Abstract
Transient receptor potential vanilloid 1 (TRPV1) activation by capsaicin binding increased intracellular calcium influx and stimulated adipocyte-to-adipocyte communication, leading to lipolysis. Generally, enhancement of π-stacking capabilities improves certain binding interactions. Notably, nitroarenes exhibit strong binding interactions with aromatic amino acid side chains in proteins. New capsaicinoid analogs were designed by substitution of the OCH3 group with a nitrogen dioxide (NO2) group on the vanillyl ring to investigate how π-stacking interactions in capsaicinoid analogs contribute to lipolysis. Capsaicinoid analogs, nitro capsaicin (
Keywords
The increasing number of overweight individuals has become a major global concern for maintaining healthy lifestyles. In 2016, in excess of 1.9 billion adults were overweight, with at least 650 million of them being obese, and the numbers continue to increase worldwide. 1 Moreover, obesity is one of the chief factors leading to noncommunicable diseases, 2 such as hyperglycemia, 3 hypertension, 4 coronary heart disease, 5 ischemic stroke, 6 respiratory complications, 7 and atherosclerosis. 8 Previous studies have revealed that excess weight and obesity are the result of energy disproportion between consumption and utilization of calories in the body, which leads to inordinate fat storage 9 -11 and increasing the numbers, size, and mass of adipocytes. 12
Generally, adipocytes, the most prevalent cells among adipose tissues, control anabolism, by transforming energy intake into triglycerides, as well as catabolism, by converting triglycerides to free fatty acids. 13 -16 Furthermore, adipocytes regulate lipid mobilization and distribution in the body. 17 Therefore, they play a key role in energy balance and body weight management. 18 Currently, functional food from natural sources is being utilized as an alternative approach for weight management. For example, coffee, tea, garlic, and soybeans have been demonstrated as being beneficial for ameliorating the energy balance in overweight people. 19 Presently, several bioactive compounds are employed as ingredients in supplementary food for weight control, including resveratrol from grapes, 20 quercetin from onions, 21 genistein from soy, 22 6-shogaol from ginger, 23 and piperine from pepper. 24 Capsaicin (CAP) from chilies, which is generally used as a flavor ingredient in spicy foods, has also found use in the context of weight control. 25 Furthermore, the beneficial role of CAP has been established for the treatment and prevention of several diseases, such as cardiovascular, 26 gastrointestinal, 27 and Alzheimer’s, 28 as well as cancers. 29 Moreover, CAP is well known as a treatment for inflammatory and pain symptoms, such as those associated with rheumatoid arthritis, posthepatic neuralgia, and osteoarthritis. 30
The mode of action of CAP involves the transient receptor potential vanilloid 1 (TRPV1) receptors. Based on the structure-activity relationships of CAP and TRPV1, CAP is generally divided into 3 sections (Figure 1): the head section (vanillyl residue), neck section (amide bond), and tail section (hydrocarbon chain). Previously, computational calculations of CAP-TRPV1 binding interactions had revealed that the vanillyl moiety and amide bond are involved through hydrogen bonding. Moreover, the unsaturated hydrocarbon chain displays nonspecific Van der Waals interactions with TRPV1 receptors. 31 Recently, further evidence has suggested that TRPV1 activation from CAP increases intracellular calcium influx, which then stimulates adipocyte-to-adipocyte communication, leading to lipolysis. 32 Enhancement of binding interactions between ligands and TRPV1 receptors has been achieved via various approaches, such as increasing the number of hydrogen bonding sites, 33 -39 alteration of the amide bond, 40 -43 and structural modification of the tail section. 44 -47

Segmentation of capsaicin (CAP) into 3 sections: (1) head section (vanillyl residue), (2) neck section (amide bond), and (3) tail section (hydrocarbon chain). Chemical structures of naturally occurring capsaicinoids: CAP, dihydrocapsaicin (DHC), and synthetic nitro capsaicinoid analogs: nitro capsaicin (5) and nitro dihydrocapsaicin (6).
