Abstract
Background
One significant consequence of sepsis and a major factor in death in critical care units is acute lung injury (ALI). Purpurin has been proven in studies to minimize oxidative stress and inflammation.
Objectives
It is yet unknown, nevertheless, how lipopolysaccharide (LPS)-induced ALI is protected against by purpurin’s anti-inflammatory and antioxidative properties.
Materials and Methods
Male Sprague-Dawley rats were administrated with an intraperitoneal injection of LPS (5 mg/kg) to create an ALI model. The animals were divided into four groups: the control animals (group I), LPS-treated control animals (group II), purpurin treated animals in a dose-based manner (groups III and IV).
Results
The animals in group II exhibited increased levels of macrophages, neutrophils, lung injury, inflammatory cytokines, myeloperoxidase (MPO), nitric oxide (NO), reactive oxygen species (ROS), and endothelial cell permeability. The level of antioxidants was reduced in the group II animals. Purpurin treatment in a dose-based manner to the LPS-treated animals considerably reduced the level of macrophages, neutrophils, lung injury, inflammatory cytokines, MPO, NO, ROS, and endothelial cell permeability. In addition, it increases the antioxidant levels in the lung tissue.
Conclusion
In summary, purpurin can ameliorate ALI induced by LPS through antioxidative and anti-inflammatory effects. Nursing care for patients with sepsis-related acute lung disease (ALD) requires monitoring vital signs, supportive care, supplemental oxygen therapy, ventilator settings, fluid balance, and pharmacological interventions like purpurin. Nutritional support, infection control, and collaboration with a multidisciplinary team are crucial.
Introduction
One of the primary causes of death in critical care units is sepsis, a systemic inflammatory reaction linked to infection. An immunological response that is dysregulated, oxidative stress, and mitochondrial dysfunction are the hallmarks of sepsis, a leading cause of mortality worldwide. Morbidity and mortality from sepsis are still quite high, making it a prevalent and dangerous clinical issue. The most common causes of death and illness in sepsis are acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) both of which include several organ injuries and dysfunction (Gill et al., 2015). Edema fluid that is high in proteins builds up in the lung’s alveolar compartment, which is a hallmark of ARDS/ALI. The emergence of ARDS/ALI is largely dependent on injury to epithelial and endothelial cells. This includes elevated vascular permeability, selection of leukocytes, overproduction of cytokines, and surfactant disorder, which can result in hypoxemia, alveolar collapse, and interstitial and alveolar pulmonary edema. The pathogenesis of ALI/ARDS is thought to include lung-tissue edema as a crucial step because of the compromised alveolar fluid clearance resulting from epithelial damage (Chen et al., 2010).
The most frequent prior diseases are described as sepsis, pneumonia, aspiration, trauma, pancreatitis, high volume blood transfusions, and inhalation of smoke or poisonous gases in more are the most epidemiological factors for ALI. The highest prevalence of ALI has been linked to severe sepsis and numerous blood transfusions; individuals with trauma or drug overdoses had the lowest risk. Lung damage risk is increased in individuals with many comorbidities, persistent alcohol misuse, or chronic lung disease (Mowery et al., 2020). The complicated interaction between soluble inflammatory mediators, like lipopolysaccharide (LPS), circulating cellular components, and various cytokines [like interleukin 1 beta (IL1β) and tumor necrosis factor alpha (TNF-α)] on various pulmonary cellular targets results in the pathophysiology of ALI/ARDS in sepsis. Proinflammatory cytokines get overexpressed as a direct reaction to infection or trauma, or they may be an indicator of continued cellular damage. Baseline and continuously higher levels of plasma interleukin IL-6, IL-8, and TNF-α were found to be highly predictive of death (Parekh et al., 2011).
