Abstract
Background
Kidney cancer, also called renal cancer, is a kind of malignancy that develops in the kidneys. Kidney cancer is a major health issue that has garnered increasing attention in the medical community due to its growing prevalence and complex challenges associated with its treatment.
Objectives
The current study evaluated the growth-inhibitory and apoptotic-inducing potentials of xanthohumol against renal cancer A498 cells.
Materials and Methods
The cytotoxicity of xanthohumol at varying dosages against kidney cancer A498 cells and non-malignant Vero cells was studied using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The accumulation of endogenous reactive oxygen species (ROS) and apoptotic cell death were assessed utilizing their respective fluorescent staining assays. Oxidative stress biomarkers, pro- and anti-apoptotic proteins, cyclin D1, and proteins associated with the PI3K/AKT/mTOR signaling were assessed in the untreated and treated A498 cells using appropriate assay kits.
Results
The present results have exhibited that the growth of A498 cells was markedly diminished following treatment with xanthohumol, whereas Vero cell growth remained unaffected by the same concentrations of xanthohumol. The xanthohumol treatment markedly increased ROS generation and promoted apoptosis in A498 cells, which is evidenced by the fluorescent staining results. The xanthohumol treatment increased thiobarbituric acid reactive substances (TBARS) levels and decreased antioxidant levels in A498 cells. Moreover, xanthohumol elevated the pro-apoptotic proteins while diminishing the levels of Bcl-2, cyclin D1, and PI3K/AKT/mTOR proteins in the kidney cancer A498 cells.
Conclusion
The findings of this work highlight that xanthohumol considerably inhibits cell proliferation and activates apoptotic mechanisms in kidney cancer A498 cells. Consequently, it possesses effectiveness as an anti-cancer candidate for the treatment of kidney cancer.
Introduction
Renal cancer is a major health issue that has garnered increasing attention in the medical community due to its growing prevalence and complex challenges associated with its management (Wilson et al., 2024). Renal cancer arises in the kidneys, small, bean-shaped organs situated in the posterior abdomen, tasked with filtering waste and surplus water from the bloodstream. Renal cancer can take several forms, with the most general type being clear cell renal cell carcinoma (ccRCC), which is the foremost subtype, accounting for 80% of all renal cancer, and is responsible for high mortality rates. The development of renal cancer is often linked to various risk factors, including smoking, obesity, and exposure to certain chemicals and toxins (Vrieling et al., 2024). The complications associated with renal cancer can be diverse and challenging to manage. One of the primary concerns is the potential for metastasis, where the cancer spreads to other organs, significantly reducing the chances of successful treatment. Additionally, the refractoriness of ccRCC, a major subtype, to conventional chemotherapy and radiotherapy poses a major problem in the clinical management of the disease (Koguchi et al., 2023). The global prevalence of renal cancer has been on a concerning rise in recent decades. The World Health Organization reports that the annual mortality rate for kidney cancer is 140,000–170,000 individuals globally. This concerning trend extends beyond high-income nations, as the prevalence of chronic kidney disease, a major risk factor for renal cancer, is disproportionately elevated in low- and middle-income nations, where healthcare access and preventative medicines are often restricted (Bukavina et al., 2022).
Renal cell carcinoma (RCC) is a prevalent tumor of the urinary system, following bladder and prostate cancer in frequency, with its prevalence consistently rising annually. Furthermore, it is highlighted that roughly 30% of RCC patients have distant metastases during early detection, and an additional 40% of patients develop distant metastases after undergoing surgical intervention (Chawla et al., 2023). The onset of renal cancer is complex and multifaceted, implicating a combination of genetic, epigenetic, and environmental factors. Genetic alterations, like mutations in the Von Hippel–Lindau gene, have been strongly implicated in the development of ccRCC, the most common and aggressive subtype of the disease. Additionally, it has been highlighted that the deregulation of various pathways, including the PI3K/Akt/mTOR and hypoxia-inducible factor pathways, play a crucial role in the growth, survival, and metastasis of tumor cells (Ballesteros et al., 2021).
