Abstract
Background
Lonicerae japonicae flos, a representative kind of Traditional Chinese Medicine (TCM) for clearing heat and detoxifying, is known as Jinyinhua in China. The determination of multiple components’ content, especially multiple pharmacological components’ content, is often used to evaluate the quality of TCM, which could objectively evaluate the quality of TCM.
Objectives
The ultra high-performance liquid chromatography–mass spectrometry/mass spectrometry (UPLC–MS/MS) method was used to determine the content of chlorogenic acid, isochlorogenic acid A, isochlorogenic acid C, and luteoloside in the current study.
Materials and Methods
15 batches of Lonicera japonica flos were collected from different sources. 75% methanol was taken as a solvent, and the solution ratio was 1 (mg:mL). The ultrasonic method was utilized to prepare the sample solution for 30 min, which was centrifuged for 10 min at 10,000 rpm. Then, the supernatant was taken for reserve. C18 was chosen as the fixed phase, and acetonitrile (A)–0.1% phosphate acid solution (B) was chosen as the mobile phase. The flow rate was 0.3 mL.min−1. The column temperature was 40°C, and the sample injection was 1 µL. In the negative ion mode, data were collected using multiple reaction monitoring (MRM) scan mode.
Results
The scope of chlorogenic acid, isochlorogenic acid A, isochlorogenic acid C, and luteoloside was in the range of 62.5–6,250 ng. mL−1, which had good linearity (r2 = 0.9997). The content of the four components showed some differences among different sources.
Conclusion
The method was fast and sensitive, which could effectively evaluate and control the quality of Lonicera japonica flos.
Keywords
Introduction
Lonicerae japonicae flos, one of the herbal medicines, is named Jinyinhua in Traditional Chinese Medicine (TCM). Jinyinhua is originated from Lonicera japonica Thunb., which is recorded in Chinese Pharmacopoeia (2020 edition) (Li et al., 2023). Jinyinhua has multiple pharmacology activities, such as antitumor, antibacterial, and immune enhancement (Delalat et al., 2022; Wang et al., 2017; Yuan et al., 2020). Jinyinhua has strong adaptability, is suitable for planting and survival, and has high profits. For example, the organic acids, flavonoids, and glycosides in honeysuckle have anti-inflammatory effects, such as chlorogenic acid, isochlorogenic acid A, isochlorogenic acid C, and luteoloside. They could inhibit the production of inflammatory cytokines and reduce the degree of inflammation (De Stefano et al., 2021; Hong et al., 2013; Kwon et al., 2015). Therefore, it is widely distributed in various regions in Shandong province and Henan province. Many natural conditions influence the plants’ growth, including sunshine, soil property, and climate. These factors could lead to uneven quality and even affect medicinal effects. Therefore, it is necessary to conduct a quality control study to ensure the safety of clinical medication.
In China, many quality evaluation methods could be applied to evaluate TCM’s quality, including trait identification, physical and chemical identification, inspection, and content determination. The authenticity and quality evaluation are carried out based on appearance characteristics, physical and chemical properties, inspection of non-medicinal components (impurities, harmful substances, and so on), and content determination of indicator components. Currently, the study focuses on the active ingredients to evaluate quality connotation. However, the characteristics of TCM’s roles present “multiple components,” and a single evaluation index may not objectively reflect the quality connotation of TCM. Therefore, multicomponent analysis is gradually being applied to the quality evaluation of TCM.
Standard methods include thin-layer chromatography, high-performance liquid chromatography (HPLC), gas chromatography (GC), mass spectrum, and nuclear magnetic resonance (Gu et al., 2022; Xiong et al., 2022; Zhang et al., 2023; Zhu et al., 2016). Due to the complexity of multiple components in TCM, it is difficult to determine the content of every ingredient. Besides, there may be specific interactions among various elements, which could affect the detection and analysis of components. Hyphenated chromatography techniques, such as GC–mass spectrometry (MS) and LC–MS, could achieve a qualitative and quantitative analysis of complex components in TCM, which have high sensitivity and resolution characteristics. Therefore, the study established a method to determine the content of chlorogenic acid, isochlorogenic acid A, isochlorogenic acid C, and luteoloside in Jinyinhua based on the ultra high-performance liquid chromatography (UPLC)–MS method. The analysis was conducted by Jinyinhua from different regions, which provided the basis for the quality evaluation of Jinyinhua.
Materials and Methods
Instruments and Equipment
Agilent G6465A HPLC (Agilent, USA), Agilent Ultivo Triple Quad LC/MS (Agilent, USA), SCQ-200 ultrasonic cleaner (AB SCIEX, USA), SCQ-200 ultrasonic cleaner (Shanghai Spectroscopic Ultrasonic Equipment Factory), AE-240 electronic balance (Mettler Toledo Shanghai Co., Ltd.), KDM type temperature regulating electric heating jacket (Heze Mudan Junteng Electronic Instrument Co., Ltd.), MDF-492 ultra-low temperature refrigerator (Sanyo company of Japan), SK-1 rapid mixer (Jiangsu Jintan Medical Device Factory).
