Abstract
Background
Nasopharyngeal carcinoma (NPC) is an uncommon type of epithelial carcinoma, characterized by unique features and a higher prevalence in certain populations. Traditional medicines are commonly employed in the treatment of various cancers, including NPC. Early-stage NPC patients who undergo radiotherapy alone or in combination with chemotherapy can experience significant improvements in clinical outcomes; however, these treatments often come with substantial side effects. Given that NPC is typically diagnosed at an advanced stage, there is a significant likelihood of local recurrence, distant metastasis, and drug resistance, which limits treatment options and results in poor clinical outcomes. Consequently, the development of highly active antitumor drugs with minimal side effects has become a major focus in current cancer research.
Objectives
This study aims to summarize the current clinical treatments for NPC and explore the role of the phosphoinositide 3-kinase/protein kinase B (PI3K/AKT) signaling pathway in NPC. Additionally, it reviews the effects of traditional Chinese medicine (TCM) compounds and monomers on NPC through the PI3K/AKT signaling pathway.
Methodology
The main keywords chosen for this study were “Traditional Chinese Medicine (TCM)”, “nasopharyngeal carcinoma (NPC),” “natural plant,” and “PI3K/AKT signaling pathway.” We conducted a comprehensive literature search using multiple online databases, including CNKI, Web of Science, and PubMed, along with other publication resources.
Results
The findings suggest that current first-line treatments for NPC fall short of fully addressing clinical needs. The PI3K/AKT signaling pathway is crucial in the development and progression of NPC. Several components of TCM have been shown to impact NPC development and progression through the modulation of the PI3K/AKT signaling pathway.
Conclusion
The regulatory mechanisms of NPC involve multiple signaling systems, with the PI3K/AKT pathway identified as a critical mechanism for effective modulation of the disease. This review highlights traditional medicines that target the PI3K/AKT pathway, demonstrating promising anti-NPC effects by inhibiting cell growth, inducing apoptosis, and modulating the PI3K/AKT/mammalian target of rapamycin (mTOR) pathway. The review concludes by underscoring the potential of the PI3K/AKT pathway as a therapeutic target for NPC and the importance of continued research to optimize treatment strategies.
Introduction
Nasopharyngeal carcinoma (NPC) is an epithelial malignancy originating in the mucosal lining of the nasopharynx, commonly presenting in the pharyngeal recess, particularly the fossa of Rosenmüller. Although it shares its epithelial origin with other head and neck tumors, NPC exhibits unique characteristics distinguishing it from these malignancies. On a global scale, NPC is a rare form of cancer, with a typical incidence rate of less than one case per 100,000 person-years. However, the Cantonese community in southern China reports significantly elevated rates, while indigenous groups in Southeast Asia, the Arctic, North Africa, and the Middle East experience moderate rates. NPC causes over 65,000 deaths annually worldwide, with its incidence varying significantly by region. Although NPC is uncommon in North America and Europe, it is prevalent in endemic regions, with incidence rates exceeding 20 cases per 100,000 people in areas such as Southern China, Southeast Asia, and the Middle East/North Africa (Chang & Adami, 2006; Ferlay et al., 2010).
Etiology and Contributing Factors
The World Health Organization classifies NPC into three subtypes: keratinizing squamous, basaloid squamous, and non-keratinizing. The keratinizing subtype is rare worldwide, while the non-keratinizing subtype, which accounts for over 95% of cases, is primarily linked to the Epstein-Barr virus (EBV) in regions where the disease is endemic. Key risk factors for NPC include EBV infection, genetic susceptibility, and environmental exposures (Figure 1) (Lawrence et al., 2014; Pathmanathan et al., 1995; Wang et al., 2016).
The Key Risk Factors Associated with Nasopharyngeal Carcinoma.
