Abstract
Hyperglycemia-induced oxidative stress plays important roles in the development of non-alcoholic fatty liver disease (NAFLD), which is a common complication in diabetic patients. The Nrf2-Keap1 pathway is important for cell antioxidant protection, while its role in exogenous antioxidant mediated protection against NAFLD is unclear. We thus, postulated that antioxidant treatment with allopurinol (ALP) may attenuate diabetic liver injury and explored the underlying mechanisms. Control (C) and streptozotocin (STZ)-induced diabetes rats (D) were untreated or treated with ALP for 4 weeks starting at 1 week after diabetes induction. Serum levels of alanine aminotransferase (ALT) and aspartate transaminase (AST), production of lipid peroxidation product malondialdehyde (MDA), and serum superoxide dismutase (SOD) were detected. Liver protein expressions of cleaved-caspase 3, IL-1β, nuclear factor-erythroid-2-related factor-2 (Nrf2), heme oxygenase-1 (HO-1), P62, Kelch-like ECH-associated protein 1 (Keap1), and LC3 were analyzed. In vitro, cultured rat normal hepatocytes BRL-3A were grouped to normal glucose (5.5 mM, NG) or high glucose (25 mM, HG) and treated with or without allopurinol (100 µM) for 48 h. Rats in the D group demonstrated liver injury evidenced as increased serum levels of ALT and AST. Diabetes increased apoptotic cell death, enhanced liver protein expressions of cleaved-caspase 3 and IL-1β with concomitantly increased production of MDA while serum SOD content was significantly reduced (all P < 0.05 vs C). In the meantime, protein levels of Nrf2, HO-1, and P62 were reduced while Keap1 and LC3 were increased in the untreated D group as compared to control (P < 0.05 vs C). And all the above alterations were significantly attenuated by ALP. Similar to our findings obtained from in vivo study, we got the same results in in vitro experiments. It is concluded that ALP activates the Nrf2/p62 pathway to ameliorate oxidative stress and liver injury in diabetic rats.
Introduction
Non-alcoholic fatty liver disease (NAFLD) often occurs in association with metabolic diseases such as diabetes mellitus and non-alcoholic steatohepatitis (NASH).1,2 It is due to excessive accumulation of fatty acids in liver cells. 3 Recently NAFLD is observed in patients with both type 1 and type 2 diabetes mellitus (T1DM, T2DM)4,5 and also has been shown to be relevant to the development of cardiovascular diseases. 6 NAFLD is becoming more and more common globally.7,8 However, the relationship between T1DM and NAFLD and especially the underlying mechanism remains unknown. 9 Oxidative stress is an important contributor to the pathogenesis of diabetic complications due to the formation of hyperglycemia-induced glycation products and reactive oxygen species production, 10 suggesting that treatment strategies against oxidative stress may be particularly valuable for the prevention and treatment of diabetic liver injury.11,12 To date, study on antioxidant therapeutic strategies for NAFLD is constantly emerging, but the protective mechanisms of antioxidant therapy are largely unclear.13,14
The Nrf2-Keap1 pathway is important for cell antioxidant protection. 15 Upon activation, Nrf2 in the cytoplasm is transported into the nucleus to bind with antioxidant response elements (ARE) and promote the transcription of antioxidant enzymes such as heme oxygenase-1 (HO-1) and superoxide dismutase (SOD).16,17 Recent study has shown that the activation of Nrf2 played important roles in the protection of mouse liver from oxidative damage caused by dietary NASH 18 and in the protection of diabetic renal injury. 19 Also, it has been demonstrated that activation of the Nrf2 pathway alleviates oxidative stress and proinflammatory response, and improves lipid metabolism in NAFLD. 20 At present, Nrf2 is a therapeutic target for regulating plasma glucose levels and diabetic complications, and its importance is steadily increasing. 15 As one of the activators of Nrf2-Keap1 pathway, 21 autophagy adaptor protein p62 plays important roles in the pathogenesis of several liver diseases including NAFLD. 19 P62/SQSTM1 is a target gene of the transcription factor Nrf2, which can generate a positive feedback loop by inducing the transcription of genes driven by ARE. 22 p62 interacts with Keap1 through a conserved sequence motif. When autophagy is impaired, p62 will accumulate and self-activate Nrf2 by chelation of Keap1 into inclusion bodies, and recent studies have demonstrated that the Keap1-Nrf2 system plays a key role in maintaining cellular homeostasis.23,24 However, the role of Nrf2-Keap1 system in the development of diabetes-induced NAFLD and in particular in exogenous antioxidant mediated protection against NAFLD is unclear and merits intensive research.
