Abstract
Objective
The aim of this study was to examine the association between serum complement 1q (C1q) and the associated factors of acute ischemic stroke in patients with type 2 diabetes (T2DM).
Methods
The baseline clinical variables of the participants were collected, and the levels of blood lipids, blood sugar, inflammatory cytokines, and C1q in the three groups were then compared. The variables which affected the associated factors of acute ischemic stroke in T2DM cases were determined.
Results
The levels of C1q in the DAIS group were increased significantly compared with those in the T2DM group. Receiver operating characteristic curve analyses showed that the AUC for C1q and the combined diagnosis of acute ischemic stroke were 0.830 (95%CI 0.747–0.914), with a sensitivity of 0.854 and specificity of 0.780. The results of Pearson’s correlation analyses demonstrated that C1q was associated positively with low-density lipoprotein cholesterol (LDL-C), fasting blood glucose (PBG), 2-h postprandial blood glucose (2h PG), and high-sensitive C reaction protein (hs-CRP) (all p < .05). Stratified analysis showed that there was a positive relationship between C1q and the associated factors of acute ischemic stroke for partial LDL-C, and hs-CRP strata. Logistic model analysis suggested that C1q was an independent risk factor for acute ischemic stroke in patients with T2DM. After adjusting for potential confounders, a one-standard deviation (SD) increase in C1q level was strongly related to an approximately 1.5-fold increased risk of acute ischemic stroke in cases with a hs-CRP ≥1.78 mg/L.
Conclusion
In DAIS patients, the levels of C1q were increased significantly and were an independent associated factor which affected the occurrence of acute ischemic stroke.
Introduction
Type 2 diabetes mellitus (T2DM) has become a major public health problem in China, and is diagnosed according to progressive insulin resistance and hyperglycemia. 1 T2DM is caused by multiple reasons which result in insufficient insulin secretion in the body or insulin not being used effectively, leading to a continuous increase in blood sugar levels. 2 T2DM is also considered as an independent risk factor for cerebrovascular disorders, including acute ischemic stroke (AIS). 3 Stroke as a severe cerebrovascular event is a health burden on families and society. 4 Despite the advanced treatment available for stroke patients, stroke is the major cause of mortality and acquired disability in adults. 5 Stroke includes ischemic and hemorrhagic stroke, the former accounting for 80–87% of all stroke events. 6 Accurate diagnosis and appropriate management of AIS are therefore important to save the lives of patients.
The complement system is an important part of our immune defense against infections. It consists of a cascade of serum proteins which could be activated by three different cascades that each use pathway-specific recognition molecules, including complement 1q (C1q). 7 Complement activation can occur through three routes, the classical, the lectin and the alternative pathway. Both the three cascades converge at component C3, a central molecule in the complement system which ultimately triggers complement functions. 8 The classical pathway is activated by the recognition of an antigen-antibody complex by C1q. Upon binding, C1r cleaves C1s which in turn cleaves C2 and C4 into a small (C2b, C4a) and a large fragment (C2a, C4b). C2a and C4b together form the C3 convertase. Complement activation, especially C1q, plays an important role in the pathogenesis of inflammatory disorders including ischemia/reperfusion injury, sepsis, acute lung injury, allergy and asthma.8,9 Complement C1q is a key cofactor of both nonspecific and humoral immunity and is involved in the immunoregulation of various tumors. 8 Moreover, C1q activation has been found to associated with the presence and severity of diabetic nephropathy, indicating that its activation is involved in the development of renal pathology in patients with diabetes. 9 In addition, in a mouse of mild traumatic brain injury (TBI), C1q expression specifically in the corticothalamic system chronic up-regulate, 10 suggesting it is correlated to the brain injury.
Ischemic stroke leads to neuroaxonal injury and subsequent cerebral artery occlusion. 11 Therefore, the extent of neuroaxonal damage due to AIS may induce the expression of C1q protein into the peripheral blood. Previous studies have demonstrated that diabetes is associated with higher serum C1q levels, 12 and long-term outcomes after ischemic stroke. 13 C1q levels were shown to increase significantly in acute cerebral small vessel disease stroke and remain elevated for at least 3 months post-stroke. 14 A previous study also reported that C1q may exacerbate brain injury, which is associated with stroke, by modulating mitochondrial reactive oxygen species (ROS) generation. 15 However, it remains unclear whether C1q is a promising biomarker for diagnosing diabetic acute ischemic stroke (DAIS). We hypothesized that in T2DM patients without a history of stroke, those with high C1q levels would have a higher risk of AIS than those with lower levels. Therefore, the aim of the present study was to examine the association between C1q and the risk of acute ischemic stroke in patients with T2DM.