Additionally, nondestructive and noncovalent π-stacking interactions are an important phenomenon that has been extensively investigated for application in chemistry and molecular biology, especially regarding binding interactions in enzymes and receptors. 48 Computational calculations have established that the attachment of nitrogen dioxide (NO2) groups on aromatic rings results in strong π-stacking interactions, particularly with tryptophan, tyrosine, phenylalanine, and histidine, which consequently significantly promotes binding interactions with proteins. 49
Structurally, the vanillyl residue represents a crucial pharmacophore that contributes to TRPV1 binding. Removal of the OH group, which acts as a hydrogen bond donor on the vanillyl residue, clearly diminishes binding interactions, thereby reducing TRPV1 activation.
50
To avoid alteration of the OH group while promoting binding affinity with TRPV1 receptors, an appropriate approach for the modification of CAP in order to increase lipolysis involves the adjustment of π-stacking interactions of the vanillyl residue. The NO2 group was therefore chosen to substitute the OCH3 group on the vanillyl residue, as its introduction onto the aromatic ring can be accomplished employing relatively straightforward chemistry. It should be noted that a CAP-like analog containing this modification of the aromatic region, as well as other structural modifications, has been shown previously to bind to the TRPV1.
33
To explore this hypothesis, nitro capsaicin (
The construction of
First, commercially available 4-hydroxybenzaldehyde (
Next,

Synthetic route of preparation of capsaicinoid analogs 5 and 6 via amide coupling between an aromatic amine and fatty acids.
A previous report
55
had established that CAP, the most pungent of the capsaicinoid analogs, induced apoptosis of 3T3-L1 adipocytes in the concentration range from 50 to 250 µM after 24 hour exposure. Unlike CAP, the nitroarene capsaicinoid analogs are less pungent, which was evident during the synthesis. Therefore, CAP was selected for the initial screen to establish a suitable concentration for use in the biological studies. With the mentioned criteria, we explored the appropriate concentration of CAP using its cytotoxicity with differentiated 3T3-L1 adipocytes, as determined using 3-(4,5-dimethylthiazol-2-yl)−2,5-diphenyltetrazolium bromide (MTT) assay. Differentiated 3T3-L1 adipocytes were tested with various concentrations of CAP (5, 10, 25, 50, and 100 µM) for 24 hours. The results shown in Figure 2(A) demonstrate that concentrations of CAP under 10 µM exhibited no significant cytotoxicity to differentiated 3T3-L1 adipocytes in comparison with the vehicle control (no CAP treatment). In order to observe a distinct change in biological activities, 10 µM CAP, DHC,
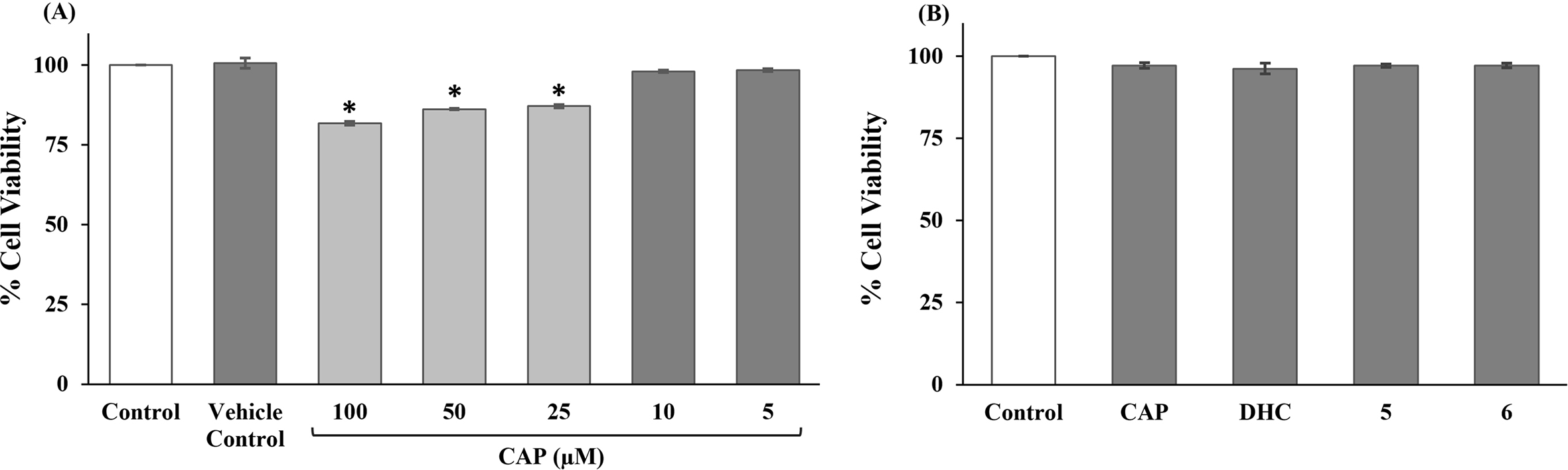
(A) Effect of CAP at 5 different concentrations (5, 10, 25, 50, and 100 µM) on cell viability. (B) Effect of CAP, DHC, 5, and 6 at 10 µM on cell viability. Differentiated 3T3-L1 adipocytes were treated for 24 hours. The cell viability was quantified and compared with control by measurement using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay at an absorbance of 595 nm. Data are expressed as mean ± SE from 3 independent experiments. *P < 0.05 versus the vehicle control. CAP, capsaicin; DHC, dihydrocapsaicin.
