Abstract
Background
Acute lung injury (ALI) is associated with increased inflammation and oxidative stress. Estradiol is produced by the ovaries and is the most active hormone of estrogen. Our aim was to investigate whether estradiol contributes to protect against lipopolysaccharide (LPS)-induced ALI via induction of aquaporins AQP1 and AQP5 and the underlying mechanisms.
Introduction
Sepsis-induced acute lung injury (ALI) is an acute inflammatory disorder associated with oxidative stress and exhibits high rates of morbidity and mortality. 1 Therefore, research into the identification of new pathological mechanisms and novel treatments for ALI remain challenging.
Studies have shown that estrogen plays an important role in post-injury infection. 2 Deficiency or lack of estrogen increases the risk of infection and shock in patients and animals. 3 Estrogen can reduce the production of interleukin-1 (IL-1), interleukin-6 (IL-6), and tumor necrosis factor α (TNF-α) in cultured monocytes, macrophages, and osteoblasts in vitro and in serum of post-menopausal women.4,5 Further studies have shown that administering 17-estradiol (E2) to male rats after traumatic bleeding can improve cardiac function and reduce liver and lung damage. 6 The intact ovary and exogenous estradiol could inhibit LPS-induced ALI and the recruitment of polynucleated neutrophils in mice, suggesting that endogenous or exogenous estrogens had a protective effect on ALI. 7 In addition, estrogen also has a protective effect on hyperoxic lung injury, seawater aspiration-induced lung injury, and pulmonary edema.8,9 However, functions of estrogen in ALI remain largely unclear.
Aquaporins (AQPs) are a group of cell membrane transporters, and mainly four kinds of AQPs are distributed in the respiratory system, among which AQP1 and AQP5 are the most important. 10 Studies have shown that AQPs in lung tissues play an important role in the removal of lung water, especially in the removal of excess fluid in the alveolar.11,12 In addition, AQP1 and AQP5 are involved in the formation of pulmonary edema caused by ALI induced by multiple causes, including pulmonary infection, hyperoxia, hemorrhagic shock, acute kidney injury, and acute pancreatitis.12–14 The expression of AQP5 was down-regulated in pulmonary edema after LPS-induced diffuse in vascularization.15–17 Conditional knockout of pulmonary AQP1 in mice, increasing hydrostatic pressure of perfusate while reducing lung fluid volume by more than 2-fold.15,18
Current studies have shown that in the model of ALI induced by LPS, both estrogen receptor α (ERα) and estrogen receptor β (ERβ) were increased, suggesting that estrogen receptors may play an important role in ALI.19,20 However, whether AQPs mediate the anti-inflammatory and the clearance of alveolar water effects of estrogen through the estrogen receptor pathway remains unclear.
In the present study, we demonstrate that estradiol, the most active hormone of estrogen, can attenuate oxidative stress and inflammatory responses in rat model of LPS-induced ALI via induction of AQP1 and AQP5. Additionally, rats after LPS-challenge, pre-treated with estradiol, displayed greater benefit to the anti-inflammatory and antioxidant effects than estradiol associated with ERα antagonist or ERβ antagonist. Thus, estradiol may have potential therapeutic value for the treatment of ALI.
Methods
Reagents and chemicals
Estradiol was purchased from Sigma, (USA, E8875) and dissolved in ethanol (Sinopharm Chemical Reagent limited corporation, 10009218). Lipopolysaccharide (LPS) (sigma, L2630) was dissolved in saline (FeiBo, C19082108), and ERα antagonist methyl-piperidino-pyrazole (MPP) and ERβ antagonist 4-2-phenyl-5,7-bis(trifluoromethyl)pyrazolo{1,5-a}pyrimidin-3-ylphenol (PHTPP) were purchased from MCE (HY-103454, Y-103456).
