Abstract
Pterostilbene (PTE), a natural plant extract, has an anti-inflammatory effect; however, whether PTE could protect nucleus pulposus cells (NPCs) in the intervertebral disk from inflammation remains unclear. Primary NPCs isolated from Sprague-Dawley (SD) rats were cultured, and Cell Counting Kit-8 (CCK-8) analysis was used to test the cytotoxicity of PTE. The effect of PTE on interleukin-1β (IL-1β)-induced inflammation was analyzed using an enzyme-linked immunosorbent assay, real-time polymerase chain reaction (PCR), and a Griess test. Western blotting, immunofluorescence, and a nuclear factor erythroid 2-related factor 2 (Nrf2) small interfering RNA (siRNA) transfection were used to assess the involvement of Nrf2 in the anti-inflammatory mechanism of PTE on NPCs. The results of the CCK-8 analysis showed that PTE produced no cytotoxicity in NPCs at 20 μM for 24 h. PTE suppressed the production of nitric oxide (NO) and prostaglandin E2 (PGE2) and inhibited the messenger RNA (mRNA) expression of cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS) induced by IL-1β. PTE could promote the nuclear translocation of Nrf2 in NPCs. In addition, Nrf2 silence reversed the inhibitory effect of PTE on the production of NO and PGE2 and the expression of COX-2 and iNOS. These results indicate that PTE inhibits inflammation in the rat nucleus pulposus by promoting the nuclear translocation of Nrf2.
Introduction
Intervertebral disk degeneration (IDD) is an important cause of back pain and is the pathophysiological basis of various degenerative spinal diseases, such as intervertebral disk herniation, spinal stenosis, and lumbar spondylolisthesis.1,2 Nucleus pulposus cells (NPCs) are the only constitutive cell of the nucleus pulposus tissue. They have an important role in the intervertebral degeneration process. In recent studies, there is mounting evidence indicating a close relationship between IDD and intervertebral disk inflammation. 3 In the degenerative intervertebral disk, there is a high level of expression of inflammatory mediators such as interleukin-1β (IL-1β), cyclooxygenase-2 (COX-2), nitric oxide (NO), prostaglandin E2 (PGE2), and tumor necrosis factor-α (TNF-α).3,4 The increase in inflammatory mediators in the nucleus pulposus tissue can cause an increase in different matrix metalloproteinases (MMPs), which can cause degradation of the nucleus pulposus extracellular matrix (ECM) and the degeneration of the intervertebral disk. Therefore, relieving inflammation in NPCs is a critical step for effective treatment, but an effective medicine is not yet available.
As a natural constituent of blueberries, pterostilbene (PTE) is a phytoalexin similar to resveratrol, and it has significant biological activity. 5 According to recent studies, PTE has anti-inflammatory, anti-oxidative, and anti-apoptotic effects. For instance, PTE can inhibit the generation of IL-1β, TNF-α, MMPs, and the synthesis of reactive oxygen species in endothelial cells. 6 It has also been reported that PTE has certain helpful effects in cancer treatment and hyperlipidemia treatment. 7 However, the effect of PTE in NPCs has not yet been studied. IL-1β is a common inflammatory mediator that induces inflammation of the intervertebral disk. 8 It is used in this study to induce inflammation in NPCs.
Nuclear factor erythroid 2-related factor 2 (Nrf2) is a transcription factor of the class I basic leucine zipper. It regulates the expression of antioxidant proteins that protect against oxidative damage triggered by injury and inflammation. 9 When cells are stimulated with inflammation, Nrf2 in the cytoplasm begins nuclear transcription and promotes transcription related to the antioxidant response element, such as that of heme oxygenase-1, nicotinamide adenine dinucleotide phosphate (NAD(P)H), and catalase. 10 The protective effect of PTE in the liver, brain, and heart has already been reported, whereas its effect in the intervertebral disk is unknown. It has been reported that Nrf2 plays a role in the anti-apoptotic and anti-oxidative properties of PTE, but how this relates to anti-inflammatory processes in NPCs is not known.
Materials and methods
The animal experiments in this study were approved by the Fudan University Zhongshan Hospital Animal Ethics Committee. This article does not contain any studies with human participants performed by any of the authors.
Reagents and antibodies
PTE and collagenase (type II) were purchased from Sigma. Nrf2, β-actin, Lamin B, and PGE2 enzyme-linked immunosorbent assay (ELISA) kits were all purchased from Abcam. Immunofluorescence-related reagents, western blotting–related reagents, 4′,6-diamidino-2-phenylindole (DAPI), CelLytic™ NuCLEAR™ Extraction Kit, and a Nitric Oxide Assay Kit were all purchased from Beyotime; Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), and 0.25% trypsin–ethylenediaminetetraacetic acid (EDTA) were purchased from Gibco; Cell Counting Kit-8 (CCK-8) was purchased from Dojindo; TRIzol and SYBR Premix Ex Taq were purchased from Takara; and a complementary DNA (cDNA) synthesis kit was purchased from MBI Fermentas.
