Abstract
Background
Neurodegenerative diseases (NDs) have become a significant public health problem, and oxidative stress (OS) serves as a pivotal factor in the pathological progression of NDs. Pharmacological research indicates that Astragalus polysaccharide (APS, extract of Astragalus membranaceus) has antioxidant and antiaging effects, closely related to NDs.
Objectives
The potential of APS in treating NDs remains uncertain. Herein, the neuroprotective effect of APS was evaluated.
Materials and Methods
The H2O2-induced PC12 cell damage model was established to assess the neuroprotection of APS. Cell viability was determined by MTT. Cell images were observed using ImageXpress Micro Confocal analysis. Apoptosis of the cells and intracellular levels of reactive oxygen species (ROS) were detected using flow cytometry. The expression of Klotho, nuclear factor erythroid 2-related factor 2 (Nrf2), heme oxygenase-1 (HO-1), thioredoxin reductase 1 (TrxR1), and NAD(P)H quinone oxidoreductase 1 (NQO1) was determined by Western blot (WB).
Results
APS exerted significant protection against H2O2-caused cell death dose-dependently, and APS could significantly reduce the intracellular ROS and relieve cell apoptosis. Furthermore, we found that APS could upregulate the expression of Klotho and Nrf2 dose-dependently, and the expression levels of Nrf2-mediated antioxidant proteins increased significantly, including HO-1, TrxR1, and NQO1. Moreover, we found that APS upregulated Klotho and Nrf2-mediated antioxidant proteins time-dependently. The expression of klotho peaked at 6 hours after treatment with APS (400 mg/L). Subsequently, the protein level of Nrf2 reached the maximum at 12 hours, and the downstream NQO1, HO-1, and TrxR1 levels peaked at 24 hours.
Conclusion
This study demonstrates that APS is promising for further development against oxidative stress-related NDs.
Introduction
Neurodegenerative diseases (NDs), including Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD), amyotrophic lateral sclerosis (ALS), and other similar diseases, are distinctively marked by the degeneration and subsequent loss of neuronal cells. Projections indicate that by the year 2024, NDs are anticipated to emerge as the second leading cause of mortality globally (Buga & Oancea, 2023; Rehman et al., 2023). NDs share a common feature: They enhance oxidative stress (OS) and inflammation, which cause cellular death. OS plays a pivotal role in the pathophysiological processes of most neurodegenerative states (Buga & Oancea, 2023). OS in AD affects glutamate transport, neuronal glucose, mitochondrial membrane potential, and neuronal Ca2+ homeostasis, factors that cause neuronal dysfunction and damage (Ionescu-Tucker & Cotman, 2021; Olufunmilayo et al., 2023). OS is also considered to be very closely related to dopaminergic neuron degeneration (Dias et al., 2013). Significantly elevated levels of OS markers, including 8-hydroxy-deoxyguanosine, 8-hydroxy-guanosine, and 4-hydroxyl-2-nonenal, were observed in PD brains (Olufunmilayo et al., 2023). Oxidative damage to proteins, lipids, and deoxyribonucleic acid (DNA) was also found in postmortem ALS neuronal tissue (Olufunmilayo et al., 2023). OS-induced mitochondrial calcium dyshomeostasis and mitochondrial DNA damage also play critical roles in the pathogenesis of HD (Brustovetsky, 2016; Olufunmilayo et al., 2023). Given the established fact that OS plays an important role in nerve cell damage and death, targeting antioxidant systems may be a good therapeutic strategy against various OS-related neurodegenerative disorders.
As a popular traditional Chinese medicine renowned for its health-promoting properties, the dried root of Astragalus membranaceus, commonly known as Huangqi, has been extensively utilized as an immunomodulating agent for the treatment of diverse diseases and bodily disorders, including common cold, diarrhea, fatigue, and anorexia (Jina et al., 2014). It is reported that A. membranaceus was confirmed to be able to ameliorate stroke by antioxidant action (Cao et al., 2014). Pharmacological research indicates that the extract components of A. membranaceus (astragalosides, polysaccharides, flavonoids, etc.) have antioxidant and antiaging effects (Jina et al., 2014; Liu et al., 2017; Yu et al., 2018). Astragalus polysaccharide (APS) is one of the main components of A. membranaceus and is derived from the extracts of its dried roots. As a water-soluble heteropolysaccharide, the components of APS are complex and diverse, and it is mainly composed of polymerized carbohydrates that are formed through the linkage of monosaccharides via alpha-type glycosidic bonds (Jina et al., 2014; Li et al., 2020). APS exerts multiple bioactive effects, such as the regulation of blood glucose (Jina et al., 2014), regulation effect on immunity (Li, Wang, et al., 2019; Li et al., 2018), anti-inflammatory effects (Huang et al., 2013; Ren et al., 2018), antiaging effects (Liu et al., 2017), antitumor effects (Huang et al., 2016; Yu et al., 2018), antioxidant effects (Cao et al., 2014; Jina et al., 2014; Li, Liu, et al., 2019; Ma et al., 2011), antiviral effects (Guo et al., 2014), and so on.
