Abstract
Background
Altitude sickness is often prone to occur during tourism or work in high-altitude areas. In China, traditional Tibetan medicines have a long history of preventing or treating altitude sickness, especially altitude hypoxia, which may lead to myocardial cell apoptosis and myocardial hypoxia-reoxygenation injury.
Purpose
This study investigated the effect of a Tibetan-origin edible Chinese herbal prescription (named C18) on protecting H9C2 cardiomyocytes from cobalt chloride-induced hypoxia injury and its potential mechanism.
Materials and Methods
In this study, a hypoxic injury model of H9C2 cardiomyocytes induced by cobalt chloride was established first. Then the cell viability, relevant antioxidant indicators malondialdehyde (MDA), superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and protein expression (hypoxia-inducible factor 1 alpha (HIF-1α), phosphoinositide 3-kinase (PI3K), phosphorylated protein kinase B (p-AKT)) were measured after pretreatment with or without C18. At last, the specific PI3K/AKT inhibitor LY294002 was applied to verify the antihypoxia signaling pathway.
Results
C18 could significantly promote normal H9C2 cardiomyocyte proliferation and inhibit apoptosis of hypoxic H9C2 cardiomyocytes, reduce the release of lactate dehydrogenase and MDA, and increase the levels of SOD and GSH-Px antioxidant enzymes. In addition, C18 could significantly downregulate the expression of HIF-1α protein and upregulate the expression of intracellular p-AKT. Moreover, these effects of C18 can be blocked by the specific PI3K/AKT inhibitor LY294002.
Conclusion
C18 protects H9C2 cardiomyocytes from cobalt chloride-induced hypoxia injury through the PI3K/AKT signaling pathway.
Keywords
Introduction
Plateau areas above 3,000 m are easy to lead to hypoxia in the human body, causing altitude sickness including acute altitude sickness, high altitude pulmonary edema, high altitude brain edema, and so on. In a short period of time, entering the plateau above 3,000 m can produce headache, dizziness, palpitations, shortness of breath, and other reactions, and the heavy ones also have appetite loss, nausea, vomiting, insomnia, fatigue, abdominal distension, and chest tightness. Hypoxia caused by a low oxygen environment at high altitudes is the main cause of acute altitude sickness. Acute altitude sickness is a kind of stress reaction of the body to the plateau environment, and it is a clinical symptom of the process in which the various functions of the body reach a new balance on a new basis through the regulation of neurohumoral. Some people, through the long-term uninterrupted adjustment process, always cannot achieve balance on a new basis, so the formation of a plateau dysfunction phenomenon shows a series of clinical symptoms; this situation is called chronic altitude sickness. Chronic altitude sickness is also known as “body dysfunction.” Altitude sickness is a problem that cannot be ignored, affecting not only foreign workers and tourists physically but also seriously affecting their lives and work, so the prevention and improvement of altitude sickness have become very important issues (Fu et al., 2021; Luo et al., 2013).
High altitude hypoxia environment is prone to cause hypoxia damage to cardiac tissue, which is clinically manifested as palpitations, accelerated heartbeat, weakness, and fatigue (Oliver et al., 2012). Long-term hypoxia environment can lead to compensatory hypertrophy of the heart (Imray et al., 2010). In addition, when myocardial cells undergo hypoxia-reoxygenation, they can produce large amounts of oxygen-free radicals, which damage important structures such as myocardial cell membranes and mitochondria, leading to myocardial cell apoptosis and myocardial hypoxia-reoxygenation injury. Hypoxia-inducible factor 1 alpha (HIF-1α) (Rius et al., 2008) is considered to be the main regulator of hypoxia and plays a key role in regulating the response to hypoxia homeostasis. The phosphoinositide 3-kinase/protein kinase B (PI3K/AKT) signaling pathway serves a crucial function in regulating cell metabolism, growth, proliferation, and survival. Studies have shown that activating the PI3K/AKT signaling pathway can protect body tissues from damage caused by hypoxia (Wang et al., 2021; Zhang et al., 2021).