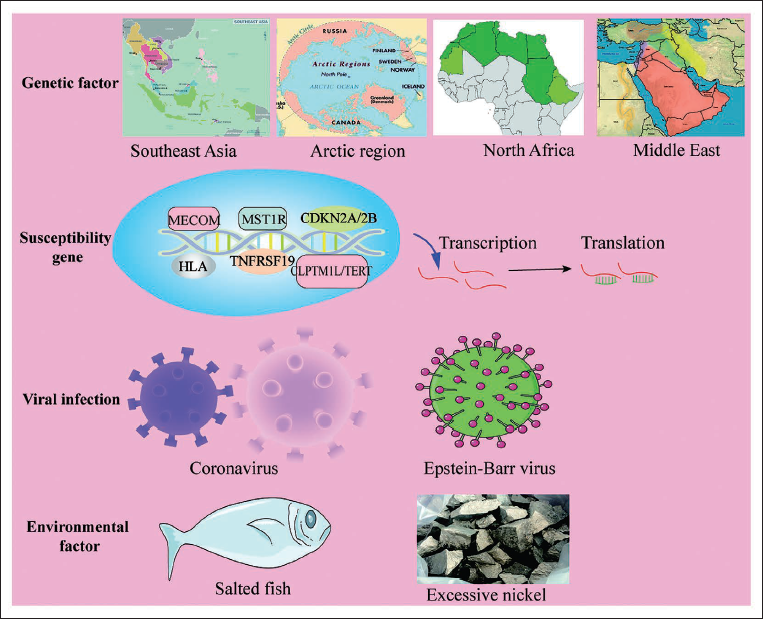
Genetic research has pinpointed HLA genes on chromosome 6p21 as significant risk loci for NPC. Genome-wide association studies (GWAS) have uncovered additional loci linked to NPC, including TNFRSF19, MECOM, CDKN2A/2B, and CLPTM1L/TERT. Variants in the MST1R gene are linked to early-onset NPC, highlighting its potential for population screening (Bei et al., 2010; Fachiroh et al., 2012; Ng et al., 2009). Recent studies have uncovered several genomic alterations in NPC, such as mutations in NF-κB-negative regulators, loss of CDKN2A/CDKN2B, amplification of CCND1, mutations in TP53, and aberrations in the PI3K/MAPK pathways. Alterations in chromatin modification and DNA repair mechanisms have also been observed (Lin et al., 2014). LMP1, an EBV oncoprotein, activates NF-κB signaling. A notable mutual exclusivity exists between LMP1 overexpression and NF-κB pathway aberrations, suggesting that both somatic and virus-mediated NF-κB signaling are integral to NPC pathogenesis. The primary etiological factors for NPC include host genetics and EBV infection, with additional risk factors such as family history, tobacco use, consumption of preserved foods and alcohol, and poor oral hygiene (Zhang et al., 2017).
Stages and Prognosis
The diagnosis of NPC involves evaluating symptoms and staging using the UICC/AJCC TNM system. Molecular biomarkers, such as pretreatment plasma EBV DNA, DNA methylation, and miRNAs, enhance prognosis and potential treatment benefits. Immunotherapy explores PD-1/PD-L1 expression, with tumor-infiltrating lymphocytes (TILs) suggesting favorable outcomes (Liu et al., 2012). Pretreatment plasma EBV DNA significantly improves predictive performance (Tang et al., 2018; Wang et al., 2018). Ongoing studies assess its role in risk-stratified treatment adaptation and posttreatment surveillance. However, assay standardization challenges persist, urging harmonization efforts for consistent measurements in prospective studies. Recommendations include targeting two EBV sequences for improved sensitivity and exploring advanced technologies like next-generation sequencing or digital PCR. Laboratories are optimizing assays to enhance NPC staging and facilitate widespread clinical use (Chan et al., 2018; Le et al., 2013).
Radiotherapy Treatment
NPC is treated with radiotherapy, evolving from conventional to intensity-modulated techniques (IMRT). IMRT have notably improved locoregional control, lowered 5-year failure rates to 7.4%, and reduced radiation-induced toxicities (Mao et al., 2016). IMRT demonstrated superior 5-year local control and overall survival compared to 2D or 3D radiation, as evidenced by a meta-analysis (Zhang et al., 2015). While IMRT is now favored, there is growing interest in proton or carbon ion radiotherapy for NPC, aimed at enhancing the therapeutic ratio. The use of proton and carbon ion methods allows for a rapid decrease in radiation dosage, enabling higher doses to be delivered to the tumor while minimizing the impact on surrounding healthy tissues. Intensity-modulated proton therapy (IMPT) offers superior dosimetric improvements over IMRT by precisely minimizing radiation exposure to surrounding healthy tissues (Lewis et al., 2016). Initial studies on IMPT and intensity-modulated carbon ion therapy (IMCT) report promising outcomes, including high survival rates. However, more comprehensive studies with extended observation periods are essential to confirm these findings and investigate any potential adverse effects (Hu et al., 2018). Despite improved radiotherapy techniques, precise delineation and accurate dose delivery are crucial for NPC treatment success. Consensus guidelines exist for clinical target volume (CTV) and OAR delineation, but high inter-observer variability persists (Lee et al., 2018; Sun et al., 2014). Efforts to improve the process of delineating the main GTV include using deep learning methods, such as neural networks with convolution, to automate the process and promote accuracy, consistency, and efficiency in radiation management (Lin, Dou, et al., 2019).