As mentioned above, APS has been used for antioxidants and antiaging. NDs are closely related to OS, and aging is a key predisposing factor for some types of NDs, such as AD, PD, and HD (Luo et al., 2020; Querfurth & LaFerla, 2010). The above reports made us think that APS may have neuroprotective effects by alleviating OS. To our knowledge, there are no reports on the application of APS in the treatment of NDs. Klotho is a well-known antiaging gene (Chu et al., 2021; Kurosu et al., 2005; Wang et al., 2020; Zeng et al., 2018). Some studies have reported that klotho could induce nuclear factor erythroid 2-related factor 2 (Nrf2)-mediated antioxidant defenses (Cui et al., 2019; Ji et al., 2020; Maltese et al., 2017). As a key cellular defense protein against OS, Nrf2 fights against OS by increasing the expression levels of anti-OS proteins, such as thioredoxin reductase 1 (TrxR1), heme oxygenase-1 (HO-1), and NAD(P)H quinone oxidoreductase 1 (NQO1) (Baird & Dinkova-Kostova, 2011; Baird et al., 2013; Ma, 2013). Activating the Nrf2 pathway is an attractive strategy for treating NDs (Calkins et al., 2009; de Vries et al., 2008; Johnson & Johnson, 2015). Herein, we try to confirm the therapeutic effect of APS on NDs.
Materials and Methods
Cell Cultures
PC12 cells (Shanghai Institute of Biochemistry and Cell Biology) were cultured in Dulbecco’s modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 µg/mL streptomycin. The cells were incubated in a controlled environment, maintaining a humidified atmosphere at 37℃ and CO2 concentration of 5%.
Cell Viability Assay
PC12 cells in the log phase were digested, centrifuged, and diluted with medium. They were counted and transferred to 96-well plates (1 × 104 cells/well) for 24 hours culture; cells were then subjected to APS or H2O2 for another 24 hours. APS (98.1%) was prepared as described in the Supplemental material. Cell viability was measured by the MTT assay. For the H2O2 injury model, PC12 cells were seeded and cultured for 24 hours, then treated with APS for 12 hours. Medium was discarded, followed by stimulation with H2O2 (400 µM) in fresh medium for another 12 hours. Finally, cell viability was determined by MTT.
Preparation of Cell Samples for Flow Cytometry Assay
PC12 cells (3 × 105 cells per well) were plated in 6-well plates and cultured for 24 hours. Subsequently, these cells were subjected to APS for 12 hours. The original medium was discarded, followed by stimulation with H2O2 (400 µM) in fresh medium for another 12 hours. Finally, the cells were digested with 0.05% trypsin, centrifuged, collected, and rinsed thrice with phosphate-buffered saline (PBS) buffer.
Detection of Intracellular Level of Reactive Oxygen Species (ROS)
2′,7′-Dichloro-fluorescein diacetate (DCFH-DA, a fluorogenic dye) was utilized to assess the intracellular level of ROS. Cells were stained with DCFH-DA (10 µM) in an FBS-free medium at 37°C for 30 minutes, then centrifuged to remove the supernatant, resuspended, and rinsed thrice with PBS. Finally, the cell pellets were resuspended with 500 µL FBS-free medium, and the ROS levels were detected by BD-Accuri-C6 flow cytometry.
Apoptosis Assay
The Annexin-V/7-AAD staining apoptosis detection kit (Keygen Biotech, #KGA1016) was utilized to identify apoptotic PC12 cells. Briefly, prepared cell samples were stained with Annexin-V/7-AAD dyes at 25℃ for 15 minutes and were immediately analyzed by BD-Accuri-C6 flow cytometry. The apoptosis rate was analyzed with BD Cell Task software.
