Abstract
Background
The legal quality control of Cangzhu is used the single component as an evaluation indicator, which would not be enough to reflect the authenticity of quality. It is necessary to select several typical chemical components as the basis for quality control of medicinal materials and decoction pieces to reflect the content of effective ingredients.
Purpose
Based on UPLC-MS/MS, the study aimed to build the content determination method of atractyloidin, atractylidin A, atractylenolide I, and atractylenolide III in Cangzhu from different sources.
Materials and Methods
Agilent SB-C18 (2.1 × 100 µm, 1.8 µm) was used to be the stationary phase. The mobile phase system was formed by water-formic acid (1000:1) and acetonitrile-formic acid (1000:1) in gradient elution. The column temperature set at 40℃, and the flow rate was 0.3 mL min–1.
Results
The contents of atractyloidin, atractylidin A, atractylenolide I, and atractylenolide III had good linearity in a certain range (r2 ≥ 0. 9990). The RSD of the precision, stability and repetitive test results were less than 3.00%. In addition, the content of the four components were some different among different patches of Cangzhu.
Conclusion
The method was simple, rapid and highly sensitive, which was suitable for the determination of atractyloidin, atractylidin A, atractylenolide I, and atractylenolide III in Cangzhu.
Introduction
Cangzhu is included in the first part of Chinese Pharmacopoeia (2020 edition), which originated from Atractylodes lancea (Thunb.) DC. and Atractylodes chinensis (DC.) Koidz (Liu et al., 2000). Cangzhu has effects on the syndrome, such as drying dampness, invigorating the spleen, dispelling wind, dispersing cold, and brightening the eyes. It can be used in a variety of formulations, such as ermiao pill, sanmiao pill, and cangshu powder, having good efficacy on rheumatism and arthralgia (Cheng et al., 2016). Modern pharmacological studies have found that Cangzhu has various activities of anti-infection, antitumor (Guo et al., 2019, Na-Bangchang & Karbwang, 2016), immune regulation (Qin et al., 2019), and promoting gastric emptying (Jun et al., 2018).
Cangzhu contains various types of chemical components, such as sesquiterpenoids and alkynes. In addition, it also contains triterpenoids, glycosides, polysaccharides, and other compounds (Zhang, 2021; Zhang, Zhao et al., 2021). Nowadays, Cangzhu plays an essential role in the treatment of ulcerative colitis (Zhang, Deng et al., 2021), the promotion of gastric emptying (Bai et al., 2017), the antiviral and antitumor aspects (Zhao et al., 2023), and so on. Cangzhu shows strong advantages and definite curative effects on many diseases. In recent years, quality control of Cangzhu mainly focuses on the content control of atractylotin in the drug standard by high performance liquid chromatography (HPLC). In contrast, LC–MS technology has the characteristics of being simple, quick, and highly sensitive. It combines the advantages of high-efficiency separation of the liquid phase and high-efficiency detection of the mass spectrum, which can quickly separate and analyze samples with complex components. It has been more and more widely used in the modern research of traditional Chinese medicine (TCM). Based on a large number of documents, this experiment will use the HPLC-Q-Trap-MS/MS method to determine the content of four representative components of atractylon, atractyloside A, atractylolide I, and atractylolide III in Cangzhu, which would provide the theoretical basis for comprehensive, objective, and accurate evaluation of the quality of Cangzhu.
Materials and Methods
Instruments and Equipment
Triple Quad 6500+ LC–MS/MS instrument (AB SCIEX, USA), Analyst 1.5.2 software data system (AB SCIEX, USA), SCQ-200 ultrasonic cleaner (Shanghai Spectroscopic Ultrasonic Equipment Factory), AE-240 electronic balance (Mettler Toledo Shanghai Co., Ltd.), KDM type temperature regulating electric heating jacket (Heze Mudan Junteng Electronic Instrument Co., Ltd.), MDF-492 ultra-low temperature refrigerator (Sanyo, Japan), SK-1 rapid mixer (Jiangsu Jintan Medical Device Factory).