Animals
Animal experiments were performed following the guidelines in the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (National Institutes of Health publication No. 85-23, revised 1985). The protocol was approved by the Institutional Animal Care and Use Committee of the Hangzhou Hibio Technology Co.Ltd. (IACUCprotocol number: HBFM3.68-2015). SD rats (female, oestrum) were obtained from SHANGHAI SLAC LABORATORY ANIMAL CO. LTD. Rats were housed in the same room under specific pathogen-free conditions at 18–24°C and 40–70% humidity, with a 12 h light–dark cycle. All rats were handled in accordance with guideline of the Institutional Animal Care.
LPS-induced ALI and estradiol, MPP and PHTPP pre-treatment
SD rats 14 days after oophorectomy (
Lung wet-to-dry weight ratio
To measure the lung edema after LPS-induced ALI in rats, right lung tissues were collected (
Total protein concentration of bronchial lavage fluid and pulmonary permeability
After LPS administration for 6 h, bronchial lavage fluid (BALF) was collected (
Histology and immunohistochemistry
Changes in left lung morphology (
Electron microscopy
As described previously,
22
fractions (1 mm3) of the left lung tissues (
Real-time PCR
Total RNA was isolated from lung tissues ( AQP1 forward 5’-TCACTTGGCCGAAATGACCTG-3’, reverse 5’- GTCCCACCCAGAAAATCCAGT-3’. AQP5 forward 5’- TCCAGGACCACACCAGAAAG-3’, reverse 5’- ATAAAATAGCACTCCGTGAGCC-3’. β-actin forward 5’- ACTGCCGCATCCTCTTCCTC-3’, reverse 5’- GAACCGCTCATFGCCGATAGTG-3’.
Reaction specificity was confirmed by melting curve analysis. The ratio for the AQP1 and AQP5 mRNA was normalized with β-actin and expressed as mean ± SEM as previously described. 23
Western blot
Protein from lung tissues (
ELISA
BALF was collected (
Determination of MDA and SOD
Supernatant from lung tissues (
Statistical analyses
Data got from this study were expressed as mean ±SEM. Statistical analyses were performed using one-way ANOVA, followed by Newman–Keuls post-test using GraphPad Prism 7.0 (Graph Pad Software, San Diego, CA).
Results
Estradiol protects against LPS-induced acute lung edema