Allopurinol, a xanthine oxidase inhibitor that is widely used in clinical practice, has been demonstrated to reduce hepatic oxidative stress. 25 A study shows that ALP can attenuate acute liver injury through adjusting the cellular redox-sensitive transcription factors in rats, suggesting that ALP can play a role in hepatocyte protection by interacting with Nrf2 signaling pathway. 26 And, a latest study shows that ALP can enhance the therapeutic effects of metformin and vitamin E on fructose-induced NAFLD, 27 but it does not clarify the potential molecular mechanism of ALP in attenuating NAFLD in rats. Furthermore, ALP has been shown to alleviate type 1 diabetes-associated NAFLD by reducing liver inflammation and lipid accumulation under hyperglycemic environment. However, the molecular mechanism by which ALP attenuates NAFLD in type 1 diabetes remains largely unknown. 28 We, thus, designed in vivo and in vitro experiments to test the hypothesis that ALP activates the Nrf2/p62 pathway to ameliorate oxidative stress and to attenuate NAFLD in type 1 diabetes rats.
Materials and methods
Cell culture and treatment
Normal rat hepatocytes BRL-3A was bought from Shanghai Cell Biology, Chinese Academy of sciences (Shanghai, China). Cells were cultured in DMEM medium containing 10% FBS, supplemented with 100 U/mL penicillin and 100 μg/mL streptomycin in a humidified environment in 5% CO2 at 37°C. Cells were plated for 24 h and then processed for different assays at the specified concentrations. The BRL-3A cells were maintained in DMEM containing normal glucose (5.5 mM, as control) or various concentrations of high glucose (15, 25, 35, 45, and 55 mM) and the finally selected concentration of glucose treatment with or without the presence of allopurinol (100 µM).
Determination of cellular injury
Cell viability was tested using Cell Counting Kit-8 (Dojindo, Japan) and Cell apoptosis was tested using DeadEnd™ Fluorometric TUNEL System (Promega, USA) following the producer’s protocol as described. 29
Animals and induction of diabetes
Male adult Sprague-Dawley rats (250 ± 10 g, 7 ± 1 weeks) were obtained from Guangdong Medical Laboratory Animal Center. All rats were placed in a room with controlled temperature (22–24°C), light–dark cycle for 12 h, and offered free access to standard feed and water. Type 1 diabetes was induced as described. 30 In brief, after the rats were anesthetized, streptozotocin (STZ) dissolved in 0.1 M citrate buffer (pH 4.5) was injected through the tail vein at a dose of 65 mg/kg body weight (Sigma-Aldrich, St. Louis, MO), or 0.1 M citrate buffer alone injected in the control group. One week after STZ injection, rats manifesting hyperglycemia (blood glucose 16.7 mmol/L) were considered diabetic rats and subjected to subsequent research. All experimental protocols used in this study were approved by the Committee on Animal Research and Ethics of Guangzhou Medical University and carried out in compliance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (8th Edition, 2011; https://www.ncbi.nlm.nih.gov/books/NBK54050/).
Animal experimental protocol
Rats were divided into three groups at random (n = 6, per group): control (C), diabetes (D), diabetes treated with allopurinol (D + ALP). Allopurinol (Sigma-Aldrich, St. Louis, MO, USA) was dissolved in drinking water and was administrated for 4 weeks starting 1 week after the induction of diabetes. A dose of ALP at 100 mg/kg/day was selected based on the finding that this dose of ALP can reduce liver damage in diabetic rats. 26 Meanwhile, we regularly monitored the blood glucose, body weight, water intake, and food consumption of rats. After the completion of determent, rats were then anesthetized with ketamine (100 mg/kg body weight) and Xylazine (10 mg/kg body weight). Blood was collected by cardiac puncture and centrifuged in tubes at 1000 × g for 15 min and then stored at −80°C until being analyzed. The liver tissue of each rat was rapidly removed and frozen in liquid nitrogen for later assays.
