Abstract
To elucidate the effect of tricin in cerebral ischemia/reperfusion (I/R) injury and examine its possible underlying mechanisms. Rats were randomly divided into Sham (exposed the right internal carotid arteries), I/R, and tricin (administered at various doses) groups. After the cerebral I/R injury model was established, a Morris water maze test and a tetrazolium chloride assay were performed. Apoptosis and autophagy were assessed in the nerve cells of hippocampus tissue, and the levels of inflammatory markers within animal serum were detected. Proteins related to apoptosis and the PI3K/Akt pathway were evaluated. To further investigate the mechanisms by which tricin affects brain damage, mouse neuroblastoma cells N2a were divided into control, oxygen-glucose deprivation and reoxygenation (OGD/R), tricin, PI3K/Akt activator, and tricin + PI3K/Akt inhibitor groups. The cell viability, apoptosis, inflammatory factors, and PI3K/Akt pathway related proteins in N2a cells were also detected. The results revealed that I/R-induced learning and memory dysfunction was improved by tricin treatment. The area of cerebral infarction, the levels of apoptosis and autophagy in nerve cells, and the serum inflammatory marker content were all decreased following tricin treatment. Additionally, the expression of Beclin-1 protein was downregulated, while the expression of Bcl-2 protein, p-PI3K/PI3K and p-Akt/Akt was upregulated after tricin treatment. Mechanistically, tricin or PI3K/Akt activator ameliorated OGD/R-induced apoptosis, autophagy, and inflammation. However, these effects were reversed following PI3K/Akt inhibitor treatment in OGD/R-induced N2a cells. In summary, this study suggested that tricin can against I/R-induced brain injury by inhibiting autophagy, apoptosis and inflammation, and activating the PI3K/Akt signaling pathway.
Introduction
Ischemic cerebrovascular disease is the main type of cerebrovascular disease and is associated with numerous disorders.1–3 Cerebral ischemia is associated with high mortality and morbidity rates, and represents a major socioeconomic burden worldwide. 4 Thus, the development of effective therapies for patients with cerebral ischemic injury is urgently required.
Flavonoids have been shown to have a variety of biological effects and tricin (3’,5’-dimethoxyflavone) is the active component of Njavara rice bran. Previous studies have demonstrated that tricin not only exhibits significant anti-inflammatory, anti-atherogenic, and anticancer biological activities, but also exerts anti-allergic effects.5,6 Moreover, evidence has revealed that tricin exerts protective effects on the brain, alleviating cerebral ischemia/reperfusion (I/R) injury and attenuating blood-brain barrier (BBB) breakdown. 7 In this study, middle cerebral artery occlusion (MCAO) was used to establish a rat model of cerebral ischemic injury in order to determine whether the tricin can improve the learning and memory of model rats. The possible mechanisms underlying these effects were also investigated.
Apoptosis is the main form of cell death in the pathogenesis of cerebral ischemia. 8 Autophagy is a lysosomal degradation pathway, which serves an important role in the removal of protein aggregates and the maintenance of cell homeostasis by damaged or excessive organelles. 9 A previous study demonstrated that autophagy and apoptosis usually occur in different types of brain injury. 10 Furthermore, Miao et al. 11 revealed that inflammation served a major role in the mediation of cerebral ischemia-induced brain injury. The PI3K/Akt pathway is one of the most frequently variable signaling networks in humans, serving a central role in cell proliferation, metabolism, survival and motility. 12 The PI3K/Akt pathway mediates autophagy and participates in a variety of important cellular activities in the body, including cell metabolism, apoptosis inhibition and inflammation. 13 Therefore, the purpose of the current study was to investigate whether treatment with tricin effectively improved ischemia injury-induced learning and memory dysfunction in rats, and to explore whether the mechanism underlying its action was related to the regulation of autophagy, apoptosis and inflammation mediated by the PI3K/Akt pathway.
Materials and methods
Experimental animal and ethics statements
60 Male Sprague Dawley (SD) rats (weighing 250 ± 30g and aged 8-10 weeks) were kept in a clean environment with 55 ± 5% humidity and 23 ± 2°C. The current study was carried out in accordance with the guidelines of the National Institutes of Health (NIH Pub. No. 85–23, revised 1996) and approved by the Animal Protection and Use Committee of Yantai Affiliated Hospital of Binzhou Medical University (No: 20200101019).