Western Blot Assay
PC12 cells were incubated with APS for the indicated time, then trypsinized and harvested by centrifugation. The collected cell pellets were resuspended in RIPA lysis buffer (Beyotime, # P0013C) and centrifuged at 4°C at 12,000 rpm for 10 minutes to collect the supernatant containing total proteins.
BCA protein assay kit (Beyotime, # P0010S) was used for protein quantification. An aliquot of proteins was separated by SDS-PAGE gels (8, 10, or 12%), subsequently transferred to polyvinylidene difluoride (PVDF) membranes, and blocked with nonfat dry milk [5% in tris buffered saline with Tween-20 (TBST)] for 90 minutes. Next, PVDF membranes were rinsed thrice with TBST and incubated with primary antibodies specific for Klotho (Proteintech, #28100-1-AP), Nrf2 (Proteintech, #16396-1-AP), NQO1 (Abcam, #Ab80588), TrxR1 (Abcam, #124954), HO-1 Abcam, #Ab52947), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (Abcam, #Ab181602) at 4°C overnight. Subsequently, the PVDF membranes were rinsed thrice with TBST buffer and labeled with a secondary specific antibody tagged with horseradish peroxidase (Yeasen, #33101ES60) at 25℃ for 2 hours. Finally, the PVDF membranes were rinsed thrice with TBST. Bound immunocomplexes were visualized by the ChemiDOC XRS+ system (Bio-Rad).
Statistical Analysis
GraphPad Prism 5 was used for data analysis. Data were expressed as means ± standard error of the mean (SEM). Differences between control and sample groups were statistically evaluated using a t-test. p < 0.05 was considered statistically significant. The following notations were used to indicate levels of significance: *p < 0.05, **p < 0.01, and ***p < 0.001 vs control group; ^p < 0.05, ^^p < 0.01, and ^^^p < 0.001 vs H2O2 group.
Results and Discussion
Establishment of Cell Damage Model
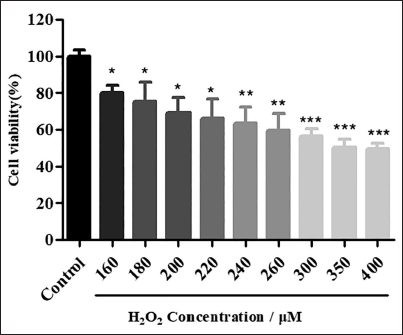
To assess the neuroprotection of APS, a PC12 cell damage model induced by H2O2 was established, which is a widely acknowledged cell model for the study of nerve injury (Ji et al., 2020; Qu et al., 2021). Briefly, PC12 cells were subjected to H2O2 (0– 400 µM) for 12 hours. It was observed that the cell death rate rose with increasing H2O2 concentration. Approximately 50% cell death was observed at H2O2 concentration of 400 µM (Figure 1). The specific concentration of H2O2 (400 µM) was chosen for further study.
Cell Damage is Caused by Different Concentrations of H2O2.
Neuroprotection of APS Against H2O2-induced PC12 Cells Damage Dose-dependently
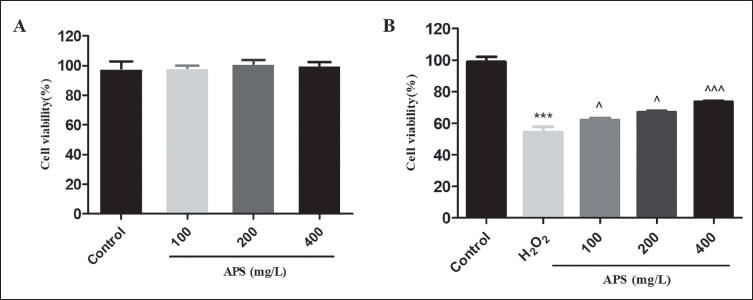
First, the cytotoxicity of APS was measured by MTT. As shown in Figure 2A, APS was not toxic to PC12 cells at the maximum concentration (400 mg/L). To assess the neuroprotection of APS, PC12 cells were coincubated with APS (100, 200, 400 mg/L) for 12 hours and subsequently subjected to H2O2 (400 µM) for another 12 hours. As shown in Figure 2B, about 50% cell death was observed after treatment with H2O2 for 12 hours compared to the control group. Pretreatment with APS (100, 200, 400 mg/L) for 12 hours before stimulation with H2O2 could improve the survival rate of PC12 cells dose-dependently. Cell viability increased to about 62%, 67%, and 74%, respectively. Furthermore, we observed many round and floating PC12 cells in the culture medium of the H2O2 model group. Pretreatment of PC12 cells with APS caused a dose-dependent decrease in round and floating cells (Figure 3). The results indicated that APS could effectively prevent the neurotoxicity caused by H2O2.
