Abstract
The aim of the study was to investigate the protective effect of pyrrolidine dithiocarbamate (PDTC) against methotrexate (MTX)-induced testicular damage in rats. Forty Wistar albino male rats were divided into equally four groups: Control group (saline solution, IP), PDTC group (100 mg/kg PDTC,IP, 10 days), MTX group (20 mg/kg MTX, IP, single dose, on the 6th day) and MTX + PDTC group (100 mg/kg PDTC, IP, 10 days and 20 mg/kg MTX, IP, single dose, on the 6th day). After 10 days, testicular tissues were excised for morphometric, histological and immunohistochemical evaluations. Serum testosterone, follicle stimulating hormone (FSH), luteinizing hormone (LH) and prokineticin 2 (PK2) levels were determined. Body and testicular weights were measured. Testicular damage was assessed by histological evaluation. Nuclear factor kappa B (NFkB), nuclear factor erythroid 2 related factor 2 (Nrf2) and PK2 immunoreactivities were evaluated by HSCORE. Body and testicular weights, serum FSH, LH, testosterone levels, seminiferous tubule diameter and germinal epithelial thickness were significantly decreased in the MTX group. However, serum PK2 level, histologically damaged seminiferous tubules and interstitial field width were significantly increased. Additionally, there was an increase in NFkB and PK2 immunoreactivity, whereas there was a significant decrease in Nrf2 immunoreactivity. PDTC significantly improved hormonal, morphometric, histological and immunohistochemical findings. Taken together, we conclude that PDTC may reduce MTX-induced testicular damage via NFkB, Nrf2 and PK2 signaling pathways.
Keywords
Introduction
According to the World Health Organization, infertility is major health challenge that affects 10–15% of couples and 50–80 million people worldwide. 1 It is thought that 50% of infertile couples are male factor infertility. Only male factor infertility is responsible for 20% of infertility cases, whereas 30% of infertility cases are accompanied by male and female factor infertility. 2 Chemotherapy-related gonadal dysfunction is one of the etiological factors implicated in the development of male factor infertility. It may result in temporary or permanent infertility depending on the type of drugs and the cumulative dosage administered. 3
Methotrexate (MTX), a folic acid antimetabolite, is a chemotherapeutic agent widely used in broad spectrum treatment of malignant and non-malignant diseases, due to its antitumoural, anti-inflammatory, antimicrobial and immunosuppressive properties. 4 MTX demonstrates its antiproliferative effect by causing cytotoxicity during the active periods of cell proliferation. Therefore, it has a toxic effect not only on cancer cells, but also on bone marrow, gastrointestinal mucosa, hair follicles and spermatogenic cells with high proliferation ability. 5 Toxicity of spermatogenic cells to the testis in which it is produced is important and this is because it may cause gonadal dysfunction and subsequent infertility. 3 Accordingly, MTX-induced testicular toxicity has been shown to rapidly cause increasing in abnormal sperm morphology and decreasing in sperm count.4-6 It has also been reported to cause a decrease in testicular weight, follicle stimulating hormone (FSH), luteinizing hormone (LH) and testosterone levels.6-8 It also causes testicular degeneration.9-12
The exact mechanisms of MTX toxicity are still elusive. A well-known mechanism of MTX toxicity is the inhibition of dihydrofolate reductase (DHFR).13,14 It has been reported that MTX not only inhibits DHFR inhibition but also inhibits non-DHFR dependent metabolic pathways, thereby triggering oxidative stress in cells. 13 Studies have shown that MTX enhances the effects of reactive oxygen species (ROS) by reducing the antioxidant capacity of cells. 15-17 Antioxidant defence decreases with the decrease in expression of nuclear factor erythroid 2 related factor 2 (Nrf2), which acts as an important nuclear transcription factor that upregulates antioxidant/detoxification genes transcription to scavenge ROS, thus preventing or ameliorating oxidative stress.11,18 Accordingly, increased oxidative stress cells trigger inflammation via activation of nuclear factor kappa B (NFkB), which acts as a transcription factor for proinflammatory genes. In addition, prokineticin 2 (PK2), an adipocytokine induced by oxidative stress, has mainly angiogenic and mitogenic survival roles, although it has attracted attention due to its proinflammatory properties in recent years.20-23
Given these mechanisms, new agents are therefore required to reduce the organ damage caused by oxidative stress and oxidative stress–induced inflammation caused by cancer cells and chemotherapeutic agents. Pyrrolidine dithiocarbamate (PDTC) is among the dithiocarbamates (DTCs) class of thiol compounds.24,25 As a specific inhibitor of NFkB, it is a metal chelator with strong anti-inflammatory properties. It is also a potent antioxidant.24-27 Studies have reported that PDTC protects and improves histopathology by reducing oxidative stress in organ damage caused by various agents.25,28,29 To the best of our knowledge, no study has examined the protective effect of PDTC on testicular damage caused by MTX.
We hypothesized that MTX induces testicular damage by triggering oxidative stress and consequent inflammation however; PDTC may ameliorate this damage. In the present study, we purposed to evaluate the immunoreactivity of NFkB, Nrf2 and PK2, as well as testicular histology in order to investigate the protective effect of PDTC on testicular damage induced by MTX. Furthermore, serum reproductive hormones (FSH, LH and testosterone) and proinflammatory PK2 level were assayed.
Materials and methods
Chemicals
MTX (50 mg/mL flk) was purchased from Kocak Pharma (Tekirdag, Turkey), and PDTC was obtained from Sigma-Aldrich (St. Louis, MO, USA).