In China, traditional Tibetan-origin edible Chinese herbs, such as Urtica dioica Linn., Hippophae tibetana Schlechtend., Rhodiola crenulata (Hook. f. et Thoms.) H. Ohba, Potentilla anserina L., Lycium ruthenicum Murr., Brassica rapa L., Polygonatum cirrhifolium (Wall.) Royle., and so on, have a long history of preventing or treating altitude sickness (Wang et al., 2022). Tibetan medicine compounds are also widely used to treat altitude sickness and cardiovascular and digestive diseases. For example, Sanguo Tang San is used for antioxidant and antiplateau polycythemia (Fu et al., 2020). The 15-flavored agarwood powder and the 20-flavored agarwood powder in the Tibetan medicine compound can prevent hypoxic injury caused by altitude sickness (Zhou et al., 2010).
Rhodiolae crenulatae radix et rhizome, transliterated as “Suolomaobu” in Tibetan (Panossian et al., 2010), is the dried root of the plant (R. crenulata (Hook. f. et Thoms.) H. Ohba.) with pharmacological activities of antihypoxia, antifatigue, anti-inflammation, and improvement of cardiovascular diseases (Gupta et al., 2009). It is regarded as a natural “adaptation” abroad (Panossian et al., 2010). H. tibetana Schlechtend, transliterated as “Sadar” in Tibetan, is the dry and mature fruit of Elaeagnaceae plants with pharmacological activities of antihypoxia and antioxidant (Suryakumar & Gupta, 2011). L. ruthenicum Murry, translated into Tibetan as “Pangna Zhai Wu.” is the dried ripe fruit of the Solanaceae plant with pharmacological activities of antihypoxia, antioxidant, and antifatigue (Hou et al., 2019; Ni et al., 2013). U. dioica Linn., a perennial herb belonging to the genus Urtica in the family Urticaceae (Sayhan et al., 2012; Semalty et al., 2017), with pharmacological activities of antihypoxia, antioxidant (Joshi et al., 2015), and inhibition of cell apoptosis. Four kinds of plants with medicinal and edible properties are recorded in the Tibetan medical classics “Si Bu Yi Dian” and “Jing zhu Materia Medica.” Medicinal and edible homologous plants in Tibet have the advantages of being edible, mild in nature, low-toxic, and with few side effects. However, there is little research on the compound prescription of antihypoxia medicinal food from Tibet. Prescription C18, including these four edible herbals (R. crenulatae radix et rhizome, H. tibetana Schlechtend., L. ruthenicum Murr., B. rapa L., U. dioica Linn) mentioned above, is a folk recipe for antihypoxia used in Tibet for hundreds of years. In this study, a model of H9C2 cardiomyocyte hypoxia injury was established using cobalt chloride (CoCl2), a classic chemical hypoxia mimetic agent (Munoz-Sanchez & Chanez-Cardenas, 2019). Then the antihypoxia activity and potential mechanism of C18 were studied.
Materials and Methods
Materials
The Tibetan medicine in this experiment was purchased from the Chinese herbal medicine market in Shigatse, Tibet, and identified by Professor Zhu Quan. Retention specimens were stored in the pharmaceutical laboratory, School of Medicine, Foshan University. Before the experiment, the C18 water extract was dissolved in phosphate buffer saline (PBS) to a final concentration of 100 mg/mL solution. The solution was filtered in sterile conditions and stored in a refrigerator at 4°C for subsequent dilution during the experiments. CoCl2 was purchased from Shanghai Macklin Biochemical Technology Co., Ltd. Rat cardiomyocyte cell line H9C2 was obtained from the Chinese Center for Type Culture Collection. A Cell Counting Kit-8 (CCK-8) was purchased from Beyotime Biotechnology. Fetal calf serum was obtained from Biological Industries. Tips, dishes, test tubes, etc., for cell culture, were bought from Thermo Fisher Scientific (Waltham, MA, USA). Primary antibodies: PI3K (1:1,000; No. #4249; Cell Signaling Technology, USA), phosphorylated protein kinase B (p-AKT) (1:1,000; No. #4058; Cell Signaling Technology, USA), AKT (1:2,000; No. 10176-2-AP; Proteintech, China), HIF-1α (1:1,000; No. 340462; Zen-Bio, China), β-actin (1:10000; No. 200068-8F10; Zen-Bio, China).