Model construction
Rats were divided into sham, I/R, and tricin [administered at different dosages and named tricin-low (TRI-L), tricin-medium (TRI-M) and tricin-high (TRI-H)], 12 rats per group. The rat model of cerebral I/R injury was established according to a previously described method. 14 In short, a 2 cm longitudinal incision was made to expose right internal carotid arteries and external carotid artery. Then, a nylon filament was inserted into the internal carotid artery from the external carotid artery to block the middle cerebral artery for 2 h. After 2 h of ischemia, the rats were re-perfused with nylon filaments removed. The right internal carotid arteries of sham group rats were exposed; however, embolization and artery occlusion were not subsequently performed. The I/R model was established via MCAO for 2 h. Tricin (>98% purity; SynInnova Laboratories Inc., Edmonton, AB, Canada) was dissolved in DMSO and added in water (<0.5% v/v DMSO) for experiments. 15 Rats of the TRI-L (75 mg/kg), TRI-M (100 mg/kg) and TRI-H (150 mg/kg) groups were received tricin by oral administration 2 h, 4 h and 6 h after reperfusion.16,17 The sham and I/R groups received the same volume of DMSO solution (<0.5% v/v DMSO).
Morris water maze test
At 24 h after cerebral I/R induction, a Morris water maze test was conducted. The apparatus consisted of a circular water tank with a water temperature of 24 ± 2°C. The escape platform, which was used for place-learning acquisition, was located in the middle of the southeast quadrant, 2 cm below the water surface. Rats were randomly placed in one of the four starting positions and left to swim freely in order to locate the platform. Rats were required to remain on the platform for at least 15 sec. If the rats were unable to locate the platform within 90 sec, they were guided to the platform and placed there for 15 sec. Training was conducted for five consecutive days, after which time the experimental results were recorded on day 6.
Tetrazolium chloride (TTC) staining
After Morris water maze test evaluation, the rats were anesthetized with intraperitoneal injection of sodium pentobarbital (40 mg/kg). Then, animals were euthanized via decapitation and placed into an ice bath ready for brain dissection. Five coronal sections with a thickness of 2 mm were cut from the anterior to the posterior side of the brain. After staining with TTC solution at 37°C for 15 min in the dark, PBS solution containing 4% paraformaldehyde was used to fix and preserve tissues and the infarct area was calculated.
Immunofluorescence staining
LC3, also known as LC3B-I, is a microtubule-associated protein that localizes to and accumulates in autophagosomes. 18 The hippocampi were cut into 20-μm coronal sections, and incubated overnight with LC3 primary antibody (Abcam, UK) at 4°C. After incubation with goat anti-rabbit IgG antibody (Cell Signaling Technology, USA) at room temperature for 2 h, nuclei were counterstained with 4′,6-diamidino-2-phenylindole and observed under a fluorescence microscope (Leica, Wetzlar, Germany).
Transmission electron microscopy
Hippocampal tissue was fixed with 2.5% glutaraldehyde solution, rinsed with PBS and further fixed with 1% osmium acid. After dehydration with gradient acetone and embedding in resin, hippocampal tissue was sliced into 1-μm sections. The sections were then stained with uranyl acetate and lead citrate. Electron microscopy (JEM-1400/JEOL, Nippon Electronics Corporation, Japan) was used to assess the formation of vacuoles.
TUNEL assay
Hippocampal tissue was fixed in 4% paraformaldehyde, embedded in paraffin and coronally sliced into 4-μm sections. After conventional xylene dewaxing and gradient ethanol dehydration, a TUNEL assay (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) was performed. Results were assessed under an optical microscope (BX50; Olympus Corporation, Japan) and the number of apoptotic cells was quantified.
Cell culture and modeling
The mouse neuroblastoma cells N2a (ATCC, USA) were cultured in DMEM (HyClone, USA) containing 10% FBS with 1% penicillin/streptomycin in incubator (5% CO2, 37°C).
The model of in vitro I/R was established by oxygen-glucose deprivation and reoxygenation (OGD/R). 19 N2a cells were cultured in glucose-free DMEM and placed in an anaerobic chamber containing 95% N2 and 5% CO2 at 37°C. After 3 h, N2a cells were returned to normal medium and incubated under normal conditions to terminate the OGD and start reperfusion. Cells were divided into control (N2a cells were cultured normally), OGD/R, tricin (TRI, OGD/R-induced N2a cells were cultured with 50 μM tricin 16 ), PI3K/Akt activator insulin-like growth factor 1 (IGF-I, N2a cells were cultured with IGF-I for 30 min before they were subjected to OGD 20 ), and tricin + PI3K/Akt inhibitor LY294002 (TRI + LY, N2a cells were cultured with LY294002 for 30 min before they were subjected to OGD and then cultured with tricin 20 ).