Animals
Approval of the study was obtained from the Experimental Animals Local Ethics Committee (Protocol no: 2017/34). A total of 40 Wistar albino male rats (age = 3 months; weight = 250–300 g) with similar biological and physiological features and living under standard laboratory conditions at Trakya University Experimental Animal Research Unit (22 ± 10C room temperature, 60% humidity, 12 h light/dark rhythm) were randomly selected.
Experimental design and treatment
The rats were divided into four groups of 10 animals each.
The control group was intraperitoneally (IP) administered saline twice daily for 10 days; for the PDTC group, PDTC (Sigma-Aldrich) was dissolved in saline, and 100 mg/kg PDTC was IP administered for 10 days28,30,31; The MTX group was IP administered a single dose of 20 mg/kg MTX (50 mg/mL flk, Kocak Pharma) on the 6th day8,32; for the MTX + PDTC group, PDTC was dissolved in saline, 100 mg/kg PDTC was IP administered for 10 days and a single dose of 20 mg/kg MTX (50 mg/mL flk) was IP administered on the 6th day. Daily total dose of 100 mg/kg PDTC was divided into two equal dose and IP administered.
Sample collection
Twenty-four hours after the last dose of PDTC, blood samples were collected from the animals by cardiac puncture prior to their sacrifice under the general anesthesia with intramuscular 5 mg/kg xylazine (Basilazin, Bavet, Bösensell, Germany) and 50 mg/kg ketamine (Ketasol, Richter Pharma AG, Wels, Austria). Serum samples were obtained by centrifuging blood samples at 4000 r/min for 10 min and stored/frozen at −80°C for Enzyme-Linked Immunosorbent Assay (ELISA). After sacrifice by cervical dislocation, the testes of the animals were excised and fixed with neutral 10% formalin (Sigma-Aldrich). Routine histological tissue procedure steps were performed, and paraffin blocks were obtained. Five micrometer (5 μm) sections were placed on slides from these paraffin blocks. The slides were stained with hematoxylin and eosin (H&E). H&E stained preparations were evaluated histologically under a light microscope (Olympus BX51, Tokyo, Japan), and findings were photographed at different magnifications (Olympus DP20 microscopic digital camera system).
Measurement of serum reproductive hormones and PK2 level
Ready-made kits were procured and used to measure FSH, LH and PK2 (Elabscience, Houston, TX, USA) as well as total testosterone (MyBioSource, San Diego, CA, USA) levels in serum samples of experimental animals by ELISA. Analysis of each hormone was carried out according to the manufacturer’s instructions.
Morphometric analysis
The photographs of H&E stained preparations were added to Imaging Analysis System (version 2.11.5.1, Camera-Argenit, Istanbul, Turkey). Morphometric analysis was performed by measuring seminiferous tubule diameter, germinal epithelial thickness and interstitial field width between the two seminiferous tubules at ×100 or ×400 magnification. For the seminiferous tubule diameter and thickness of germinal epithelium, 100 tubules randomly selected near each round or round of each animal were measured and averaged by two different researchers. The interstitial field width was similarly performed by both researchers with 100 different measurements of distance between the two seminiferous tubules and averaged. The measurement of each parameter was performed by evaluating five testicle sections and 20 different areas for each animal.19,33
Evaluation by histology and immunohistochemistry