Subjects and methods
Patient cohort and study design
From January 2020 to December 2021, 48 T2DM patients with first-ever acute ischemic stroke were enrolled at the emergency department of our hospital (DAIS group), 50 patients with simple T2DM were also enrolled (T2DM group), and 51 healthy volunteers without dyslipidemia, hypertension conditions, a history of T2DM and acute ischemic stroke who were age- and gender-matched with the above two groups enrolled as the control group. These cases were collected within 48 h of suffering from a new focal or global neurological disorder. T2DM and acute ischemic stroke were diagnosed based on the World Health Organization criteria. The mean age of the T2DM group was 59.22 ± 11.06 years, with 28 (56.0%) men; the mean age of the DAIS group was 58.71 ± 12.51 years, with 48 (52.1%) men; and the mean age of the control group was 58.63 ± 10.52 years, with 27 (54.0%) men. The exclusion criteria were as follows: cases with malignancies, intracerebral hemorrhage, renal insufficiency, febrile diseases, a history of recent surgery or trauma, autoimmune diseases, or systemic infections during the period of the study. The study was approved by the ethics committee of the Central Hospital of Enshi Tujia and Miao Autonomous Prefecture. All participants provided written informed consent before enrollment.
Clinical data collection
The baseline clinical characteristics of all the subjects were collected including age, sex, body mass index (BMI), duration of diabetes, NIH stroke scale (NIHSS) score, history of smoking and alcohol intake, assessment of vascular risk factors (hypertension, atrial fibrillation, hypercholesterolemia, coronary heart disease, and family history for stroke), and biochemical factors (fasting blood glucose [PBG], 2-h postprandial blood glucose [2-h PG], fasting insulin [Fins], total cholesterol, triglycerides, high-density lipoprotein cholesterol [HDL-C], low-density lipoprotein cholesterol [LDL-C], glycosylated hemoglobin [hemoglobin A1C, HbA1c], high-sensitive C reaction protein [hs-CRP]), and C1q. The serum levels of C1q were measured using immune transmission turbidimetry.
Neuroimaging
Brain imaging (CT or MRI) was completed within 24–48 h after enrollment. Stroke was diagnosed according to the results of imaging based on the International Classification of Disease, 10th revision. Intracranial hemorrhage was excluded by CCT examination.
Statistical analyses
SPSS 20.0 software was used to analyze the data. The data were first tested to determine whether they had a normal distribution, with continuous variables expressed as mean ± standard deviation. Additionally, if data had no normal distribution, with continuous variables expressed as median (the interquartile range (IQR)). The acceptability of sample size was assessed by power analysis. Statistical differences between the groups were analyzed using an independent sample t test, Mann–Whitney U test or two-way ANOVA. Enumeration data were expressed as percentages, and the chi-square test was used to analyze the differences. The associations between C1q and each risk factor were assessed by Pearson’s analysis. Univariate and multivariate logistic model analyses of clinical factors affecting acute ischemic stroke were performed. A receiver operating characteristic (ROC) curve was constructed to evaluate the diagnostic value of C1q for DAIS. In addition, the associations between a one-SD change in C1q levels and DAIS were also tested using logistic regression analysis. p values <.05 were considered to show statistically significant differences in the data.
Results
Comparison of baseline clinical characteristics in the participants
Baseline characteristics and comparison of the three patient groups.

TC: total cholesterol; TG: triglycerides; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; PBG: fasting blood glucose; 2h PG: 2-h postprandial blood glucose; HbA1c: hemoglobin A1C; hs-CRP: high-sensitive C reaction protein.
a P < 0.05, compared with control group;
b P < 0.05, compared with T2DM group.
