Abstract
Background
Stroke is a collection of disorders that develop due to the rapid rupture of blood vessels in the brain or due to the blockage of blood flow. Ischemic stroke (IS) has high mortality as well as morbidity rates.
Objectives
The current work aimed to investigate the neuroprotective activities of pinobanksin against cerebral ischemic/reperfusion (C I/R) injury in a rat model through its anti-inflammatory properties.
Materials and Methods
Male Wistar albino rats underwent the middle cerebral artery occlusion (MCAO) method to initiate I/R injury. The experimental rats were orally treated with pinobanksin at various doses after 2 hours of MCAO operation. The infarct size, neurological score, edema and water content, and Evans blue assay were assessed. The inflammatory cytokines and biomarker (nuclear factor-kappa B (NF-κB), cyclooxygenase-2 (COX-2), and prostaglandin E2 (PGE2)) levels were studied using commercial kits. Histopathological analysis was performed on the brain tissues.
Results
The treatment with pinobanksin successfully mitigated the neurological deficits, edema and water content, infarct size, and Evans blue leakage in the C I/R-induced rats. The pro-inflammatory cytokine levels were reduced and interleukin-10 was increased by the pinobanksin in the I/R rats. The pinobanksin also effectively reduced NF-κB, PGE2, and COX-2 levels in I/R rats. The pinobanksin treatment effectively attenuated the brain histological alterations.
Conclusion
Altogether, this work highlighted the neuroprotective activity of pinobanksin in the C I/R injury in rats. The present findings could aid in the development of pinobanksin as a novel therapeutic intervention to prevent or reduce IS-induced brain injury.
Introduction
Ischemic stroke (IS) is a highly debilitating neurological condition that impacts a significant number of individuals and is the second foremost cause of mortality globally. Stroke is a collection of disorders that develop when there is a rapid blood vessel rupture in the brain or when the brain tissue is injured due to blood flow blockage (Saini et al., 2021). It is well known for its high rates of death and disability. Based on epidemiological data, the worldwide prevalence of stroke is projected to be around 80.1 million incidences. In 2016, 13.7 million new cases of stroke were reported (GBD 2019 Stroke Collaborators, 2021). Research has established that cerebral ischemia negatively affects the ability to learn and memory in patients with IS. Additionally, it has been found that 25–30% of individuals who survive an IS experience either sudden or delayed cognitive impairments (Kalaria et al., 2016; Vicentini et al., 2017).
Neuroinflammation has been known as the primary pathogenic cause of cerebral ischemic/reperfusion (C I/R) injury following an IS. The inflammation following cerebral ischemia contributes to neuronal apoptosis, dysregulation of the blood–brain barrier (BBB), and subsequent impairment of brain tissue and neural function (Shi et al., 2019). The significance of ongoing inflammatory processes during ischemia is well known, which involve inflammatory cell recruitment in the circulation, pro-inflammatory cytokine release, and the activation of microglia cells (Jin et al., 2010). Previous data suggest that inflammation can cause severe tissue damage, including the activation of harmful enzymes in brain tissue and the accumulation of free radicals (Gill et al., 2010). Studies have indicated that C I/R can cause damage to the cell membranes of neurons and trigger the activation of microglia cells. This activation generates pro-inflammatory markers, which worsen the damage to the neurons. Therefore, by suppressing the inflammatory response, it is possible to reduce neuronal ischemia injury and minimize the extent of the infarction (Kaushal & Schlichter, 2008; Salman et al., 2017).
There are currently no specific strategies that can effectively mitigate cognitive impairments caused by C I/R injury. The increased inflammatory response in the brain is a major cause of the cognitive impairments caused by C I/R injury (Kandasamy et al., 2020; Pantoni, 2017). Reperfusion, which refers to the restoration of blood supply, is the sole method for treating cerebral ischemia. Nevertheless, the process of reperfusion unavoidably results in extensive cell death and tissue damage inside the perfusion region, a condition known as C I/R injury. Recombinant tissue-type plasminogen activator (r-tPA) is the most efficient method to reestablish blood flow in IS. However, only a small number of patients with stroke, approximately 3–5%, can be adequately treated because of the limited time frame for r-tPA treatment. Neuroprotective drugs may be advantageous for patients with IS during the blood flow restoration during the late stage (Powers et al., 2018).
