Abstract
Background
Aristolochic acid is naturally found in plants of the Aristolochiaceae family and possesses antitumor, anti-infection, and anti-inflammatory properties.
Objectives
This study investigated the protective effect of apigenin (API) on aristolochic acid I (AAI)-induced renal damage in mice and its underlying mechanisms.
Materials and Methods
Male C57BL/6J mice were assigned to the control group, model group, and API low, medium, and high-dose groups (10, 20, 40 mg/kg). On the 5th day, 2 h after administration, except for the control group, all other groups were intraperitoneally injected with AAI (10 mg/kg). Following final administration, 24-h urinary protein levels, serum creatinine (Scr), and blood urea nitrogen (BUN) levels were assessed along with hematoxylin–eosin (HE) staining of kidneys and terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining, as well as analysis of malondialdehyde (MDA) levels and superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) activity.
Results
When compared with the model group, mice in API treatment groups exhibited significantly reduced 24-h urinary protein levels and serum levels of Scr and BUN (p < 0.05) with an improvement in renal tissue histopathological changes and cell apoptosis as well as decreased MDA and increased SOD and GSH-Px (p < 0.05). Tumor necrosis factor alpha protein (TNF-α)/interleukin (IL)-1β/IL-6 levels showed a reduction (p < 0.05). Nuclear factor erythroid 2-related factor 2 (Nrf2) and heme oxygenase-1 (HO-1) were upregulated, while NF-κB/Cleaved Caspase-3 were downregulated (p < 0.01).
Conclusion
API protects against renal damage induced by AAI possibly through Nrf2/HO-1 pathway, which in turn inhibits oxidative stress damage, inflammatory responses, and cell apoptosis.
Keywords
Introduction
Aristolochic acid is naturally found in plants of the Aristolochiaceae family and possesses antitumor, anti-infection, and anti-inflammatory properties. Traditional Chinese medicines and pharmaceutical preparations containing aristolochic acid have been widely used in clinical practice (Fan et al., 2020; Zhang et al., 2019). A study has indicated a significant association between exposure to aristolochic acid and a heightened risk of kidney disease and upper urinary tract cancer (Pu et al., 2017). Aristolochic acid I (AAI), as one of the main components of aristolochic acid, also exhibits nephrotoxicity and carcinogenic properties (Li et al., 2021). Previous researchers have found that high doses of aristolochic acid can cause direct damage to renal proximal tubular epithelial cells within a short period, leading to cell degeneration, apoptosis, and necrosis (Liu, Wu, et al., 2017; Zeng et al., 2017). The clinical pathological manifestations include progressive atrophy of proximal tubules, marked interstitial fibrosis, and rapid progression to end-stage kidney disease (Kanaan et al., 2016). Under AAI’s influence, levels of tumor necrosis factor alpha protein (TNF-α)/IL-1β/IL-6 increase within the kidneys (Liu et al., 2011; Xie et al., 2017). This will further result in oxidative stress, alterations in renal microstructure, and renal function abnormalities within the kidneys (Honarpisheh et al., 2018). Thus, it is evident that the nephrotoxicity of AAI is closely associated with the renal inflammation response caused by this compound. Despite national restrictions on the use of products containing aristolochic acid, new cases of aristolochic acid nephropathy continue to emerge. Currently, there is no effective medication available to reverse the kidney damage caused by AAI. Therefore, investigating the pathogenic mechanisms of AAI-induced renal damage, identifying specific drug targets, and developing effective treatment strategies represent the primary directions for researching AAI-induced kidney injury.
Oxidative stress and inflammation play a crucial role in kidney diseases. Nuclear factor-erythroid 2 (Nrf2) is known as the leucine zipper transcription factor activator family and is a key factor in cellular defense against various stress injuries. Biliverdin can then be converted into bilirubin, and both substances serve as potent free radical scavengers within the body. Nrf2/heme oxygenase-1 (HO-1) plays a crucial regulatory role in oxidative stress and inflammatory responses. Studies have shown that oleuropein exhibits inhibitory effects on inflammation in RAW264.7 cells via Nrf2/HO-1 signaling (Ryu et al., 2015). Similarly, oleuropein suppresses oxidative stress-induced cardiomyocyte damage via Nrf2/HO-1 signaling (ALHaithloul et al., 2019). Research by Wang et al. (2019) found that naringin, by enhancing Nrf2/HO-1 signaling, alleviated renal damage in pregnancy-induced hypertension in rats. Similarly, Chen et al. (2019) discovered that the addition of 1% tert-butylhydroquinone (tBHQ) in the diet could inhibit oxidative stress damage in the kidneys of type 1 diabetic mice through the Nrf2/HO-1 pathway.