Cell culture
A total of 30 male Sprague-Dawley (SD) rats (3 months old, 250–300 g) were administered 3.5 mg of chloral hydrate per kilogram of body weight to perform euthanasia. Under aseptic conditions, the L1–L6 lumbar vertebrae were dissected. The excess muscular tissue was removed after cleaning using phosphate-buffered saline (PBS). The gelatinous nucleus pulposus tissue was isolated from each segment and processed using 0.1% class II collagenase at 37°C for 4 h. After that, the samples were filtered using a 200 µm filter. The cells were maintained in DMEM in the presence of 1% penicillin–streptomycin and 10% fetal bovine serum at 37°C with 5% carbon dioxide. The cells used in the following experiments were all second generation.
Experimental design
To analyze the inhibiting effect of PTE on NPC inflammatory mediators, the cells were divided into a control group, an IL-1β group, a PTE group, and an IL-1β + PTE group. To analyze the effect of Nrf2 on the PTE inhibition of inflammatory mediators, the cells were divided into an IL-1β group, an IL-1β + PTE group, an IL-1β + PTE + control small interfering RNA (siRNA) group, and an IL-1β + PTE + Nrf2 siRNA group. PTE was added to the culture medium 2 h prior to the IL-1β. PTE was dissolved in dimethylsulfoxide and then diluted in medium to treat cells.
CCK-8 analyses
NPCs were incubated overnight in 96-well culture plates at a density of 5 × 103 cells per well. After that, 1, 5, 10, 20, 40, 60, or 80 μmol/L of PTE was added for 24 h or 20 μmol/L of PTE was added for 2, 4, 6, 12, 24, 36, or 72 h. The medium in each well was then exchanged for 100 µL of fresh DMEM. After that, 10 μL of the CCK-8 reagent was added into each well and incubated at 37°C for 1 h. Then, the optical density was recorded using an enzyme mark instrument at a 450 nm wavelength. The results are presented as a relative value based on the control.
Immunofluorescence
After incubation with 10 or 20 μmol/L of PTE for 24 h, the NPCs were fixed using 4% paraformaldehyde for 10 min at 4°C. Then, the cells were washed thrice using PBS. After washing, the cells were incubated for 30 min with 2% Triton X-100 to permeabilize the cell membranes and were then incubated for 30 min with 5% bovine serum albumin to block non-specific antigens. Then, the cells were incubated overnight with an Nrf2 (1:100 dilution) primary antibody at 4°C. After that, the cells were washed thrice using PBS and incubated with a fluorescein isothiocyanate (FITC)-labeled secondary antibody for 2 h at room temperature. Finally, the NPCs were observed using fluorescence microscopy after DAPI nuclear staining and a PBS wash.
Western blotting
Using the method suggested by the manufacturer (Beyotime), the proteins from the cell nucleus and the cell plasma were extracted, and the protein concentration was determined using a bicinchoninic acid (BCA) assay. The nuclear proteins and the plasma proteins underwent electrophoretic separation on a sodium dodecyl sulfate–polyacrylamide gel. Then, the proteins were transferred onto a polyvinylidene difluoride membrane (Bio-Rad) using the wet phase inversion method. The membranes were incubated in 5% skim milk for 2 h and then incubated overnight at 4°C with an Nrf2 primary antibody (1:1000 dilution). They were then washed thrice using Tris-buffered saline (TBS) containing 0.1% Triton X-100 (TBST) and incubated with a horseradish peroxidase–labeled secondary antibody for 2 h. Then, the membranes were exposed on an imaging system (PerkinElmer) with an electrochemical luminescence (ECL) reagent (Thermo). The semi-quantitative analysis of the protein bands was performed using AlphaEaseFC 4.0 software.