Reagents
Atractylon, atractyloside A, atractylolactone I, atractylolactone III (Chengdu Plant Standard Chemical Pure Biotechnology Co., Ltd., Batch numbers 191207, 200417, 191026, 200301), methanol, acetonitrile (UPLC grade, Thermo Fisher, USA), formic acid (UPLC grade, Tianjin Kemi European Chemical Reagent Co., Ltd.), purified water (Watson’s Drinking Water Co., Ltd.). Cangzhu (Liaoning, fried, Inner Mongolia (Tongrentang), Zhejiang, Inner Mongolia, fried with bran, three, four, five, Heilongjiang, etc., numbered 1 to 10, a total of 10 batches). Cangzhu was identified by Mr. Sunjikai at Qiqihar Medical University.
Methods
Analysis Conditions
Chromatography Conditions.
ZPRBAX SB-C C18 (100 mm × 2.1 mm, 1.8 µm), the mobile phase was 0.1% formic acid water (A) and 0.1% formic acid acetonitrile (B). Gradient elution was as follows: 0 min, 95.0% A, – 2.5 min, – 50.0% A, – 5 min, – 5% A, – 7 min, – 5% A, – 7.5 min, – 95.0% A. Flow rate was 0.30 mL min–1, and column temperature was 40℃. The injection volume was 1 µL.
Conditions of the Mass Spectrum
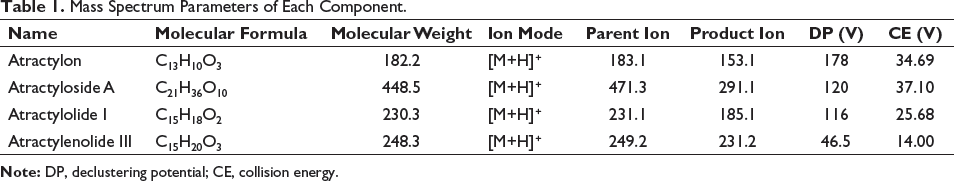
The electric spray ion source was electrospray ionization (ESI). Multiple reaction monitoring (MRM) mode was set in the positive ion collection mode. Curtain air was 25.00 psi. The gas ion source temperature was 55.00℃. The spray voltage was 4,500 kV. The atomization temperature was 550.0℃; the mass spectrum parameters of the four components are shown in Table 1.
Mass Spectrum Parameters of Each Component.
Preparation of Reference Solution
All of the standard substrates, namely, atractylon, atractyloside A, and atractylolactone I as well as atractylolactone III, were accurately weighed, and they were diluted with methanol 10 times. Then, we prepared them in the concentration of 100 µg mL–1 mixed reference solution.
Preparation of Test Solution
According to the relevant provisions under the “content determination” in the Chinese Pharmacopoeia (2020 edition), we prepared the sample solution of No. 1-10. Methanol was the solvent used to prepare two parallel samples following the certain solid–liquid ratio (1:10). Ultrasonic extraction was used to extract for 40 min. After cooling, methanol was used to supplement the weight loss, and centrifuge at 1,500 rpm for 5 min. The supernatant was taken to pass 0.22 µm microporous membrane.
Methodology Validation
Repeatability Test.
1 µL of the same sample solution was taken under “2.3” to inject 6 times. The peak area was measured according to the chromatography conditions under “2.1,” and the precision of the instrument was inspected. After calculation, the relative standard deviation (RSD) values met the requirements. The RSD values of atractylon, atractyloside A, atractylolactone I, and atractylolactone III were 2.6%, 1.7%, 1.0%, and 2.2%, respectively. The result indicated good repeatability.