Apigenin (API) is a flavonoid compound in plants of Rutaceae, Scrophulariaceae, and Cupressaceae families. It is widely distributed in vegetables and fruits in temperate and tropical regions, with celery having a higher content. API exhibits notable biological activities such as antioxidation and anti-inflammation (Zhou et al., 2019) and can also inhibit cell apoptosis (Lu et al., 2010). Research has shown that API protects mice with cisplatin-induced renal damage by inhibiting oxidative stress damage, inflammation, and apoptosis.
Materials and Methods
Animals
Fifty male C57BL/6 mice with specific pathogen-free (SPF) status, weighing between 18 and 22 g, were purchased from Beijing Huafukang Biotechnology Co., Ltd, with license number SCXK (Beijing) 2019-0008. They were acclimated for 1 week under environmental conditions at (25 ± 2)°C, where they had access to food and water.
Pharmaceuticals and Reagents
API (purity: ≥98%, CAS: 520-36-5) and AAI (purity ≥95%, CAS: No. 313-67-7) were purchased from Dosf Biotechnology Co., Ltd (Nanjing, China). Hematoxylin–eosin (HE) staining solution (CAS: D006-1-1), urinary protein measurement reagent kit (CAS: 20190611), serum creatinine (Scr) measurement reagent kit (CAS: 20190514), blood urea nitrogen (BUN) measurement reagent kit (CAS: 20181226), TNF-α measurement reagent kit (CAS: 20190115), IL-1β measurement reagent kit (CAS: 20190321), IL-6 measurement reagent kit (CAS: 20190406), superoxide dismutase (SOD) measurement reagent kit (CAS: A001-3-2), malondialdehyde (MDA) measurement reagent kit (CAS: A003-1), and glutathione (GSH) measurement reagent kit (CAS: A005-1-2) were from Nanjing Jiancheng Bioengineering Institute. Dapi (batch no. G1012), antifluorescence quenching tablet (batch no. G1401) were purchased from Wuhan Xavier Biotechnology Co., Ltd, and Tunel Kit (batch no. 11684817910) was purchased from Roche Co., Ltd. Rabbit antimouse Nrf2 antibody (CAS: 201903019), HO-1 antibody (CAS: 201905024), nuclear factor-κB (NF-κB) antibody (CAS: 201812005), Cleaved Caspase-3 antibody (CAS: 201901031), goat antirabbit immunoglobulin G (IgG) antibody (CAS: A0208).
Animal Treatment
The mice were separated into the control group, model group, and API-L, API-M, and API-H groups (10, 20, 40 mg/kg) (n = 10/each group). Each treatment group received the respective drug via oral gavage for 7 consecutive days. On the 5th day, 2 h after the last oral administration, except for the control group, all other groups received an intraperitoneal injection of AAI (10 mg/kg) for 3 days to establish an acute kidney injury model. After final administration, mice fasted for 12 h but had access to water. The protocols were approved by the Animal Experiment Committee of Wenzhou Medical University.
Measurement of 24-h Urinary Protein, Scr, and BUN Levels
After the final administration, 24-h urine samples were collected from each group of mice, and centrifuged to obtain the supernatant. Following the instructions provided with the assay kit, the 24-h urinary protein level was determined using the Coomassie brilliant blue method. Subsequently, the mice were anesthetized, and blood was drawn from the abdominal aorta, and centrifuged to obtain serum, which was used to measure Scr and BUN levels.
Renal Histopathological Examination and Detection of Cellular Apoptosis
After blood collection, the left kidney was placed in a 4% paraformaldehyde solution for fixation for 72 h. Following dehydration and clearing with xylene, the kidney was embedded to obtain sections with 3 µm thickness. Pathological examination: HE staining, xylene clarification, mounting with neutral resin, and observation of renal tissue pathological changes under a microscope. Cell apoptosis detection: The paraffin sections were dewaxed, dehydrated, rinsed, and rehydrated with distilled water, mixed with proteinase K and terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) reaction solution, incubated at 37°C for 2 h, and the nuclei were counterstained with Dapi, fluorescence microscope images, and photographs.