Reagents
Chlorogenic acid, isochlorogenic acid A, isochlorogenic acid C, luteoloside (Chengdu Plant Standard Chemical Pure Biotechnology Co., Ltd., batch numbers 200118, 200122, 200812, and 201010), methanol (HPLC grade, Merck, USA), acetonitrile (HPLC grade, Merck, USA), formic acid (HPLC grade, Tianjin Kemi European Chemical Reagent Co., Ltd.), purified water (Watson’s Drinking Water Co., Ltd.), Jinyinhua (Hebei Julu, Fengqiu, Henan, Shandong Zibo, Henan Xinxiang, Shandong Huantai, Shandong Pingyi, Shandong Heze, Henan Xinxiang No. A220806, Henan Xinxiang No. A220718, Henan Xinxiang No. A211124, Shandong Weifang, Laizhou Zhacun Town, Hebei Province Dongguang County, Henan Qingfeng). The Chinese medicinal materials were signed from 1 to 15. Xieran identified Jinyinhua in the Zibo Integrated Traditional Chinese and Western Medicine Hospital.
Methods
Analysis Conditions
Chromatographic Conditions. The mobile phase for ZPRBAX SB-C C18 (100 × 2.1 mm, 1.8 µm) was 0.1% formic acid water (A) and 0.1% formic acid acetonitrile (B). Gradient elution was as follows: 0 min, 80.0% A, – 1.5 min, – 70.0% A, – 3 min, – 50% A, – 3.5 min, – 50% A, – 5.0 min, – 60.0% A. The flow rate was 0.30 mL.min−1, and the column temperature was 40°C. The injection volume was 2 µL.
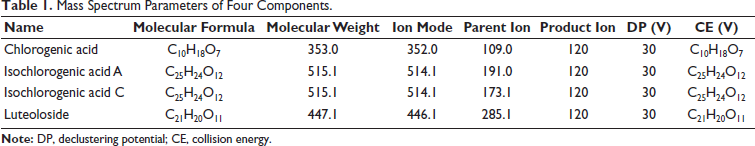
Conditions of the Mass Spectrum. The electric spray ion source was ESI. Multiple reaction monitoring (MRM) mode was set in the positive ion collection mode. Curtain air was 20.00 psi. The gas ion source temperature was 50.00°C. The spray voltage was –3,500 kV. The atomization temperature was 550.0°C. The mass spectrum parameters of the four components are shown in Table 1.
Mass Spectrum Parameters of Four Components.
Preparation of Standard Solution
All standard substrates, namely chlorogenic acid, isochlorogenic acid A, isochlorogenic acid C, and luteolin, were accurately weighed and diluted with methanol 10 times. Then, the solutions were prepared in a concentration of 1 mg.mL−1 mixed reference solution. A specific volume of 75% methanol was added to prepare the series of standard solutions with concentrations of 62.5, 125, 250, 500, 1,250, 2,500, and 6,250 ng.mL−1.
Preparation of Test Solution
According to the Chinese Pharmacopoeia (2020 edition), we prepared the sample solution from No. 1 to No. 15. Two parallel samples were prepared at the solid–75% methanol (1:10) ratio. Then, we extracted the solution for 40 min using the ultrasonic method. And 75% methanol was added to supplement the weight loss after cooling. The solution was centrifuged at 1,500 rpm for 5 min under 4°C. The supernatant was taken by 0.22 µm microporous membrane.
Methodology Validation
Linearity
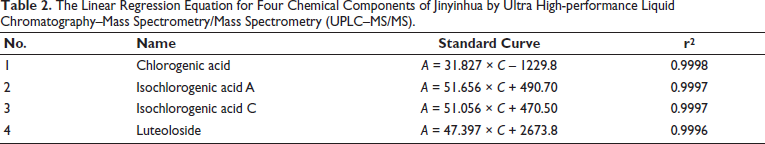
The reference solution was accurately measured and mixed by diluting it with methanol to prepare a series of reference solutions. Then, the solution was determined under the analysis conditions of “2.1.” Take the concentration of each reference substance (ng.mL−1) as the abscissa and the response intensity as the ordinate and draw the standard curve, as shown in Table 2.
The Linear Regression Equation for Four Chemical Components of Jinyinhua by Ultra High-performance Liquid Chromatography–Mass Spectrometry/Mass Spectrometry (UPLC–MS/MS).
Limit of Quantification
The limits of quantification of chlorogenic acid, isochlorogenic acid A, isochlorogenic acid, and luteoloside were 1.7, 3.3, 3.3, and ng.mL−1, respectively, using the signal-to-noise ratio method within S/N = 10.
Stability Investigation
The No. 1 Jinyinhua sample solution was accurately measured at 0, 2, 4, 6, 8, 10, 12, and 24 h, and 2 µL was injected each time, according to the preparation method in the part of “2.3.” RSD values of chlorogenic acid, isochlorogenic acid A, isochlorogenic acid C, and luteoloside were 0.50%, 0.26%, 0.27%, and 0.17%, respectively. The results indicated that Jinyinhua samples were stable based on the concentration of chlorogenic acid, isochlorogenic acid A, isochlorogenic acid C, and luteoloside.