Renal cancer is a complex and challenging disease that has seen significant advancements in treatment options in recent years. However, the long-term prognosis for many patients remains poor, with over 30% of those who undergo nephrectomy (surgical removal of the kidney) developing metastatic disease. One of the significant challenges in treating renal cancer is its resistance to traditional chemotherapy and radiation therapy (Mikami et al., 2020). The current standard of care for localized renal cancer is surgical removal of the primary tumor, but this approach has limited efficacy in cases of metastatic disease. While these advancements have been encouraging, the long-term prognosis for many renal cancer patients remains poor, highlighting the need for additional studies to develop new therapies. One promising avenue of investigation is the use of plant-derived bioactive compounds, which have been shown to possess anti-tumor properties with relatively low toxicity (Bajalia et al., 2022). Xanthohumol is a prominent prenylated flavonoid compound found in the hops plant (Humulus lupulus) and is recognized for its extensive range of biological actions, including anti-arthritic (Wang, Chen, et al., 2023; Wang, Deng, et al., 2023), hepatoprotective (Fernández-García et al., 2019), anti-oxidative (Yao et al., 2015), and neuroprotective (Huang et al., 2018) properties. Furthermore, xanthohumol effectively inhibited the viability and promoted apoptosis in pancreatic cancer cells (Saito et al., 2018), nasopharyngeal cancer (Hsieh et al., 2022), breast cancer (Gupta et al., 2022), glioblastoma (Ho et al., 2020), and oral cancer (Li et al., 2020) cells. However, the apoptotic-inducing potentials of xanthohumol on renal cancer cells have not been studied yet. Hence, the current study evaluated the growth-inhibitory and apoptotic-inducing potentials of xanthohumol against renal cancer A498 cells.
Materials and Methods
Chemicals
Xanthohumol, Dulbecco’s modified Eagle medium (DMEM), antibiotics, dimethyl sulfoxide (DMSO), and additional chemicals were acquired from Sigma–Aldrich, USA. The diagnostic kits for quantifying the biochemical markers were acquired from Abcam and Elabscience, USA, respectively.
Cell Culture
A498 human kidney carcinoma cells were acquired from ATCC, USA, and cultivated in DMEM with 10% FBS and 1% anti-mycotic mixture in a 5% CO2 incubator. The mature cells were harvested upon reaching 80% confluency and utilized for additional experiments.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium Bromide (MTT) Assay
The xanthohumol’s effects on the viability of A498 and Vero cells were studied utilizing the MTT assay. Both cells were individually cultivated on a 96-well plate and were subsequently treated with xanthohumol for 24 h at varying dosages of 1, 2.5, 5, 7.5, 10, 12.5, and 15 µM/mL. Subsequent to the treatment, 20 µL of MTT reagent was combined with 100 µL of DMEM in the wells and incubated for 4 h. After dissolving the formazan deposits with DMSO (100 µL), the absorbance was then taken at 570 nm.
Propidium Iodide (PI) Staining
The apoptotic occurrences in the untreated and xanthohumol-treated A498 cells were assessed using PI staining. The cells were cultivated in a six-well plate and subsequently treated with 7.5 µM/mL of xanthohumol and/or 2 µg/mL of doxorubicin (DOX) for 24 h. Later, a 5 µL solution of PI dye was added and maintained for 20 min in a dark place. The resultant fluorescence was further examined utilizing a fluorescence microscope.
DCFH-DA Staining
The reactive oxygen species (ROS) production in untreated and xanthohumol-treated A498 cells was analyzed by 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) staining. The A498 cells were cultured in a six-well plate and subsequently administered with xanthohumol at a 7.5 µM/mL concentration and/or 2 µg/mL of DOX for 24 h. Afterward, DCFH-DA (10 µL) dye was mixed to the wells and incubated for 10 min. The fluorescence microscope was employed to study the intensity of the developed fluorescence, which correlates directly with the amount of ROS.
Analysis of Oxidative Stress Biomarkers
The cell lysate was prepared by lysing untreated and xanthohumol-treated A498 cells using a lysis buffer, and the lysate was centrifuged and utilized to evaluate the oxidative stress biomarkers. The glutathione (GSH), superoxide dismutase (SOD), and thiobarbituric acid reactive substances (TBARS) concentrations were evaluated in the untreated and xanthohumol-treated A498 cells. The tests were conducted using kits (Elabscience, USA), following the methods described by the kit’s manufacturer.