ALI is thought to be primarily caused by LPS, a substance of gram-negative bacterial endotoxins that causes proinflammatory cytokines in host cells and acute inflammation. LPS causes ALI in experimental animals by increasing pulmonary microvascular permeability and entering the lung with activated neutrophils and macrophages. The LPS-induced ALI animal approach is now widely utilized in pathogenesis research and medication development (Hwang et al., 2019). There are still no proven ways to lower the high death rate linked to ALI/ARDS, despite recent research in this area. Consequently, it is imperative to find novel medications and create effective ALI/ARDS treatment plans. One natively available anthraquinone that was extracted from Rubia cordifolia roots is called purpurin. It has been utilized as a red dye in the past. Nevertheless, the biological impacts and photosensitizing characteristics have elucidated its unique applicability. In both in vivo and in vitro studies, purpurin has antioxidant activity in conjunction with antigenotoxic, anticancer, neuromodulator, and antimicrobial properties. Most of purpurin’s pharmacological actions are caused by its strong antioxidant properties. By lowering oxidative stress, it has anti-inflammatory properties. This is important since disorders involving endoplasmic reticulum and mitochondrial stress are often targeted by these properties (Singh et al., 2021).
To explore the mechanism behind purpurin’s therapeutic impact on sepsis-induced ALI, the current study created LPS-induced rat models. Our research offers proof of purpurin’s anti-inflammatory effects on pulmonary damage, suggesting that it might be a potential target for treatment in the clinical management of ALI. Purpurin, a natural anthraquinone compound from madder plant roots, has shown potential in treating acute lung disease (ALD) caused by sepsis. Nursing care involves monitoring vital signs, providing supportive care, and administering purpurin, antibiotics, and anti-inflammatory agents. However, clinical trials are needed to determine its safety and efficacy. Nurses can improve outcomes for patients.
Materials and Methods
Materials
Purpurin, LPS, and other chemicals required for the current study were acquired from Sigma–Aldrich, all of which were of analytical grade.
Experimental Model
Male Sprague-Dawley rats, weighing 215 ± 10 g (approximately 3 months of age), were utilized for this investigation. The experimental animals were housed in a laboratory under controlled conditions, with a room temperature of 21 ± 2°C, a humidity level of 60–65%, and a light/dark cycle of 12h, respectively. They were provided with standard rat chow and had free access to drinking water ad libitum. The animals were housed in polycarbonate cages. All procedures involving animals were approved by the institutional animal care and use committee.
Experimental Design
The animals were randomly assigned to four groups, each consisting of six rats:
Group I: Control group, administered 0.5 mL of normal saline.
Group II: LPS-treated control group, receiving LPS (5 mg/kg body weight) intraperitoneally to induce lung damage.
Group III: LPS-treated group, receiving LPS (5 mg/kg body weight) and purpurin (20 mg/kg body weight).
Group IV: LPS-treated group, receiving LPS (5 mg/kg body weight) and purpurin (40 mg/kg body weight). Following the experimental period, the animals were anesthetized and subsequently euthanized. Their lung tissues were immediately harvested and stored in a freezer at −80°C for subsequent analyses.
Determination of Endothelial Cell Permeability
To assess endothelial cell permeability, the rats were administered an intravenous injection of Evans Blue (20 mg/kg). After 30 min, the left lung was excised from each euthanized rat. The lung tissue was homogenized in formamide, followed by centrifugation at 8,000g at 37°C. The absorbance (A) of the supernatant was measured at 620 nm using a spectrophotometer to determine the quantity of Evans blue in the lung tissue (Han et al., 2013).
Estimation of Oxidative Stress in Lung Tissues
The overall antioxidant capacity of the pulmonary tissues was evaluated using commercial test kits according to the manufacturer’s instructions. The levels of reactive oxygen species (ROS) and the activities of antioxidant enzymes, specifically glutathione peroxidase (GPx) and superoxide dismutase (SOD), were assessed. The ROS assay Kit was utilized for quantifying ROS, while the SOD assay Kit was used to measure SOD levels. Absorbance was measured using a microplate reader.