Oil Red O staining of differentiated 3T3-L1 adipocytes treated with 10 μM CAP, DHC,

Effect of CAP, DHC, 5, and
In addition to lipid accumulation, intracellular lipolysis metabolism of differentiated 3T3-L1 adipocytes was examined by measuring triglyceride levels, using the Cayman triglyceride assay kit. Differentiated 3T3
Effect of CAP, DHC, 5, and
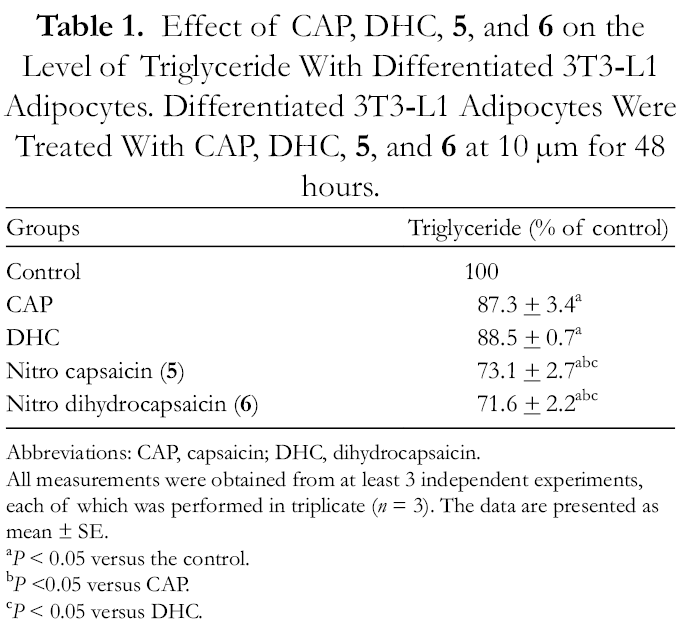
Abbreviations: CAP, capsaicin; DHC, dihydrocapsaicin.
All measurements were obtained from at least 3 independent experiments, each of which was performed in triplicate (n = 3). The data are presented as mean ± SE.
a P < 0.05 versus the control.
b P <0.05 versus CAP.
c P < 0.05 versus DHC.
π-Stacking interactions can be manipulated by attenuating hydrophobicity, surface area of substituents, electronegativity, and dipole moment of the participating residues.
56
The inclusion of a NO2 group presumably increases the dipole moment on
The significant increase in the calculated dipole moment in the model compounds bearing a nitro group resulted from the appreciable negative charge of the NO2 group. The HOMO orbital plot illustrates that the electrons are predominantly localized on the carbon atom of the aromatic ring for both model compounds. However, only the model structure for the nitro-containing compounds

Calculated molecular structures, dipole moments (blue arrow), and HOMO orbitals of the model compounds representing (
Not only were the calculated dipole moment values and predicted electron distribution on the model structure for the nitro-containing compounds
The total number of docking poses for

The best docking pose of 5 (red stick) and CAP (yellow stick) in the vanilloid binding site of the TRPV1 receptor. The structures are represented by superposition of docking poses and the corresponding key amino acid residues (red and yellow lines) interacting with the TRPV1 receptor (green and blue ribbon for chain D and B, respectively) are illustrated. CAP, capsaicin; TRPV1, transient receptor potential vanilloid 1.