Real-time polymerase chain reaction
After performing the experiments as described above, the total RNA from NPCs in six-well culture plates was extracted using the TRIzol method. After a concentration and purity test, cDNA was synthesized through reverse transcription using 1 μg of total RNA. The reaction mixtures were prepared prior to the start of the reaction as required. A total of 20 μL of the reaction solution was used in this experiment. The reaction mixture contained 2 μL of 1:2 diluted cDNA, 10 μL of SYBR Premix Ex Taq solution, 0.2 μmol/L of primers, and the rest consisted of enzyme-free sterile water. The primers for COX-2, inducible nitric oxide synthase (iNOS), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) are shown in Table 1. The above 20 μL reaction system was added to eight tubes and placed in a LightCycler real-time polymerase chain reaction (PCR) system (Roche). The reaction procedure was 95°C pre-denaturation for 10 min, 95°C denaturation for 15 s, 56°C annealing for 1 min, and 72°C extension for 1 min for a total of 40 cycles. After the reaction, the Ct value was derived and normalized according to the internal reference gene GAPDH. The semi-quantitation of the messenger RNA (mRNA) was performed using the 2−ΔΔCt method.
Primer sequences for COX-2, iNOS, and GAPDH.

COX-2: cyclooxygenase-2; iNOS: inducible nitric oxide synthase; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
NO level determination
The NO level of the culture medium serum was determined using the classic Griess method. After performing the experiments as described above, the supernatant was collected using the method suggested by Beyotime, and then, it was added to a 96-well culture plate. The Griess reagents I and II were each added to the supernatant in the 96-well culture plate and incubated for 3 min at 37°C. The absorbance was detected using a microplate reader at a 540 nm wavelength. The NO level was calculated using a standard curve.
ELISA analysis
After performing the experiments as described above, the supernatants were collected from the cells. The PGE2 concentration was assessed according to the ELISA method suggested by Abcam. The sensitivity of the PGE2 ELISA reagent was 13.4 pg/mL.
Nrf2 siRNA transcription
Nrf2 siRNA was designed and the details are listed in Table 2. The siRNA was synthesized by Shanghai GenePharma. A total of 2 × 105 NPCs were added to each well of a six-well culture plate and incubated overnight. The transfection was conducted using Lipofectamine 2000 according to the instructions provided by Invitrogen. Non-specific non-targeting siRNA was used as control RNA. After transfection, nuclear proteins were extracted for western blotting to examine the effects of the transfection.
The sequences of small interfering RNAs (siRNAs).

Statistical analysis
The differences between the groups were analyzed using analysis of variance (ANOVA) using the variance analysis with the SPSS15 software package (SPSS Inc.). If the variance analysis was statistically significant, the Bonferroni method was used for the analysis of the differences between two groups. P < 0.05 was considered statistically significant.
Results
The influence of PTE on the cell viability of primary NPCs
A CCK8 assay showed that the cell viability of NPCs treated with PTE at an 80 µmol/L concentration was significantly decreased after 24 h (P < 0.05, Figure 1(a) compared with the control group, and 1–60 µmol/L PTE produced no obvious change in the cell viability of NPCs. When PTE was administered at 20 µmol/L, it was found that the cell viability significantly decreased at 72 h only (P < 0.05, Figure 1(b)), suggesting that a 20 µmol/L PTE treatment has no effect on NPC viability at 0–48 h. Therefore, a 20 µmol/L PTE treatment for 24 h was set as the dosage and treatment duration for the follow-up experiments.
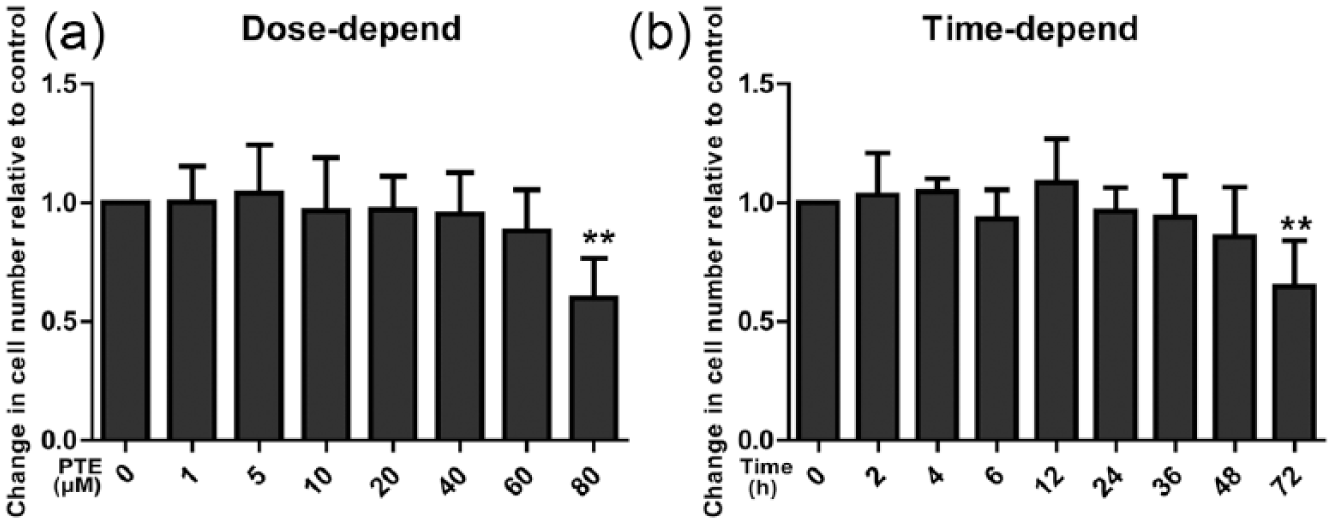
Effect of pterostilbene (PTE) on the viability of nucleus pulposus cells (NPCs). (a) NPC viability after PTE treatment for 24 h. (b) NPC viability after a 20 µmol/L PTE treatment for different times (**P < 0.01, compared with control group, n = 6.)