Six testis H&E stained preparations for each animal were examined by histology according to the following criteria at ×100 magnification under a light microscope (Olympus BX51): detachment (separation of spermatocyte cell mass from the seminiferous epithelium); sloughing (release of germ cell cluster from the seminiferous epithelium to the lumen) and vacuolization (appearance of empty spaces in the seminiferous tubules). The average percentage of normal or damaged tubules for each parameter was determined. The average percentage was calculated for each section by dividing the number of normal or damaged (detachment, sloughing and vacuolization) round tubules by total round tubules in the same field and results were multiplied by 100. These evaluations were performed on three different fields on each section and averaged. 34
For evaluation by immunohistochemistry, 5 μm testes sections placed on positive charged slides were incubated overnight at 56°C and were left to cool for 10 min at room temperature. The slides were deparaffinized with toluene (Merck Millipore, Darmstadt, Germany) for 20 min twice and were passed through decreasing dilution alcohol series (100%, 96%, 90%, 80% and 70%). Afterwards, they were rehydrated for 5 min in distilled water. The slides were placed in citric acid buffer solution (10 mM, pH 6.0, Thermo Scientific/Lab Vision) and boiled for 20 min in a microwave for antigen retrieval. Afterwards, they were left to cool for 20 min at room temperature. The slides were washed thrice for 5 min with phosphate buffer solution (phosphate buffer saline, PBS, Sigma-Aldrich) and around the testis sections were circumscribed with hydrophobic PAP Pen (Sigma-Aldrich). They were placed in a humidity chamber at room temperature for the next steps and first treated with hydrogen peroxide (Thermo Scientific/Lab Vision) for 15 min to prevent endogenous peroxidase activity. After the washing with PBS, blocking solution (Ultra V Block, Thermo Scientific/Lab Vision) was dropped for 5 min in order to block non-specific antigens. After removal of the blocking solution without washing, NFkB/p65 primary antibody (polyclonal rabbit antibody at 1/100 dilution, [Rel A] Ab-1 Cat. #RB-1638 P1, Thermo Scientific/Lab Vision) was dropped and incubated for 1 h at room temperature. Nrf2 and PK2 primer antibodies (polyclonal rabbit antibody at 1/400 dilution, [ab137550], [ab76747], Abcam, Cambridge, MA, USA) were also incubated overnight at 4°C. Negative controls were obtained by dropping PBS into sections instead of primary antibodies. After incubation of the primary antibodies was completed, they were washed with PBS thrice for 5 min. Then, biotinylated secondary antibody (Biotinylated Goat Anti-Polyvalent, Thermo Scientific/Lab Vision) was added for 10 min at room temperature. After washing with PBS, streptavidin peroxidase (Streptavidin Peroxidase, Thermo Scientific/Lab Vision) was added for 10 min at room temperature. The sections were chromogenized with 3-amino-9-ethylcarbazole (AEC, Thermo Scientific/Lab Vision). Immunoreactivity was observed within 5–10 min in each slide and slides were immersed in distilled water, and reaction was stopped. No immunoreactivity was observed in the negative control sections. Afterwards, opposite staining was performed with hematoxylin, and slides were washed with tap water. After slides were dried, they were covered up cover slip with water-based solution (Thermo Scientific/Lab Vision).28,35