Apoptotic Protein Marker Levels
The concentrations of apoptotic proteins such as Bax, Bcl-2, caspase-3, -9, and cyclin D1 were analyzed in the cell lysate of the untreated and xanthohumol-treated A498 cells. The assays were conducted utilizing assay kits (Elabscience, USA) as per the recommended instructions of the manufacturer.
Analysis of PI3K/AKT/mTOR Proteins
The protein concentrations of PI3K, AKT, and mTOR were evaluated in untreated and xanthohumol-exposed A498 cells utilizing commercial kits. The assays were conducted according to the specifications provided by the manufacturer (Abcam, USA).
Statistical Analysis
The statistical tests were done using Prism software, and results are portrayed as a Mean ± Standard Deviation (SD) of triplicate assays. The values are studied utilizing one-way analysis of variance (ANOVA) and Duncan’s multiple range test (DMRT), with p < .05 as significant.
Results
Effect of Xanthohumol on the Viability of A498 and Non-malignant Vero Cells
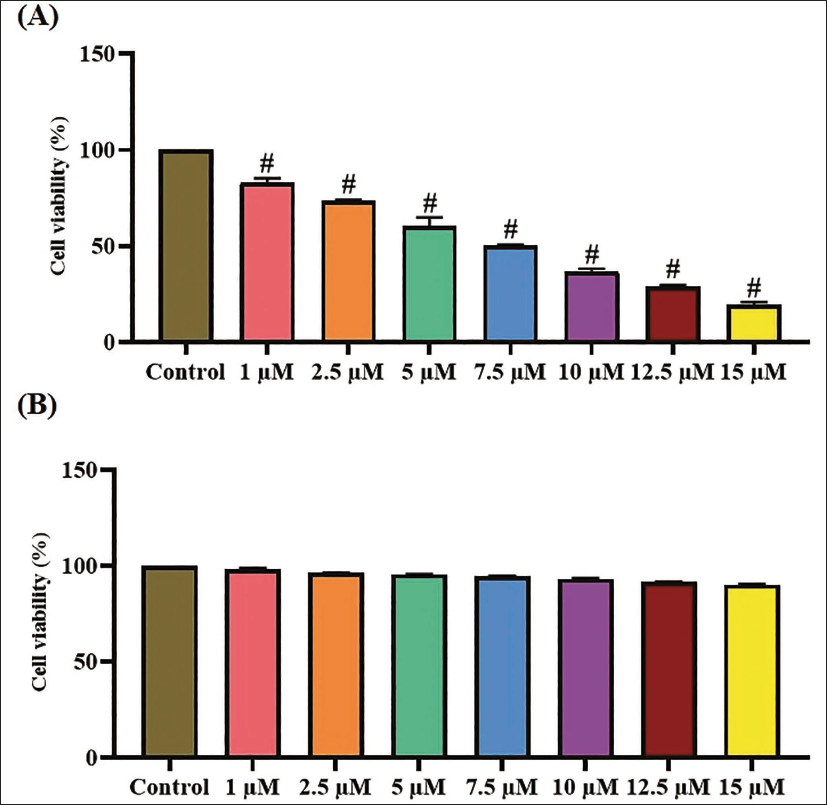
Figure 1 illustrates the results of the MTT assay, demonstrating the result of xanthohumol on the growth of both A498 and non-malignant Vero cells. The treatment with xanthohumol at varying concentrations (1–15 µM/mL) markedly diminished the viability of A498 cells. Whereas, the xanthohumol treatment at the same concentrations did not disturb the growth of Vero cells. The xanthohumol at increasing concentrations exhibited a drastic diminution in the A498 cell growth (Figure 1). The half maximal inhibitory concentration (IC50) concentration of xanthohumol was determined to be 7.5 µM/mL against A498 cells, and this dosage was selected for following investigations.