Determination of Myeloperoxidase (MPO) Activity
MPO activity was measured spectrophotometrically in lung homogenates following the protocol established by Desser. Lung tissue samples were homogenized in 0.02 M EDTA and centrifuged at 20,000 × g for 15 min. The resulting pellets were then resuspended in 0.1 M sodium phosphate buffer containing 0.5% hexadecyltrimethylammonium bromide. This suspension was centrifuged again at 20,000g for 15 min to obtain the supernatant. Subsequently, 25 µL of the supernatant were mixed with 1 mL of 0.1 M sodium phosphate buffer (pH 7.0) containing 0.0005% hydrogen peroxide and 1 µL of guaiacol as substrates. MPO activity was determined at 470 nm using a spectrophotometer. The protein content of the tissue was measured using the Biuret method, and results were expressed in milligrams of protein (Werner et al., 2002).
Determination of Nitric Oxide (NO) Activity
NO levels were evaluated using the Griess method to measure nitrite and nitrate levels as indicators of tissue NO content. Tissue samples were homogenized in a 5 mM Tris–HCl buffer (pH 7.4) with 2 mM EDTA and centrifuged at 2,000g for 5 min. Nitrate was reduced to nitrite using copper-coated calcium granules in glycine buffer (pH 9.7) after deproteinization with Somogyi reagent. Following 45 min at room temperature in the presence of Griess reagent, samples were analyzed at 545 nm using a spectrophotometer. Nitrite levels were quantified using a standard curve made from sodium nitrite dilutions. The Biuret method was employed to determine tissue protein concentrations (Cortas & Wakid, 1990).
Determination of Inflammatory Mediators
The concentrations of inflammatory cytokines, specifically IL-6, IL-1β, and TNF-α, in tissue were measured using commercially available ELISA kits, according to the manufacturer’s instructions.
Hematoxylin–Eosin (H&E) Staining
For histological assessment, the upper lobe of the right lung was fixed in 10% buffered formalin for 24 h, embedded in paraffin, and sectioned at a thickness of 4–5 µm. H&E staining was performed following deparaffinization and dehydration. A blinded observer evaluated each section under a light microscope, and lung damage was scored based on parameters such as edema, hemorrhage, alveolar wall thickness, and neutrophil infiltration into the vessel wall or airspace (Wu et al., 2007).
Statistical Analysis
Statistical analyses were conducted using GraphPad Prism 5.0 software. The mean and standard deviation (SD) were calculated for each dataset (n = 6). One-way analysis of variance (ANOVA) was employed to determine statistical differences between groups, and post hoc Bonferroni tests were used to validate the findings. A p value of less than 0.05 was considered statistically significant.
Results
Effect of Purpurin on Pulmonary Injury
Figure 1 depicts the effect of purpurin on pulmonary injury. Compared to control rats, LPS animals had greater pulmonary tissue damage scores and higher levels of neutrophils and macrophages. The experimental animals administrated with purpurin in a dose-dependent manner (groups III and IV) exhibited a substantially decreased inflammatory score and a higher number of macrophages and neutrophils levels were decreased in comparison to the LPS group.
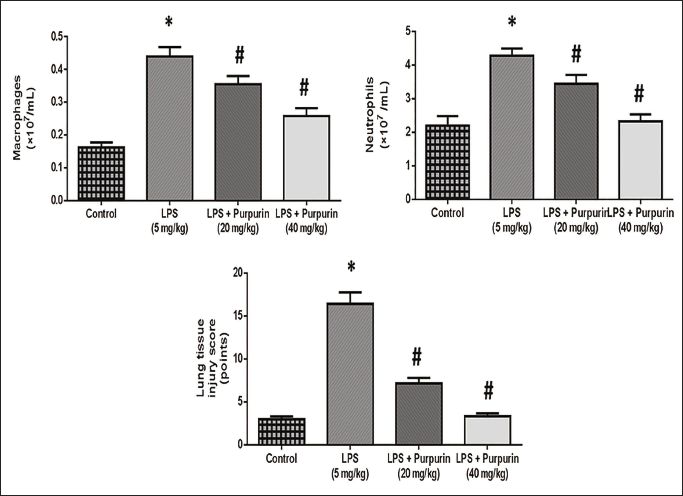
Effect of Purpurin on Lung Endothelial Cell Permeability
Figure 2 shows the effect of purpurin on lung endothelial cell permeability. Compared to control rats, the lung tissue of LPS-treated animals (group II) had a considerably larger amount of EB. The lung tissues of the animals treated with purpurin in a dose-dependent manner (groups III and IV) exhibited a reduced amount of EB in comparison to the LPS group.