As a result of immunohistochemical staining, NFkB, Nrf2 and PK2 immunoreactivities were semi-quantitatively evaluated under light microscope at ×400 magnification by the HSCORE method. HSCORE value was calculated by the formula: HSCORE = ∑ i x Pi. For this scoring, it was calculated by summing the percentages of cells stained at each intensity score (Pi) and multiplying the value by degree of staining (i; 0 = no staining, 1 = weak staining, 2 = moderate staining, 3 = intense staining) for each animal. In each slide, five randomly selected areas were evaluated by two independent observers and averaged. 35
Statistical analysis
Statistical Package for the Social Science (SPSS) version 20 for Windows (IBM SPSS Statistics; Armonk, NY, USA) was used for all statistical analysis. Values were expressed as mean ±standard deviation (±SD). A value of p < 0.05 was considered statistically significant. The suitability of data to normal distribution was determined with a single sample by the Shapiro–Wilk test. One-way analysis of variance was used to compare values among groups. When there was a significant difference among groups, Tukey’s or Tamhane’s multiple comparison tests were applied. Tukey’s test was applied where group variances are homogeneous, while Tamhane’s multiple comparison test was used where variances are not homogeneous.
Results
Effect of PDTC on body weight, total testis weight and testis/body weight ratio in MTX-induced rats
Comparison of control and experimental groups’ body weight (BW) changes, total testicular weight (TW) and testicular weight/body weight ratios (TAI = TA/VA × 100).

Values are presented as mean ± SD.
∗p < 0.001, ∗∗p < 0.01, ∗∗∗p < 0.05, versus control group.
ap < 0.001, bp < 0.01, cp < 0.05, versus PDTC goup.
dp < 0.05, versus MTX group.
Effect of PDTC on serum FSH, LH, Testosterone and PK2 levels in MTX-induced rats
FSH, LH and testosterone levels
The mean FSH levels were 24.24 ± 4.81 ng/mL in the control group, 25.66 ± 5.8 ng/mL in the PDTC group, 8.14 ± 1.76 ng/mL in the MTX group and 18.62 ± 2.78 ng/mL in the MTX + PDTC group. The mean LH levels were 7.89 ± 1.27 mIU/mL in the control group, 7.98 ± 0.43 mIU/mL in the PDTC group, 2.64±0.79 mIU/mL in the MTX group and 5.35 ± 0.68 mIU/mL in the MTX + PDTC group. The mean testosterone levels were 7.65 ± 1.06 ng/mL in the control group, 7.97 ± 1.68 ng/mL in the PDTC group, 3.05 ± 0.64 ng/mL in the MTX group and 5.60 ± 1.32 ng/mL in the MTX + PDTC group. Serum FSH, LH and testosterone levels were significantly decreased in the MTX-treated groups compared to control and PDTC groups (p < 0.001). FSH, LH and testosterone levels were significantly increased in the MTX + PDTC group compared to the MTX group (Figure 1). The effect of pyrrolidine dithiocarbamate on serum follicle stimulating hormone, luteinizing hormone, testosterone and prokineticin 2 levels in methotrexate-treated rats (mean ± SD). (∗p < 0.001, ∗∗p < 0.01, ∗∗∗p < 0.05, vs control group), (†p < 0.001, ††p < 0.01, †††p < 0.05, vs PDTC group), (#p < 0.001, ##p < 0.01 vs MTX group).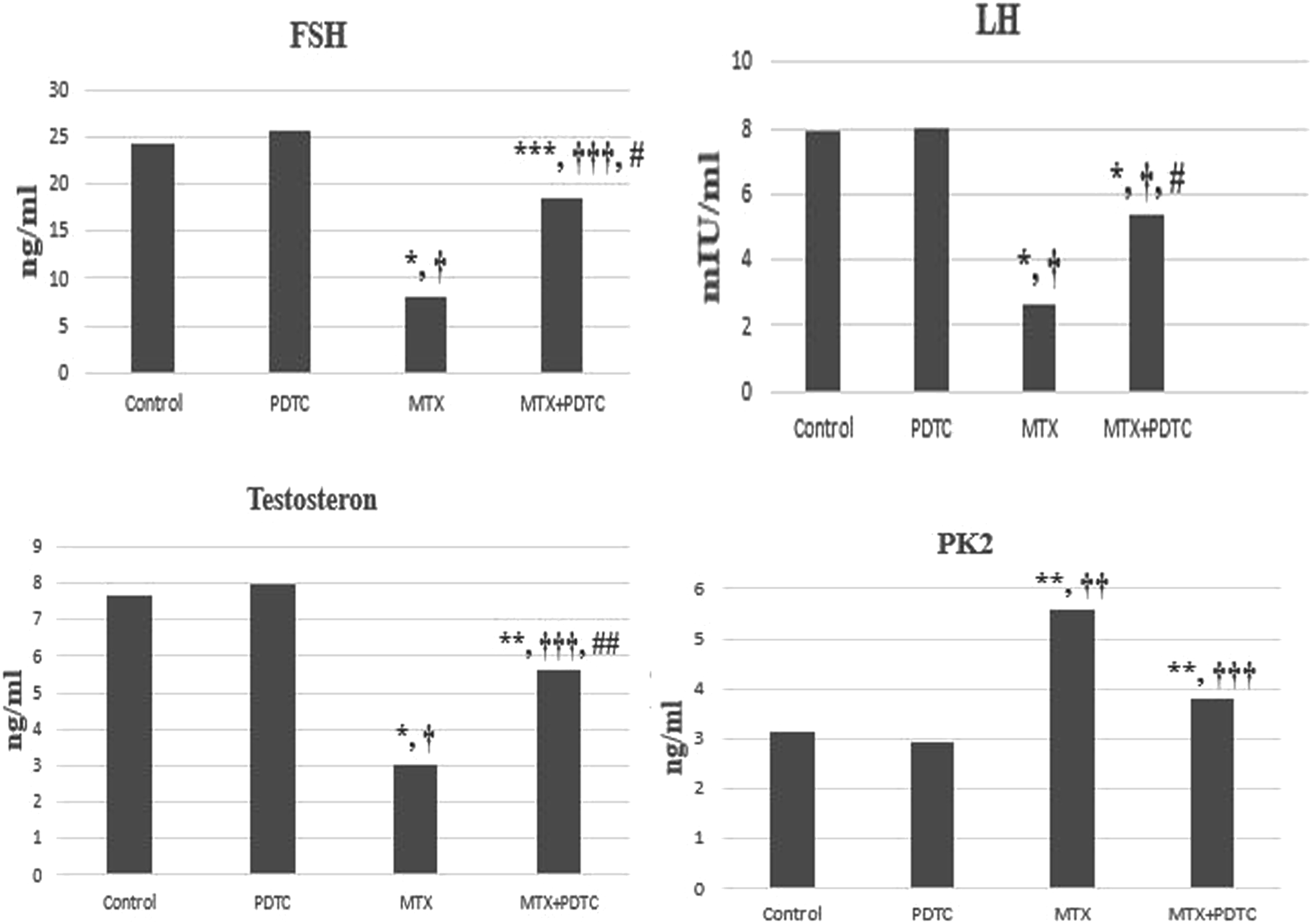
PK2 levels
The mean PK2 levels were 3.16 ± 0.22 ng/mL in the control group, 2.94 ± 0.71 ng/mL in the PDTC group, 5.58 ± 1.69 ng/mL in the MTX group and 3.82 ± 0.43 ng/mL in the MTX + PDTC group. Proinflammatory PK2 levels were significantly increased in MTX groups compared to control and PDTC group (p < 0.01, p < 0.05, respectively). Serum PK2 levels were decreased in the MTX + PDTC group compared to the MTX group, but this decrease was not significant (p = 0.057) (Figure 1).
Morphometric analysis
Comparison of seminiferous tubule diameter, germinal epithelium thickness and interstitial field width of control and experimental groups.
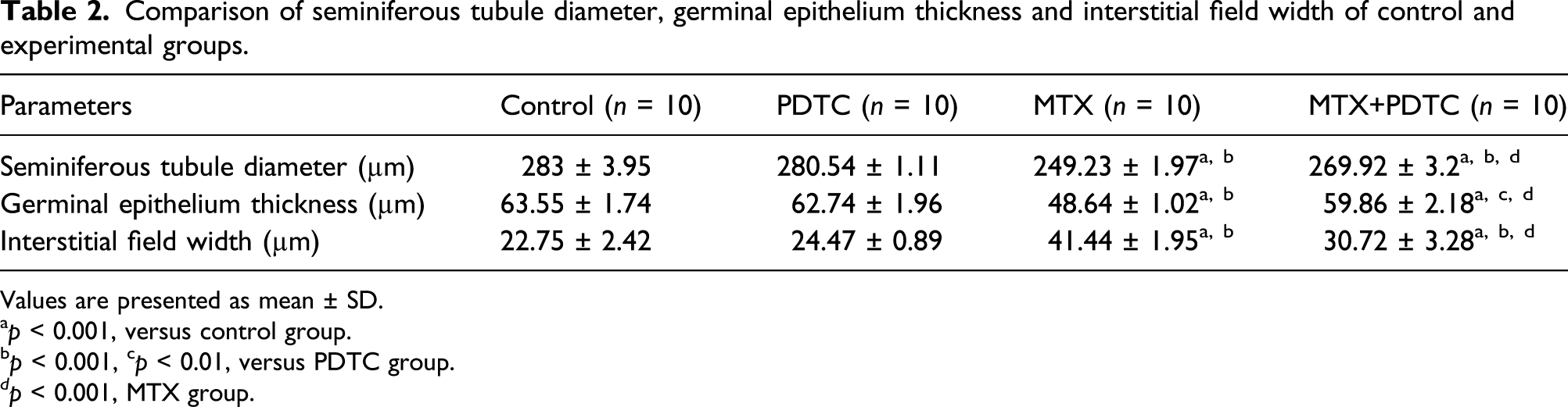
Values are presented as mean ± SD.
ap < 0.001, versus control group.
bp < 0.001, cp < 0.01, versus PDTC group.
dp < 0.001, MTX group.