(A) Cytotoxicity of Astragalus Polysaccharide (APS) by MTT Assay. (B) APS Exerted Neuroprotective Effect Against H2O2 Caused Cell Damage Dose-dependently.
The Cells were Observed Using ImageXpress Micro Confocal Analysis. Scale bar: 100 µM.

Preclusion of Intracellular ROS Accumulation in PC12 Cells
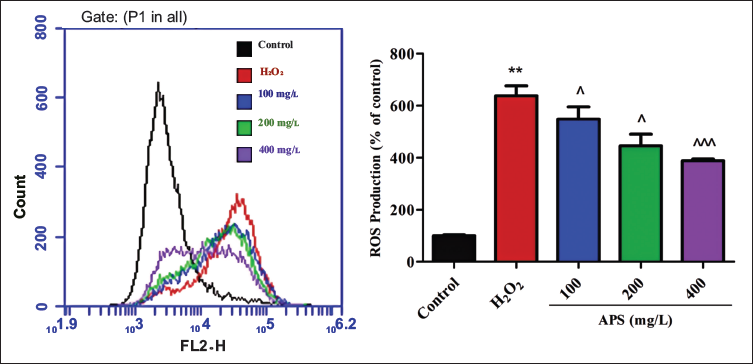
OS is one of the key factors of neurodegeneration (e.g., PD, AD) (Dias et al., 2013; Querfurth & LaFerla, 2010). In the presence of H2O2, the production of ROS exceeds the cellular capacity to eliminate oxidants, ultimately leading to cell death. Based on related reports, we postulated that APS may exert neuroprotective effects by alleviating OS (Huang et al., 2013; Li, Liu, et al., 2019). We quantified the intracellular levels of ROS using flow cytometry (Figure 4). When the cells were stimulated with H2O2, intracellular ROS levels increased more than six-fold compared to the control group. While pretreatment with APS (100, 200, 400 mg/L) for 12 hours before stimulation with H2O2, the intracellular ROS level was reduced significantly and dose-dependently compared to the model group. This result indicated that APS could efficiently reduce the intracellular ROS.
Astragalus Polysaccharide (APS) Reduced the Accumulation of Intracellular Reactive Oxygen Species (ROS) Detected by 2′,7′-Dichloro-Fluorescein Diacetate (DCFH-DA).
Alleviation of PC12 Cell Apoptosis Induced by H2O2
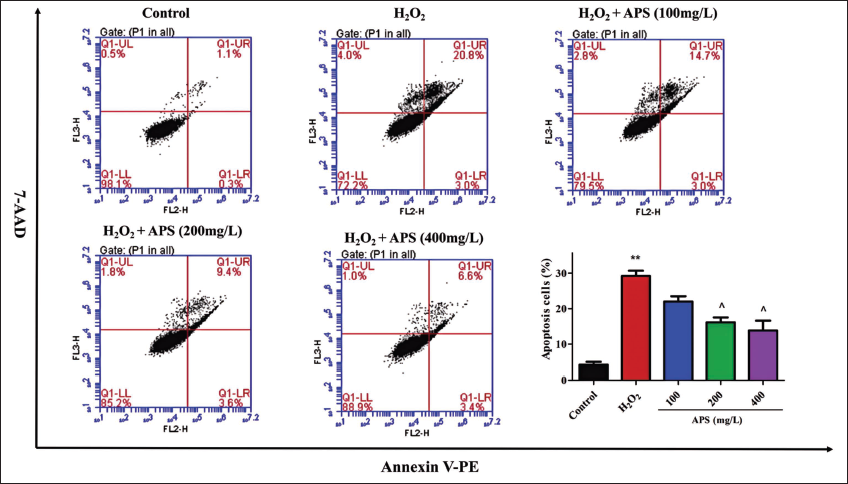
OS is a critical factor in neuron apoptosis, a key feature of neurodegeneration (Lin & Beal, 2006). To confirm APS’s antiapoptotic ability, apoptotic PC12 cells were labeled with Annexin-V/7-AAD dyes and detected using flow cytometry (Luo et al., 2023). Upon incubation with 400 µM H2O2, we observed an increase in the apoptosis rate from 1.9% to 27.8% compared to the control group (Figure 5). The apoptosis rate was reduced to 20.5%, 14.8%, and 11.1% dose-dependently after treatment with APS (100, 200, 400 mg/L). This result indicated that APS could efficiently relieve H2O2-induced apoptosis.