Histology detection
Randomly selected liver tissues were fixed with 4% paraformaldehyde in phosphate buffered saline (PBS). Subsequently, tissue samples were embedded in paraffin to obtain 5 μm thick sections, and then being processed for immunohistochemistry and hematoxylin and eosin (H&E) staining. At last, all the stained slides were examined using an optical microscope (80i, Eclipse, Nikon®, Tokyo, Japan).
Apoptotic cell death determination
Terminal deoxynucleotidyl transferased UTP nick-end labeling (TUNEL) reaction was assessed using the DeadEnd™ Fluorometric TUNEL System (Promega, USA). The tissue sections were observed under an optical microscope and five randomly selected areas of each slide were analyzed, and the apoptosis index was calculated as the percentage of apoptotic nuclei relative to the total nuclei. 31
Biochemical analyses
The activities of alanine aminotransferase (ALT), aspartate transaminase (AST), malondialdehyde (MDA), and SOD content in serum were detected by the specific enzyme-linked immunosorbent assay (ELISA) kits (Jianglaibio, Shanghai) as described. 32
Nucleocytoplasmic separation
About 20–100 mg of liver tissue was cut into small pieces and placed in a microcentrifuge tube and then washed with PBS. The liver tissue was then centrifuged at 500 × g for 5 min, and the supernatant was carefully removed and discarded, leaving cell pellet as dry as possible. The liver tissue was homogenized using a Dounce homogenizer or a tissue grinder in the appropriate volume of CER I according to the producer’s instruction. At last reagents as indicated in the producer’s instructions (Thermo, Chicago, IL) were used to proceed cytoplasmic and nuclear protein extraction. The adherent BRL-3Acells were harvested with trypsin-EDTA and then centrifuged at 500 × g for 5 min. The cells were then washed by suspending the cell pellet with PBS. An number of 1–10 × 106 cells were transferred to a 1.5 mL microcentrifuge tube and centrifuged at 500 × g for 2–3 min. The supernatant was carefully removed and discarded as the supernatant, leaving the cell pellet as dry as possible before being added with ice-cold CER I. Finally, specific volumes of reagent as indicated in the producer’s instructions was used to proceed to cytoplasmic and nuclear protein extraction.
Western blotting
The rat liver tissue and BRL-3A cell lysates were prepared by incubation with lysis buffer (0.1 mM EDTA, 0.1 mM PMSF, 0.1 mM Na3PO4, and 0.1 mM NaF plus protease and phosphatase inhibitor cocktails (Beyotime, Shanghai)) on ice and centrifuged at 3000×g for 15 min at 4°C. The supernatant was collected, and the protein concentration was measured by using the Enhanced BCA Protein Assay Kit (Beyotime, Shanghai). Cell nuclear and cytoplasmic protein were isolated following the producer’s protocol described in the Nuclear and Cytoplasmic Extraction Kit (Thermo, Chicago, IL). Fifty micrograms of protein from each sample was separated by using 10% sodium dodecylsulfonate-polyacrylamide gel electrophoresis (SDS PAGE) at 100 V for 2 h at room temperature. Subsequently, the proteins were electrotransferred onto a 0.22 μm immuno-Blot PVDF membrane (Merck millipore, Germany) (0.32 A and 2 h at 4°C). The membranes were blocked with a 5% blocking solution in 1 × TBST (Tris-buffered saline with Tween-20) and skimmed milk powder (Solarbio, Beijing) for 2 h at room temperature to prevent non-specific binding to the membranes. The membranes were rinsed once with 1 × TBST, then were added with the prepared primary antibody and slowly shaked with a horizontal shaker and incubate at 4°C overnight. Then, the membranes were washed three times in 1 × TBST the next day, and incubated for 2 h at room temperature in appropriate secondary antibodies which were chosen according to the primary antibody. The primary antibodies against Nrf2, IL-1β, HO-1 were purchased from Abcam (Cambridge, MA), and primary antibodies for LC3, histone H3, GAPDH, p62, and Keap1 were purchased from Cell Signaling Technology (Beverly, MA). Immunoreactive bands were visualized by an enzymatic chemiluminescence method and the intensity of each band was analyzed by ImageJ software (NIH, MD, USA)
Statistical analysis
All of the data are shown as mean ± standard error of the mean (SEM). Statistical analysis (GraphPad Prism 6.0, USA) of data obtained in the same group of rats and between groups of rats were assessed by using one-way analysis of variance (ANOVA). Multiple comparisons of group means were then performed by using Tukey’s test. P < 0.05 was regarded as statistically significant.