The middle cerebral artery occlusion (MCAO)-induced C I/R injury model is commonly employed in analyses of cerebral ischemia because it resembles the pathophysiological characteristics observed in patients (Gharbawie et al., 2006). MCAO triggers the onset of IS, which is triggered by the blockage of brain blood flow. This results in a series of mechanisms, like neuronal cell degeneration and neuroinflammation, which thereby results in the death of neuron cells (Speetzen et al., 2013). While the precise molecular pathways of C I/R injury remain poorly known, inflammation is the foremost cause that shows significant damage to biological macromolecules, leading to damage to neurons and tissues (Durukan & Tatlisumak, 2007). Hence, a new approach to treating cognitive impairments following IS would involve specifically addressing the inflammation in the brain caused by C I/R injury.
Pinobanksin, a major bioactive flavonoid compound, is extensively found in propolis and honey. Pinobanksin has been well-reported for its antioxidant (Zheng et al., 2018), antimicrobial (Kasote et al., 2015), antifungal (Yang et al., 2011), antiproliferation (Chen et al., 2023), and antiangiogenic (Bang & Ahn, 2021) effects. An earlier study reported that pinobanksin exerted neuroprotective effects on experimental vascular dementia in rats through its antioxidant properties (Liu et al., 2015). Hence, we hypothesized that pinobanksin can show neuroprotective activities against C I/R injury. Thus, the current work aimed to evaluate the neuroprotective activities of pinobanksin against C I/R injury in a rat model through its anti-inflammatory properties.
Materials and Methods
Experimental Rats
The present work employed male Sprague-Dawley rats weighing between 210 and 250 g. The rats were sustained in controlled environments. The temperature was set at a constant 22 ± 2°C, and humidity was kept at 55 ± 5%. The rats were exposed to a 12-hour light/dark sequence. The institutional ethics committee for animal experiments verified and approved all animal experimental protocols.
Establishment of MCAO-induced C I/R Injury Model
The rats were administered anesthesia using 1% pentobarbital sodium (0.45 mL/kg). They were then kept anesthetized using isoflurane (1.5%) and oxygen (80%) through an evaporator. The middle cerebral artery was surgically taken and a filament measuring 18 mm was inserted by a tiny incision measuring 5 mm in the common carotid artery and extended to the internal carotid artery in order to induce blockage of the middle cerebral artery, following the previous method (Longa et al., 1989). Following 120 minutes of occlusion, the filament was carefully taken out, allowing blood to flow back to the brain. The identical procedure was carried out in the sham-operated rats (control) without placing the filament. During the MCAO operation, the rats’ body heat was maintained at a constant level of 37 ± 0.5°C using a thermostat surgery tray. Following the operation, the rats were placed on the surgical tray until they regained consciousness, after which they were returned to their cabins. The experimental rats were categorized into five groups, with six rats (n = 6) in each. Group I (sham-operated control), group II (MCAO-operated rats), and group III–V (MCAO + 5, 10, and 20 mg/kg of pinobanksin, respectively, treated rats). The pinobanksin treatment was given orally to the experimental rats in groups III and IV after 2 hours of the MCAO surgery.
Analysis of Neurological Deficit Score
Following 24 hours of I/R induction, the neurological score was assessed and carried out, as previously stated (Gerriets et al., 2004). The scoring method was described as grade 0, which indicates the absence of any neurological impairment. Grade 1 refers to the inability to completely extend their left forepaw. Grade 2 signifies frequent circling to the left. Grade 3 involves falling to the left. Grade 4 is characterized by the absence of spontaneous walking and a decreased degree of consciousness.
Analysis of Brain Edema and Water Content
The wet/dry approach was employed to analyze brain edema. Following a neurological deficit assessment, the rats were sacrificed and their brains were promptly retrieved and measured for their weight (wet weight). Following 72 hours of drying at a temperature of 70°C, the brains were reweighed to assess their dry weight. The level of cerebral edema and/or water content was assessed using the formula: wet weight − dry weight/wet weight × 100% (Gerriets et al., 2004).
Analysis of Cerebral Infarction
The retrieved brains were immersed in a dish filled with saline, and the protective membranes surrounding the brain were separated and divided into five cross-sectional slices, each measuring 2 mm in width. The brain sections were submerged in a 0.5% 2,3,5-triphenyltetrazolium chloride (TTC) solution for 15 minutes and subsequently treated with 4% paraformaldehyde for fixation. The brain tissues were analyzed based on their color as follows: The color white was used to represent the infarct area, whereas the color red was used to represent the noninfarcted area (Popp et al., 2009). The brain infarct area was calculated using the ImageJ program.