Content Determination
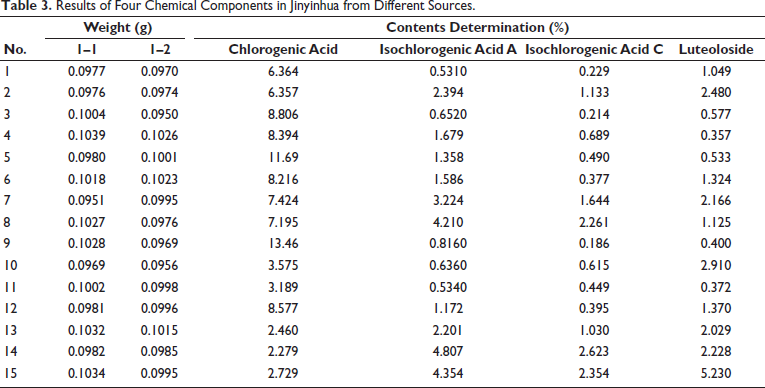
The samples were measured under the condition of “2.3,” and the contents of four components were determined in Jinyinhua from different sources from No. 1 to 15. According to the standard curve, the contents of chlorogenic acid, isochlorogenic acid A, isochlorogenic acid, and luteoloside were listed in Table 3, which were in the solution of test product No. 1–15. After calculation, all of the RSDs were less than 3.0%. The results showed that chlorogenic acid content was highest in No. 9. According to Chinese Pharmacopeia (2020 edition), chlorogenic acid was the quality index of Jinyinhua, which suggested that No. 9 may be the best group. However, luteolin was highest in No. 15. In this study, four compounds were chosen as indexes to evaluate the quality. Therefore, we used the sum of the content of the four ingredients for the analysis.
Results of Four Chemical Components in Jinyinhua from Different Sources.
Discussion
LC–MS/MS technology has been widely used in the identification of TCM, and MS technology provides information on dissolution, which would ensure the analytical characterization of the prototype components and metabolic compounds of TCM. In the current study, chlorogenic acid was used as the representative component of organic acids. The extraction rate was high by ultrasonic extraction, and the extraction rate was related to the length of the alcohol-carbon chain and the number of hydroxyl groups. During MS cleavage, ESI was used as an ion source, and the corresponding intensity was higher in the negative ion mode than in the positive ion mode. The structure of chlorogenic acid contains multiple phenolic hydroxyl groups, which are prone to [M–H]− formation. Thus, signal acquisition was selected in the negative ion mode. In this mode, characteristic fragment ions were produced at m/z 191.0550 of chlorogenic acid, possibly because of the presence of caffeic acid substituents in chlorogenic acid compounds.
The chlorogenic acid content was highest in No. 9, and the content of both isochlorogenic acid A and isochlorogenic acid C were highest in No. 14, while No. 15 had the highest value of luteolosiden. The study suggested that the content of the four components was distinct in different origins. During the screening process, the study found many kinds of literature to design the reference solution’s concentration gradient, which would be verified by testing. Finally, the concentration range was determined by ultra high-performance liquid chromatography–mass spectrometry/mass spectrometry (UPLC–MS/MS), and the regression equation was utilized in the study. It provided the basis for establishing a quality evaluation method in vivo. The method was sensitive, specific, and reliable in this experiment, which could provide a basis for further pharmacokinetic studies.
Conclusion
In this study, the HPLC–MS/MS method was used to determine the contents of chlorogenic acid, isochlorogenic acid A, isochlorogenic acid C, and luteolin in Jinyanhua. The results showed that the total content was highest in Jinyinhua from Henan Xinxiang (No. 9, A220806), but the different batches of Jinyinhua did not consist of the same regions, such as No. 9, No. 10, and No. 11. The result illustrated that the storage time may affect the content of chemical components in Jinyinhua. Additionally, the different content of compounds may also be affected by factors such as sealing degree and storage environment light. Jinyinhua’s genuine producing areas were mainly in Shandong Province and Henan Province. The results showed that the method was simple, rapid, and sensitive, which was suitable for determining Jinyinhua, which would provide a reference for the quality evaluation of Jinyinhua from different sources.
Footnotes
Abbreviations
UPLC-MS/MS: Ultra high-performance liquid chromatography–mass spectrometry/mass spectrometry; MRM: Multiple reaction monitoring; RSD: Relative standard deviation; TCM: Traditional Chinese medicine; HPLC: High-performance liquid chromatography; MS: Mass spectrometry; DAD: Diode array detector; DP: Declustering potential; CE: Collision energy.
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
According to the review of the Experimental Animal Ethics Committee of Qiqihar Medical University, the animal experimental research content and experimental design meet the international ethical standards for the use of experimental animals and the provisions formulated by the Experimental Animal Ethics Committee of Qiqihar Medical University, so the animal experimental research is approved. In this study, there was no animal in the experiment.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Province Traditional Chinese Medicine Science and Technology Project (M-2022020).