Results and discussion
Results
Allopurinol activates the Nrf2 signaling pathway and effectively alleviated the damage and apoptosis of BRL-3A cells caused by high glucose
As shown in Figure 1a, rat liver cells BRL-3A were cultured for 48 h at different concentrations of glucose (5, 15, 25, 35, and 45 mM). Compared with normal glucose (5 mM, NG) culture, the survival rate of BRL-3A cells was significantly reduced when cultured for 48 h with 25–45 mM glucose. Based on the above experimental results, we selected a fixed concentration (25 mM) glucose medium as the high glucose culture. For purpose of testing whether allopurinol exhibited protection on the viability of the BRL-3A cells against high glucose-induced cytotoxicity, cells were treated with 25 mM glucose with or without allopurinol at 100 μM. The results indicated that allopurinol significantly suppressed high glucose-induced cytotoxicity (Figure 1b). High glucose increased the accumulation of the lipid peroxidation end product malondialdehyde (MDA) and allopurinol prevented this change (Figure 1c). In addition, allopurinol treatment restored SOD content in cells exposed to high glucose (Figure 1d). Moreover, ALP treatment reduced high glucose induced inflammatory factor IL-1β protein expression in BRL-3A cells, as shown in Figure 1e. Furthermore, allopurinol treatment in high glucose (HG)-exposed BRL-3A cells effectively repaired the Nrf2 signaling pathway. As shown in Figure 2, allopurinol treatment reverted high glucose induced reductions of hepatic protein expressions of HO-1, nuclear Nrf2, and P62, and significantly attenuated high glucose induced elevations of cleave-caspase 3, Keap1, and LC3 II/I in BRL-3A cells (Figure 2a–e).

Effect of allopurinol on high glucose-induced BRL-3A cells’ cytotoxicity. Cell viability of BRL-3A cells that were treated with different concentrations of glucose (a). Cell viability of BRL-3A cells exposed to 25 mM glucose with or without 100 μM allopurinol (b). The content of MDA and SOD in the BRL-3A cells’ culture supernatant of (c and d). Protein expression of IL-1β in BRL-3A (e). Data are shown as means ± SEM of two independent experiments, each conducted in triplicate.

Changes of signaling proteins in BRL-3A cells exposed to 25 mM glucose with or without 100 μM allopurinol. Allopurinol treatment increased protein expression of nuclear Nrf2, HO-1, and P62, while decreased Keap1 and LC3 II/I in high glucose-exposed BRL-3A cells. Cytosolic and nuclear Nrf2 protein expression in BRL-3A (a and b). Protein expression of HO-1 and cleave-caspase3 in BRL-3A (c). Protein expression of P62 and LC3 II/I in BRL-3A (d). Protein expression of Keap1 in BRL-3A (e). Data are shown as means ± SEM of two independent experiments, each conducted in triplicate.
General features of control and diabetic rats treated with or without ALP
As shown in Table 1, 5 week STZ-induced diabetic rats showed symptoms of hyperglycemia, polydipsia, and polyphagia with significant increase in plasma glucose, water intake, and food consumption (P < 0.05 D vs C) and significant weight loss (P < 0.05 vs C). ALP treatment significantly decreased water intake in diabetic rats (D + ALP group), but did not significantly affect their blood glucose levels, food consumption and body weight. The changes of the serum specific markers related to hepatic damage were displayed in Figure 3a and b. Both serum AST and ALT were significantly increased in diabetic group compared with the control group (P < 0.05 vs C), and were significantly reduced by ALP treatment (P < 0.05 vs D). It was observed that the liver structure of rats in C group was clear with little fat droplets, and the liver structure of rats in D group was unclear and filled with fat droplets of different sizes, while the liver structure of diabetic rats treated with ALP (D + ALP group) was improved and fat droplets were decreased (Figure 3c). The above changes indicate that ALP effectively alleviated the histological changes in the liver of diabetic rats. In addition, tissue section TUNEL assay showed that the apoptosis of liver tissue in D group was significantly increased (P < 0.05 vs C), while the apoptosis of liver tissue was significantly decreased in diabetic rats with ALP treatment (Figure 4a and b). Moreover, the protein expression of cleave-caspase 3 was significantly increased in D group compared with C group, and its overexpression was significantly decreased in diabetic rats with ALP treatment (Figure 4c). Besides, serum levels of MDA was significantly increased in STZ-induced diabetic rats (P < 0.05 vs C, Figure 5a) while SOD was significantly decreased (P < 0.05 vs C, Figure 5b), but with ALP treatment, the MDA level was reduced and SOD was upregulated in STZ – induced diabetes (Figure 5a and b). The expression of inflammatory factor IL-1β was upregulated in the liver of D rats, and its overexpression was inhibited after treatment with ALP (Figure 5c).