Evans Blue Extravasation Assay
To examine alterations in BBB permeability, the rats were subjected to intraperitoneal injection with 2% Evans blue at a 4 mL/kg concentration. After 24 hours of reperfusion, regular saline was introduced for heart perfusion. Next, the brain tissues that had been dissected were processed through homogenization and centrifugation to get supernatants. These supernatants were then assessed using a spectrophotometer at 620 nm.
Analysis of Inflammatory Markers
The inflammatory cytokine levels were assessed in the brain tissue and serum samples of the experimental rats. The tissues were mechanically disrupted in a Tris buffer solution containing Triton X-100 (1%), glycerol (10%), NaCl (150 mM), Tris-HCl (50 mM), pH 7.4, and a protease inhibitor combination for 20 minutes. Afterward, the suspension was centrifuged at 10,000 g for 20 minutes. The resultant supernatant was utilized to quantify the tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and interleukin-1 beta (IL-1β). These marker levels were studied using commercially available assay kits. The procedure provided by the manufacturer (Elabscience, USA) was followed for the estimations in triplicate.
Analysis of Prostaglandin E2 (PGE2), Cyclooxygenase-2 (COX-2), and Nuclear Factor-Kappa B (NF-κB) Levels
The commercial assay kits were employed to measure the influence of pinobanksin treatment on the levels of PGE2, NF-κB, and COX-2 in the serum of the experimental rats. The guidelines provided by the manufacturer (Abcam, USA) were followed for the experiments in triplicate.
Histological Analysis
The brain tissues were taken out, dehydrated using graded ethanol, and fixed in Bouin’s fixative buffer for 24 hours. Afterward, the tissues were paraffinized and cut into 4–6 µm size. These tissue slides were then stained using eosin and hematoxylin and assessed using an optical microscope.
Statistical Analysis
The results are reported as a mean ± SD of triplicate assays. The statistical assays were performed using GraphPad Prism 5.0 software. Group variations were studied using a one-way analysis of variance (ANOVA) and a Newman–Keuls test. Significance was indicated by “*” p < 0.01 and “#” p < 0.05.
Results
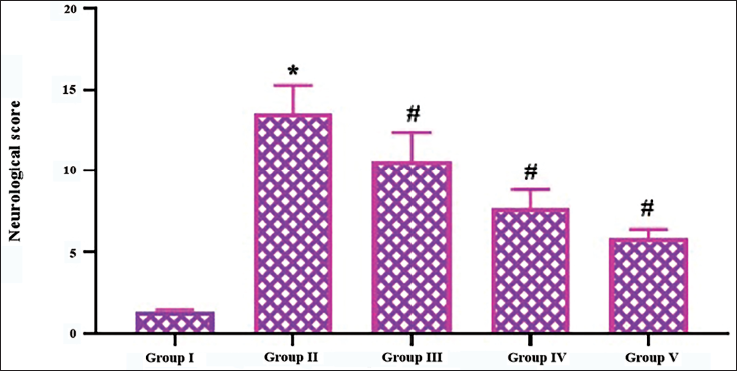
Effect of Pinobanksin on the Neurological Deficit Scores of Experimental Rats
Figure 1 demonstrates the effect of pinobanksin treatment on the neurological scores in C I/R injury-operated rats. The IS-induced rats by ischemic/reperfusion injury exhibited significantly elevated neurological scores, which were determined due to the presence of frequent circling and falling to the left, the absence of spontaneous walking, and decreased consciousness. Remarkably, the pinobanksin treatment at concentrations of 5, 10, and 20 mg/kg in the C I/R injury-induced rats resulted in a considerable reduction in neurological deficit scores (Figure 1).