Recovery Test
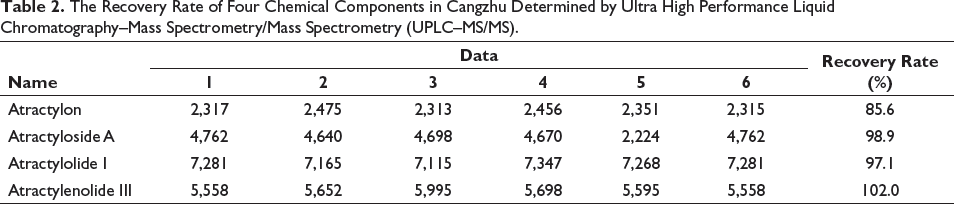
No. 2 sample was accurately weighed at 0.2538 g to prepare the sample solution according to the preparation method of “2.3,” and the reference substance was precisely added within the content of 1:1 into the test sample. Under the conditions of “2.1,” data were determined, and the recovery rate was calculated. The results showed that the recovery rate of atractylon was 87.4%, the recovery rate of atractyloside A was 115.7%, the recovery rate of atractylolactone I was 107.5%, and the recovery rate of atractylolactone III was 105.0%. The recovery rates were in the range of 75.0–125.0%, which was considered to meet the requirements. The standard is according to guidelines for single laboratory validation of chemical methods for dietary supplements and botanicals (Table 2).
The Recovery Rate of Four Chemical Components in Cangzhu Determined by Ultra High Performance Liquid Chromatography–Mass Spectrometry/Mass Spectrometry (UPLC–MS/MS).
Linearity
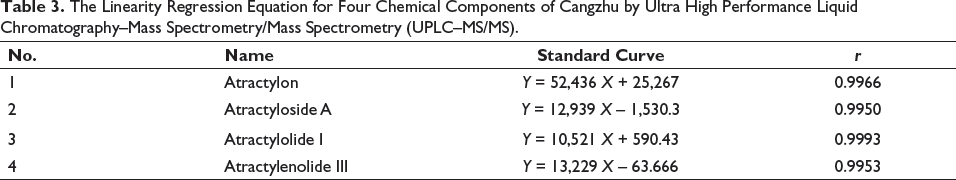
The reference solution was accurately measured and mixed by diluting it with methanol to prepare a series of reference solutions. Then, the solution was determined under the analysis conditions of “2.1.” Take the concentration of each reference substance (ng mL–1) as the abscissa and the response intensity as the ordinate, and draw the standard curve as shown in Table 3. The linearity of atractylon was 62.50 ng mL–1, 125.0 ng mL–1, 250.0 ng mL–1, 500 ng mL–1, 1,000 ng mL–1. The linearity of atractyloside A is 12.50 ng mL–1, 25.00 ng mL–1, 1,000 ng mL–1, 1,500 ng mL–1, 2,000 ng mL–1. Concentration range of atractylolide I was 31.25 ng mL–1, 62.50 ng mL–1, 125.0 ng mL–1, 250.0 ng mL–1, 500.0 ng mL–1. Atractylolactone III had a good linearity relationship within the linear range of 62.50 ng mL–1, 125.0 ng mL–1, 250.0 ng mL–1, 500.0 ng mL–1, and 1,000 ng mL–1.
The Linearity Regression Equation for Four Chemical Components of Cangzhu by Ultra High Performance Liquid Chromatography–Mass Spectrometry/Mass Spectrometry (UPLC–MS/MS).
Limit of Quantification
The limits of quantification of atractylon, atractylodes A, atractylodes I, and atractylodes III were 0.6305 ng mL–1, 1.387 ng mL–1, 10.50 ng mL–1, and 9.241 ng mL–1, respectively, which were determined using the signal-to-noise ratio method within S/N = 10.
Stability Investigation
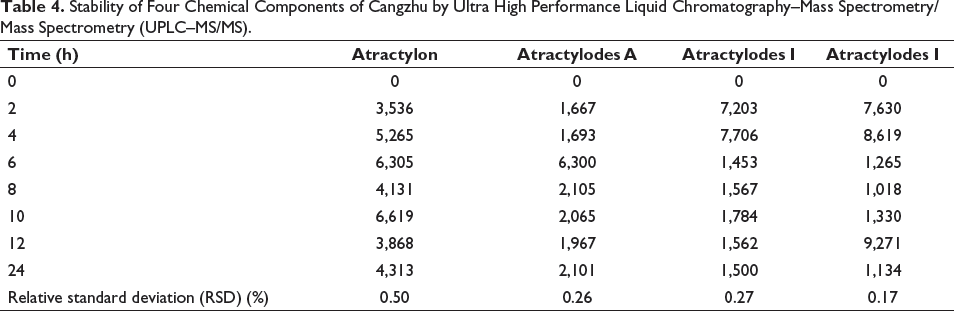
The No. 2 Cangzhu sample solution was accurately measured at 0 h, 2 h, 4 h, 6 h, 8 h, 10 h, 12 h, and 24 h, and 1 µL was injected each time, according to the preparation method in the part of “2.3.” RSD values were 0.50%, 0.26%, 0.27%, and 0.17%, respectively. The results indicated that Cangzhu samples were stable based on the concentration of atractylon, atractylodes A, atractylodes I, and atractylodes III. The standard is according to guidelines for single laboratory validation of chemical methods for dietary supplements and botanicals (Table 4).