MTT assay
Cells were plated into 96-well plates at a density of 1×105 cells per 100 μL for 24 h. After treated accordingly and incubating for 24 h, cells were cultured with MTT solution (Sigma-Aldrich, USA). 4 h later, after removed the medium, 100 mL of DMSO was added and the absorbance was measured by a microplate reader (Molecular Devices, California, USA) at 490 nm. 21
Flow cytometry analysis
1 x binding buffer (Beyotime Biotech, Shanghai, China) and 5 μL of FITC-labeled annexin-V (Beyotime Biotech, Shanghai, China) were added to the cells in different group. After incubation with 5 μL of propidium iodide (PI) at room temperature, 200 μL one x binding buffer was added. The apoptosis was analyzed by flow cytometer (Beckman Coulter, Brea, CA, USA).
ELISA
After homogenized in RIPA Lysis buffer (Solarbio Science& Technology Co) and centrifugated at 4°C, the serum of rats or supernatant of N2a cells was collected, and quantitatively assayed for the levels of TNF-α, IL-6 and IL-1β were measured according to the instructions of ELISA kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China).
Western blotting
The tissue or cells homogenates were prepared as previously described method. 22 A bicinchoninic acid (BCA) protein assay kit (Solarbio Science and Technology, Co, Ltd, Beijing, China) was used to determine the protein concentration in hippocampal tissues or N2a cells. Then, the proteins were separated and transferred. After blocking with 5% fat-free milk, the following primary antibodies were applied: LC3B (Abcam, UK), Beclin-1 (MilliporeSigma, USA), Bcl-2 (Abcam, UK), PI3K (Biorbyt Ltd, Cambridge, UK), phosphorylated (p)-PI3K (Biorbyt Ltd, Cambridge, UK), Akt (Biorbyt Ltd, Cambridge, UK), p-Akt (Biorbyt Ltd, Cambridge, UK) and β-actin (Abcam, UK) was used. After incubation with horseradish peroxidase-labeled goat anti-rabbit IgG (Cell Signaling Technology, Inc.), the membranes were visualized with an enhanced chemiluminescence system (Amersham; Cytiva, USA) and the gray scale was calculated using Image J software (National Institutes of Health).
Statistical analysis
The data were presented as means and standard deviations (mean ± SD) and the results were analyzed by SPSS 21.0 software. Differences were considered statistically significant at p < 0.05. One-way ANOVA followed by LSD analysis was performed for pairwise comparisons.
Results
Effect of tricin on the cognition and cerebral infarction area in rats with cerebral I/R injury
As presented in Figures 1(a) to (c), the escape latency of the I/R group was prolonged, and the times of crossing the platform was reduced when compared with the sham group (p < 0.05), indicating that the cognitive function of the rats in the I/R group was decreased. However, compared with the I/R group, the escape latency in the tricin groups was significantly reduced, and the times of crossing the platform was significantly increased (p < 0.05). Additionally, white infarcts were clearly visible in I/R rats, and the ratio of infarct to non-infarcted area was markedly decreased in the tricin groups than in I/R group (p < 0.05; Figures 1(d) and (e)). Meanwhile, the TRI-H group achieved the best result than TRI-L and TRI-M groups (p < 0.05; Figure 1). The results indicated that tricin ameliorated cognitive function and reduced brain damage in cerebral I/R rats. Effect of tricin on the cognition and cerebral infarction area in rats with cerebral I/R injury. (a) Navigational path map of rats in each group on the fourth day of the water maze experiment. (b) The time required to locate the hidden platform. (c) Number of times across the platform. (d) Tetrazolium chloride staining was performed on rat brain tissue. (e) Percentage of infarct area in the brains of rats. **p < 0.01 against Sham; #p< 0.05, ##p< 0.01 against I/R; ∧p < 0.05, ∧∧p < 0.01 against TRI-L; $p< 0.05 against TRI-M. I/R, ischemia-reperfusion; TRI-L, tricin-low; TRI-M, tricin-medium; TRI-H, tricin-high.