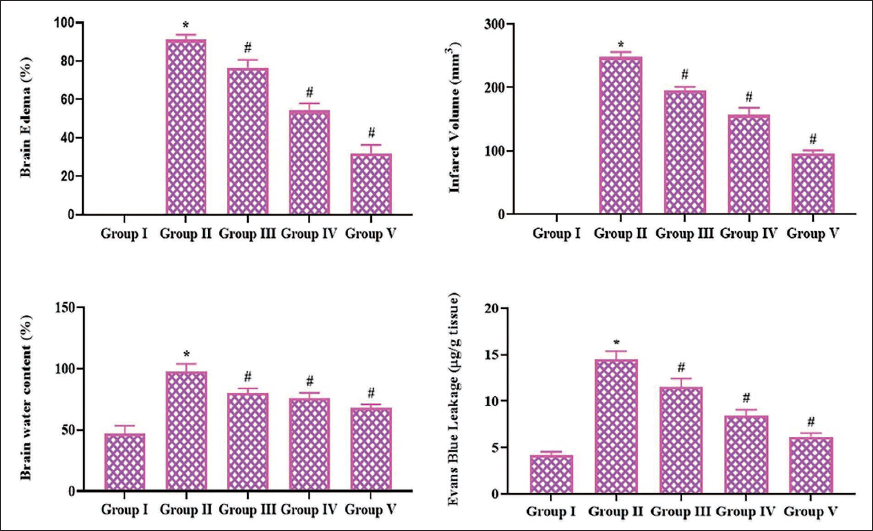
Effect of Pinobanksin on the Brain Edema and Water Content, Infarct Volume, and Evans Blue Extravasation in Experimental Rats
The levels of brain edema and water content, infarct size, and Evans blue extravasation in the experimental rats were presented in Figure 2. The C I/R injury-induced rats exhibited a considerable elevation in brain edema and water content, infarct size, and Evans blue extravasation levels when compared to the control. Whereas, the treatment of pinobanksin at various concentrations (5, 10, and 20 mg/kg) effectively diminished the levels of edema and water content, infarct size, and Evans blue leakage in the C I/R rats (Figure 2).
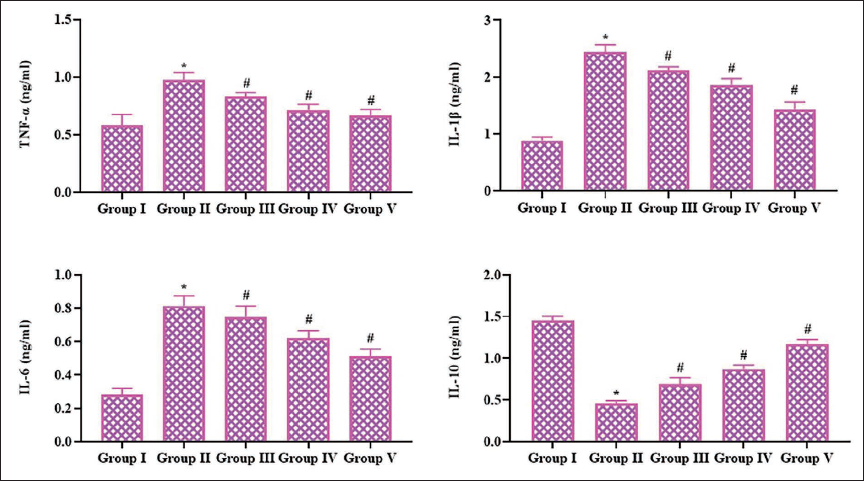
Effect of Pinobanksin on the Inflammatory Cytokine Levels in the Serum of Experimental Rats
Figure 3 illustrates the inflammatory cytokine levels in the serum of experimental rats. The findings demonstrate a significant increase in IL-6, TNF-α, and IL-1β and a reduction in interleukin-10 (IL-10) in the C I/R injury rats. Conversely, the 5, 10, and 20 mg/kg of pinobanksin treatment in the I/R rats resulted in a diminution of IL-6, TNF-α, and IL-1β and boosted the IL-10 levels in their serum.