ALI caused by sepsis is a common critical disease, and acute pulmonary edema is the pathological process of acute lung injury. To investigate the effects of estradiol, estradiol associated with ERα antagonist, or ERβ antagonist on LPS-induced lung edema, we first established LPS-induced ALI model in SD rats 14 days after oophorectomy. The lung edema was measured by wet-to-dry weight ratio and permeability of lung and total protein concentration of BALF. Administration of LPS caused severe lung edema in rats, reflected by increasing wet-to-dry weight ratio and permeability of lung and total protein concentration of BALF. Pre-treatment with estradiol reduced LPS-induced lung edema compared to the OVXM group in rats. Additionally, estradiol was more effective than estradiol associated with ERα antagonist or ERβ antagonist pre-treatment (Figure 1). Estradiol attenuates wet/dry ratio and permeability of lung and total protein concentration of BALF. (a) The quantitative analysis of wet/dry weight ratio of lung, 
Estradiol protects against LPS-induced ALI
To investigate the effects of estradiol, estradiol associated with ERα antagonist, or ERβ antagonist on LPS-induced ALI, lung histology was determined by HE staining. Administration of LPS caused severe lung injury in rats, reflected by edema, congestion, and thickening pulmonary septal of lung tissue. Pre-treatment with estradiol reduced LPS-induced lung injury compared to the OVXM group. Additionally, estradiol was more effective than estradiol associated with ERα antagonist or ERβ antagonist pre-treatment (Figure 2(a)). Estradiol reduces the pathological changes of lung tissues. (a) Representative images of lung tissues by HE staining at 6 h after LPS administration in rats, scale bar = 100 μm. The red arrows indicate hyperemia, the green arrows indicate inflammatory invasion, and the black arrows indicate thickening of the alveolar septum. (b) Representative images of lung ultrastructure by electron microscopy at 6 h after LPS administration, and the red arrows indicate swallowing vesicles, scale bar = 1 μm. Data represent mean ± SEM for groups of four rats.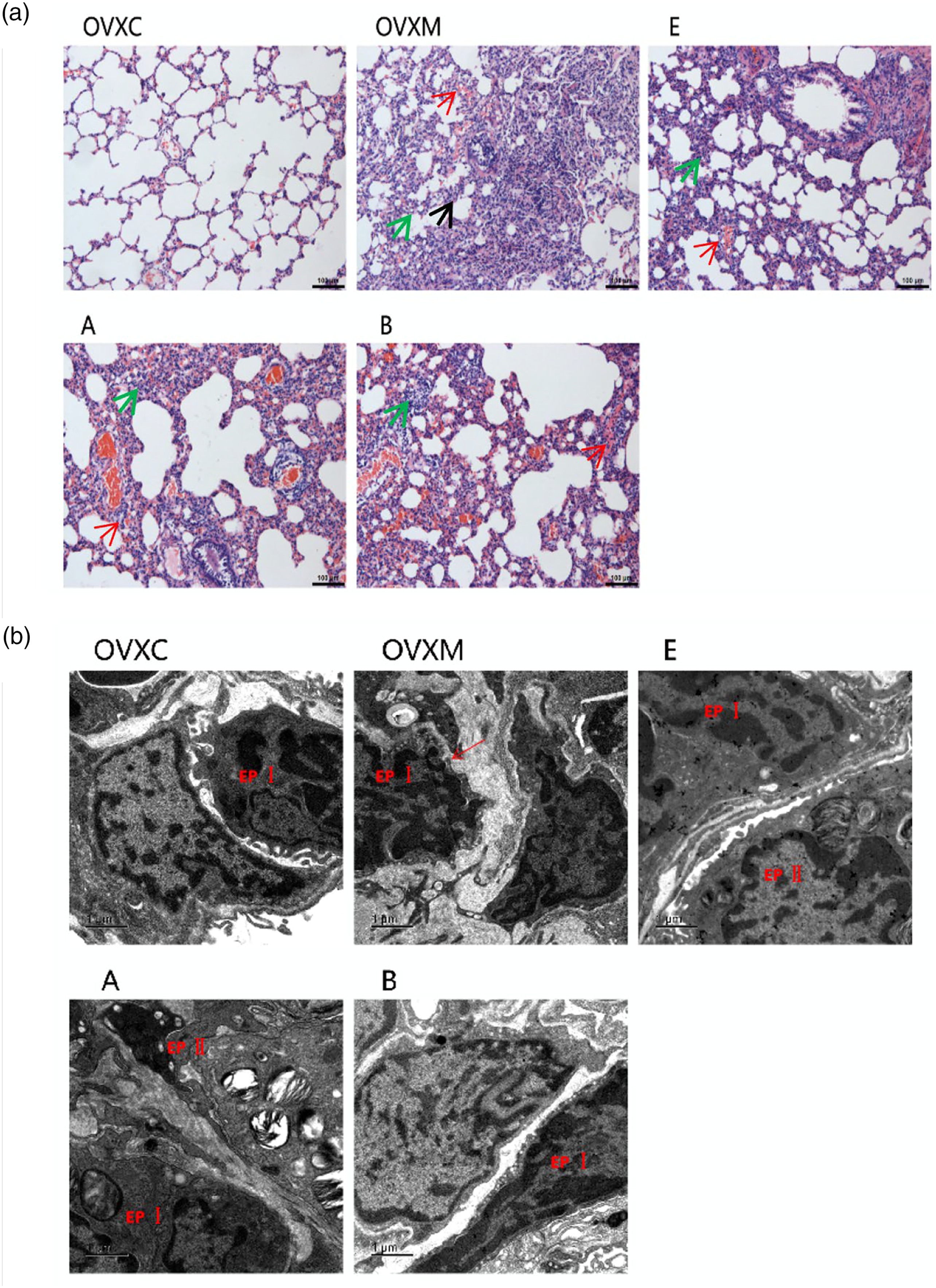
Furthermore, lung ultrastructure was determined by electron microscopy of lung sections separately. Administration of LPS caused severe lung injury in rats, reflected by enlarging intercellular space and increasing swallowing vesicles in the cytoplasm of lung tissues. Pre-treatment with estradiol reduced LPS-induced lung injury compared to the OVXM group. Additionally, estradiol was more effective than estradiol associated with ERα antagonist or ERβ antagonist pre-treatment (Figure 2(b)).