Chemotherapy and Adjuvant Chemotherapy Treatment
Several studies have consistently demonstrated that concurrent chemoradiotherapy offers a survival advantage over radiation alone in individuals with locoregionally advanced NPC, regardless of whether adjuvant chemotherapy is administered (Al-Sarraf et al., 1998; Chan et al., 2005; Kwong et al., 2004). “A recent meta-analysis shows that there are significant benefits in overall survival when using both contemporary plus adjuvant therapy (HR 0.65 [95% CI 0.56–0.76]) or concurrent radiation therapy alone (HR 0.80 [0.70–0.93]).” On the other hand, there is no notable advantage shown when using adjuvant treatment alone (HR 0.87 [0.68–1.12]) or induction therapy only (HR 0.96 [0.80–1.16]) (Blanchard et al., 2015). The debate on whether adjuvant chemotherapy post-concurrent chemoradiotherapy provides additional survival benefits persists. Preliminary results from a phase 3 trial show no significant improvement in outcomes with adjuvant chemotherapy added to concurrent chemoradiotherapy. Consequently, concurrent chemoradiotherapy remains the primary treatment for locoregionally advanced disease.
Cisplatin, commonly used in concurrent chemotherapy, can be administered through various dosing regimens. The recommended cumulative dose is 200 mg/m2 when no prior induction chemotherapy is administered, and 160 mg/m2 when induction therapy is included. Alternative concurrent agents that can be used include the combination of uracil and tegafur, as well as oxaliplatin and nedaplatin (Lv et al., 2018; Tang, Chen, et al., 2018). Administering adjuvant chemotherapy with cisplatin (80–100 mg/m2) and fluorouracil (800–1,000 mg/m2 per day over 4–5 days) every 4 weeks for a total of three cycles does not appear to significantly improve survival when used independently. Moreover, subsequent chemotherapy following chemoradiotherapy does not seem to provide additional advantages in the treatment of advanced-stage disease. In a phase 3 clinical trial, high-risk patients with detectable levels of EBV DNA following radiotherapy were allocated either to an observation group or to receive six cycles of adjuvant chemotherapy with cisplatin and gemcitabine (Zhang et al., 2016). Despite this biomarker-based selection and the use of a potent drug combination, there was no significant improvement in 5-year relapse-free survival (49% vs. 55%) or overall survival (64% vs. 68%) (Chan et al., 2018). The ongoing NRG-HN001 trial is currently investigating whether adjuvant therapy with gemcitabine and paclitaxel offers better outcomes compared to cisplatin and fluorouracil, guided by post-radiotherapy plasma EBV DNA levels. However, the poor tolerance of conventional chemotherapy (cisplatin combined with either fluorouracil or gemcitabine) within the first 6 months post-radiotherapy, with compliance rates between 50% and 76%, may diminish the efficacy of adjuvant chemotherapy. Metronomic oral chemotherapy (e.g., capecitabine) offers an alternative with benefits like high compliance, low toxicity, and convenience (A et al., 2016). Retrospective analyses conducted by Chen et al. (2017) have demonstrated enhanced survival rates in high-risk patients receiving a 12-month metronomic regimen of uracil and tegafur. Additionally, an ongoing phase 3 clinical trial (NCT02958111) is currently assessing the efficacy of metronomic capecitabine compared to observation in patients with high-risk NPC, with the results highly anticipated.
Induction chemotherapy has shown better tolerance and a quicker eradication of micrometastases compared to adjuvant treatment. Consequently, the combination of induction chemotherapy with concurrent chemoradiotherapy has emerged as a promising approach in the IMRT era for treating NPC (Chua et al., 2005; Ribassin-Majed et al., 2017). The variability in outcomes across randomized trials could be linked to sample size discrepancies or variations in induction regimens. However, pooled analyses from endemic regions have confirmed a survival advantage, largely due to improved distant metastasis control. Reflecting its increasing significance, the 2018 NCCN Guidelines have elevated the evidence supporting induction chemotherapy combined with concurrent chemoradiotherapy, particularly for advanced NPC in the IMRT era. High-risk patients for distant metastasis might gain additional benefits from this induction chemotherapy approach (Chen et al., 2018).