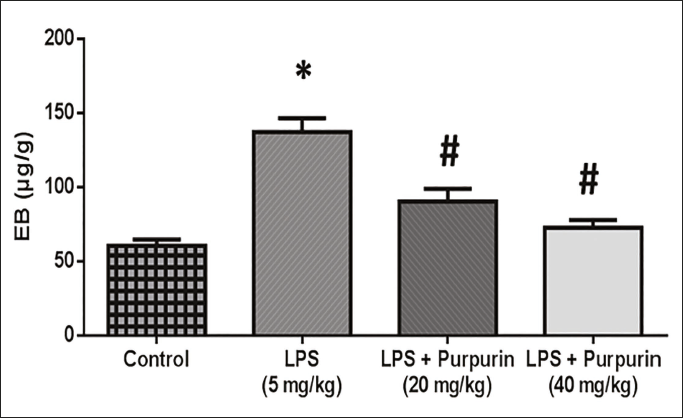
Potent of Purpurin on Oxidative Stress
The potential outcome of purpurin on the antioxidant enzyme and ROS is illustrated in Figure 3. The assessment of oxidative stress encompassed tests of SOD and GPx, which are the principal constituents of the natural antioxidant defense system. The LPS-treated group had a statistically significant decrease in the activities of GPx and SOD levels and increased levels of ROS in comparison to control rats. The treatment of purpurin in a dose-dependent manner considerably increased the SOD and GPx levels in the lungs as well as reduced the ROS levels in comparison to the LPS group.
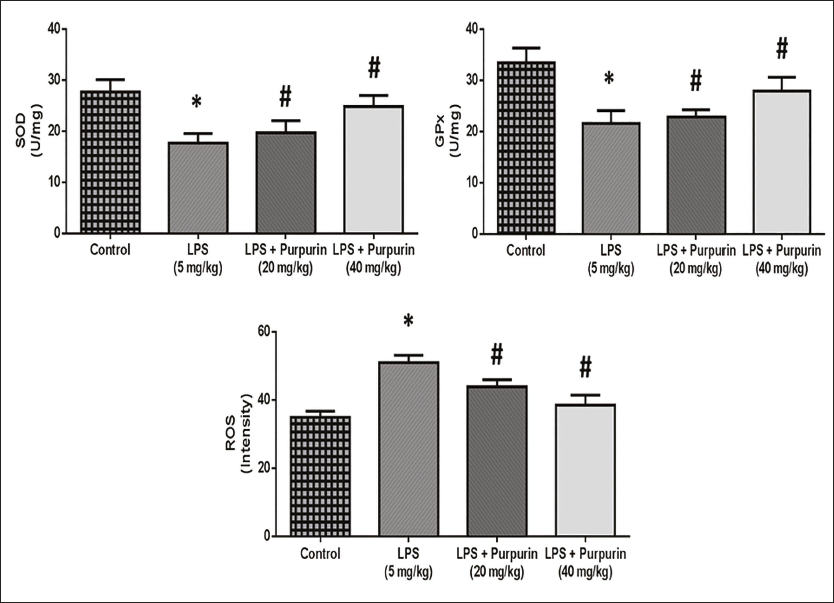
Effect of Purpurin on MPO and NO Levels
Figure 4 depicts the effect of purpurin on the MPO and NO levels in the lung tissue of the animals. The levels of MPO and NO were observed to be elevated in the LPS-treated animals compared to the control animals. Animals treated with purpurin in a dose-based manner (groups III and IV) had a considerable reduction in the MPO and NO levels compared to the LPS-treated animals.