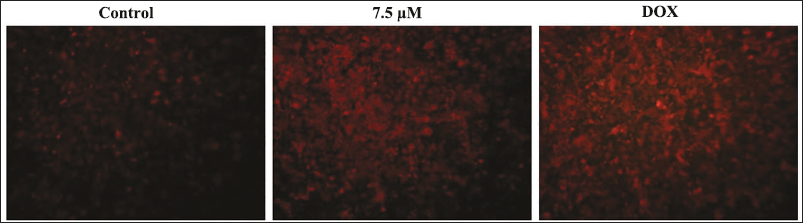
Effect of Xanthohumol on the Apoptosis in A498 Cells
The extent of apoptosis in untreated and xanthohumol-treated A498 cells was assessed via PI staining, with results presented in Figure 2. The A498 cells subjected to xanthohumol at a 7.5 µM/mL dosage exhibited intense red fluorescence, which evidences more apoptotic incidence when compared with the control. A concentration of 7.5 µM/mL xanthohumol markedly increased apoptosis and led to a reduction in cell counts in A498 cells, as corroborated by the outcomes of the DOX treatment.
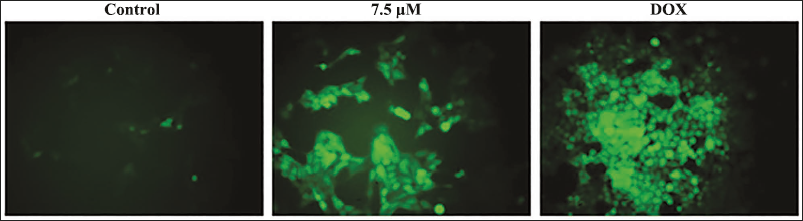
Effect of Xanthohumol on the ROS Production in the A498 Cells
Figure 3 depicts the effect of xanthohumol on endogenous ROS generation in A498 cells. The A498 cells exposed to 7.5 µM/mL of xanthohumol exhibited increased green fluorescence than the control. The intensified green fluorescence signifies the increased endogenous buildup of ROS in A498 cells exposed to xanthohumol. These findings are also validated by the results of the DOX treatment.
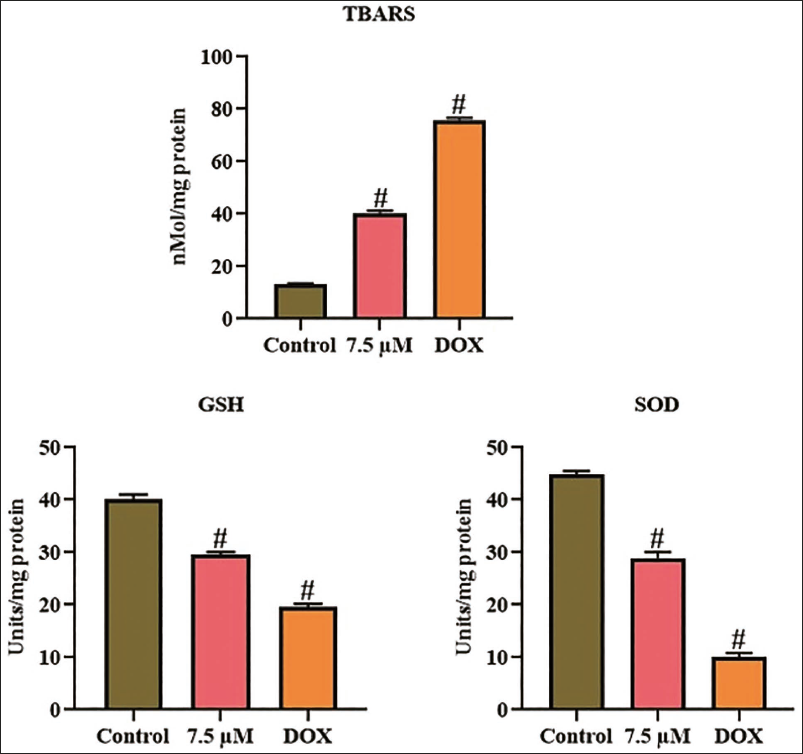
Effect of Xanthohumol on the Oxidative Stress in the A498 Cells
Figure 4 illustrates the oxidative stress biomarkers in untreated and xanthohumol-treated A498 cells. The control cells had decreased TBARS and elevated SOD and GSH concentrations. Whereas, the treatment with 7.5 µM/mL of xanthohumol demonstrated a drastic elevation in the TBARS level and subsequent diminution in SOD and GSH concentrations compared with the control. These findings indicate that xanthohumol induces oxidative stress, hence promoting apoptosis in A498 cells.