PTE inhibited the level of inflammatory mediators induced by IL-1β in NPCs
NO, PGE2, COX-2, and iNOS are common inflammatory mediators, which were used in this study to determine the role of PTE in NPC inflammation. IL-1β significantly stimulated NPCs to produce a significant quantity of iNOS, the key enzyme for the production of NO (P < 0.05, Figure 2(c)) and COX-2 and PGE2 (P < 0.05, Figure 2(a) and (b)). Furthermore, it also increased the expression of COX-2 mRNA (P < 0.05, Figure 2(d)). When the PTE and IL-1β were administered separately, it was found that 20 µmol/L of PTE alone had no effect on the inflammatory mediators. However, when the combination of IL-1β and PTE was administered, PTE significantly inhibited the expression of the inflammatory mediators induced by IL-1β (P < 0.05, Figure 2(a)–(d)).

Effect of PTE on inflammatory mediators in nucleus pulposus cells (NPCs) treated using IL-1β. (a) NO levels in the cell supernatant. (b) PGE2 levels in the cell supernatant. (c) COX2 mRNA expression in NPCs. (d) iNOS mRNA expression in NPCs (*P < 0.05, **P < 0.01, n = 6).
PTE promotes the nuclear translocation of Nrf2 in NPCs
In order to study the mechanism of PTE inhibition of NPC inflammation, further analysis of Nrf2 nuclear translocation was undertaken. A western blot analysis found that with treatment from 10 to 40 µmol/L of PTE for 24 h, as the concentration of PTE was increased, Nrf2 expression in the nucleus also increased, while the expression of Nrf2 in the cytoplasm gradually decreased, especially at the concentrations of 20 and 40 µmol/L of PTE (P < 0.05, Figure 3(a)–(d)), suggesting that PTE can promote the nuclear expression of Nrf2 for nuclear transcription. A cell immunofluorescence study showed that Nrf2 is mainly distributed in the cell cytoplasm and barely appears in the nucleus in untreated cells. Following treatment with 10 µmol/L of PTE for 24 h, the expression of Nrf2 in the nucleus increased and decreased in the cytoplasm gradually. Following treatment with 20 µmol/L of PTE for 24 h, Nrf2 was mainly expressed in the nucleus and expression decreased significantly in the cell cytoplasm, which further illustrates that PTE promoted the nuclear translocation of Nrf2 in NPCs.

Stimulatory role of PTE in the nuclear translocation of Nrf2 in nucleus pulposus cells (NPCs). (a) Nuclear Nrf2 expression in NPCs. (b) Cytoplasmic Nrf2 expression in NPCs. (c) Semi-quantitative analysis of nuclear Nrf2 expression. (d) Semi-quantitative analysis of cytoplasmic Nrf2 expression (*P < 0.05 and **P < 0.01, compared with control group). (e)–(m) Immunofluorescence analysis for the expression of Nrf2 (n = 6).
The protection produced by PTE against inflammation in the NPCs was inhibited by downregulating Nrf2
To further study the role of Nrf2 in the PTE-mediated reduction of inflammation in NPCs, RNA interference (RNAi) was used to downregulate the expression of Nrf2 in the nucleus. After transfection using Nrf2 siRNA for 48 h, the cells were then treated using 20 µmol/L of PTE for 24 h. A western blot analysis showed that the expression of Nrf2 in the nucleus was significantly decreased compared with the control siRNA group, whereas in the group without PTE treatment it was also markedly decreased (P < 0.05, Figure 4(a) and (b)). On treatment using IL-1β and PTE, the expression of NO, PGE2, COX-2, and iNOS was increased significantly in the silenced Nrf2 group compared with untreated cells. Therefore, downregulation of Nrf2 could reverse the inhibitory effects of PTE on these inflammatory mediators.