Treatment of Traditional Chinese Medicine (TCM)
With the continuous advancements in modern pharmacological research, targeted drug therapies for cancer have achieved remarkable progress. As a significant component of pharmaceuticals, TCM has been extensively studied at both the basic and clinical levels (Lin, Cao, et al., 2019; Xu et al., 2003). Numerous studies have demonstrated that various Chinese herbal medicines possess antitumor properties (Chen & Liu, 2024). These herbs primarily exert their anticancer effects by suppressing tumor cell growth and metastasis, triggering autophagy, apoptosis, and necrosis in cancer cells, facilitating the differentiation of cancer stem cells, and regulating key signaling pathways within tumor cells (Cai et al., 2023). The research into the anticancer effects of individual compounds derived from TCM has provided substantial support for the development of new cancer therapies. Specifically, the focus on targeting abnormally expressed kinases and aberrantly activated signaling pathways in tumors with TCM monomers has become a hot topic in drug development, offering new hope for cancer treatment (Xiang et al., 2019).
The broad clinical use of TCM as a complementary therapy has been demonstrated to significantly reduce various side effects associated with radiotherapy. These reactions include skin redness, dry mouth and nose, nasal bleeding, oral mucosal ulcers, and reductions in white blood cell and platelet counts (Del Re et al., 2019; Wu et al., 2021). When combined with radiotherapy and/or chemotherapy, TCM significantly enhances treatment outcomes by improving patient quality of life, reducing acute side effects, boosting immune function, and increasing radiotherapy sensitivity (Mao et al., 2014b; Zhu et al., 2020). Moreover, TCM has demonstrated a marked ability to lower both overall mortality and cancer-specific mortality in patients with advanced NPC (Wang, Wang, et al., 2020). A case-control study conducted in the Guangxi and Guangdong regions in China demonstrated that the consumption of herbal soups may lower the risk of NPC. Furthermore, a systematic review of 22 randomized controlled trials revealed that integrating traditional Chinese herbal treatments with standard NPC therapies notably improved survival rates at 1, 3, and 5 years, boosted tumor response, and decreased the likelihood of distant metastasis. These results suggest that TCM serves as an effective complementary therapy for NPC (Kim et al., 2015).
Role of PI3K/AKT in NPC
The dysregulated activation of the phosphoinositide 3-kinase/protein kinase B (PI3K/AKT) signaling pathway in NPC leads to a range of biological effects, primarily through the phosphorylation of downstream targets such as BCL2, Bad, mammalian target of rapamycin (mTOR), and glycogen synthase kinase-3 (GSK3). The activation of the PI3K/AKT signaling pathway enhances cellular growth and proliferation, inhibits apoptosis in response to various stressors, disrupts normal cell cycle regulation, and modulates cell motility (Glaviano et al., 2023). Additionally, the PI3K/AKT pathway is integral to angiogenesis and is a key contributor to the development of tumor multidrug resistance, which results in resistance to both chemotherapy and radiotherapy. Mutations in the PIK3CA gene, encoding the p110α catalytic subunit of PI3K, are frequently observed in various human cancers (Bredemeier et al., 2017; Or et al., 2006). These mutations result in the constant activation of the PI3K/AKT signaling pathway, which facilitates oncogenic processes including unchecked cell growth, proliferation, and survival. The frequent occurrence of PIK3CA mutations highlights the significant role of this gene in cancer progression, positioning it as a promising target for therapies aimed at inhibiting the PI3K/AKT pathway (Samuels et al., 2004).
The aberrant activation of the PI3K/AKT signaling pathway drives various biological processes by phosphorylating key downstream proteins, including BCL2, Bad, mTOR, and GSK3 (Peng et al., 2022). This activation promotes cell growth and division, inhibits apoptosis, disrupts cell cycle regulation, and enhances cell motility. Additionally, this pathway is critical in supporting angiogenesis and plays a major role in tumor drug resistance, diminishing the effectiveness of chemotherapy and radiation (Li et al., 2022; Sun et al., 2021).