Stability of Four Chemical Components of Cangzhu by Ultra High Performance Liquid Chromatography–Mass Spectrometry/Mass Spectrometry (UPLC–MS/MS).
Content Determination
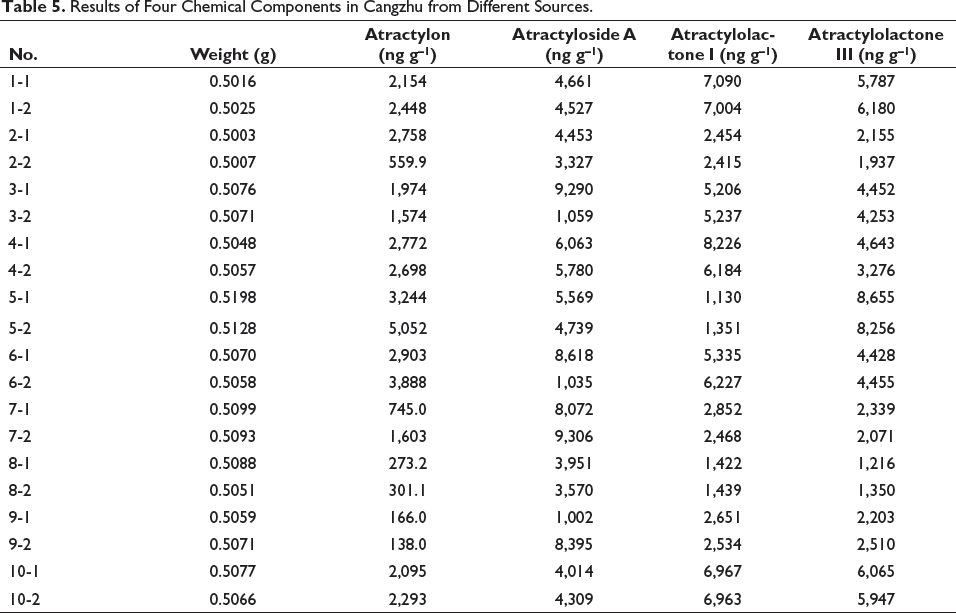
The samples were measured under the condition of “2.3,” and the contents of four components were determined in Cangzhu from different sources from No. 1–10. According to the standard curve, the contents of atractylon, atractyloside A, atractylolactone I, and atractylolactone III were listed in Table 5, which were in the solution of test products No. 1–10. After calculation, all RSDs were less than 3.0%. The results showed that the contents of both atractylon and atractylolactone I were relatively higher in No. 5, and the content of atractyloside A was relatively higher in No. 7. The content of atractylolactone I was relatively higher in No. 4. The results showed that the content of four components in Cangzhu (No. 6) after standardized processing was higher than that of Cangzhu derived from medicinal materials market, suggesting the necessity of standardized operation. In terms of growth years, the results suggested that the content of various components of Cangzhu born in four years (Qiqihar) was relatively high.
Results of Four Chemical Components in Cangzhu from Different Sources.
Discussion
In the early stage of the study, mobile phase ratio and mass spectrum conditions were investigated and optimized. Three elution procedures were investigated, among which, procedure 1. Three elution procedures were investigated, and the separation of atractyloside I from atractyloside A was poor and the baseline was unstable. Program 2 provided that the baseline was unstable, the peak interval between atractylolactone I and atractylon, and the total time was too long.