Protective effect of PDTC on testicular tissue histology
When testicular tissue sections of the control and PDTC groups were examined under a light microscope, the seminiferous tubules with properly borders and lined with stratified germinal epithelium had a normal appearance. The germinal serial cells located in the seminiferous tubule wall were properly lined up and displayed normal organization. In the interstitial field, various connective tissue cells, blood vessels and Leydig cells in groups were observed in a normal histological structure (Figures 2(a) and (b)). In the MTX group, disruption in organization of the germinal series cells and vacuolization as a result of cell loss were observed in the seminiferous tubules. Detachment from the basement membrane was observed starting with primary spermatocytes in most of the seminiferous tubules. Sloughed immature cells were found in the lumens of some damaged tubules. Irregularity, cell loss, minimal or moderate congestion of blood vessels and interstitial oedema were observed in the interstitial field (Figures 2(c) and (d)). It was observed that normal seminiferous tubule percentage increased significantly and continued spermatogenesis actively in these seminiferous tubules in the MTX + PDTC group. However, in some regions, the presence of detached or sloughed tubules was found at varying rates. It was observed that interstitial oedema and congestion regressed, with irregularity and cell loss in the interstitial field (Figure 2(e)). When the testicular tissue histological structure was evaluated, it was observed that the damage regressed significantly (Figure 2(f)). Light microscopy of testicular tissue in different groups. A, B. control (A) and pyrrolidine dithiocarbamate (B) groups showed that seminiferous tubules (asterisks) and interstitial field (IF) were observed as normal testis tissue histological structure. C, D. In the MTX group, detachment (De), sloughing (Sl) and vacuolization (V) were observed in the seminiferous tubules. The interstitial field (IF) damaged including cell loss, congestion (Co) in the blood vessels and marked interstitial oedema (Oe) were also observed. E. MTX+PDTC group exhibited mild degeneration in the seminiferous tubules, mostly detachment (De). Also, it was observed that spermatogenesis continued actively most of in these seminiferous tubules (asterisks), and congestion in the blood vessels and interstitial oedema were noticed to regress in the interstitial field. Hematoxylin and Eosin staining, original magnification, ×200, scale bar = 100 μm. F. Effect of pyrrolidine dithiocarbamate on histological structural changes of seminiferous tubules in methotrexate-treated rats (mean ± SD). (∗p < 0.001, vs control group), (†p < 0.001, vs PDTC group), (#p < 0.001, vs MTX group).
Protective effect of PDTC on immunohistochemical findings
NFkB immunoreactivity findings
NFkB immunoreactivity demonstrated weak staining in the testicle seminiferous tubule sections of control and PDTC groups. Similarly, no staining was observed in the interstitial area (Figures 3(a) and (b)). When the seminiferous tubule sections of MTX group were examined, intense staining was observed mainly in primary spermatocytes, and moderate or intense staining was observed in spermatids and other cells of the germinal epithelium. Leydig cells were intensely stained in interstitial field (Figure 3(c)). NFkB expression regressed with PDTC treatment in the MTX + PDTC group (Figure 3(d)). NFkB immunoreactivity in MTX and MTX + PDTC groups increased significantly compared to control and PDTC groups (p < 0.001, Figure 3(f)). NFkB immunoreactivity in the MTX + PDTC group decreased significantly compared to the MTX group (p < 0.001; Figure 3(f)). Nuclear factor kappa B immunoreactivity of testis among the groups. A, B. Primary spermatocytes (PS) and spermatids (S) showed weak staining in the seminiferous tubules in the control and pyrrolidine dithiocarbamate groups. C. In the MTX group, primary spermatocytes (PS) showed intense staining, and spermatids (S) and other cells of the germinal epithelium showed moderate or intense staining. Intense immunoreactivity is also observed in the interstitial field where Leydig cells are located (arrowhead). D. In the MTX+PDTC group, primary spermatocytes (PS) and spermatids (S) showed moderate to weak staining, and the interstitial field (arrowhead) showed weak immunoreactivity. E. No staining was observed in the negative control. Hematoxylin counterstain, original magnification, ×400, scale bar = 50 μm. F. Effect of pyrrolidine dithiocarbamate on the nuclear factor kappa B immunoreactivity of testis in methotrexate-treated rats (mean ± SD). (∗p < 0.001, vs control group), (†p < 0.001, vs PDTC group), (#p < 0.001, vs MTX group).