Effect of tricin on the autophagy flow and formation of mitochondrial vacuoles in rats with cerebral I/R injury
As shown in Figure 2(a), compared with the sham group, the autophagy flow was significantly increased after I/R injury (p < 0.05). However, when compared with the I/R group, the autophagy flow was significantly reduced in the tricin groups (p < 0.05). Additionally, the structure of mitochondria in the sham group was obvious, while the mitochondria in the I/R group were vacuolated (Figure 2(b)). Whereas, the degree of mitochondrial vacuole gradually weakened in the tricin groups than in I/R group (Figure 2(b)). The results suggested that tricin reduced autophagy in a dose-dependent manner in cerebral I/R rats. Effect of tricin on the autophagy flow (A, 400×) and formation of mitochondrial vacuoles (B, 200×) in rats with cerebral I/R injury. **p < 0.01 against Sham; ##p< 0.01 against I/R; ∧∧p< 0.01 against TRI-L; $$p < 0.01 against TRI-M. I/R, ischemia-reperfusion; TRI-L, tricin-low; TRI-M, tricin-medium; TRI-H, tricin-high.
Effect of tricin on the apoptosis of neurons and expression of apoptosis-related proteins in hippocampal tissue
The number of apoptotic neurons in the I/R group was markedly increased compared with that in sham group (p < 0.05; Figure 3(a)). Additionally, when compared with the I/R group, the number of apoptotic neurons in the tricin groups were markedly decreased (p < 0.05; Figure 3(a)). Meanwhile, compared with TRI-L and TRI-M groups, the TRI-H group produced the lowest result (p < 0.05; Figure 3(a)). The results demonstrated that tricin can inhibit neuronal apoptosis in cerebral I/R rats. Compared with the sham group, the protein expression of Beclin-1 was significantly upregulated in the I/R group, while the expression of Bcl-2 was significantly downregulated (p < 0.05; Figure 3(b)). Furthermore, the protein expression of Beclin-1 was downregulated and the protein expression of Bcl-2 was upregulated after tricin administration. All the results suggested that tricin inhibited apoptosis in the hippocampal tissue of cerebral I/R rats. Effect of tricin on the apoptosis of neurons and apoptosis-related proteins in rats with cerebral I/R injury. (a) TUNEL assay (400×). (b) Western blotting. **p < 0.01 against Sham; ##p < 0.01 against I/R; ∧∧p < 0.01 against TRI-L; $$p < 0.01 against TRI-M. I/R, ischemia-reperfusion; TRI-L, tricin-low; TRI-M, tricin-medium; TRI-H, tricin-high.
Effect of tricin on serum inflammatory factors and ratio of p-PI3K/PI3K and p-Akt/Akt in rats with cerebral I/R injury
Figure 4 shown that compared with the sham group, the levels of TNF-α, IL-6 and IL-1β were markedly increased in the I/R group, while the ratio of p-PI3K/PI3K and p-Akt/Akt were markedly decreased (p < 0.05). However, the TNF-α, IL-6 and IL-1β levels were markedly decreased and the ratio of p-PI3K/PI3K and p-Akt/Akt were markedly increased in the tricin groups than in the I/R group, and the TRI-H group produced the best result than TRI-L and TRI-M groups (p < 0.05). The results indicated that tricin treatment reduced the level of cerebral I/R injury-induced inflammatory factors and activated the AKT signaling pathway. Effect of tricin on the serum levels of inflammatory factors and expression of PI3K/Akt signaling pathway-related proteins in rats with cerebral I/R injury. (a) ELISA. (b) Western blotting. **p < 0.01 against Sham; ##p < 0.01 against I/R; ∧∧p < 0.01 against TRI-L; $p < 0.05, $$p < 0.01 against TRI-M. I/R, ischemia-reperfusion; TRI-L, tricin-low; TRI-M, tricin-medium; TRI-H, tricin-high.
Effect of tricin on the cell viability and apoptosis of N2a cells
OGD/R was found to cause a dramatical decrease of cell viability and a significant increase of apoptosis in N2a cells (p < 0 .05; Figure 5). However, compared with OGD/R group, the cell viability was significantly higher and the apoptosis was significantly lower in tricin or IGF-I treatment group (p < 0.05; Figure 5). Whereas, the effect of tricin was reversed by LY294002 treatment (p < 0.05; Figure 5). These findings demonstrated that tricin promotes cell viability and inhibits apoptosis of N2a cells by activating the PI3K/Akt pathway. Effects of tricin on the cell viability and apoptosis of N2a cells. (A) MTT assay. (B) Flow cytometry analysis. **p < 0.01 against Control; ##p < 0.01 against OGD/R; ∧p < 0.05, ∧∧p < 0.01 against TRI; $$p < 0.01 against IGF-I. OGD/R, oxygen-glucose deprivation and reoxygenation; TRI, tricin; IGF-I, insulin-like growth factor 1.
