy after LPS challenge has been demonstrated in mice and rats.20–22 Histopathological changes observed in ALI and septic cardiomyopathy include the accumulation of activated neutrophils and macrophages, apoptosis, a proinflammatory response, and increased pulmonary capillary permeability. 23 Leukocyte adhesion, followed by excessive local nitric oxide and reactive oxygen species production, plays a major role in this harmful effect. 20 IL-6, which is produced in an autocrine manner by cardiomyocytes and recruited myeloid cells (neutrophils and macrophages), upregulates intercellular adhesion molecule 1 on cardiomyocytes, leading to neutrophil binding and stimulation of cytotoxic activity.24,25 In the present report, all four patients showed significantly elevated levels of neutrophils, leukocytes, and hs-CRP after activation of LPS. Although a large number of neutrophils recruited to the myocardium can be beneficial in clearing bacterial infections, the associated tissue damage and contribution to acute inflammation can be detrimental to the myocardium.24,25
LDH and CK are two reliable markers indicating the death of primary cardiac myocytes. 26 In the present study, two patients had elevated LDH levels after LPS inhalation and one patient had significantly elevated CK and CK-MB levels. In addition, substantial amounts of tumor necrosis factor‐α, IL-6, and IL-1β produced by cardiac myocytes and immune cells exposed to LPS can lead to myocardial injury via the reactive oxygen species‐mediated PI3K/Akt/mTOR pathway. 8 Notably, cardiac dysfunction or sepsis-induced cardiomyopathy in LPS-treated mice is reversible.27,28 However, failure to follow-up on the abnormal myocardial enzyme levels after treatment was one limitation of this study.
Previous studies have demonstrated that LPS plays a significant role in causing sepsis and contributes to the pathogenesis of sepsis-associated acute kidney injury, which may lead to cytokine storm, intensified oxidative stress, low blood pressure, renal hypoperfusion, and finally, a gradual decline in renal function via the TLR4/NLRP3 signaling pathway in a dose- and time-dependent manner. 9 The serum creatinine and urea nitrogen levels of the four patients in our study were within the normal range. However, we did observe abnormal findings in the routine urinalysis of Patient 4, with 22 epithelial cells/µL and 10 tubular cells/µL. This suggests that LPS exposure may have influenced the rat kidney tubular epithelial cells (NRK-52E cell line) via the connexin 43 channel regulating the content of reactive oxygen species between the neighboring cells, ultimately influencing the development of renal injury. 29 Conversely, lipoxin A4 exerts renoprotective effects by the peroxisome proliferator-activated receptor-γ/NF-kB pathway. 30
Conclusions
Our study revealed flu-like symptoms in healthy individuals inadvertently exposed to LPS gas. Significant increases in the serum concentrations of inflammatory cells and cytokines were observed. To the best of our knowledge, this study is the first to report on three patients experiencing acute myocardial dysfunction and one patient showing urine routine injury after LPS exposure. Despite active anti-inflammatory and antioxidant treatment, one patient’s condition still progressed to mild pulmonary fibrosis. Notably, the patient with the highest inflammatory response, elevated myocardial death markers, and abnormal urinalysis results showed the most severe multi-organ injury, highlighting the potential dose-dependency of LPS-induced damage.
This report emphasizes the importance of early diagnosis and treatment of significant LPS exposure, highlighting that this is particularly crucial in the period of the most rapidly increasing inflammatory response. It also suggests that personalized treatment approaches are necessary to manage LPS poisoning. Patients with suspected LPS-related lung injury should undergo extended observation because the initial clinical presentation may not fully reveal the extent of organ injury. However, because of the lack of serum concentration monitoring and standardized treatment guidelines, the diagnosis and treatment of LPS-related organ injury warrant further exploration.
Footnotes
Acknowledgements
We appreciate the help of all our colleagues in our hospital.
Author contributions
YD analyzed the data and wrote the first draft of the manuscript. DL designed the research. HYC analyzed the test results. YD edited and reviewed the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Data availability statement
Data are available from the corresponding author upon reasonable request.
Declaration of competing interests
The authors declare no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