Biochemical Parameter Analysis of Renal Tissue
The right kidney was rinsed with precooled physiological saline at 4°C, minced, and homogenized in nine times the volume of precooled lysis buffer at 4°C. Tissue homogenates were centrifuged to collect the supernatant. Following instructions provided with the assay kit, the MDA content, SOD, and glutathione peroxidase (GSH-Px) activities were measured. TNF-α/IL-1β/IL-6 levels were detected using ELISA.
Detection of Protein Expression in Renal Tissue
The homogenate of the right kidney tissue was centrifuged to extract total protein, which was quantified and separated by 12% SDS-PAGE gel electrophoresis for western blot using antibodies (Nrf2 at 1:500, HO-1 at 1:1,000, NF-κB at 1:500, Cleaved Caspase-3 at 1:1,000, β-actin at 1:2,000). The grayscale values of each protein band were measured.
Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR)
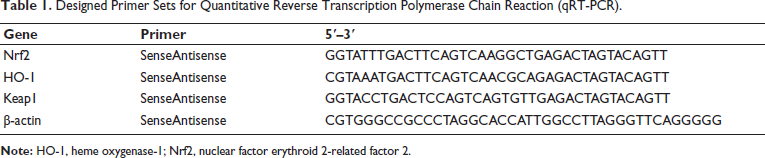
Ribonucleic acid (RNA) was isolated and synthesized into complementary deoxyribonucleic acid (cDNA), followed by polymerase chain reaction (PCR) amplification using primers, as shown in Table 1.
Designed Primer Sets for Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR).
Statistical Method
SPSS software analyzed data presented as mean ± standard deviation (SD) and assessed by Shapiro–Wilk test or analysis of variance (ANOVA). p < 0.05 refers to a difference.
Results
The 24-h Urinary Protein Levels and Scr and BUN Levels
A schematic diagram of the research route is shown in Figure 1. Model mice exhibited increased 24-h urinary protein levels and serum Scr and BUN levels (p < 0.01), which were decreased in API-M and API-H groups (p < 0.05) (Table 2).
Comparison of 24-h Urinary Protein Levels and Serum Scr and BUN Levels in Each Group of Mice (n = 10).
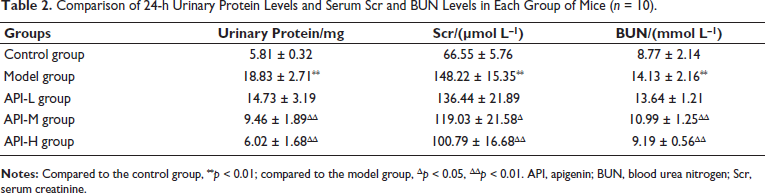
Schematic Diagram of Research Route.
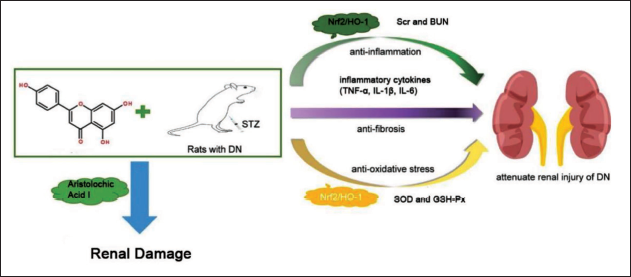
Renal Histopathological Examination
In the control group of mice, the renal glomeruli and tubular structures were clear, and the cellular matrix was evenly distributed. No abnormal findings were observed. The renal glomeruli showed swelling and a blurred capsule structure in the model group. Mouse protein casts were visible in the renal tubules, and there was inflammatory cell infiltration in the renal interstitium. Cell membrane rupture, nuclear condensation, and disappearance of cells were also observed. In the API-treated groups, the pathological changes in the kidneys of mice induced by AAI were improved. In the API-H group, there were no significant pathological changes such as glomerular swelling, protein casts, or inflammatory cell infiltration (Figure 2).
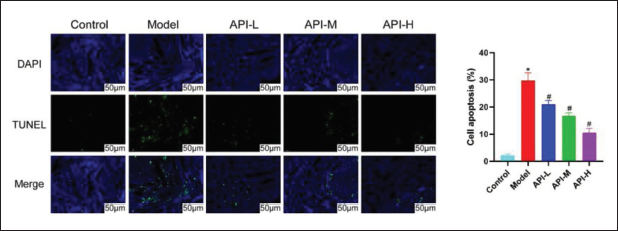
Apoptosis Status of Renal Cells
As shown in Figure 3, nuclei after Dapi counterstaining were blue upon UV excitation, and positive apoptotic nuclei were green. Compared with the control group, the number of apoptotic tubular epithelial cells in the model group was increased and decreased in Jingfang granule groups.