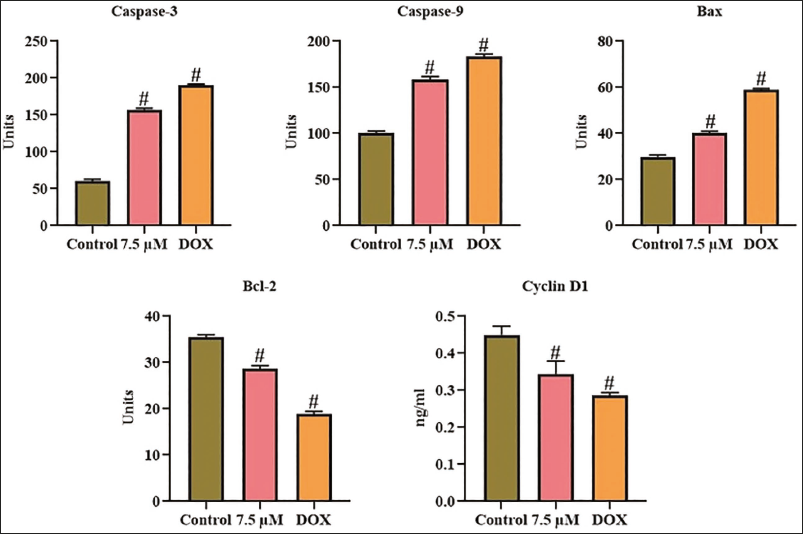
Effect of Xanthohumol on Apoptotic Protein Levels in A498 Cells
The concentrations of apoptotic proteins in untreated and xanthohumol-exposed A498 cells were analyzed, with results depicted in Figure 5. In control cells, the concentrations of Bax and caspase-3/-9 were diminished, while the concentrations of Bcl-2 and cyclin D1 were elevated. However, the administration of 7.5 µM of xanthohumol exhibited a remarkable elevation in Bax, caspase-3, and -9 concentrations, while reducing the concentrations of Bcl-2 and cyclin D1. In addition, the findings of DOX treatment corroborated these results, which confirms that xanthohumol treatment successfully promotes apoptosis in A498 cells.
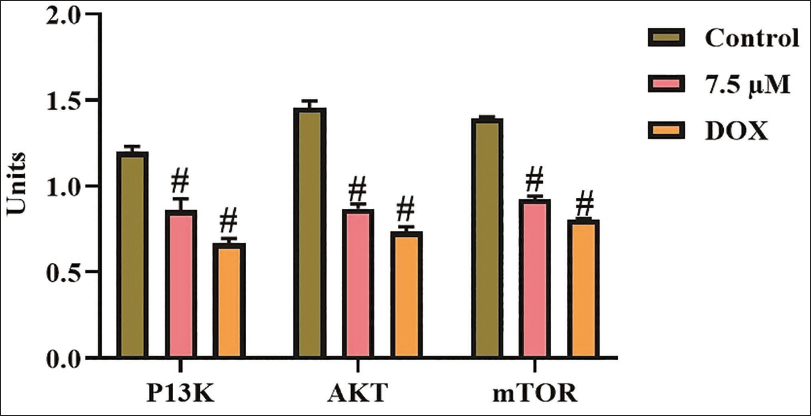
Effect of Xanthohumol on PI3K/AKT/mTOR Levels in A498 Cells
The PI3K, AKT, and mTOR concentrations were examined in untreated and xanthohumol-treated A498 cells, and the findings are presented in Figure 6. In control cells, the PI3K, AKT, and mTOR concentrations were elevated. However, the 7.5 µM/mL of xanthohumol treatment revealed considerable diminution in PI3K, AKT, and mTOR concentrations compared with the control. Furthermore, the outcomes of the DOX treatment supported these findings, as it similarly diminished the PI3K/AKT/mTOR concentrations in the A498 cells. Consequently, it was evident that xanthohumol may inhibit cell growth in A498 cells via downregulating the PI3K/AKT/mTOR pathway.