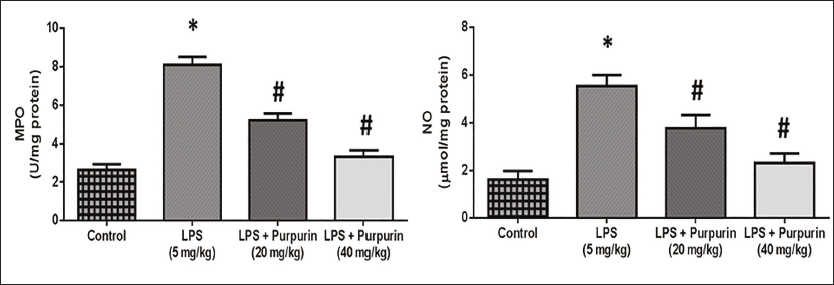
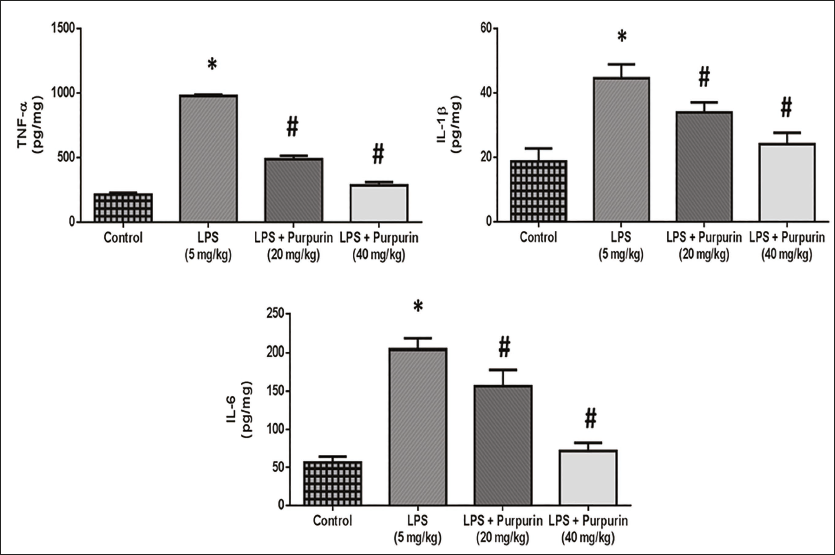
Effect of Purpurin on Inflammatory Cytokine Levels
Figure 5 depicts the effect of purpurin on inflammatory cytokines levels in LPS-triggered animals. The lung tissue of LPS-triggered animals exhibited notably higher levels of TNF-α, IL-6, and IL-1β expression in comparison to the control group. On the other hand, animals treated with purpurin in a dose-based manner pointedly reduced TNF-α, IL-1β, and IL-6 expression in comparison to the LPS animals.
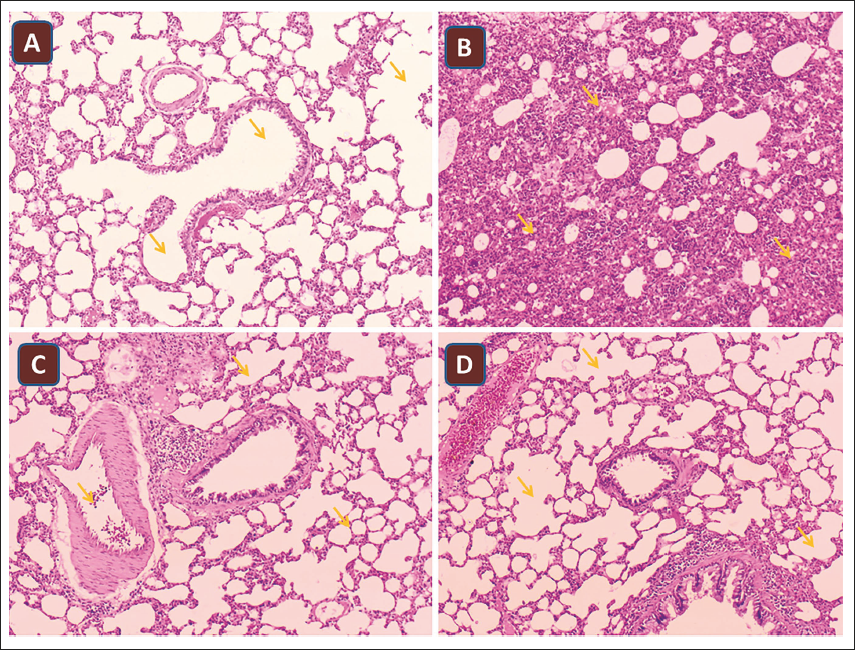
Histopathological Analysis
Figure 6 shows the lung tissue architecture of all the groups. The results demonstrated that there were no evident pathological changes in the control animals and purpurin-treated animals (groups III and IV). However, pathological lung injury was apparent in LPS-treated animals (group II), characterized by large numbers of macrophages and neutrophils, and clear congestion. However, there was a significant reduction in lung injury in purpurin-treated animals in a dose-based manner.