EBV in NPC cells displays type II latency, expressing latent viral proteins and noncoding RNAs, including LMP1, LMP2A/B, BARF1, EBNA1, BARTs, EBER1/2, and miR-BARTs. The LMP1 and LMP2 genes play a pivotal role in regulating several signaling pathways that facilitate tumor survival and metastasis, contributing to the poor prognosis of NPC (Chou et al., 2008; Dawson et al., 2012; Hariwiyanto et al., 2010). The primary pathogenic pathways implicated include NF-κB, PI3K/AKT, RAS/RAF/MEK/ERK, JNK/c-Jun, JAK/STAT, and PLC/PKC signaling, with particular emphasis on the PI3K/AKT pathway due to its significant impact (Figure 2).
The Mechanism of Phosphoinositide 3-Kinase (PI3K)/Protein Kinase B (Akt) Signaling Pathway in the Nasopharyngeal Carcinoma.
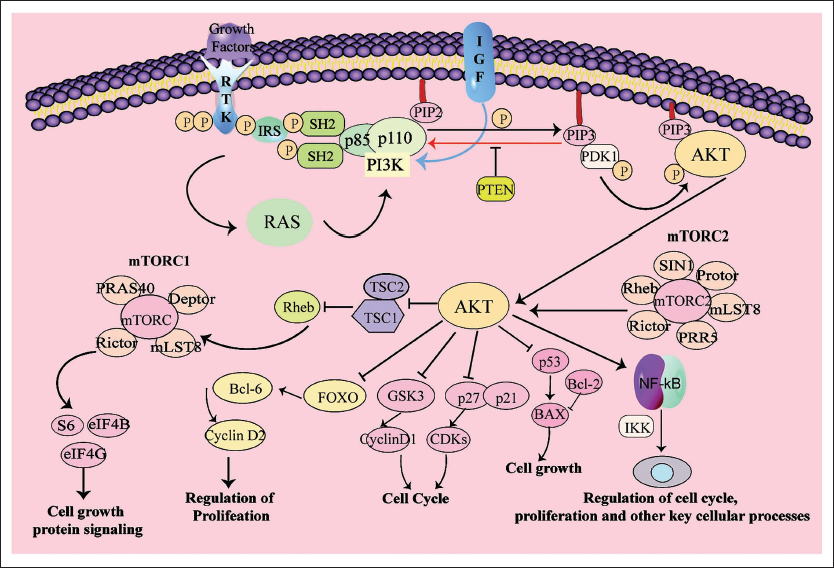
Research highlights the pivotal involvement of the PI3K/AKT pathway in the pathogenesis of EBV-associated NPC, contributing to tumor progression through mechanisms such as enhanced cell proliferation, inhibition of apoptosis, genomic instability, and alterations in cytoskeletal dynamics. LMP1 activation of PI3K/AKT significantly impacts cell survival and migration in EBV-positive NPC (Dai et al., 2019; Fei et al., 2019). Antagonists could potentially alleviate these effects, emphasizing the pivotal role of PI3K activation in EBV-induced carcinogenesis. PI3K, driven by LMP2A, modulates the expression of genes related to glucose metabolism in NPC samples (Ma et al., 2015; Qin et al., 2015). LMP2A induces EMT and cancer stem cell phenotype via PI3K/AKT activation. LMP1 activates PI3K/AKT, increasing Bcl-2 expression, suppressing Par-4 activity, inhibiting DNA repair, and influencing cytoskeleton regulation. The PI3K/AKT pathway may enhance cdc2 activity, affecting microtubule regulation (Chen et al., 2008).
Traditional Drugs Modulate NPC Progression by Targeting the PI3K/AKT Signaling Pathway
With continuous advancements in modern pharmacological research, targeted therapies for cancer have made significant strides. TCM, as a vital component of pharmaceutical science, has been extensively investigated at both fundamental and clinical levels. Numerous studies have revealed that various Chinese herbal medicines possess antitumor properties (Zhang et al., 2021). The primary mechanisms through which these herbs exert their anticancer effects include inhibiting tumor cell proliferation and metastasis, inducing autophagy, apoptosis, and necrosis in tumor cells, promoting the differentiation of cancer stem cells, and regulating tumor cell signaling pathways (Wang, Zhang, et al., 2020; Xu et al., 2022). As TCMs, these compounds have provided substantial support for the development of new anticancer drugs. Targeting abnormally expressed kinases and aberrantly activated signaling pathways in tumors with TCM monomers has become a hot topic in drug development, offering new hope for cancer treatment (Zhu et al., 2023).