Estradiol exhibits anti-inflammatory effect to protect against LPS-induced ALI
Estradiol attenuates inflammatory cells of BALF in rats.

OVXC = ovariectomy without LPS as control group; OVXM = ovariectomy with LPS-induced ALI model group; E = ovariectomy with LPS-induced ALI and pre-treated with estrogen group; A = ovariectomy with LPS-induced ALI and pre-treated with estrogen associated with ERα antagonist group; B = ovariectomy with LPS-induced ALI and pre-treated with estrogen associated with ERβ antagonist group. Data represent mean ± SEM for groups of six rats.
Previous studies have demonstrated that endotoxin-induced pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6 can contribute to the development of ALI.
24
Moreover, activation of circulating neutrophils and their transmigration into the alveolar airspace are associated with development of ALI. Thus, we next assessed the expression of TNF-α, IL-1β, and IL-6 in BALF. As expected, estradiol significantly attenuated the expression of TNF-α, IL-1β, and IL-6 in BALF. Additionally, estradiol was more effective than estradiol associated with ERα antagonist or ERβ antagonist pre-treatment (Figure 3). Estradiol decreases TNF-α, IL-1β, and IL-6 of BALF. The quantitative analysis of TNF-α, IL-1β, and IL-6 in BALF. Data represent mean ± SEM for groups of six rats (##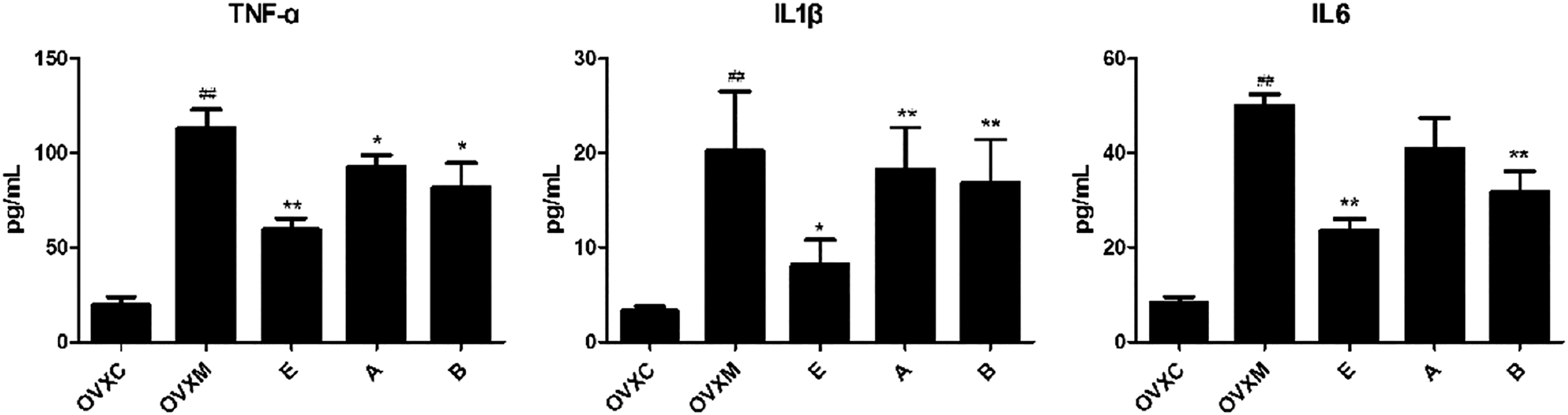
Estradiol exhibits antioxidant effect to prevent LPS-induced ALI
Next, to investigate the effects of estradiol on LPS-induced pulmonary oxidative stress, lung tissues were analyzed at 6 h after LPS administration. We found that administration of LPS caused serious pulmonary oxidative stress in rats, reflected by increasing the level of MDA while decreasing the level of SOD in lung tissues. Pre-treatment with estradiol reduced LPS-induced pulmonary oxidative stress by decreasing the level of MDA while increasing the level of SOD in lung tissues. Additionally, estradiol was more effective than estradiol associated with ERα antagonist or ERβ antagonist pre-treatment (Figure 4). These data suggest that estradiol, estradiol associated with ERα antagonist, or ERβ antagonist pre-treatment can exhibit therapeutic effects in LPS-induced ALI. Estradiol decreases MDA while increasing SOD of lung tissues. The quantitative analysis of MDA and SOD in lung tissues. Data represent mean ± SEM for groups of six rats (##
Estradiol upregulates AQP1 and AQP5 mRNA and protein expression