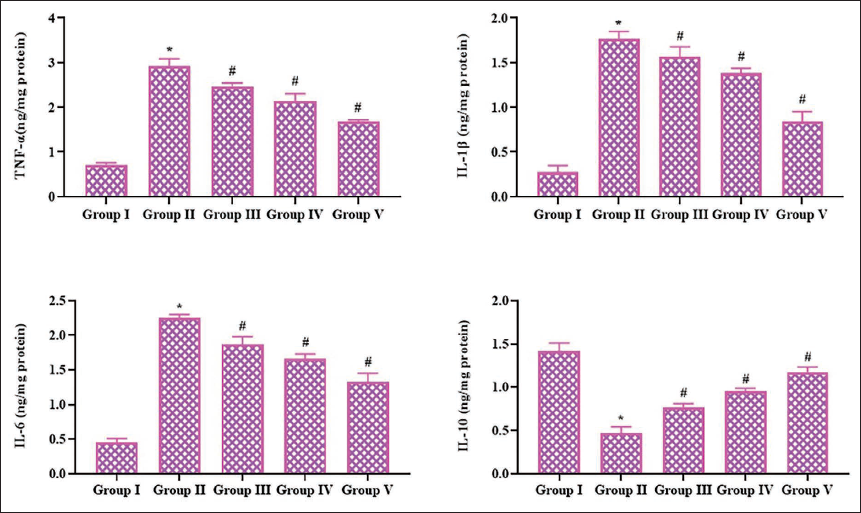
Effect of Pinobanksin on the Inflammatory Cytokine Levels in the Brain Tissue Homogenate
Figure 4 reveals the impact of pinobanksin treatment on the concentrations of IL-1β, IL-6, TNF-α, and IL-10 in the brain tissue homogenates of the rats. These cytokine levels were elevated in the brain tissues of the rats with C I/R injury. The C I/R rats also exhibited a reduced level of the anti-inflammatory cytokine IL-10 in their brain tissues. Notably, the pinobanksin (5, 10, and 20 mg/kg) treatment successfully reduced these cytokine levels while improving the IL-10 levels in the brain tissues of the C I/R rats.
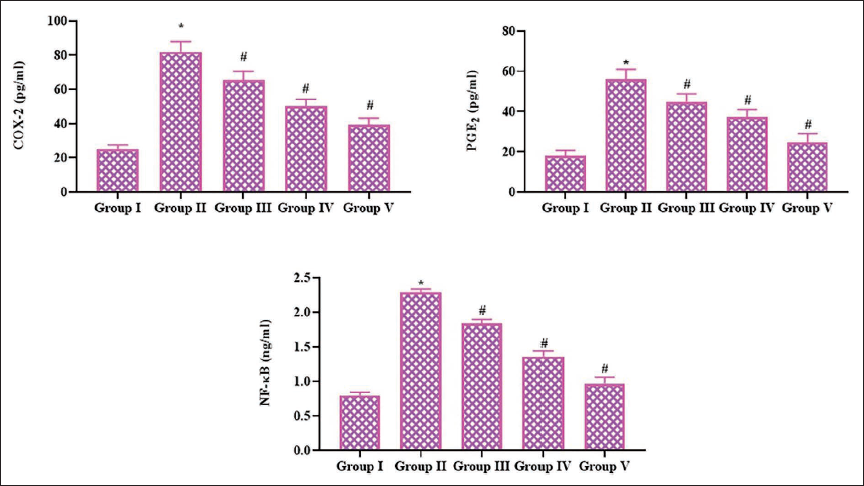
Effect of Pinobanksin on the Inflammatory Biomarker Levels of Experimental Rats
The PGE2, COX-2, and NF-κB levels were measured in the serum of experimental rats, and the findings are given in Figure 5. The rats with C I/R injury exhibited an elevation in the levels of PGE2, NF-κB, and COX-2 in the brain tissues compared to the control. Nevertheless, the treatment of pinobanksin at various concentrations (5, 10, and 20 mg/kg) effectively diminished the PGE2, COX-2, and NF-κB levels in their serum (Figure 5).