The overall binding free energies of
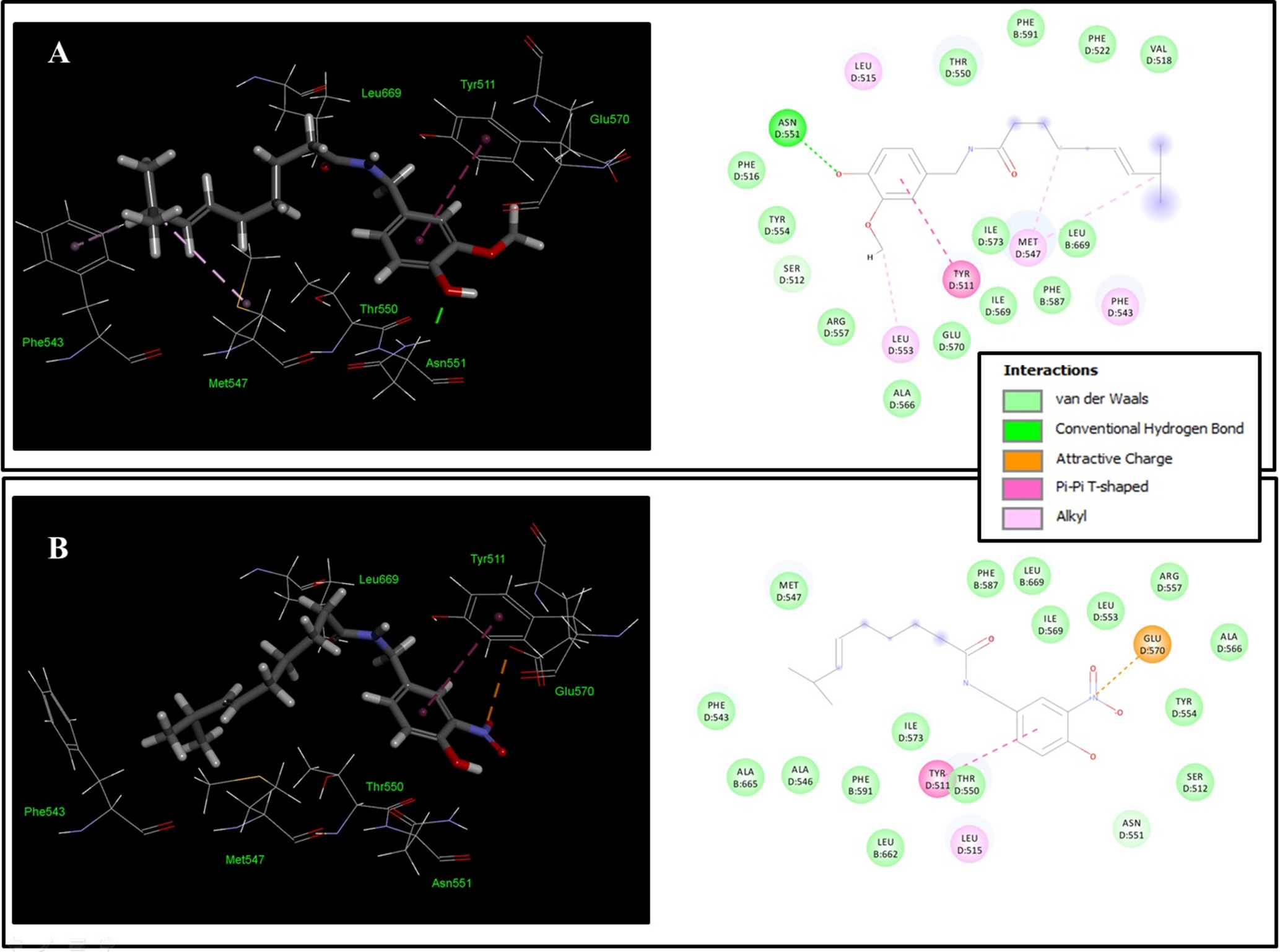
Molecular docking diagram of (A) CAP and (B)
The amide group of the
The binding conformation of
Interaction Energies Per-Residue Within 4 Å of CAP and

Abbreviations: CAP, capsaicin; TRPV1, transient receptor potential vanilloid 1; VDW, Van der Waals.