Inhibitory role of Nrf2 silence in the protective effect of PTE against inflammatory mediators in nucleus pulposus cells (NPCs). (a) Nuclear Nrf2 expression in NPCs. (b) Semi-quantitative analysis of nuclear Nrf2 expression. (c) NO levels in the cell supernatants. (d) PGE2 levels in the cell supernatants. (e) COX2 mRNA expression in NPCs. (f) iNOS mRNA expression in NPCs (*P < 0.05, **P < 0.01, n = 6).
Discussion
Lower back pain is one of the most common musculoskeletal diseases, and IDD is the main cause of lower back pain. Therefore, relieving IDD has an important clinical significance. Numerous studies have focused on relieving IDD, but the mechanism of IDD remains unclear, and a reliable and effective drug to treat IDD is not available. In recent studies, inflammation in NPCs and the ECM has been reported as a major factor in IDD. In this study, PTE, a natural anti-inflammatory and anti-catabolic agent, was used to inhibit the production of inflammatory mediators and prevent the production of MMPs and subsequent degradation of the ECM. Therefore, it is meaningful to explore the effect of PTE on inflammation in NP cells.
Since this is the first study investigating the influence of PTE on primary NPCs, the effect of PTE on the viability of NPCs was investigated. It was observed that there was no influence on cell viability when 20 µmol/L of PTE was applied for 24 h. Likewise, Nikhil et al. 11 found that there was no cytotoxic effect in rat macrophages when 1–100 µmol/L of PTE was applied for 24 h, but when applied for 72 h, 30 µmol/L of PTE inhibited the macrophage viability.
IL-1β is a classic cytokine that promotes inflammation and catabolism in NPCs. In this study, it was discovered that IL-1β can promote the expression of COX-2, NO, PGE2, and iNOS. COX-2 and PGE2 are important mediators of discogenic pain caused by inflammation. 3 The potential for PTE to inhibit the expression of inflammatory mediators induced by IL-1β makes PTE a possible drug to relieve discogenic pain.
There are multiple signaling pathways involved in the protective effect of PTE such as deacetylase sirtuin 1, extracellular signal–regulated kinase, and nuclear factor kappa B (NF-ĸB). 12 However, Nrf2 is considered a signaling pathway with more translational potential. As an endogenous protective mechanism, the expression of Nrf2 features in multiple diseases involving inflammation and oxidative stress. At affected sites of the brain in Alzheimer’s disease patients, the expression of Nrf2 in the cell nucleus decreases significantly, whereas Nrf2 is highly expressed in the cell cytoplasm of healthy subjects. 13 Inflammation and oxidative stress in the rat liver caused Nrf2 in the cell nucleus to decrease significantly. 14 At the hypertensive mesenteric artery, the nuclear translocation of Nrf2 decreased significantly and accompanied an increase in oxidative stress. 15
Under normal physiological conditions, Nrf2 is mainly located in the cytoplasm and is integrated with Kelch-like ECH-associated protein-1 (Keap1). Therefore, separating Nrf2 from Keap1 is the first critical step to activate Nrf2 and induce its nuclear translocation. 16 According to the National Cancer Institute’s Thesaurus, the nuclear translocation of Nrf2 is a process in which the Nrf2 was transported in the nuclear, leading to the increase in nuclear expression and decrease in cytoplasmic expression of Nrf2. Using a luciferase reporter gene, Ramkumar et al. 16 discovered that PTE can disturb the relationship between Keap1 and Nrf2 and induce nuclear translocation of Nrf2. In this study, it was demonstrated that PTE can induce the nuclear translocation of Nrf2 in NPCs. After nuclear transcription, Nrf2 integrates with the antioxidant response element and forms a dimer and then combines with transcription c-Jun protein and Maf protein family members (G/F/K) to facilitate the transcription of anti-oxidation genes in order to work against the products of oxidative reactions, such as free oxygen radicals, so that inflammatory reactions can be prevented. 10 In this study, it is likely that the activation of Nrf2 also prevented inflammatory reactions through anti-oxidation effects.
In summary, PTE does not affect the viability of NPCs within a certain concentration range, and PTE can significantly inhibit the formation of NO and PGE2 induced by IL-1β and decrease levels of COX-2 and iNOS mRNA. PTE inhibits these inflammatory mediators by inducing the nuclear translocation of Nrf2. Therefore, PTE has considerable protective effects against inflammation in NPCs and has significant potential as a treatment for IDD.
Footnotes
Acknowledgements
S.L., L-B.J, and S-H.L. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The research was supported by the National Natural Science Foundation of China (81371376).