Discussion
The widespread inflammatory reaction driven on by an infectious condition is known as sepsis. Organ malfunction (e.g., tissue hypoxia, hypoperfusion, lung damage, etc.) is a sign of severe sepsis. According to the conventional understanding, the pathophysiology of sepsis is caused by hyperinflammatory reactions that lead to the overproduction of cytokines and abnormal immune system activation. Both result in organ failure and tissue damage (Shimaoka & Park, 2008). The pulmonary symptoms of an acute systemic inflammatory disease are known as ALI. Sepsis (severe sepsis from a pulmonary source is the most prevalent cause), trauma, aspiration, repeated blood transfusions, acute pancreatitis, inhalation damage, and specific medication toxicity are major causes of ALI/ARDS (Tsushima et al., 2009).
LPS, a substance found in the cell walls of gram-negative bacteria, constitutes one of the main contributors to sepsis. Cytokines and ROS, including hydroxyl radicals, peroxynitrite, and superoxide, are produced in response to LPS. These LPS effects have the potential to cause oxidative damage to proteins and DNA as well as lipid peroxidation (Duan et al., 2020). Purpurin’s anti-inflammatory characteristics lead us to believe that it might be a useful therapeutic drug for sepsis treatment. We investigated the molecular mechanisms of sepsis and confirmed the role of purpurin in the illness by employing the LPS-induced model, the most frequently utilized and well-established animal model of sepsis.
ALI caused by sepsis is characterized by the elevated release of neutrophils and macrophages, and a high lung tissue injury score. Neutrophils are the first immune cells to be drawn to the site of damage in ALI, and they produce cytotoxic chemicals. A prior investigation demonstrated that removing neutrophils might lessen the severity of ALI (An et al., 2019). In the present investigation, the animals treated with LPS (group II) exhibited high levels of neutrophils, macrophages, and lung injury scores. Animals treated with purpurin in a dose-dependent manner (groups III and IV) exhibited lower levels of macrophages, neutrophils, and lung injury scores in comparison to the LPS-treated control animals (group II).
The severe inflammatory reaction in ALI alters the architecture of the alveoli, increases alveolar and endothelial permeability, and reduces alveolar fluid clearance, all of which seriously compromise lung function (Huang et al., 2015). In this study, when compared to the control animals (group I), the treated animals (group II) exhibited a high amount of EB, corresponding to increased endothelial permeability. Animals treated with purpurin in a dose-dependent way (groups III and IV) exhibited low levels of EB in comparison to the LPS-treated animals (group II). Thus, purpurin-treated animals had less endothelial permeability. This demonstrates the anti-inflammatory properties of purpurin (Zeng et al., 2023).
LPS has the inherent ability to increase ROS generation in lung tissue, intensifying the inflammatory response and damaging the lungs. While the innate immune system requires a certain level of ROS to operate, excessive ROS levels can cause necrosis, apoptosis, and tissue damage. Thus, oxidative stress reduction may stop LPS-induced ALI (Chen et al., 2021). In the previous study, it was reported that in LPS-induced ALI models, there was a reduction in the amounts of GPx and SOD in lung tissue (Mao & Huang, 2017). As a result, a similar observation was observed in the current study, where the level of SOD and GPx was reduced and the ROS level was high in the LPS-treated animals (group II). Purpurin-treated animals in a dose-based way (groups III and IV) showed elevated levels of SOD and GPx and the level of ROS was downregulated in comparison to the LPS-treated animals (group II). The antioxidative effect of purpurin aided in the upregulation of antioxidants and downregulation of ROS in the animals (Nam et al., 2017).