Discussion
Kidney cancer is a type of malignancy that originates in the kidneys. Kidney cancer is a major type of cancer, with a rising global incidence in recent years. RCC is the most predominant type of kidney cancer, which is responsible for 80% of all kidney cancer. This particular subtype is characterized by its resistance to traditional therapies like chemotherapy and radiotherapy, making surgical intervention the primary treatment option. Efforts to develop more effective diagnostic and treatment modalities are crucial for improving patient outcomes and reducing the mortality associated with this disease (Liu et al., 2023). Apoptosis, a highly regulated form of programmed cell death, has emerged as a crucial aspect of cancer therapy. This process of controlled self-destruction plays a fundamental role in maintaining the equilibrium between cell proliferation and cell death, which is critical for the homeostasis of multicellular organisms. Disruptions in the apoptotic machinery can result in the escape from cell death, a phenomenon of tumor, and the subsequent uncontrolled proliferation of malignant cells. In the case of renal cancer, the dysregulation of apoptosis is a crucial factor contributing to the resilience and growth of tumor cells (Markowitsch et al., 2024). One of the emerging techniques in the analysis of apoptosis in tumor cells is the use of dual staining and PI staining. This approach provides a more comprehensive understanding of the cellular changes occurring during the different stages of the apoptotic process, facilitating the evaluation of the efficacy of apoptosis-inducing therapies. PI staining, on the other hand, is a broadly utilized technique to detect cell death (Grubb et al., 2022). In the present work, the apoptosis in the xanthohumol-treated A498 cells was assessed via dual-staining and PI-staining techniques. The findings of these fluorescent staining assays clearly proved that the A498 cells subjected to xanthohumol treatment exhibited increased apoptotic incidence. These findings evidenced the apoptotic-inducing potentials of xanthohumol against renal cancer cells. The apoptotic effects of xanthohumol were comparable to those induced by DOX, a standard chemotherapeutic agent. Notably, xanthohumol exhibited a similar potency to DOX in inducing apoptosis in A498 cells. This suggests that xanthohumol may possess similar anti-cancer efficacy to DOX. The comparable efficacy of xanthohumol and DOX highlights the potential of xanthohumol as a novel anti-tumor candidate.
One of the key factors implicated in the initiation of apoptosis is the accumulation of excessive ROS. ROS is a byproduct of cellular metabolism and can have both helpful and adverse effects on cellular function. At lower concentrations, ROS can act as signaling agents, mediating important cellular processes like proliferation and differentiation. However, in excessive amounts, ROS can result in oxidative stress, which can trigger the initiation of cell death pathways, including apoptosis. The mechanisms by which increased ROS production triggers cell death by apoptosis are complex and multifaceted (Aggarwal et al., 2019). ROS can directly destruct cellular macromolecules, like proteins, lipids, and deoxyribonucleic acid (DNA), leading to their dysfunction and the accumulation of oxidative damage. This oxidative stress can disrupt the delicate balance of pro- and anti-apoptotic genes, ultimately leading to apoptotic cell death (Zhao et al., 2023). Moreover, ROS can trigger the activation of death receptor-mediated apoptotic pathways. Specifically, ROS has been shown to participate in the signal transduction mechanisms of TNF-α, a potent inducer of apoptosis. The strategies that target the excessive production of ROS or the reduction of cellular antioxidant defense systems have been explored as therapeutic interventions to treat cancer (Wang, Chen, et al., 2023; Wang, Deng, et al., 2023). The results of this work have evidenced the increased green fluorescence in the xanthohumol-treated A498 cells. The results of this study demonstrate that xanthohumol significantly enhances ROS accumulation in A498 cells, which is comparable to the effects of DOX, a standard chemotherapeutic agent. The similarity in ROS-inducing effects between xanthohumol and DOX suggests that xanthohumol may possess similar anti-cancer properties. The comparable efficacy of xanthohumol and DOX in inducing ROS production highlights the potential of xanthohumol as a novel anti-cancer agent. Overall, the results of this study demonstrate the potential of xanthohumol to induce ROS-mediated apoptosis in kidney cancer cells, warranting further investigation into its therapeutic potential. Additionally, the comparison with DOX provides a useful benchmark for evaluating the efficacy of xanthohumol.