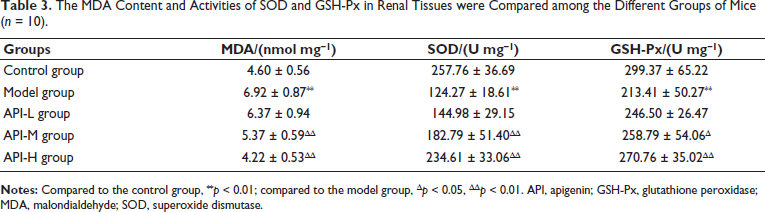
MDA/SOD/GSH-Px in the Renal Tissues
MDA content in renal tissues of the model group significantly increased, while SOD and GSH-Px activities decreased (p < 0.01), with opposite changes in API-M and API-H groups (p < 0.05) (Table 3).
The MDA Content and Activities of SOD and GSH-Px in Renal Tissues were Compared among the Different Groups of Mice (n = 10).
TNF-α/IL-1β/IL-6 Levels
Model mice exhibited upregulated TNF-α/IL-1β/IL-6 in renal tissues (p < 0.01), which were downregulated in API-M and API-H groups (p < 0.05) (Table 4).
The Levels of TNF-α, IL-1β, and IL-6 in Renal Tissues were Compared among the Different Groups of Mice (n = 10).
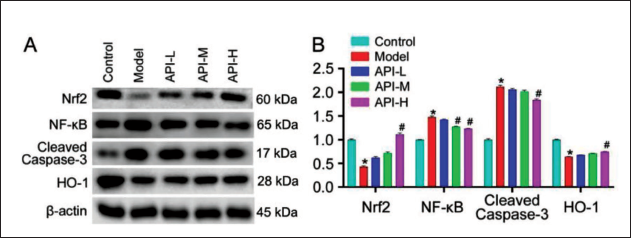
The Protein Expression of Nrf2/HO-1/NF-κB/Cleaved Caspase-3
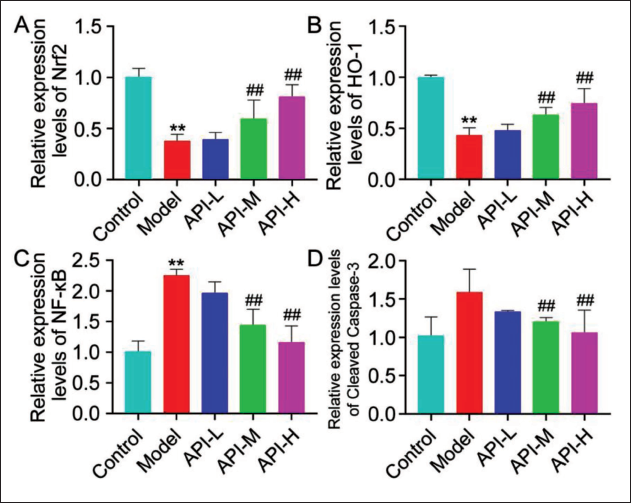
Nrf2 and HO-1 in renal tissues of the model group were downregulated, while NF-κB/Cleaved Caspase-3 was upregulated (p < 0.05), with opposite changes in API-M and API-H groups (p <0.05) (p < 0.05) (Figure 4).
Nrf2/HO-1/NF-κB/Cleaved Caspase-3 Gene Expression
Nrf2 and HO-1 gene levels in the model group were downregulated, while NF-κB/Cleaved Caspase-3 was downregulated (p < 0.01), with opposite changes in API-M and API-H groups (p < 0.01) (Figure 5).
Discussion
Chinese herbal medicines and patent medicines containing aristolochic acid are widely used in clinical practice and possess significant pharmacological activities (Cai et al., 2019; Mashour et al., 1998; Shaw, 2010). As one of the primary components of aristolochic acid, the nephrotoxicity of AAI has been widely recognized, posing a significant obstacle to the safe use of Traditional Chinese Medicine (Debelle et al., 2008). Currently, there is no established long-term, stable, and effective treatment regimen for AAI-induced kidney damage in clinical practice. We established a mouse model of kidney injury by intraperitoneal injection of AAI to preliminarily investigate the mechanisms by which API alleviates AAI-induced kidney injury.