Effect of ALP on general characteristics in controls and rats with STZ – induced diabetes.
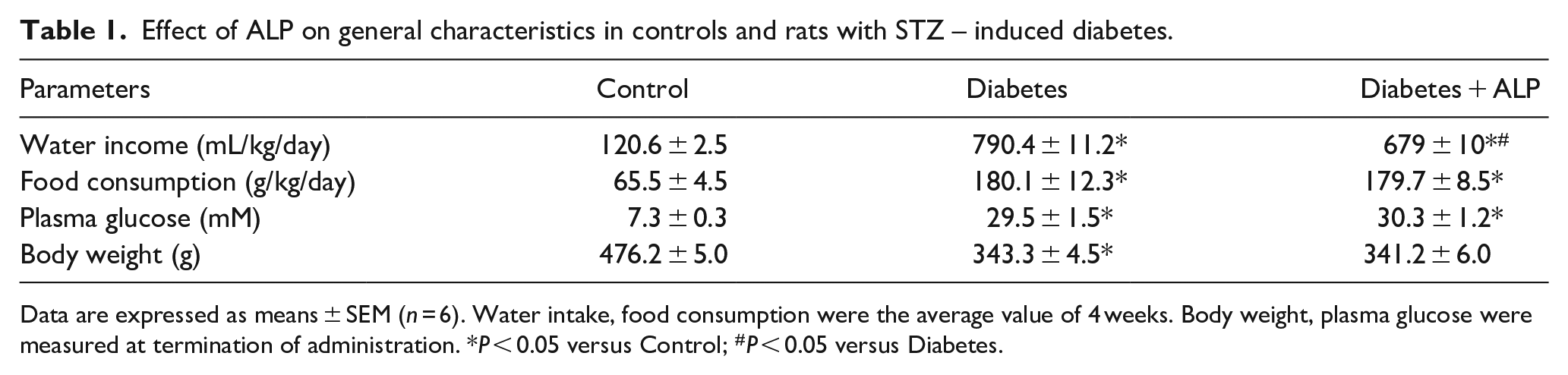
Data are expressed as means ± SEM (n = 6). Water intake, food consumption were the average value of 4 weeks. Body weight, plasma glucose were measured at termination of administration. *P < 0.05 versus Control; #P < 0.05 versus Diabetes.

Effects of allopurinol on liver function and pathology in STZ-induced diabetic rats. Rats’ serum ALT (a) and AST (b). Histological changes of liver sections of rats in each group were observed by staining with hematoxylin and eosin (magnification, 200×) (c). Values are expressed as means ± SEM (n = 6/group. *P < 0.05, **P < 0.01).

Allopurinol reduced liver oxidative stress and cleave-caspase3 expression in STZ-induced diabetic rats. Apoptotic level was assessed using TUNEL staining (magnification, 200×) (a and b). Liver protein expression of cleave-caspase 3 was detected by Western blotting (c). Values are expressed as means ± SEM (n = 6/group. *P < 0.05, **P < 0.01).

Allopurinol reduced MDA and IL-1β levels while upregulated SOD levels in STZ-induced diabetic rats. Serum MDA and SOD content in rats (a and b). Rat’s liver protein expression of IL-1β was detected by Western blotting (c). Values are expressed as means ± SEM (n = 6/group. *P < 0.05, **P < 0.01).