Oxidative stress, characterized by an imbalance between ROS generation and the capacity of cells to neutralize their detrimental effects by antioxidant mechanisms, is a major contributor to numerous pathological conditions, including tumors. In the context of cancer, the interplay between increased levels of TBARS and reduced antioxidant defenses can play a critical role in triggering apoptotic mechanisms. The increased oxidative stress can lead to oxidative injury to major cellular macromolecules, which can ultimately result in cell dysfunction and death. Specifically, the accumulation of TBARS, which are markers of lipid peroxidation, is a hallmark of increased oxidative stress (Li et al., 2024). Concurrently, the depletion of antioxidant defenses, such as SOD and GSH, can further exacerbate oxidative stress within cancer cells. SOD is a crucial enzyme that catalyzes the transformation of superoxide radicals into O2 and H2O2, while GSH is a potent antioxidant that can neutralize various ROS. The downregulation or inactivation of these antioxidant systems can render cancer cells more susceptible to oxidative insults, ultimately leading to their death through apoptosis (Iqbal et al., 2024). Cancer cells often exhibit a dysregulated redox balance, with increased levels of GSH and SOD that help them evade apoptosis. However, the accumulation of intracellular ROS, beyond a certain threshold, can overwhelm these protective mechanisms, triggering the disruption of mitochondrial integrity and the stimulation of caspase signaling, ending in cancer apoptosis. The oxidative stress-mediated mechanism of cell death represents a potential avenue to develop novel cancer therapies that target the redox imbalance within tumor cells (Galiniak et al., 2023). In this work, xanthohumol effectively augmented the TBARS and diminished the antioxidant concentrations in the kidney cancer A498 cells. These results indicate that xanthohumol can activate oxidative stress-mediated apoptosis in kidney cancer cells. The oxidative stress-inducing effects of xanthohumol in A498 cells were comparable to those of the DOX treatment. Xanthohumol treatment resulted in a significant increase in TBARS levels and a decrease in antioxidants, indicating the induction of oxidative stress. Similarly, DOX has been shown to induce oxidative stress in cancer cells, leading to apoptosis. The comparable effects of xanthohumol and DOX on oxidative stress biomarkers suggest that xanthohumol may possess similar anti-cancer properties.
The regulation of apoptosis is a crucial aspect of cancer biology, as the dysregulation of this process is a hallmark of cancer development and progression. Several key apoptotic proteins play pivotal roles in the initiation and regulation of apoptosis in tumor cells. Bax and Bcl-2 are members of the Bcl-2 family of proteins, which are key mediators of the intrinsic apoptosis mechanisms (Tian et al., 2024). Bax is a pro-apoptotic gene that enhances the discharge of cyt-c from the mitochondria, leading to the activation of the caspase signaling and eventual apoptosis. In contrast, Bcl-2 is an anti-apoptotic protein that can antagonize the pro-death function of Bax, thereby inhibiting apoptosis (Vigneswara & Ahmed, 2020). Caspase-3 and -9 are key executioner caspases that are activated during the apoptotic process and are responsible for cleaving the cellular components, leading to the characteristic morphological changes and cell death. Cyclin D1 is a cell cycle regulator that can also influence apoptosis, as its overexpression was shown to enhance viability and block apoptosis in certain tumor types (Chota et al., 2021). The intricate balance between pro- and anti-apoptotic genes, and the stimulation of the caspase signaling, is critical for the initiation of apoptosis in tumor cells. Disruption of this balance, often through the upregulation of anti-apoptotic genes or the inhibition of pro-apoptotic genes, can result in the escape of apoptosis and lead to the initiation of tumors (Singh et al., 2019). Therefore, pharmacological agents that selectively increase Bax and caspase-3/-9 expressions while decreasing Bcl-2 and cyclin D1 expressions may be effective in initiating apoptosis in cancer cells, leading to diminished tumor growth and enhanced patient outcomes (Wani et al., 2023). In a similar manner, the results of this work have evidenced the remarkable increase in the Bax, caspase-3/-9 concentrations, while decreasing Bcl-2 and cyclin D1 concentrations in the xanthohumol-treated A498 cells. These findings may provide evidence that xanthohumol treatment can promote apoptosis in renal cancer cells. The effects of xanthohumol on apoptotic protein levels in A498 cells were comparable to those of DOX. Xanthohumol treatment resulted in an elevation in pro-apoptotic proteins (Bax, caspase-3, and -9) and a diminution in anti-apoptotic proteins (Bcl-2 and cyclin D1). Similarly, DOX treatment also modulated apoptotic protein levels, confirming that xanthohumol promotes apoptosis in A498 cells through a similar mechanism. These findings suggest that xanthohumol may possess similar anti-cancer efficacy to DOX. The comparable effects of xanthohumol and DOX on apoptotic protein levels specify the capacity of xanthohumol as a new anti-cancer candidate.