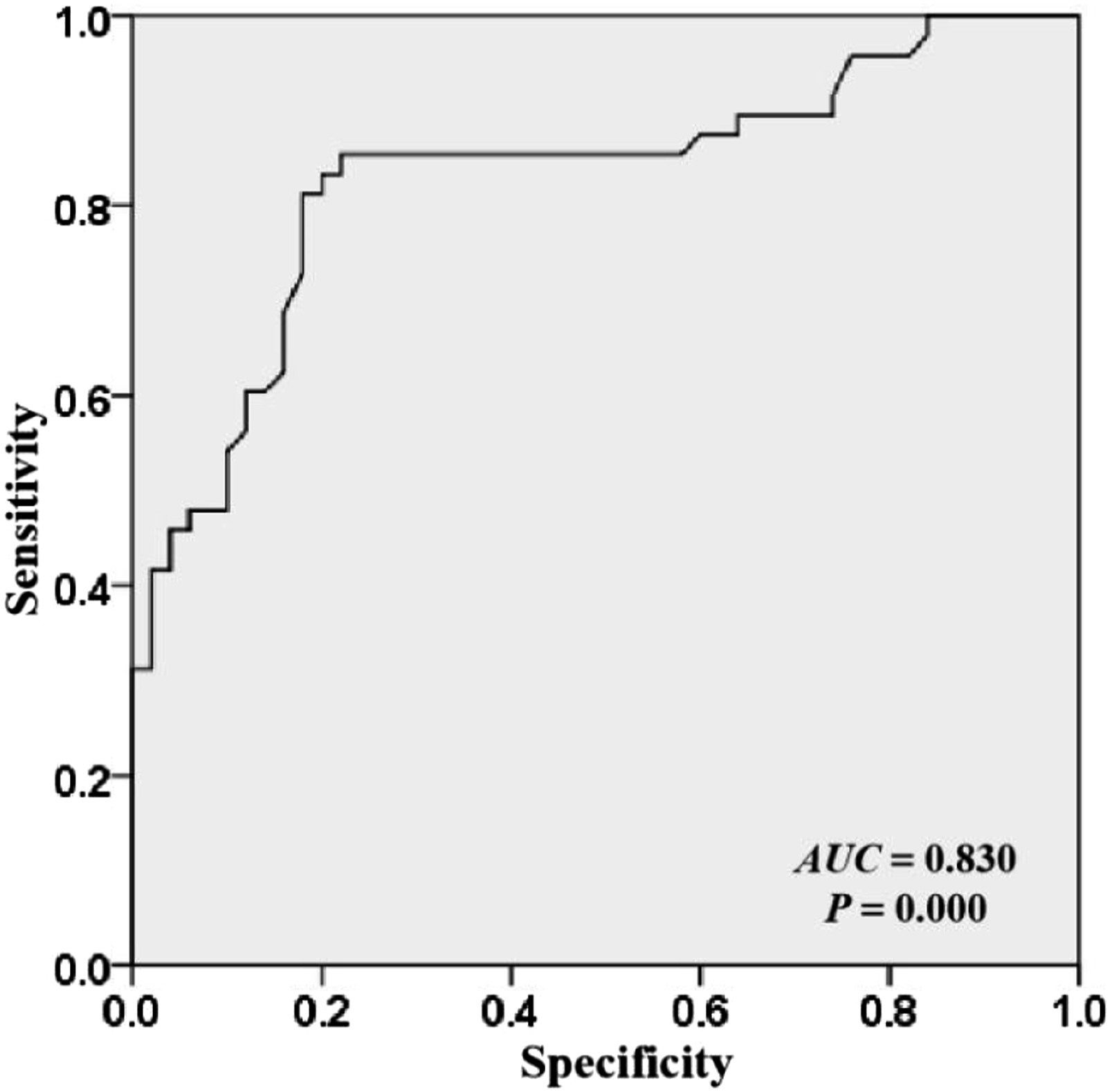
Diagnostic efficacy of C1q for DAIS
The levels of C1q in the T2DM, DAIS, and control groups were (225.92 ± 23.91), (357.10 ± 31.37), and (144.08 ± 16.87) mg/L, respectively. Our data showed that the levels of C1q in the DAIS and T2DM groups were increased significantly compared with those in the control group, while the levels of C1q in the DAIS group were markedly higher than those in the T2DM group (all p < .05, Figure 1). We also performed a ROC curve analysis to assess the diagnostic value of C1q for DAIS. As shown in Figure 2, the AUC was 0.830 (p = .000, 95%CI 0.747–0.914), with a sensitivity of 0.854 and specificity of 0.780. The cut-off value of C1q was calculated as 275.24 mg/L with a Youden index of 0.634. Comparison of C1q levels in the three groups. ROC curve of C1q in the diagnosis of DAIS.
Correlation between C1q level and blood lipid, blood sugar, and inflammatory cytokines
Association between C1q levels and blood lipids, blood sugar, and inflammatory cytokines.

TC: total cholesterol; TG: triglycerides; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; PBG: fasting blood glucose; 2h PG: 2-h postprandial blood glucose; HbA1c: hemoglobin A1C; hs-CRP: high-sensitive C reaction protein.
Correlations between C1q and acute ischemic stroke across categories of blood lipids, blood sugar, and inflammatory cytokines.