Current studies have shown that estradiol reduces the formation of pulmonary edema fluid, improving pulmonary edema via estrogen receptors to regulate the expression of AQP1 and AQP5.25,26 Subsequently, we evaluated the expression of AQP1 and AQP5 in lung tissues. We found that administration of LPS in rats significantly reduced the mRNA of AQP1 and AQP5 compared with the OVXC group (Figure 5(a)). Interestingly, pre-treatment with estradiol increased the mRNA expression of AQP1 and AQP5 compared to the OVXM group in rats. Additionally, estradiol associated with ERα antagonist or ERβ antagonist pre-treatment could also reduce the mRNA expression of AQP1 and AQP5, but the ERβ antagonist was more effective than ERα antagonist. Moreover, we found that pre-treatment with estradiol increased the protein expression of AQP1 and AQP5 compared to the OVXM group in rats by immunohistochemistry and Western blot. Additionally, estradiol associated with ERα antagonist or ERβ antagonist pre-treatment could also reduce the protein expression of AQP1 and AQP5 inordinately (Figure 5(b)–(d)). These data suggest that estradiol pre-treatment can exhibit therapeutic effects in LPS-induced ALI. Estradiol induces mRNA and protein of AQP1 and AQP5 of lung tissues. (a) The quantitative analysis of AQP1 and AQP5 in lung tissues, 
Discussion
In this study, we found that estradiol was protective and had therapeutic potential for LPS-induced ALI. This was supported by the findings that administering rats with LPS developed severe ALI with lung edema, inflammation, and oxidative stress damage, which were reversed by a pre-treated study with supplement of exogenous estrogen with estradiol. Importantly, we found that pre-treatment with estradiol combined with ERα antagonist or ERβ antagonist is less protective against lung injury than estradiol alone. Thus, results from this study revealed that estradiol is protective and has therapeutic potential for ALI.
Estrogen has been reported in cardiovascular diseases,27,28 cancer, 29 brain. 30 Estrogen is involved in the formation of brain edema 31 and has therapeutic effect on injured tissues. The administration of estrogen to male rats after traumatic hemorrhage can improve the cardiac function and reduce the damage of liver and lung. 6 In addition, mice treated with exogenous estrogen can inhibit LPS-induced ALI and the recruitment of polynucleated neutrophils. 7 Thus, estrogen may have therapeutic effect on injured tissues. We sought to identify the novel therapeutic effect of estrogen in LPS-induced ALI. To assess the therapeutic effect of estrogen on LPS-induced ALI animal model, we pre-treated rats with estradiol, the most active hormone of estrogen, before LPS administration. Rats after stimulation with LPS 6 h, the therapeutic effects of estradiol were evident in significantly attenuating pathological changes of lung tissues (Figures 1 and 2). These results strongly demonstrate that estradiol exerts not only preventive but also therapeutic effects on LPS-induced ALI. Moreover, we also found that pre-treatment with estradiol combined with ERα antagonist or ERβ antagonist is less protective against lung injury than estradiol alone (Figures 1 and 2). These results indicate that estrogen protects against ALI through the estrogen receptor pathway.