Nrf2 immunoreactivity findings
In the control and PDTC groups, while Nrf2 immunoreactivity demonstrated intense staining in mostly spermatids, it showed moderate to weak staining in interstitial field and Sertoli cells (Figures 4(a) and (b)). Weak or very weak Nrf2 immunoreactivity was observed in the MTX group (Figure 4(c)). The MTX + PDTC group showed increased Nrf2 immunoreactivity in spermatids (Figure 4(d)). Nrf2 immunoreactivity in MTX and MTX + PDTC groups decreased significantly compared to control and PDTC groups (p < 0.001; Figure 4(f)). There was a significant increase in Nrf2 immunoreactivity in the MTX + PDTC group compared to the MTX group (p < 0.01; Figure 4(f)). Nuclear factor erythroid 2 related factor 2 immunoreactivity of testis among the groups. A, B. Nuclear factor erythroid 2 related factor 2 immunoreactivity showed intense staining in mostly spermatids (S) in the control and pyrrolidine dithiocarbamate groups. Moderate to weak immunoreactivity was observed in the interstitial field where Leydig cells are located (arrowhead). C. In the MTX group, spermatids (S) showed weak immunoreactivity (De: detachment, Sl: Sloughing). D. In the MTX+PDTC group, spermatids (S) showed moderate to weak immunoreactivity. Moderate–weak immunoreactivity is also observed in the interstitial field where Leydig cells are located (arrowhead). E. No staining was observed in the negative control. Hematoxylin counterstain, original magnification, ×400, scale bar = 50 μm. F. Effect of pyrrolidine dithiocarbamate on the nuclear factor erythroid 2 related factor 2 immunoreactivity of testis in methotrexate-treated rats (mean ± SD). (∗p < 0.001, vs control group), (†p < 0.001, vs PDTC group), (#p < 0.001, ##p < 0.01, vs MTX group).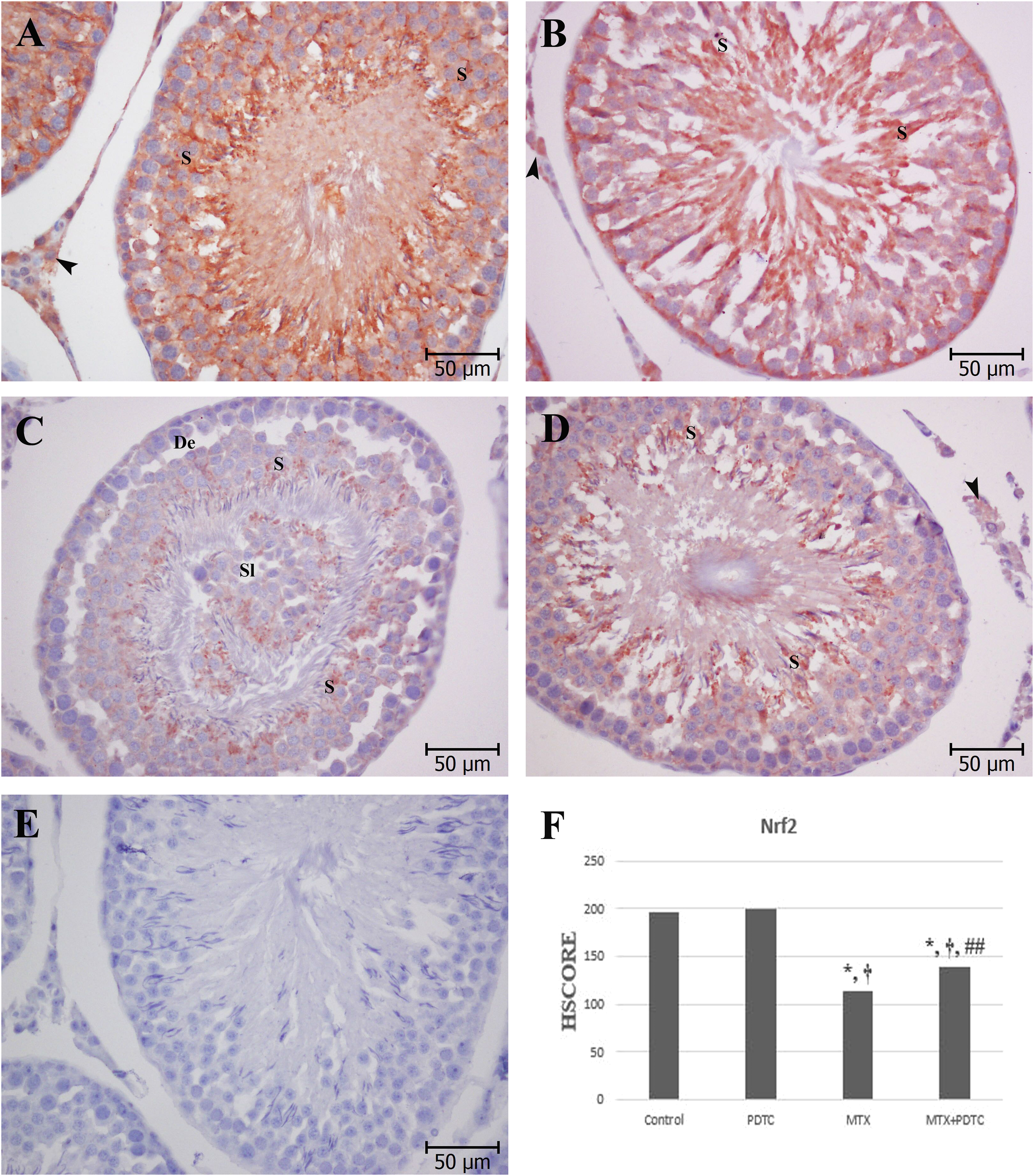
PK2 immunoreactivity findings
When PK2 immunoreactivity was examined in the control and PDTC groups, very weak immunoreactivity was observed in basal parts of the seminiferous tubules. However, moderate immunoreactivity was observed in the interstitial field (Figures 5(a) and (b)). In the MTX group, intense ring-shaped PK2 immunoreactivity was observed around the nucleus in the mainly primary spermatocytes. Besides, in the other cells of the germinal serial cells (GSC), moderate or intense immunoreactivity was also observed in a similar pattern. In the interstitial field, intense immunoreactivity was observed, especially in Leydig cells (Figure 5(c)). In the MTX + PDTC group, moderate to weak staining was observed in all cells of the germinal epithelium, and moderate staining was observed in the interstitial field (Figure 5(d)). PK2 immunoreactivity in MTX and MTX + PDTC groups increased significantly compared to control and PDTC groups (p < 0.001; Figure 5(f)). The PK2 immunoreactivity in the MTX + PDTC group was significantly reduced compared to the MTX group (p < 0.001; Figure 5(f)). Prokineticin 2 immunoreactivity of testis among the groups. A, B. Basal parts of the seminiferous tubule showed very weak staining in the control group and pyrrolidine dithiocarbamate groups. Interstitial field where Leydig cells are located (arrowhead) showed moderate immunoreactivity. C. In the MTX group, intense ring-shaped prokineticin 2 immunoreactivity was observed around the nucleus mainly in the primary spermatocytes in the seminiferous tubule section where immature cells sloughing (Sl). Besides, in the other cells of the germinal serial cells (GSC), moderate or intense immunoreactivity were also observed in a similar pattern. Widespread and intense immunoreactivity was observed in the interstitial field, especially in Leydig cells (arrowhead). D. In the MTX+PDTC group, moderate to weak prokineticin 2 immunoreactivity was observed in all cells of the germinal epithelium, and moderate staining was observed in the interstitial field where Leydig cells are located (arrowhead). E. No staining was observed in the negative control. Hematoxylin counterstain, original magnification, ×400, scale bar = 50 μm. F. Effect of pyrrolidine dithiocarbamate on the prokineticin 2 immunoreactivity of testis in methotrexate-treated rats (mean ± SD). (∗p < 0.001, ∗∗p < 0.01, vs control group), (†p < 0.001, ††p < 0.01, vs PDTC group), (#p < 0.001, vs MTX group).