Cangzhu has many pharmacological activities, and the content determination of the index component is named atractylon. It is a polyethylene–alkyne component, which is selected as a representative of the identified compounds for mass spectrum analysis. Take atractylon and atractyloside A for example: In the positive ion mode, atractylon excimolecule ion peak m/z 183.07, the molecular formula is C13H10O, atractylon structure total furan ring is unstable, easy to open the ring, loss of –CH3, –CO and other groups, forming daughter ion fragments, among them, m/z 153.06 daughter ion fragments in the determination of high ionic strength. It should be [M+H–CH2O]+. In addition, atractylon may also undergo α-cleavage to form m/z = 67.93 fragment ions, but the ionic strength is low. Therefore, the ion pairs selected are m/z 183.0 and m/z 153.06 in the experiment. Atractyloside A combines with sodium ions in the positive ion mode to form the molecular ion peak [M+Na]+, m/z 471.30. Under the action of high voltage, the cracking occurs, and the loss of –H2O and the removal of a molecule of glucose group, resulting in m/z 291.15 fragment ion, which is relatively stable and has high strength. Therefore, the above two ions are selected.
In the study, ultra high performance liquid chromatography–mass spectrometry/mass spectrometry (UPLC–MS/MS) technology was used to determine the contents of atractylon, atractyloside A, atractylolactone I, and atractylolactone III in Cangzhu. In this experiment, different gradient elution conditions were investigated, and it was found that the components measured under the No. 3 elution condition had symmetrical peak shape, stable peak time, and good repeatability. After mass spectrum and liquid chromatography were connected, the ion source temperature, atomization temperature, and gas curtain pressure were optimized. Meanwhile, according to the mass spectrum analysis of atractylon, the ion pairs selected were m/z 183.0 and m/z 153.06 in the experiment.
The results showed that the contents of atractylon, atractylolactone III, atractyloside A, and atractylolactone I were relatively high in Cangzhu derived from Inner Mongolia, Qiqihar Medical College, and Zhejiang Province, respectively. The contents of four components in the stir-dried bran (A Rong Qi) product after standardized processing were higher than those from the market, suggesting the necessity of standardized operation. In terms of growth years, the results suggested that the contents of various components in Cangzhu born at four years (Qiqihar) were relatively high.
The methodological study showed that the method was simple, rapid, and sensitive, which was suitable for the determination of Cangzhu’s content, and could provide reference for the quality evaluation of Cangzhu from different sources.
Conclusion
In this study, the UPLC–MS/MS method was used to determine the contents of atractylon, atractylolactone III, atractyloside A, and atractylolactone I in Cangzhu. The results showed that both atractylon and atractylolactone III were relatively higher in Cangzhu produced in Inner Mongolia, and the content of atractyloside A was relatively higher in three-year-old Cangzhu (Qiqihar Medical University). The content of atractyloside A was relatively higher in atractyloside A. The content of atractylolactone I was relatively higher in Cangzhu produced from Zhejiang. The results showed that the content of four components in the stir-fried bran (A Rong Qi) product after standardized processing was higher than that of Cangzhu derived from the medicinal market, which suggested the necessity of standardized operation. In terms of growth years, the results suggested that the various components may be bigger in three-year-old Cangzhu (Qiqihar). The results showed that the method was simple, rapid, and sensitive, which was suitable for the determination of Cangzhu, which would provide a reference for the quality evaluation of a Cangzhu from different sources.
Abbreviations
UPLC-MS/MS: Ultra high performance liquid chromatography–mass spectrometry/mass spectrometry; MRM: Multiple reaction monitoring; RSD: Relative standard deviation; TCM: Traditional Chinese medicine; HPLC: High performance liquid chromatography; MS: Mass spectrometry; DP: Declustering potential; CE: Collision energy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Qiqihar Science and Technology Bureau joint guidance project (LHYD-202009). It was also supported by the Postdoctoral Funding from Heilongjiang Province (LBH-Z23300).