Conclusions
Herein we demonstrate a facile synthetic route for synthesizing the respective CAP and DHC analogs
The introduction of the nitro group in the CAP skeleton had a significant effect on the lipolysis process. The nitro-substituted vanilloid ring could potentially improve the lipolytic activity due to the optimal π-π stacking and attractive charge interactions present between the substituted vanilloid ring and the Tyr511 and Glu570 residues, respectively. This specific interaction could increase the binding affinity and stability.
These preliminary findings suggest that the nitroarene capsaicinoid analogs can be used to reduce the triglyceride levels in cells. They may also help in overcoming obesity. Presently, a comprehensive study on the pungency of the nitroarene capsaicinoid analogs is being carried out. Their potential role as skin irritants is also being explored. These factors must be accounted for before the compounds can be developed as supplements for topical applications.
Experimental
General
CAP, DHC, 4-hydroxybenzaldehyde, MTT, Oil Red O solution, DMSO, and human recombinant insulin were obtained from Sigma-Aldrich (St. Louis, MO, USA); high glucose Dulbecco’s Modified Eagle’s Medium (DMEM), fetal bovine serum (FBS), bovine calf serum (BCS), phosphate-buffered saline (PBS), antibiotics (100 unit/mL penicillin and 100 µg/mL streptomycin) from GIBCO (Grand Island, NY, USA); dexamethasone (DEX) and 3-isobutyl-1-methyl xanthine (IBMX) from Merck (Kenilworth, NJ, USA); HOBt from Acros Organic (Geel, Belgium); EDC.HCl from GL Biochem (Shanghai, China); Triton X-100 from VWR international (Radnor, PA, USA); 3T3-L1 preadipocytes (ATCC CL-173) from the American Type Culture Collection (Manassas, VA, USA); and the triglyceride colorimetric assay kit from Cayman Chemical (Ann Arbor, MI, USA). All chemical reagents were used as received from vendors unless otherwise noted.
1H NMR and 13C NMR spectra were recorded on a 400 MHz NMR spectrophotometer (AVANCE, Bruker, Switzerland) in appropriate deuterated solvents. Chemical shifts (δ) are reported in parts per million (ppm) relative to either tetramethylsilane or the residual protonated solvent signal as a reference. Mass spectra were measured in positive ion mode with a high-resolution mass spectrometer (micrOTOF, Bruker, Switzerland) with electrospray ionization using sodium formate for calibration. IR spectra were obtained on a Fourier-transform infrared (FT-IR) spectrophotometer (Model 1600, Perkin Elmer, USA) in the wavenumber range of 4000–400 cm-1. The absorbance was measured using a microplate reader (Synergy H1 Hybrid Multi-Mode Reader, BioTek, USA).
Synthesis of 4-Hydroxy-3-Nitrobenzylamine Hydrochloride (2)
4-Hydroxybenzaldehyde (
The dried yellow precipitate was reacted with ammonium formate (0.38 g, 5.98 mmol) in a round-bottom flask connected to a sealed reflux apparatus. The reaction mixture was then refluxed at 180 °C for 4 hours, after which time it was cooled to room temperature and conc. HCl (2 mL) was added. The reaction mixture was then refluxed at 100 °C for 1 hour. The mixture was cooled to room temperature and the formed precipitate was collected by filtration, washed with ice-cold absolute ethanol (3 × 5 mL), and recrystallized from absolute ethanol to give compound
Compound
Synthesis of (E)-8-Methyl-6-Nonenoic Acid (3)
Unsaturated fatty acid
Compound
Synthesis of 8-Methylnonanoic Acid (4)
Saturated fatty acid
Compound
Synthesis of (E)-N-(4-Hydroxy-3-Nitrobenzyl)-8-Methylnon-6-Enamide (5) and N-(4-Hydroxy-3-Nitrobenzyl)-8-Methylnonanamide (6)
EDC.HCl (0.73 mmol) and HOBt (0.73 mmol) were added to a solution of
Compound
Compound
Cell Culture and Differentiation
3T3-L1 Preadipocytes (ATCC CL-173) were cultured in DMEM supplemented with 3.7 g/L sodium bicarbonate, 10% BCS, and antibiotics at 37 °C in a humidified atmosphere of 5% CO2. The medium was changed every 3 days. The cells were subcultured after reaching 80% confluence, seeded in a 96-well plate at a density of 2 × 103 cells/well, and cultured in maintenance medium (DMEM containing 10% BCS and 1% antibiotics) until 90%-95% confluence. After reaching the desired confluence (defined as day 0), the cells were further induced with initiation medium (1 µM DEX, 0.5 mM IBMX, 10 µg/mL insulin in 10% FBS-DMEM) for 3 days. At the end of the third day, the initiation medium was replaced with the progression medium (10 µg/mL insulin in 10% FBS-DMEM). The media was changed every 2 days until day 9, and then the cells were differentiated into a rounded shape with lipid droplets. The morphology of mature adipocytes was examined using a microscope.