Tricin inhibits nerve cell autophagy, apoptosis and inflammation in I/R-induced brain injury by activating the PI3K/Akt signaling pathway
Compared with the control group, the levels of inflammatory marker and expression of apoptosis and autophagy related proteins were markedly increased in the OGD/R group (p < 0.05; Figure 6). However, the levels of TNF-α, IL-6 and IL-1β were all decreased following tricin or IGF-I treatment (p < 0.05; Figure 6(a)). Additionally, the expression of LC3II/I and Beclin-1 pr oteins were downregulated, while the expression of Bcl-2 protein, p-PI3K/PI3K and p-Akt/Akt was upregulated after tricin or IGF-I treatment (p < 0.05; Figure 6(b)). Moreover, these effects were reversed following PI3K/Akt inhibitor treatment (p < 0.05; Figure 6). The results revealed that the neuroprotective effect of tricin was associated with PI3K/Akt pathway activation. Tricin inhibits nerve cell autophagy, apoptosis and inflammation in I/R-induced brain injury by activating the PI3K/Akt signaling pathway. (a) ELISA. (b) Western blotting. *p < 0.05, **p < 0.01 against Control; #p < 0.05, ##p < 0.01 against OGD/R; ∧p < 0.05, ∧∧p < 0.01 against TRI; $p < 0.05, $$p < 0.01 against IGF-I. OGD/R, oxygen-glucose deprivation and reoxygenation; TRI, tricin; IGF-I, insulin-like growth factor 1.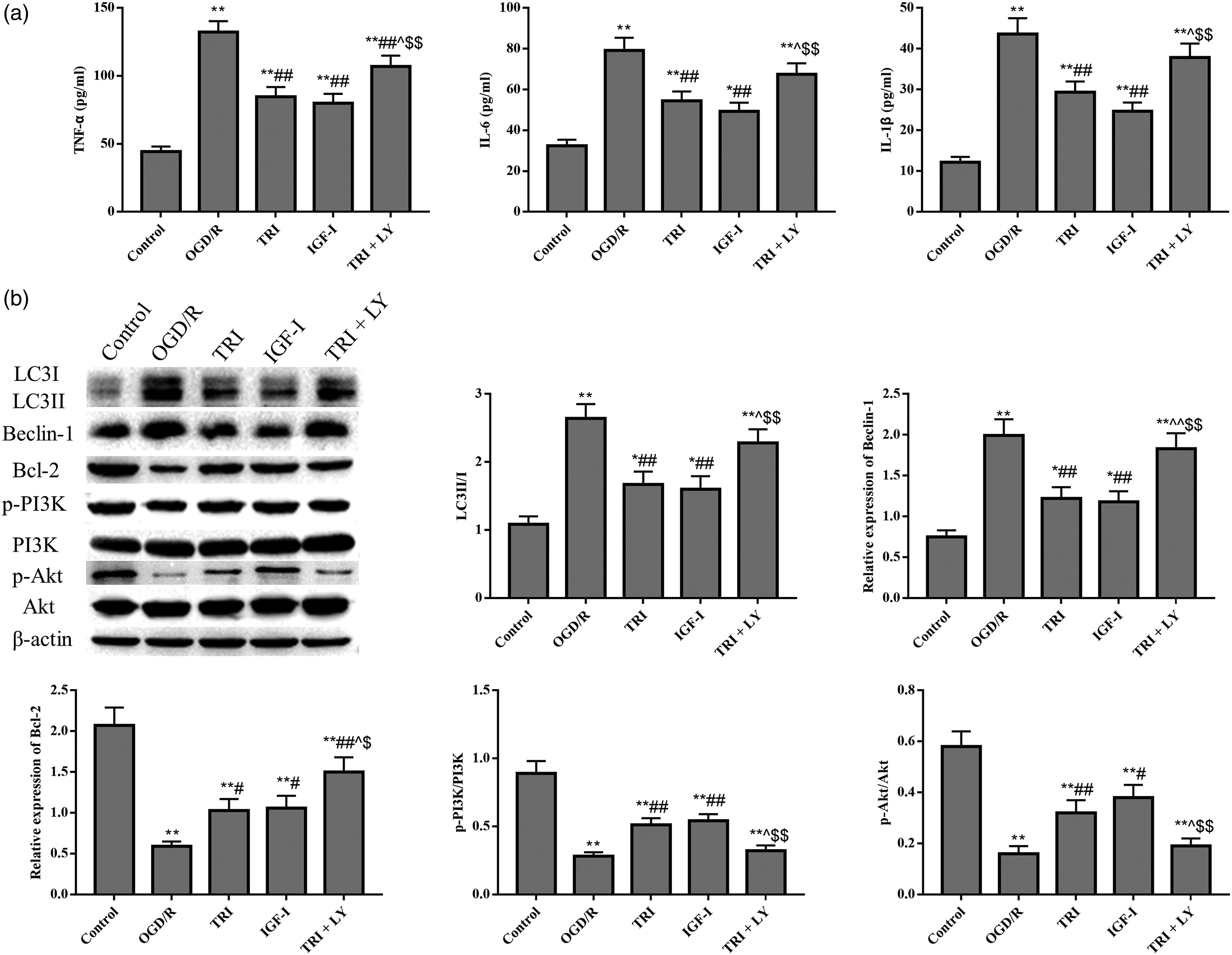
Discussion
As a major source of therapeutic drugs, plant extracts are of significant medical importance for the design of future treatments1.3,23 Tricin is a flavone isolated from rice bran and has important anti-inflammatory, anti-atherogenic, anticancer and anti-allergic properties.5–7 Although tricin has been shown to have a protective effect on the brain, the role and mechanism of tricin in I/R have not been investigated. 7 Cerebral ischemic injury is associated with high mortality and morbidity rates, which are increasing annually worldwide, and the prognosis of patients with cerebral ischemia remains unsatisfactory. 4 The results of the current study demonstrated that tricin treatment exerted neuroprotective effects against inflammation, autophagy and apoptosis by targeting the PI3K/Akt pathway in rats with MCAO-induced and in N2a cells with OGD/R-induced cerebral I/R injury.