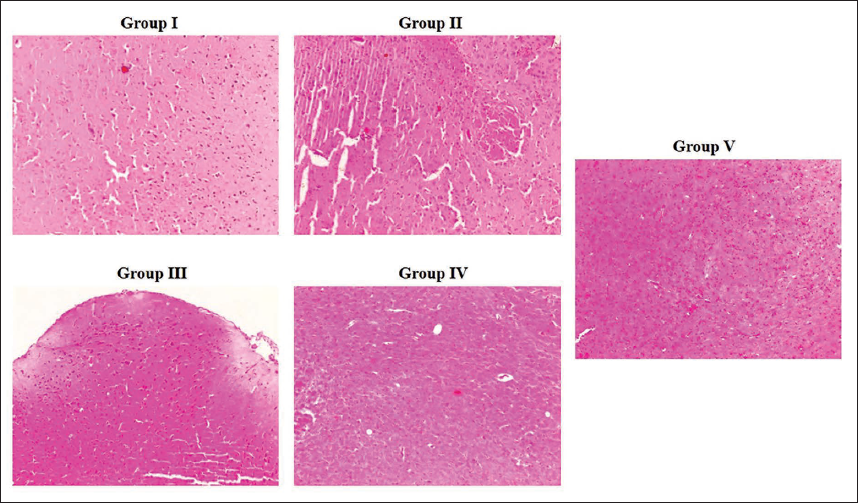
Effect of Pinobanksin on the Brain Histology of Experimental Rats
The brain tissues of the experimental rats exhibited typical tissue architecture and neuronal arrangements. On the other hand, the C I/R injury-induced rats demonstrated extensive neuronal damage, inflammatory signs, neuronal degeneration, and cell death (Figure 6). Interestingly, the pinobanksin treatment at 5, 10, and 20 mg/kg concentrations remarkably diminished neuronal degeneration, neuronal apoptosis, and other changes in the C I/R injury-induced rats, which proves the neuroprotective activities of the pinobanksin against C I/R injury.
Discussion
Stroke significantly impairs various brain functions, including locomotor activity, memory, reasoning, and language. Prompt reperfusion is an efficacious approach for managing stroke. Nevertheless, reperfusion may also cause further detrimental effects on neurological functions (Vukadinović et al., 2019). Hence, it is crucial to discover efficacious neuroprotective drugs that can effectively manage ischemic/reperfusion injury. The present work aimed to study the neuroprotective activities of pinobanksin against the MCAO-induced C I/R injury model in rats. C I/R injury has been found to be the leading cause of cerebral infarction and nerve damage. The primary signs for evaluating brain damage are neurological deficiency scores, brain water content, and edema (Orellana-Urzúa et al., 2020). In this study, the C I/R-induced rats showed an increase in the neurological score, edema, infarct size, brain water content, and BBB permeability as a consequence of C I/R injury. Reactive oxygen species (ROS) are crucial in enhancing the permeability of the BBB, resulting in tissue swelling and a subsequent rise in brain water content (Dhar et al., 2023). In this work, we observed that pinobanksin treatment substantially diminished brain edema, infarct size, water content, and BBB permeability in the I/R rats. These outcomes evidenced that the pinobanksin treatment effectively reduced the major symptoms of the C I/R injury in rats.
The pathological causes of cerebral ischemic injury are diverse and complicated. However, it has been documented that inflammation is the fundamental mechanism implicated in the pathology of IS (Herson & Traystman, 2014). Cellular inflammatory responses are known to have a considerable effect on the development of cerebral ischemia. Cerebral ischemia can lead to a decrease in the availability of oxygen and nutrients, which, in turn, disrupts the normal energy production of nerve cells, resulting in mitochondrial oxidative stress (Pawluk et al., 2022). The inflammatory response is promptly activated during ischemia, leading to the worsening of brain damage and less functional recovery of neuron cells. Oxidative stress leads to elevated levels of inflammatory cytokines and is strongly linked to damage due to the excessive release of these signaling molecules. The inflammatory reactions during C I/R can trigger a series of physiological processes that may ultimately cause neuronal cell death and neurological impairment, ultimately resulting in brain injury (Pluta et al., 2021). Inflammation also serves a significant role in the advancement of secondary brain injury due to ischemia, resulting in dysregulation of the BBB and cerebral edema formation. Thus, medicines possessing anti-inflammatory properties can be beneficial in reducing cerebral ischemia injury (García-Sánchez et al., 2020).
Proinflammatory cytokines have a critical role in the inflammatory reactions caused by C I/R injury (Dhuriya & Sharma, 2018). A report indicates that TNF-α becomes activated during cerebral ischemia. The excessive production of TNF-α is induced by the activation of cells involved in inflammation during cerebral ischemia, which leads to increased neuronal damage in brain tissues (Chen et al., 2020). The cytokine levels increase due to the leaking of leukocytes into the bloodstream, leading to the entry of neutrophils, macrophages, and microglia into the ischemic region. TNF-α causes injury to the cell linings of the blood vessels in the brain and increases the BBB permeability, which leads to brain edema development (Wang et al., 2022). Apart from IL-1β and IL-6, other cytokines and NF-κB are significant factors in brain edema development (Hou et al., 2018). The current work aimed to assess the pro-inflammatory cytokine levels in the I/R rats. The objective was to establish if pinobanksin has any anti-inflammatory properties in a rat C I/R model. The findings of our work show that pinobanksin remarkably reduced the levels of proinflammatory cytokines. This indicates that pinobanksin has an anti-inflammatory effect during MCAO-induced C I/R injury.