Astragalus Polysaccharide (APS) Relieved H2O2-induced PC12 Cell Apoptosis Detected by Annexin-V/7-AAD Dyes Using Flow Cytometry.
Activation of Klotho/Nrf2 Pathway and Related Antioxidant Proteins by APS
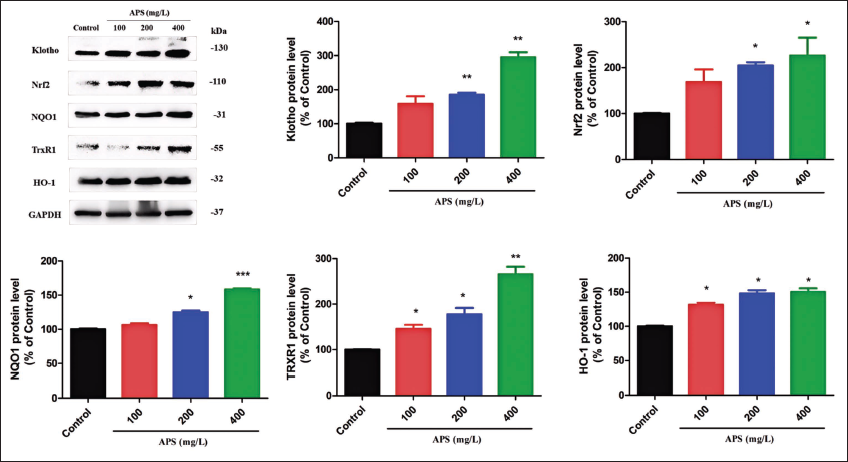
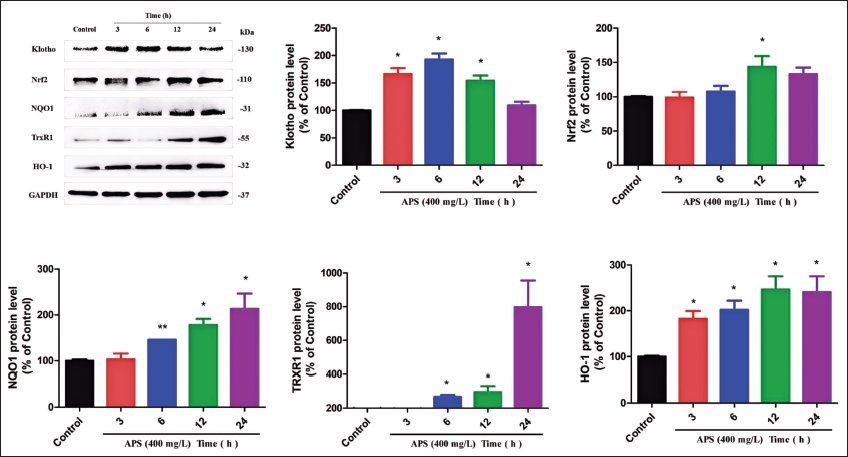
As described above, APS could protect PC12 cells from H2O2-caused cell death and alleviate ROS accumulation and cell apoptosis. Several studies have reported that A. membranaceus extracts exert antiaging and antioxidant effects (Cao et al., 2014; Liu et al., 2017). APS is one of the main components of A. membranaceus (Zheng et al., 2020). We hypothesized that the neuroprotection of APS is related to the activation of Klotho, which subsequently triggers Nrf2-mediated antioxidant defenses (Cui et al., 2019; Ji et al., 2020; Maltese et al., 2017). The expression of Klotho and Nrf2-mediated antioxidant proteins was quantified by WB. As shown in Figure 6, after pretreatment with APS (100, 200, 400 mg/L) for 12 hours, the levels of these proteins were significantly upregulated dose-dependently. Moreover, we found that APS could upregulate Klotho and Nrf2-mediated antioxidant proteins time-dependently (Figure 7). The level of klotho reached maximally at 6 hours after coincubation with APS (400 mg/L). Subsequently, Nrf2 expression reached its peak at 12 hours, followed by the attainment of maximal levels for NQO1, HO-1, and TrxR1 at 24 hours. APS may first increase the expression of Klotho within 6 hours, and then overexpressed Klotho increases the expression of Nrf2, which further activates the downstream antioxidant pathway. There are some similar results in previous Nrf2-related reports (Gu et al., 2019; Qu et al., 2021). Altogether, these results indicated that APS may exert neuroprotective effect through activating klotho and Nrf2-mediated antioxidant proteins.