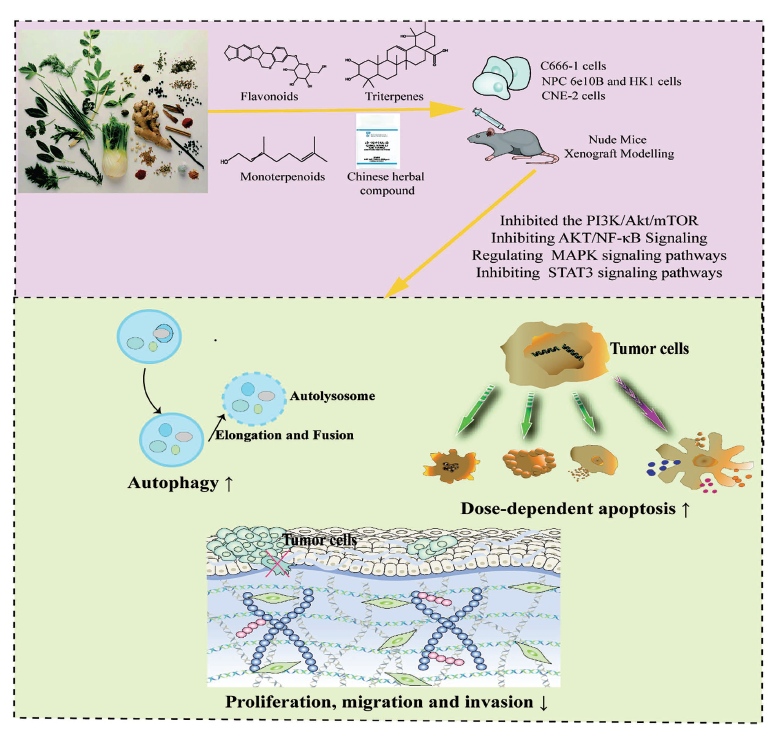
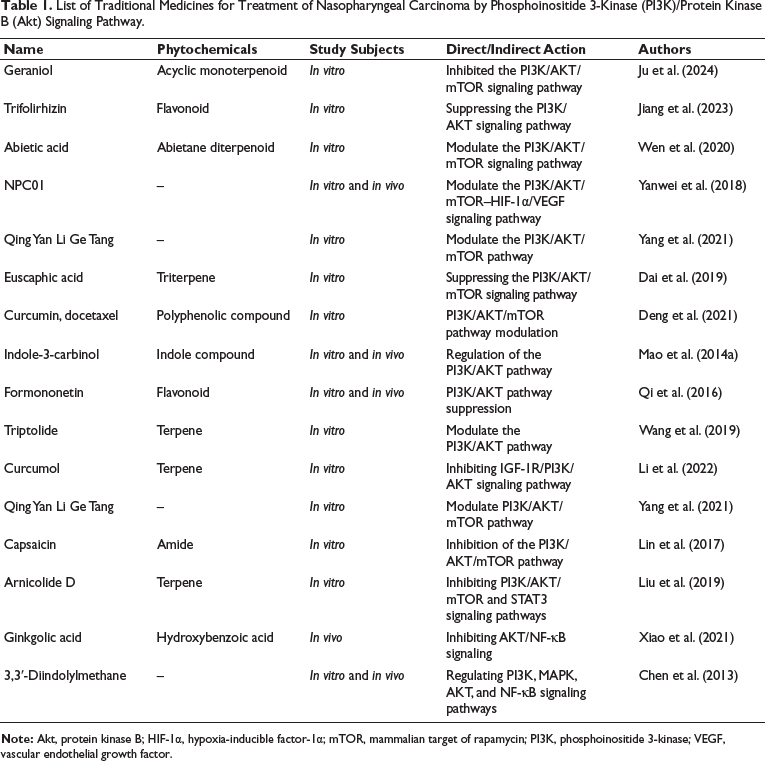
Traditional drugs have been conclusively proven effective against NPC (Figure 3). Geraniol has demonstrated notable anticancer effects in NPC by reducing the viability of C666-1 cells, as shown in MTT assays. Geraniol treatment led to decreased mitochondrial membrane potential (MMP), elevated apoptosis, and changes in oxidative stress markers. Furthermore, geraniol upregulated pro-apoptotic genes while inhibiting the PI3K/AKT/mTOR signaling pathway. These results emphasize geraniol’s potential as a potent anticancer agent for NPC, capable of inhibiting cell proliferation, inducing apoptosis, and selectively targeting the PI3K/AKT/mTOR pathway, positioning it as a promising therapeutic candidate for NPC treatment (Ju et al., 2024). Trifolirhizin, a naturally occurring flavonoid derived from leguminous plants, has attracted significant interest for its potential medicinal properties. By inhibiting the PI3K/AKT signaling pathway, it effectively suppresses the proliferation, migration, and invasion of NPC 6e10B and HK1 cells. These findings offer valuable insights into the potential application of trifolirhizin as a therapeutic agent for NPC treatment (Jiang et al., 2023). Abietic acid induced changes in MMP and caused G2/M phase cell cycle arrest through the generation of reactive oxygen species (ROS). Additionally, it inhibited cancer cell invasion and migration in NPC cells. Abietic acid also suppressed the PI3K/AKT/mTOR signaling pathway in HNE1 cells, a key target for anticancer therapies. Overall, these findings suggest that abietic acid holds promise as a therapeutic candidate for NPC treatment, as presented in Table 1 (Wen et al., 2020). Similarly, NPC01 effectively inhibited the proliferation of human NPC cell lines and induced dose-dependent apoptosis. NPC01 exhibited inhibitory effects on cell proliferation and angiogenesis in mice bearing NPC xenografts. Specifically, it reduced the levels of hypoxia-inducible factor-1α (HIF-1α) and vascular endothelial growth factor (VEGF), both of which play key roles in angiogenesis. This outcome was attributed to the inhibition of the PI3K/AKT/mTOR signaling pathway (Yanwei et al., 2018). Additionally, Qing Yan Li Ge Tang (QYLGT) suppressed NPC cell growth, colony formation, and induced the formation of vacuoles within the cells, along with the presence of autophagy-related proteins. The autophagy inhibitor 3-methyladenine (3MA) reversed QYLGT’s impact on cell viability. While QYLGT did not induce apoptosis, it activated the PI3K/AKT/mTOR pathway. PI3K and mTOR inhibitors were able to revive cell viability and reduce Atg3 expression, suggesting that QYLGT triggers autophagic cellular death in NPC cells by affecting the PI3K/AKT/mTOR pathway (Yang et al., 2021). Euscaphic acid suppressed the expression of PI3K, p-Akt, and p-mTOR in NPC cells. Conversely, the activation of the PI3K/AKT/mTOR pathway by IGF-1 promoted cell proliferation, inhibited apoptosis, and induced cell cycle arrest. In summary, euscaphic acid reduced NPC cell proliferation, induced apoptosis, and prompted cell cycle arrest by suppressing the PI3K/AKT/mTOR signaling pathway (Dai et al., 2019). The combination of curcumin (CUR) and docetaxel (DTX) effectively