BMI: body mass index; LDL-C: low-density lipoprotein cholesterol; PBG: fasting blood glucose; 2h PG: 2-h postprandial blood glucose; HbA1c: hemoglobin A1C; hs-CRP: high-sensitive C reaction protein.
Logistic model analysis of clinical factors affecting acute ischemic stroke
Univariate and multivariate logistic model analyses of clinical factors affecting acute ischemic stroke.
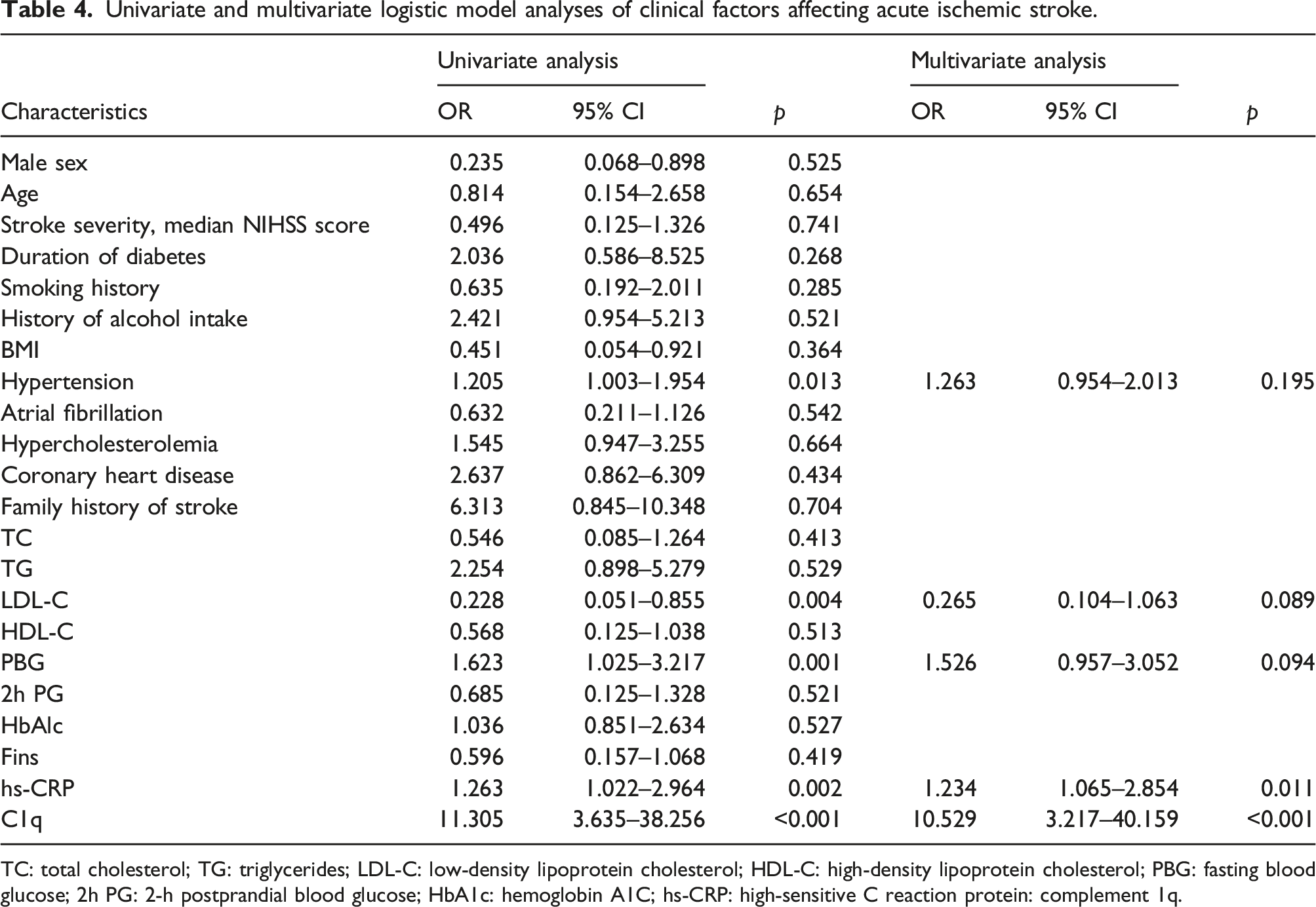
TC: total cholesterol; TG: triglycerides; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; PBG: fasting blood glucose; 2h PG: 2-h postprandial blood glucose; HbA1c: hemoglobin A1C; hs-CRP: high-sensitive C reaction protein: complement 1q.