ALP reverted the reductions in liver protein expression of nuclear Nrf2, P62 and HO-1, while attenuated the increases of Keap1 and LC3 protein expression in STZ-induced diabetic rats
Diabetes significantly increased the retention of Nrf2 protein in the cytoplasm (Figure 6a), leading to a decrease in nuclear Nrf2 protein expression in the liver (Figure 6b). Treatment with ALP significantly increased liver nuclear Nrf2 protein expression in STZ-induced diabetic rats, manifested as increased Nrf2 nuclear translocation and activation after ALP treatment (Figure 6a and b). In the meantime, liver HO-1 and P62 protein expression were significantly reduced in STZ-induced diabetic rats (Figure 6c and d), and ALP treatment significantly alleviated the reduction of protein expression of HO-1 and P62. Moreover, Keap1 protein expression was significantly increased in STZ-induced diabetic rats and the increase of Keap1 protein expression was significantly attenuated by ALP treatment (Figure 6e). Furthermore, LC3 II/I protein expression was found significantly upregulated in diabetic rats but significantly downregulated with the treatment of ALP (Figure 6e). LC3 II is a vital protein of autophagic pathway. These results indicate that diabetes may impair liver antioxidant capacity and that ALP antioxidant intervention can repair these changes by regulating autophagy.

Allopurinol treatment increased liver protein expression HO-1, nuclear Nrf2, P62 in STZ-induced diabetic rats while decreased Keap1 and LC3 II/I. Cytosolic and nuclear Nrf2 protein expression in rats’ liver (a and b). Liver HO-1 protein expression(c). Liver P62 protein expression (d). Liver Keap1 and LC3 II/I protein expression (e). Values are expressed as means ± SEM (n = 6/group.*P < 0.05, **P < 0.01).
Discussion
In the current study, we conducted in vitro experiment in BRL-3A cells derived from liver and the results showed that ALP treatment reduced the high glucose-induced oxidative stress in BRL-3A cells through Nrf2/P62 pathway. High glucose-exposure in BRL-3A cells has been shown to cause oxidative stress, inflammation, and to impair antioxidant system.15,33 Similar to our findings obtained from in vivo study, ALP effectively restrained high glucose-induced cytotoxicity and decreased the production of MDA while restored SOD content in vitro. Moreover, ALP treatment reduced high glucose induced inflammatory factor IL-1β protein expression in BRL-3A cells. These results indicated that ALP can directly work on hepatocytes.
Hyperglycemia-induced oxidative stress serves as a major contributor to the development of non-alcoholic fatty liver disease (NAFLD).1,10 We have shown in the current study that both serum AST and ALT were significantly increased that was coincident with increased apoptotic cell death and autophagy, and that serum levels of MDA were significantly upregulated while SOD was significantly reduced in STZ-induced diabetic rats. Intriguingly, the above hepatic pathology changes in STZ-induced diabetic rats were associated with concomitant reductions in nuclear translocation of Nrf2 (i.e., reduced activation of Nrf2), a key transcriptional factor for antioxidant pathway activation, while antioxidant treatment with ALP increased Nrf2 nuclear translocation and ameliorated or reversed all the above liver pathological changes. In vitro study in BRL-3A cells exposed to high glucose demonstrated significant cellular injury evidenced as reductions in cell viability and SOD content that was concomitant with increases in MDA content and IL-1 protein expression, which mirrored the changes seen in STZ-induced diabetic rats. Similarly, Nrf2 nuclear translocation was reduced in BRL-3A cells exposed to high glucose which was reversed with ALP treatment.
Oxidative stress is a key influencing factor in the pathogenetic development of STZ-induced diabetes. 10 Thus, treatment strategies targeting oxidative stress may be particularly valuable for preventing liver damage in STZ-induced diabetic rats. 34 A previous study indicated that diabetes induced liver injury is mediated by inflammatory reactions, apoptosis, and endocannabinoid system, and that these effects could be alleviated by antioxidant. 35 There are increasing evidences suggesting that induction of the intrinsic Nrf2 antioxidant pathway may protect the liver from diabetes or ischemia-reperfusion induced damage.28,36 Studies have shown that the endogenous Nrf2 antioxidant cellular pathway could be dynamically regulated in high-fat diet induced NAFLD in rats and in the tyloxapol-induced NAFLD in mice,37,38 thus, aiming at the Nrf2 pathway may represent an attractive therapy. However, the role of Nrf2 in type 1 diabetes induced NAFLD has yet to be defined.