MPO is known to be released by neutrophils and alveolar macrophages in response to acute lung inflammation. A robust marker of neutrophil invasion into the pulmonary parenchyma and alveolar cavities is elevated MPO activity. Numerous studies have utilized MPO concentration to estimate LPS-induced ALI (Wu et al., 2017). NO has a role in inflammation, memory, and vascular contraction. Specifically, there has been a correlation shown between the degree of inflammation and increased NO release through the iNOS pathway (Kang et al., 2013). In the current study, purpurin suppressed levels of MPO and NO in the treated animals (groups III and IV), thereby protecting them from lung inflammation.
IL-1β, IL-8, TNF-α, IL-18, and IL-6 are the most promising prospective molecular biomarkers among the proinflammatory cytokines to anticipate morbidity and death (Butt et al., 2016). Prior studies have demonstrated that during the initial stages of inflammation brought on by various pathogenic stimulants, such as LPS, macrophages release a great deal of proinflammatory cytokines, such as IL-1β, TNF-α, and IL-6. A high generation of proinflammatory cytokines elevates the intensity of immune system reactions, which in turn causes cascades of inflammation and tissue damage. Therefore, one potential target for anti-inflammatory pharmacological therapy is limiting inflammatory cytokine production (Zhao et al., 2017). TNF-α is particularly significant because of its wide range of activities; they involve promoting leukocyte deposition, growth, and specialization at the sites of damage and infection in addition to necrosis, angiogenesis, oxidative stress, apoptosis, and tissue remodeling (Malaviya et al., 2017). The level of TNF-α, IL-1β, and IL-6 was observed to be high in the LPS-treated animals (group II) indicating inflammation of lung tissue. Purpurin-treated animals in a dose-based manner exhibited low levels of TNF-α, IL-6, and IL-1β, indicating the anti-inflammation activity of purpurin.
Our histological results demonstrated pathological lung damage in LPS-treated animals (group II), which was consistent with the biochemical data and those from earlier investigations. This injury was marked by a high concentration of neutrophils and macrophages as well as obvious congestion. Following purpurin therapy in a dose-dependent manner, the histological alterations were significantly reduced.
Conclusion
Ultimately, our findings suggested that purpurin could protect the lung from ALD caused by LPS-triggered sepsis in rats. Purpurin treatment reduced ROS and increased antioxidant (GPx and SOD) levels in LPS-treated rats, hence reducing oxidative stress. TNF-α, IL-6, and IL-1β levels were among the additional inflammatory mediators that were reduced by purpurin treatment. Additionally, lung damage, endothelial cell permeability, MPO, NO levels, and the number of macrophages and neutrophils were all decreased by purpurin administration. Consequently, the rats under Purpurin administration are shielded from the oxidative stress and inflammation brought on by LPS. Overall, our results correspond to purpurin, which enhances prognosis in cases of ALI and sepsis caused by LPS. Nevertheless, further corroborative studies are needed to thoroughly comprehend the exact molecular mechanisms underlying the ALI and sepsis of purpurin caused by LPS.
Abbreviations
ALI: Acute lung injury; ARDS: Acute respiratory distress syndrome; BALF: Bronchoalveolar lavage fluid; DNA: Deoxyribonucleic acid; EB: Evans blue; EDTA: Ethylenediaminetetraacetic acid; ELISA: Enzyme-linked immunosorbent assay; GPx: Glutathione peroxidase; HCl: Hydrochloric acid; H&E: Hematoxylin and eosin; IL: Interleukin; LPS: Lipopolysaccharide; MPO: Myeloperoxidase; NO: Nitric oxide; ROS: Reactive oxygen species; SOD: Superoxide dismutase; TNF: Tumor necrosis factor.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The study was approved by the Ethics Committee of Shanxi Bethune Hospital, Taiyuan, China (YXLL-2023-197).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