Correlation between C1q and the occurrence of acute ischemic stroke in T2DM patients with different levels of hs-CRP
Associations between C1q and DAIS in the total population, patients with a hs-CRP < 1.78 mg/L, and patients with a hs-CRP ≥ 1.78 mg/L.

Discussion
Studies have revealed that diabetes is an independent risk factor for AIS.16,17 Our findings showed that C1q was associated significantly with the associated factors of AIS in cases with T2DM. An increased C1q level was an independent risk factor for DAIS, and correlated with inflammatory cytokines in DAIS patients. A persistent inflammatory response may be the pathological basis of DAIS. In addition, the measurement of C1q levels was helpful for diagnosing DAIS.
AIS is a common complication of T2DM and has an acute onset and high mortality. Presently, the presence and characteristics of DAIS are based on imaging examination, although cases are generally in the attack stage during these examinations. Therefore, minimally invasive, and accurate diagnostic biomarkers for DAIS would be of great value in these cases. It has been reported that hyperglycemia may result in alterations in blood rheology including decreased red blood cell deformability and elevated platelet viscosity, while in cases of T2DM these changes further induce microcirculation disturbances and increase the risk of AIS. 18 In addition, hyperglycemia, hyperlipemia, and other risk factors may induce the development of atherosclerosis (AS), which is the pathological basis of cerebrovascular disorders. 19 Studies have reported that glucose reacts with blood proteins to form glycation end products, which result in the production of ROS.20,21 C1q-mediated ROS production can induce a series of reactions and further promote the inflammatory response and chemical modification of lipoproteins, and reduce nitric oxide production, thereby elevating the risk of AS in the brain. 22 C1q is a potential biomarker for neurodegeneration, 23 although no study has evaluated its usefulness in patients with DAIS. On the other hand, C1q molecule itself has both anti-inflammatory activities and neuroprotective properties that promote resilience and resolution of injury. 24
In our study, we found that the level of C1q was associated closely with the associated factors of AIS in T2DM patients. C1q levels were increased significantly in T2DM patients with AIS compared with those with only T2DM. Our results are consistent with a previous study which demonstrated elevated C1q levels in patients with incident stroke, especially in a cohort of middle-aged and older adults with diabetes mellitus. 25 Similarly, another investigation revealed that C1q levels were also increased in cases with ischemic stroke compared with healthy controls. 15 Although the baseline levels of C1q in T2DM cases with AIS are unknown, we excluded participants with conditions that may have affected C1q levels. This included participants with malignancies, intracerebral hemorrhage, renal insufficiency, febrile diseases, a history of recent surgery or trauma, autoimmune diseases, or systemic infections during the period of our research were excluded from the analysis. Together with the above observations, our data indicate that C1q levels could be used to predict DAIS.
C1q is a specific immunoregulation protein and can be released into the blood. Some studies have reported that C1q levels are associated closely with lipid metabolism.26,27 In diseases with infiltration of immune cells, such as adult drug-resistant epilepsy, C1q activates systemic lupus erythematosus-Th17, the Th1 response in peripheral blood, and also influences the production of pro-inflammatory cytokines, including IL-17A, IL-22, TNF-α, interferon-γ, and granulocyte-macrophage colony-stimulating factor, thereby impacting disease progression. 28 Asuman et al. also reported that increased C1q levels correlated independently with diabetic microvascular complications. 29 The results of our investigation showed that serum levels of hs-CRP were increased markedly in DAIS and that hs-CRP was an independent risk factor for DAIS, suggesting that a persistent inflammatory response was associated closely with the associated factors of DAIS. In addition, C1q was greatly increased in patients with DAIS and was associated positively with LDL-C, PBG, 2h PG, hs-CRP, suggesting that C1q reflects lipid metabolism and the inflammatory status of DAIS patients to a certain extent. Moreover, the current research has some limitations. First, as our design is a cross-sectional study, we were not able to adjust the impacts of the concomitant medication history due to unavailable of nutritional data. Second, the small sample size may confound our results. Finally, C1q and other increased blood index may be increased due to stress, but our study cannot overcome.
Conclusion
In summary, C1q levels were increased significantly in DAIS patients and were an independent risk factor for the occurrence of AIS in patients with T2DM. In addition, a persistent inflammatory response and lipid disturbance might be the pathological basis of DAIS. Moreover, C1q has the potential to become a biomarker for diagnosing AIS in T2DM patients and is worthy of clinical utilization.
Footnotes
Acknowledgements
The authors thank The Central Hospital of Enshi Tujia and Miao Autonomous Prefecture.
Author contributions
ZPH and FW conceived and designed the research. YHD conducted the experiments. MY analyzed and interpreted the data. ZPH drafted the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