Astragalus Polysaccharide (APS) Upregulated Klotho and Nuclear Factor Erythroid 2-Related Factor 2 (Nrf2)-mediated Antioxidant Proteins Dose-dependently. Protein Levels were Quantified by Western Blot (WB) with Glyceraldehyde 3-Phosphate Dehydrogenase (GAPDH) as Internal Control.
Astragalus Polysaccharide (APS) Upregulated Klotho and Nuclear Factor Erythroid 2-Related Factor 2 (Nrf2)-mediated Antioxidant Proteins Time-dependently. The Protein Levels were Quantified by Western Blot (WB) with Glyceraldehyde 3-Phosphate Dehydrogenase (GAPDH) as Internal Control.
Conclusion
This study confirmed that APS exhibited a potent protective effect against the H2O2-caused cell damage dose-dependently. APS could alleviate OS and relieve H2O2-induced cell apoptosis dose-dependently. The neuroprotective mechanism studies confirmed that APS could activate Klotho and Nrf2 and increase the expression levels of related antioxidant proteins, including NQO1, TrxR1, and HO-1, dose-dependently. In addition, APS could upregulate Klotho and Nrf2-mediated antioxidant proteins in a time-dependent manner. The level of klotho reached maximally after a 6-hour coincubation with APS (400 mg/L). Subsequently, the level of Nrf2 peaked at 12 hours, and the levels of NQO1, HO-1, and TrxR1 peaked at 24 hours. Altogether, given that NDs are a major global health concern, our findings suggest that APS may be a promising therapeutic agent against OS-related NDs.
Footnotes
Abbreviations
AD: Alzheimer’s disease; ALS: Amyotrophic lateral sclerosis; APS: Astragalus polysaccharide; BCA: Bicinchoninic acid; DMEM: Dulbecco’s modified Eagle medium; FBS: Fetal bovine serum; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; HD: Huntington’s disease; HO-1: Heme oxygenase-1; NDs: Neurodegenerative diseases; NQO1: NAD(P)H quinone dehydrogenase 1; Nrf2: Nuclear factor erythroid 2-related factor 2; OS: Oxidative stress; PBS: Phosphate-buffered saline; PD: Parkinson’s disease; PVDF: Polyvinylidene difluoride; ROS: Reactive oxygen species; SDS-PAGE: Sodium dodecyl sulfate polyacrylamide gel electrophoresis; SEM: Standard error of the mean; TBST: Tris buffered saline with Tween-20; TrxR1: Thioredoxin reductase 1; WB: Western blot.
Author Contributions
Conceptualization, Lailiang Qu and Dan Yang; Data curation, Fang Bian; Formal analysis, Kuo Liu and Nannan Ding; Funding acquisition, Dan Yang; Methodology, Kuo Liu and Nannan Ding; Project administration, Ji Zhang; Supervision, Lailiang Qu; Validation, Mengying Han and Shuping Shen; Visualization, Li Chen; Writing—original draft, Lailiang Qu. All authors have read and agreed to the published version of the manuscript.
Acknowledgments
The authors thank the Tea Plant Biology Key Laboratory of Henan Province for their technical help.
Declaration of Competing Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Natural Science Foundation of Henan Province (Grant no. 242300421540), the Xiangyang Science and Technology Planning project (Grant no. 2021YL16), and the Xiangyang Central Hospital Science project (Grant no. 2023YB18).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