NF-κB is an imperative transcription factor that becomes active upon the reperfusion of cerebral ischemia. NF-κB activation participates in the initiation of different inflammatory reactions following brain ischemia. NF-κB activation induces the release of inflammatory mediators, which play a role in the brain damage that occurs during C I/R (Meng et al., 2021). Additionally, some data support the participation of NF-κB activation in the IS (Puleo et al., 2022). NF-κB controls the process of transcribing specific genes. This results in the generation and release of pro-inflammatory regulators, which worsen neuronal death and aggravate C I/R injury (Cao et al., 2023). Therefore, reducing the activity of NF-κB may help reduce brain injury caused by C I/R injury. Thus, targeting NF-κB is a new and effective method for treating cerebral ischemia. The current work found that pinobanksin considerably decreased the levels of NF-κB, indicating its potential as an anti-inflammatory agent for treating cerebral ischemia.
COX-2 and NF-κB are important inflammatory mediators that contribute significantly to the C I/R injury. During C I/R injury, the level of the aforementioned markers was increased in the brain region (Liang et al., 2021). COX is believed to play a key role in maintaining brain homeostasis. COX-2 has been detected in neuronal cells. Earlier studies indicate that there is an increased presence of COX-2 in the neurons of the infarcted region (Abd El-Aal et al., 2013). Research has extensively demonstrated that COX-2 serves a crucial role in protecting brain tissue against C I/R injury (Maida et al., 2020). Rodents with higher levels of COX-2 have larger volumes of infarcts following an experimental stroke. Pharmacologically suppressing the activity of COX-2 has been said to be an effective approach for stroke in experimental animal models. Interestingly, the findings of the present work have proved that the pinobanksin treatment significantly decreased COX-2 levels, indicating its therapeutic potential against cerebral ischemia.
Activated COX-2 is considered to initiate the development of PGs (Wei et al., 2016). Prostanoids, specifically PGE2 and PGD2, are produced via COX signaling. The activated PGE2 serves as the regulator of stroke-induced damage (Yuan & Zhang, 2021). Following cerebral ischemia, the levels of PGE2 increase, causing damage to neuronal cells and further brain injury (Li et al., 2021). Remarkably, the outcomes of this study have evidenced that pinobanksin has reduced the PGE2 levels in the C I/R-induced rats. These results highlight the therapeutic effects of pinobanksin against cerebral ischemia.
Conclusion
Altogether, the present work highlighted the neuroprotective activity of pinobanksin in the context of MCAO-induced C I/R injury in rats. Pinobanksin treatment remarkably decreased the infarct size, prevented neuronal death, reduced behavioral abnormalities, and mitigated brain histological changes. Furthermore, pinobanksin enhanced BBB integrity and reduced inflammatory responses in C I/R injury-induced rats. The present findings could assist in the development of pinobanksin as a new therapeutic agent to prevent or reduce brain injury caused by IS. In addition, additional works still need to be completed in the future to elucidate the precise therapeutic effects of pinobanksin against the IS condition.
Abbreviations
MCAO: Middle cerebral artery occlusion; C I/R: Cerebral ischemia/reperfusion; IS: Ischemic stroke; BBB: Blood–brain barrier; ROS: Reactive oxygen species; TNF-α: Tumor necrosis factor-alpha; IL-6: Interleukin-6; IL-1β: Interleukin-1 beta; IL-10: Interleukin-10; NF-κB: Nuclear factor-kappa B; COX-2: Cyclooxygenase-2; PGE2: Prostaglandin E2; TTC: 2,3,5-Triphenyltetrazolium chloride; ANOVA: Analysis of variance; SD: Standard deviation; r-tPA: Recombinant tissue-type plasminogen activator; I/R: Ischemia/reperfusion.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This work was approved by the institutional ethical committee, Xinchang Hospital affiliated to Wenzhou Medical University·Xinchang County People’s Hospital-Shaoxing Zhejiang-312500-China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