API is a natural flavonoid compound, named for its highest concentration in celery. In this study, the treatment with API significantly reduced 24-h urine protein excretion, as well as the serum levels of Scr and BUN in mice with acute kidney injury are induced by AAI. Furthermore, it notably ameliorated the pathological structural changes in renal glomeruli and tubules and inhibited renal cell apoptosis. These indicate that API has a protective effect against AAI-induced kidney injury.
Researchers have demonstrated that oxidative stress is a key mechanism through which AAI exerts its cytotoxicity (Liu et al., 2009; Wu et al., 2015). AAI can induce reactive oxygen species (ROS) generation in renal tissues, disrupting the dynamic balance and leading to oxidative stress damage (Wu et al., 2015). Excessive production of ROS leads to the overconsumption of antioxidant enzymes (SOD, GSH-Px) that use ROS as substrates, resulting in ROS surplus (Bhattacharyya et al., 2014; Lubrano & Balzan, 2015). This attacks nucleic acids, proteins, and cell membranes and leads to lipid peroxidation damage, generating biotoxic MDA (Habashy et al., 2019). In a randomized experiment, when subjects were supplemented with celery containing 3.73–4.49 mg of API in their diet, SOD and GSH-Px activity in their red blood cells increased. This indicates that API can effectively enhance the body’s antioxidant capacity (Nielsen et al., 1999). TNF-α/IL-1β/IL-6 are inflammatory chemokines that not only trigger inflammatory responses and damage renal tissues but also induce the synthesis and release of inflammatory cytokines, thereby initiating a cascade of inflammatory reactions. IL-6 can directly activate inflammatory cells, amplifying the cascade of inflammation and exacerbating renal tissue damage (Kim et al., 2015). Previous studies have found that API partially inhibits the production of IL-1β/IL-6/TNF-α in splenic dendritic cells after lipopolysaccharide (LPS) stimulation, thereby improving cellular inflammation (Liu, Xue, et al., 2017). This study found that API significantly downregulated MDA, TNF-α/IL-1β/IL-6 in renal tissues of mice with aristolochic acid-induced kidney injury. It also enhanced SOD and GSH-Px activities, indicating that API has an inhibitory effect on oxidative stress and inflammatory response in renal tissues caused by aristolochic acid.
Apoptosis is a genetically regulated, programmed, and autonomous cell death process. In this process, Caspase-3, when activated by pathological stimuli such as cytochrome C release and calcium overload, produces Cleaved Caspase-3, making it the most crucial regulatory protein in the process of apoptosis (Glushakova et al., 2018). Existing research has indicated that API can modulate the intrinsic apoptotic pathway within cells (Seo et al., 2014). Under normal conditions, Nrf2 binds to inhibitor Keap1, rendering it inactive. However, factors like ROS can induce Nrf2 dissociation from Keap1, resulting in Nrf2 activation and translocation into the cell nucleus, where it participates in regulating the transcription and expression of antioxidant enzymes. HO-1, as a downstream gene of Nrf2, helps to inhibit oxidative stress (Turpaev, 2013). The antioxidant effect of API is mediated through Nrf-2 antioxidant signaling (Kooti & Daraei, 2017). NF-κB is inactive under physiological conditions but can be activated by ROS, leading to the induction and activation of Caspase-3, TNF-α, and IL-1β expression (Cai et al., 2018). In this study, API significantly upregulated Nrf2 and HO-1 proteins in renal tissue of mice with AAI-induced kidney injury, while downregulating NF-κB/Cleaved Caspase-3. These results suggest that API has an inhibitory effect on oxidative stress, inflammatory response, and cell apoptosis in mice with AAI-induced kidney injury via the Nrf2/HO-1 pathway.
Conclusion
API protects AAI-induced kidney injury in mice. Its mechanism of action may be associated with the activation of the Nrf2/HO-1 pathway, leading to inhibition of oxidative damage, inflammatory response, and cell apoptosis.
Footnotes
Abbreviations
BUN: Blood urea nitrogen; GSH-Px: Glutathione peroxidase; HO-1: Heme oxygenase-1; MDA: Malondialdehyde; NF-κB: Nuclear factor kappa B; Nrf2: Nuclear factor erythroid 2-related factor 2; Scr: Serum creatinine; SOD: Superoxide dismutase; TNF-alpha: Tumor necrosis factor alpha protein; TUNEL: Terminal deoxynucleotidyl transferase dUTP nick-end labeling.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the ethnic committee of Cangnan Hospital Affiliated to Wenzhou Medical University.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