Overproduction of ROS has been implicated in airway and lung damage and consequently in the pathogenesis of several respiratory inflammatory diseases, including acute respiratory distress syndrome, asthma, cystic fibrosis, and chronic obstructive pulmonary disease. 32 ROS can initiate inflammatory responses in the airways and lungs through the activation of redox-sensitive transcription factors. 33 The present data showed that estradiol not only exerted anti-inflammatory function (Table 1, Figure 3) but also had antioxidant effect on lung injury reflected by decreasing the level of MDA while increasing the level of SOD (Figure 4). This is consistent with Li J and Cheng C.4,34 However, pre-treatment of estradiol associated with ERα antagonist or ERβ antagonist has less outcomes of anti-inflammatory and antioxidant against lung injury than the estradiol-only pre-treatment (Table 1, Figures 3 and 4). These results further indicate that estrogen protects against ALI through the estrogen receptor pathway.
In the mechanism study, we demonstrated that the AQP1 and AQP5 were down-regulated in LPS-induced lung tissue of rats. Estradiol pre-treatment significantly enhanced AQP1 and AQP5 expression in rats’ lung tissues (Figure 5). The previous studies are consistent with our findings,13–15,18 however, it’s not mentioned that in the study whether estradiol through estrogen receptor can protect against LPS-induced ALI via inducing the expression of AQP1 and AQP5. In our study, we have a tentative discussion that estradiol may protect against LPS-induced ALI through the estrogen receptor via upregulating the expression of AQP1 and AQP5.
Despite our findings suggesting that estradiol induces the expression of AQP1 and AQP5, the precise mechanism remains to be elucidated in the further study. For example, ENaC, Na, K-ATPase, and chloride ion channel could also affect the pulmonary edema, and we should assay them in the further study. In addition, a positive control drug is lacking in this study. This study mainly discusses the regulation effect of estrogen on aquaporins AQP1 and AQP5 in ALI. Since there is no definite positive drug in model of ALI, so we did not add positive drug in this study. However, some common positive drugs, due to their different mechanisms, may lead to different purposes and conclusions of this study. We should investigate the effect of estradiol on ALI compared to a positive control drug with the same mechanism in the further study. It is pity that we did not include the calculation and justification of the sample size selected in the method for this study. We used 10 rats per group which is in line with the statistical requirements and in accordance with the principles of animal ethics and the “3R” principle in the process of the experiment.
Conclusion
Our results demonstrate for the first time that the anti-inflammatory and antioxidant effects of estradiol are mediated by the activation of AQP1 and AQP5 and that pre-treated estradiol only is more effective than estradiol associated with ERα antagonist or ERβ antagonist in attenuating ALI. These findings establish that estradiol can exert critical protective effects on ALI via AQP1 and AQP5 induction and provide an avenue for therapeutic intervention of respiratory inflammatory diseases.
Footnotes
Authors contributions
Wang Xiaobo and Zhou xiuyun performed experiments, analyzed the data, and wrote the manuscript. Xia xiumei performed animal model, data collection, and analysis. Zhang yili designed and supervised the study and revised the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Project of Social Development of Jinhua Science and Technology Bureau of Zhejiang Province (No. 2017-3-012) and the Public Welfare Project of Science and Technology Department of Zhejiang Province (No. LGD19H150001).