Cell Viability Assay
Cell viability was determined by the MTT assay to define a suitable concentration for all testing. Differentiated 3T3-L1 adipocytes were treated with 5, 10, 25, 50, and 100 µM CAP dissolved in DMSO for 24 hours in a humidified 5% carbon dioxide (CO2) atmosphere at 37 °C. The final concentration of DMSO in the culture medium was 0.2 %v/v. The vehicle control contained the same concentration of DMSO, but no CAP. After incubation, MTT reagent in PBS solution was added to each well plate, and the plate was incubated for another 3 hours. Subsequently, the culture medium containing MTT reagent was removed. The formazan produced in the viable cells was dissolved by the addition of DMSO. The absorbance at 595 nm was measured using a microplate reader. The values were presented as a percentage of those for control cells without capsaicinoid and analogs. The effects of CAP, DHC,
Determination of Lipid Accumulation in 3T3-L1 (Oil Red O Staining)
To examine the effects of CAP, DHC,
Differentiated 3T3-L1 adipocytes were placed in a 96-well plate with CAP, DHC,
Triglyceride Assay
Differentiated 3T3-L1 adipocytes were treated with 10 µM of CAP, DHC,
Structure Preparations and Molecular Docking
In this study, the vanilloid binding site in the structure of the open TRPV1 channel in complex with RTX and DkTx (PDB ID: 3J5Q) was used as the target for docking of nitro capsaicin and CAP.
64
The -dimensional structure of this TRPV1 was retrieved from the Research Collaboratory for Structural Bioinformatics protein data bank. Before docking, the TRPV1 structure (chains B, D, E, and G) was cleaned up by removing double-knot toxin molecules (chains A, C, F, and H). The missing loop regions of the TRPV1 chains (503-LKSLF-507) were inserted based on the SEQRES data. The amino acid residues in TRPV1 were protonated at pH 7.4 according to the predicted pK values. The structure of TRPV1 was submitted for energy minimization with the conjugate gradient algorithms applying CHARMm forcefield with protein backbone constraints to reduce the atomic clash of amino acid side chains. The structures of
Statistical Analysis
The values are expressed as mean ± SE. Statistical analyses were performed using SPSS software version 17.0 (SPSS Inc, Chicago, IL, USA). Significant differences between groups were analyzed using a one-way analysis of variance followed by Tukey’s multiple comparison tests. A value of P < 0.05 indicated a significant difference.
Supplemental Material
Supplementary Material 1 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 1, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 2 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 2, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 3 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 3, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 4 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 4, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 5 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 5, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 6 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 6, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 7 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 7, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 8 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 8, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 9 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 9, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 10 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 10, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 11 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 11, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 12 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 12, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 13 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 13, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 14 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 14, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 15 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 15, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 16 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 16, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Supplemental Material
Supplementary Material 17 - Supplemental material for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs
Supplemental material, Supplementary Material 17, for Enhancement of Lipolysis in 3T3-L1 Adipocytes by Nitroarene Capsaicinoid Analogs by Thanet Laolob, Nuntavan Bunyapraphatsara, Neti Waranuch, Sutatip Pongcharoen, Wikorn Punyain, Sirirat Chancharunee, Krisada Sakchaisri, Jaturong Pratuangdejkul, Sumet Chongruchiroj, Filip Kielar and Uthai Wichai in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Thailand Research Fund (RDG6020016).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