No staining was observed in the negative controls of all three immunohistochemical staining (Figures 3(e), 4(e), and 5(e)).
Discussion
The present study demonstrated the protective effects of PDTC against MTX-induced testicular injury by evaluating changes in body and total testicular weight; serum FSH, LH, testosterone and PK2 levels; measurements of seminiferous tubule diameter, germinal epithelial thickness and interstitial field width; changes in histological structure of seminiferous tubule and interstitium and NFkB, Nrf2 and PK2 immunoreactivities. We found that PDTC treatment significantly reversed the decrease in body weight, serum FSH, LH and testosterone hormone levels caused by MTX, while significantly reducing the number of histologically damaged seminiferous tubules and interstitial field width. In addition, PDTC treatment significantly reversed the increase in NFkB and PK2 immunoreactivity and decrease in Nrf2 immunoreactivity.
MTX is widely used in solid tumours and haematological malignancies as an anticancer agent, as well as in autoimmune and inflammatory diseases as an immunosuppressive and anti-inflammatory agent, respectively.8,36 MTX has a toxic effect on spermatogenic cells with high proliferation ability, specifically due to its cytotoxic effect on active proliferating cells. 5 Treatment-related infertility is an important long-term complication in adolescent and young adult cancer patients (15–39 years of age). For this reason, chemotherapy-related gonadal dysfunctions and effects of chemotherapy on reproductive health have been accepted as the most common long-term morbidities. 3 However, MTX causes testicular tissue damage by inducing endogenous oxidant system and inflammatory pathways in the cell.8,11,13 Studies have shown that Nrf2 is an important regulator of the cell’s endogenous antioxidant defence system. 11 Deterioration in the antioxidant defence system causes accumulation of ROS in the cell, thus leading to oxidative stress. Oxidative stress activates NFkB and PK2, which have proinflammatory properties and can induce testicular damage as a result of inflammation.37,38 For this purpose, our study investigated the protective effect of PDTC on Nrf2, NFkB and PK2 expressions against testicular damage caused by MTX.
PDTC is a thiol compound in the class of DTCs. Diethyl derivatives and sulfhydryl compounds of DTCs are used in clinics. Due to its chemical properties, PDTC has a heavy metal chelating activity. Besides, it has a strong antioxidant activity due to its dithiocarboxy group. Also, it has strong anti-inflammatory properties as it is a specific inhibitor of NFkB.24,25,27 Thiol compounds such as N-acetyl-cysteine (NAC) or t-cysteine also inhibit NFkB activation. The basis of NFkB inhibition is the free oxygen radical scavenging effect of thiol groups. However, PDTC is thought to be approximately 200 times stronger because it can perform NFkB inhibition at lower concentrations than NAC. In addition, PDTC can reach higher or longer duration intracellular concentrations. 24 Studies have been conducted to understand the mechanism of action of PDTC. Virlos et al. 39 demonstrated the protective effects of PDTC via inhibition of activation by stabilization of IkBα. Ilbey et al. 28 demonstrated that PDTC induces apoptosis in tumour cells by inhibiting proteosomal activity. It has also been shown that PDTC is one of the most effective inducers of heme oxygenase-1, which protects cells against oxidative stress. 25 On the other hand, PDTC has been reported to improve histopathology and decrease oxidative stress in organ damage caused by various agents.25,28,29,40,41
In this study, a significant decrease in body weight and total testicular weight in parallel with previous studies on MTX toxicity was observed (p < 0.001 and p < 0.01, respectively; Table 1).5,8,11,12,33 The decrease in testicular weight may be due to testicular tissue degeneration and atrophy. The decrease in body weight may be the result of MTX systemic toxicity. No difference was observed when comparing the total testicular weight/body weight ratio between groups. While there are studies reporting a significant decrease in total testicular weight/body weight ratio,8,11 there are also studies suggesting that ratio remains unchanged, which is in parallel with our study.5,11,42 In this study, body weight change was significantly less when PDTC treatment was administered (p < 0.05, Table 1). Although PDTC slightly ameliorated testicular weights in the MTX + PDTC group, this change was not statistically significant.
Hypothalamic–pituitary–gonadal hormone axis is a special flow that provides the necessary hormones to be released at the right time and concentrations for the regular functioning of the male reproductive system. Any abnormality in this hormone axis can lead to infertility. 1 For this purpose, FSH, LH and testosterone levels were measured by ELISA in order to evaluate the male reproductive system functions. It was found that serum FSH, LH and testosterone levels decreased significantly in the MTX groups. Our results are consistent with those of other studies in which serum FSH, LH and testosterone were evaluated together.6,43 Significant decrease in serum testosterone levels was found, which is similar to the findings of other studies in which only testosterone levels were evaluated.7,8,33 This reduction may be due to the damage of Leydig cells. PDTC treatment significantly improved serum FSH, LH and testosterone hormone levels compared to MTX and MTX + PDTC group (Figure 1).