Allopurinol is a xanthine oxidase inhibitor, and has been demonstrated to prevent glutathione oxidization and lipid peroxidation in experimental and human type 1 diabetes. 25 The levels of serum ALT and AST are commonly considered as liver enzymes released from injured liver cells and have been generally regarded as effective quantitative biomarkers to assess hepatocellular damage. 39 In our experiment, the results showed that diabetes resulted in significant liver injury, which was manifested by elevated release of liver enzyme levels and significant histopathological changes. However, treatment with ALP attenuated the elevation of hepatic enzymes caused by diabetes to a level comparable to that in the control group and ameliorated the hepatic histopathologic changes although further detailed quantitative histopathologic assessment is merited. 40 The beneficial effects of ALP identified in the current study are in line with previous studies, which showed that ALP conferred protection against liver injury induced by various chemical hepatotoxins such as thioacetamide and carbon tetrachloride.26,41 In the present study, we have manifested that ALP, through simultaneously activating Nrf2/Keap1 and P62, activates HO-1 and alleviates liver injury in STZ-induced diabetic rats. ALP may have induced Nrf2 activation due to keap1 degradation, which concomitantly resulted in the activation of p62, an adaptor protein during autophagy. It is known that ALP confers antioxidant effects via inhibiting xanthine oxidase enzyme. However, whether or not ALP can also activate Nrf2, especially in the context of STZ-induced type 1 diabetic liver injury is unknown. To our knowledge, our current study is the first to shown that ALP treatment effectively decreased the protein expression of cytosolic Nrf2 and increased the protein expression of nuclear Nrf2, P62, and HO-1 while reduced the protein expression of Keap1 and LC3 both in the liver of STZ-induced diabetic rats and in BRL-3A cells exposed in high glucose.
Studies have emphasized that antioxidants may offer protection against liver injury by activating the Nrf2 pathway.36,42 In the current study, we have shown that ALP can activate the Nrf2 and HO-1 signaling pathway to protect the liver against diabetic liver injury. Having an important influence in maintaining antioxidant homeostasis during cellular stress, HO-1 is a part of the cytoprotection and detoxification gene family, which has antioxidant response elements in its regulatory regions. Nrf2 is a transcription factor whose activation can simultaneously enhance the expression of some antioxidant enzymes (such as HO-1). Our current study offers the experimental evidence that ALP can provide protection against liver injury by activating the Nrf2/HO-1 pathway. It is worth noting that our study involved autophagy disorders and Nrf2/p62 was first reported in type 1 diabetic liver injury. Our results provide a definitive mechanism for the liver protective effects of allopurinol on type 1 diabetes. The autophagy disorders and the alteration of Nrf2/p62 signaling in the liver of type 1 diabetic rats as identified in the current study share some similarity with the changes in hearts of type 1 diabetic rats as we recently reported, 31 however, further extensive research is needed to confirm the mechanism of allopurinol protection against diabetic liver cellular injury functions.
Conclusions
In summary, our current study indicates that ALP has beneficial antioxidant effects and can reduce the severity of liver damage in STZ-induced diabetic rats by activating the Nrf2/HO-1 pathway. It should be noted, however, although changes in the expression of Nrf2, Keap1, P62, and autophagy in diabetes are clarified in animal models or at cellular level in the current study, the Nrf2/p62 pathway activation could be involved in many other pathways of diabetic liver damage and thus further study incorporating proteomic analysis should be conducted to elucidate the potential protein-protein interrelation involved in this pathology.
Footnotes
Author contribution
Fei Zeng participated in the design of the study, carried out the Western blot, performed the statistical analysis and drafted the manuscript. Jierong Luo carried out the animal experiment. Hong Han and Wenjie Xie carried out the cell experiment. Lingzhi Wang helped the design of animal experiment. Ronghui Han and Hao Chen participated in the animal sample collection coordination, as well as animal data analyses and interpretation . Yin Cai helped rivese the manuscript. Zhengyuan Xia and Huansen Huang conceived of the study, and participated in its design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored by National Natural Science Foundation of China (NSFC, 81670770, 81970247) grant and RGC/GRF grant (17118619).
Ethics approval
Ethical approval for this study was obtained from Committee on Animal Research and Ethics of Guangzhou Medical University (GY 4276-17).
Consent to publish
The study and the publication of the work were approved by the Committee on Animal Research and Ethics of Guangzhou Medical University, and all authors have read and agreed to the submission of the manuscript in the current form for publication.
Animal research
All experimental protocols used in this study were carried out in compliance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (8th Edition, 2011; ![]() ) and was approved by the Committee on Animal Research and Ethics of Guangzhou Medical University (GY 4276-17).
) and was approved by the Committee on Animal Research and Ethics of Guangzhou Medical University (GY 4276-17).