The PI3K/AKT/mTOR pathway plays an essential role in the onset of various types of cancer. This pathway is implicated in regulating fundamental cellular mechanisms like cell growth, proliferation, survival, and metabolism (Miricescu et al., 2021). Dysregulation of this pathway has been observed in several cancers, including kidney cancer, due to genetic mutations or amplifications in the components of the pathway. Activation of the PI3K/AKT/mTOR signaling can lead to the promotion of cell growth, migration, and inhibition of apoptosis, all of which leads to the onset of kidney cancer (Rascio et al., 2021). It has been already highlighted the importance of the PI3K/AKT/mTOR pathway in tumor pathophysiology. For instance, activation of the PI3K/Akt/mTOR pathway was shown to enhance cancer cell invasion and the ability to overcome apoptosis. Furthermore, inhibition of PI3K has been linked to reduced angiogenesis and altered gene expression patterns in vascular tumors, suggesting a potential role in kidney cancer development. The PI3K/AKT/mTOR signaling also plays a role in the tumor microenvironment and angiogenesis (Badoiu et al., 2023). Given the crucial role of the PI3K/AKT/mTOR pathway in tumors, it has become a major target for cancer therapy. Various inhibitors of this signaling have been formed and are being investigated in clinical studies to treat several cancers (Ye et al., 2020; Zhou et al., 2020). In this work, the xanthohumol treatment considerably decreased the PI3K, AKT, and mTOR concentrations in the kidney cancer A498 cells. Therefore, it was evident that xanthohumol may block cell viability and activate apoptotic mechanisms in kidney cancer cells via downregulating the PI3K/AKT/mTOR signaling. The inhibitory actions of xanthohumol on the PI3K/AKT/mTOR signaling in A498 cells were comparable to the results of DOX, a standard chemotherapeutic agent. Xanthohumol treatment led to a reduction in PI3K, AKT, and mTOR levels, similar to DOX treatment. This similarity suggests that xanthohumol may possess similar anti-cancer efficacy to DOX by blocking the PI3K/AKT/mTOR pathway. The comparable effects of xanthohumol and DOX on this pathway specify the capacity of xanthohumol as a new anti-cancer candidate. These findings support the notion that xanthohumol may inhibit cell growth in A498 cells through a similar mechanism as DOX.
Conclusion
In conclusion, the findings of this work indicate that xanthohumol considerably inhibits cell proliferation and activates apoptotic mechanisms in kidney cancer A498 cells. The treatment with xanthohumol remarkably enhanced the ROS production and induced apoptotic cell death by elevating Bax and caspase-3/-9 levels while inhibiting Bcl-2 and cyclin D1 levels. Moreover, the xanthohumol treatment downregulated the PI3K/AKT/mTOR signaling in A498 cells. Consequently, it possesses the capacity to act as an anti-tumor candidate for the treatment of kidney cancer. However, further investigation is still required to elucidate the precise molecular processes implicated in xanthohumol-mediated apoptosis in kidney cancer cells.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