Prokineticins (PKs) are considered as adipokines because they are highly detected in white adipose tissue.44,45 In addition, PKs are known as cytokines/chemokines due to their structural and functional similarities. Prokineticins show proinflammatory activity. There are two isoforms of PKs: PK1 and PK2. PK2 plays an essential role in modulating the inflammatory and immune response.46,47 PK2 has been shown to exhibit more than one proinflammatory activity in inflamed tissues and play a role in regulating local and systemic inflammation. 22 PKs are thought to have angiogenic and survival factors in the testis and play a role in regulating blood testicle barrier by participating in high endothelial cell cycle. 20 PK2 can be induced by hypoxia. PK2 may play a role in protecting germ cells from apoptosis. Over-expression of PK2 can induce an inflammatory response, such as production of proinflammatory cytokines, and cause damage to the testicular tissue leading to male infertility.20,23 For this purpose, in order to evaluate inflammation, proinflammatory PK2 levels were measured by ELISA. It was found that PK2 in the MTX group increased significantly. To the best of our knowledge, no study was found evaluating serum PK2 level. Serum PK2 levels decreased with PDTC treatment; however, this decrease was not significant (p = 0.057).
In our study, in accordance with literature, seminiferous tubule diameter7,9-11,48 and germinal epithelial thickness10,33 in the MTX group decreased significantly, while interstitial field width34,50 increased significantly. Saxena et al. 49 reported that MTX could not replicate the DNA of germinal epithelial cells and significantly decrease the seminiferous tubule cellular content by inhibiting DHFR, the essential enzyme required for DNA synthesis. Accordingly, MTX toxicity can lead to a decrease in germinal epithelial thickness and tubular atrophy as a result of the decrease in germinal epithelial cell diameters and degeneration of germinal epithelial cells. In the MTX + PDTC group, PDTC treatment significantly prevented the decrease in seminiferous tubule diameter and germinal epithelial thickness and increase in interstitial field width compared to the MTX group (p < 0.001).
Histological damage to testicle by MTX which includes organization irregularity in the seminiferous tubules, detachment in the germinal epithelium, sloughing germinal epithelial cells in the tubule lumen, vacuolization in the tubules,12,48,50 interstitial field congestion 51 and oedema 48 has been similarly observed in previous studies. In our study, it was found that normal seminiferous tubules decreased significantly and damaged seminiferous tubules such as detachment, sloughing and vacuolization significantly increased in the MTX group in accordance with some studies in literature.5,7,12,42,52 When histological structure was evaluated, it was observed that testicular damage regressed significantly as a result of PDTC treatment in the MTX + PDTC group.
NFkB protein complexes can be expressed in many tissues. It plays a critical role in immune response, inflammatory process, cellular proliferation, cell survival and apoptosis regulation. 53 In our study, NFkB immunoreactivity increased due to MTX toxicity. However, significant reduction in NFkB immunoreactivity was observed with PDTC, a selective NFkB inhibitor. The increase in NFkB immunoreactivity in testicular damage caused by MTX has been previously demonstrated.6,15 Since oxidative stress activates NFkB, it has been shown to cause activation of leukocytes and many proinflammatory molecules, eventually leading to inflammation. 38
Nrf2 has been identified as an important nuclear transcription factor that provides cellular protection against oxidative stress because many endogenous protective genes can be encoded by Nrf2. 11 Nakamura et al. 54 reported that Nrf2 knock-out causes impairment of Sertoli and germ cells functions in mice. Nrf2 can play a key role in protecting the male reproductive system against oxidative stress. In our study, a significant decrease in Nrf2 was observed in the MTX group, whereas a significant increase was detected in the MTX+PDTC group compared to the MTX group. Similarly, Yu et al. 55 demonstrated that Nrf2 immunoreactivity decreased in ischaemia reperfusion injury in testis. It has been reported in accordance with our study that Nrf2 expression decreases in hepatic damage caused by MTX.56,57 However, there are some studies showing that Nrf2 expression increases as a result of stress in the testicle.58,59
Recently, it was understood that PK2 acts as a proinflammatory peptide in regulating inflammation 22 since PK2/PKR1 (prokineticin receptor 1) complex has been shown to increase IL-1β and IL-12 secretion during inflammation and decrease anti-inflammatory activity by decreasing IL-4 and IL-10 secretion.22,48 In our study, the increase in PK2 immunoreactivity caused by MTX supports the role of PK2 in regulating inflammatory response, with proinflammatory activity. A decrease in PK2 immunoreactivity was observed with PDTC treatment compared to the MTX group (Figure 5). To the best of our knowledge, there is no study evaluating PK2 expressions in MTX-induced testicular injury. However, it was observed that only a few studies on PK2 expression in testis were reported in the literature. Increased PK2 expression has been shown in experimental varicocele-induced testicular injury. 20 Chen et al. 22 found that the expression of PK2 and its receptor PKR1 increased in lipopolysaccharide-induced testicular damage. In another study, where an experimental autoimmune orchitis model was created, inflammation was induced due to increased PK2 expression in the testicle. 23
Consequently, according to our literature search, our study is the first study to evaluate the protective effect of PDTC on MTX-induced testicular damage through examinations of tissue histology, immunohistochemistry and hormone assays. In our study, as a result of testicular damage caused by MTX, an increase in NFkB and PK2 expressions and decrease in Nrf2 expressions were observed. PDTC, a specific NFkB inhibitor and potent antioxidant, has been shown to improve histological damage in the testis and hormonal parameters. We considered that PDTC plays a protective role in testicular tissue by reducing NFkB and PK2 expressions and increasing Nrf2 expressions. Therefore, we believe that PDTC can contribute to the treatment of infertility caused by MTX-induced testicular damage in the clinic.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Scientific Research Projects Coordination Unit of Trakya University (TUBAP 2018/103).