Cell survival and death are complex processes involving apoptotic and autophagic pathways. 24 Brain injury results in neuronal apoptosis, autophagic cell death and necroptosis. Previous works identified that inflammation, apoptosis and autophagy of the central nervous system are closely associated with neurological damage, learning and memory decline, cognitive impairment and other pathological conditions.25–27 As we all know, the transition of LC3B-I to LC3B-II contributes to the formation of autophagosomes and Beclin-1 participates in the regulation of neuronal autophagy. 25 The current study revealed that the expression of Bcl-2 was significantly increased, while TNF-α, IL-6 and IL-1β, LC3II/I, Beclin-1, and apoptosis levels were significantly decreased after tricin treatment.
The PI3K/Akt signaling pathway is one of the most important signal transduction pathways and is involved in cell processes via regulating cell metabolism, apoptosis and inflammation through a series of molecules. 28 Akhtar A and Sah SP found that PI3K/Akt signaling pathway is involved in neurodegeneration and Alzheimer’s disease. 29 PI3K is widely found in various cells, and a number of cytokines and stimulating factors can activate PI3K. Akt is an important downstream molecule of PI3K and serves as an important hub of various signaling pathways. To assess the molecular mechanisms by which tricin protects against cerebral I/R injury, the protein expression of PI3K, Akt and their phosphorylated forms in rats and N2a cells were detected by western blotting. The results revealed that, after tricin treatment, the expression of p-PI3K and p-AKT were increased, which indicated that tricin could activate the PI3K/Akt pathway. PI3K/Akt pathway phosphorylation protects cells via activation of the Bcl-2 family and inhibition of the caspase family. In this study, the levels of inflammatory marker and expression of apoptosis and autophagy related proteins were greatly enhanced after OGD/R treatment and tricin or IGF-I treatment led to a dramatic decrease of these factors, which was relieved by LY294002.
In conclusion, the present study revealed that tricin exerted protective effects against cerebral I/R injury by inhibiting inflammation, autophagy and apoptosis. The mechanism underlying these neuroprotective effects may be associated with PI3K/Akt signaling pathway activation. Therefore, tricin may represent a potential therapeutic approach to cerebral I/R injury, which may provide novel insight into the clinical treatment of this condition. In future studies, an in-depth study of relationship among autophagy, apoptosis, inflammation and PI3K pathways and indicate where tricin has direct effects still needs perform to clarify the cause and effect that contribute to the tricin-mediated protection.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