inhibited the viability and migration of esophageal cancer cells while inducing apoptosis. Together, CUR and DTX activated autophagy, likely by blocking the PI3K/AKT/mTOR pathway. When 3MA, an autophagy inhibitor, was added, it enhanced apoptosis and further suppressed proliferation. These findings suggest that combining CUR and DTX may offer an effectives treatment strategy for esophageal cancer by modulating apoptosis, autophagy, and the PI3K/AKT/mTOR pathway (Table 1) (Deng et al., 2021). Indole-3-carbinol (I3C) impedes the progression of NPC and promotes apoptosis, primarily by regulating the PI3K/AKT signaling pathway and its related protein synthesis. This natural, nontoxic compound has shown considerable promise as both a preventive and therapeutic option for NPC due to its anticancer efficacy. However, further studies are required to thoroughly understand the molecular pathways through which I3C exerts its inhibitory effects on NPC, providing a theoretical basis for its future clinical application (Mao et al., 2014a). Additionally, research underscores the potent anticancer activity of formononetin against human NPC, observed both in vitro and in vivo. It revealed the molecular mechanism involving MAPK signaling activation and PI3K/AKT pathway suppression, leading to apoptosis through the mitochondrial pathway. These findings offer early support for formononetin’s potential as a chemotherapeutic drug and encourage further exploration of novel anticancer agents for treating NPC clinically (Qi et al., 2016). Triptolide markedly reduced the viability of C666-1 cells. The NPC cells showed heightened sensitivity to triptolide, resulting in significant apoptosis, which was mediated through the PI3K/AKT pathway during incubation (Table 1) (Wang et al., 2019). Curcumol effectively suppresses the growth of CNE-2 cells by inducing cell cycle arrest and promoting apoptosis. Curcumol primarily inhibits the IGF-1R/PI3K/AKT/GSK-3β pathway and regulates key molecules involved in cell cycle progression and apoptosis by altering the expression of total p85 and GSK-3β. This suggests that curcumol holds promise in herbal medicine for NPC treatment. However, further research, including animal studies and testing on various NPC cell lines, is necessary to validate its therapeutic potential (Xumei et al., 2018). QYLGT demonstrates anticancer effects in NPC cells by impairing cell survival and colony formation. It also induces vacuole formation and increases the presence of autophagy-related proteins. The autophagy inhibitor 3MA reverses the effects of QYLGT, restoring cell viability and reducing Atg3 expression. This suggests that QYLGT induces autophagic cell death via the PI3K/AKT/mTOR pathway, as described by Yang et al. (2021). Capsaicin suppresses cancer cell proliferation, arresting the cell cycle in the G1 phase. It triggers autophagy through the class III PI3K/Beclin-1/Bcl-2 pathway, as indicated by elevated autophagy markers (LC3-II, Atg5), degradation of p62 and Fap-1, and increased caspase-3 activity, promoting apoptosis. The anticancer effects of capsaicin are closely linked to the inhibition of the PI3K/AKT/mTOR pathway, underscoring its potential as a therapeutic agent for NPC cells (Lin et al., 2017). Arnicolide D exhibited dose- and time-dependent reductions in NPC cell viability. It induced G2/M cell cycle arrest and apoptosis by regulating key proteins, decreasing cyclin D3, cdc2, p-PI3K, p-Akt, p-mTOR, and p-STAT3 levels, while increasing cleaved PARP, cleaved caspase-9, and Bax. Fluctuations in Cyclin B1, cdk6, and Bcl-2 expression were also observed. Arnicolide D effectively controlled cell cycle progression, activated the caspase pathway, and inhibited the PI3K/AKT/mTOR and STAT3 signaling pathways, highlighting its potential for drug development from TCMs (Liu et al., 2019). Ginkgolic acid (GA) was shown to reduce the survival and invasiveness of NPC cells, including 5–8F and CNE2, while promoting apoptosis in a dose-dependent manner. GA altered the expression of Bcl-2, Bax, PARP, caspase-9, and caspase-3, and triggered G0/G1 phase arrest. It also reduced cyclin-dependent kinase six activity and the levels of cyclins D2 and D3. Additionally, GA enhanced apoptosis when combined with 5-FU, suggesting its potential as an adjunct to chemotherapy in NPC treatment (Xiao et al., 2021). 3,3′-Diindolylmethane (DIM) inhibited NPC cell proliferation, induced apoptosis, and suppressed key molecular pathways involved in NPC development and chemotherapy resistance. Notably, DIM demonstrated no adverse effects on normal tissues, supporting its safety profile. Animal studies further confirmed DIM’s tumor-suppressing properties, indicating its potential in preventing tumor initiation and progression (Chen et al., 2013).
The Role of Traditional Drug Therapies for Nasopharyngeal Carcinoma by the Phosphoinositide 3-Kinase (PI3K)/Protein Kinase B (Akt) Signaling Pathway.
List Traditional Medicines for Treatment of Nasopharyngeal Carcinoma by Phosphoinositide 3-Kinase (PI3K)/Protein Kinase B (Akt) Signaling Pathway.
Targeting the PI3K/AKT signaling pathway is a promising therapeutic approach for NPC. This pathway’s crucial role in NPC development and progression makes it an attractive target for drug development. Ongoing research on PI3K/AKT inhibitors offers hope for new, effective treatment options for NPC patients. However, challenges such as drug resistance and patient variability highlight the need for continuous refinement of targeted therapies. As our understanding of NPC’s molecular mechanisms advances, the potential to develop more precise and effective treatments that improve patient outcomes will continue to grow.
Footnotes
Authors’ Contributions
Jitang Cai and Huimin Wang designed the study; Qiong Fang collected and analyzed the data; Qiong Fang drafted the initial manuscript; Junjie Wang and Tingting Wu revised the article critically; Yining Cai reviewed the article; Jitang Cai and Huimin Wang are co-corresponding authors. All authors approved the final manuscript.
Declaration of Conflict of Interests
The authors have stated that there are no potential conflicts of interest related to the research, authorship, or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Special Project for Research on Traditional Chinese Medicine in Henan Province (2023ZYZD08). Based on the TGF-β1/Smads signaling pathway, the mechanism of nasal rehabilitation nasal tamponade therapy in the treatment of chronic rhinosinusitis in children was investigated (2023ZYZD08).
Informed Consent
Not applicable.