Preparation of C18
100 g R. crenulatae radix et rhizome, 100 g L. ruthenicum Murr., 50 g Hippophae Fructus, and 50 g U. dioica were dried and powdered, immersed in 10 times water for 2 hours, heated for reflux extraction for 1 hour, collected the extraction solution, and then extracted twice more with 8 times water for 1 hour each, combined with the extraction solution, filtered, concentrated under pressure until dry, stored in a refrigerator at 4°C until use.
Cell Culture and Treatment
The H9C2 cardiomyocytes were cultured with 10% fetal bovine serum (FBS) Dulbecco’s modified Eagle medium (DMEM) complete medium in cell culture incubator (37°C, 5% CO2). When the adherent cells reached 80–90% confluence, the H9C2 cardiomyocytes were seeded in 96 well plates (5,000 cells/well). Then, C18 was diluted to different concentrations and added into 96 wells for 24 h incubation to figure out their safe concentrations to be used in the bioassay. The success of CoCl2 modeling and the effects of C18 treatment were screened by the CCK-8 (Beyotime, Shanghai, China) assay. Then, the OD value of the well plate at 450 nm was measured using a microplate reader (Bio-Rad, Hercules, CA, USA). Finally, the cells were pretreated with C18 (125, 250, 500 µg/mL) for 24 h prior to the intervention with CoCl2 (800 µM) for 24 h for subsequent experiments.
Cellular Biochemical Analysis
First, H9C2 cells were seeded in a 12-well plate at 1 × 105 cells/well and incubated for 12 h. Then, they were subjected to different treatments, and the media of each group were collected. The lactate dehydrogenase (LDH) release in the cell media was measured using an LDH cytotoxicity assay kit. Malondialdehyde (MDA) is a degradation product of oxygen-derived free radicals and lipid oxidation, which can reflect the damage caused by reactive oxygen species (ROS) to a certain extent. Superoxide dismutase (SOD) is a secreted enzyme that protects the extracellular matrix from degradation by active species by regulating the level of extracellular superoxide. Glutathione peroxidase (GSH-Px) not only catalyzes the decomposition of hydrogen peroxide, but also helps neutralize free radicals, protects cell membranes from oxidative damage, and ensures the structural stability and normal function of cell membranes, which is essential for maintaining the stability of the intracellular environment and resisting external stress. The levels of LDH, MDA, SOD, and GSH-Px were measured with the assay kits (Jian Cheng Bioengineering Institute, Nanjing, China), and the test process was operated according to the kit instructions.
Western Blot (WB) Analysis
H9C2 cells in logarithmic growth were seeded in 12 well plates (1 × 105 cells/well), followed by culture with 10% FBS DMEM complete medium for 12 h. The H9C2 cells were pretreated with different concentrations of C18 solution (125, 250, 500 µg/mL) for 24 h. After that, the cell culture medium was removed, gently washed two times with PBS, and incubated with CoCl2 (800 µM) solution for 24 h. Then, the culture medium containing CoCl2 was removed, and the cells were washed two times with cold PBS. The total protein in H9C2 cells was separated by radioimmunoprecipitation assay (RIPA) lysis buffer containing protease and phosphatase inhibitors (Beyotime, Shanghai, China). The protein concentration was determined using the BCA protein assay kit (Beyotime, Shanghai, China). Then, proteins (20 µg) were separated through 10% SDS-PAGE separating gel buffer and electrotransferred to polyvinylidene difluoride membranes. After being blocked with blocking buffers (Beyotime, Shanghai, China) for 30 min at 4°C, the membrane was incubated with diluted primary antibody overnight at 4°C. The primary antibodies used included PI3K, p-AKT, AKT, HIF-1α, and β-actin. The membrane was washed three times and then incubated with horseradish peroxidase (HRP)-conjugated goat antirabbit immunoglobulin G (IgG) for 1 h (Biosynthetic, Beijing, China). Band densities were detected using ImageJ 1.48 software (National Institutes of Health, USA).
Statistical Analysis
All the data are presented as the mean ± standard deviation (SD) and were analyzed by one-way analysis of variance (ANOVA) with GraphPad Prism version 8.30 (GraphPad Software, CA, USA). p < 0.05 was considered to indicate statistical significance.
Results
Hypoxia Injury Model Establishment
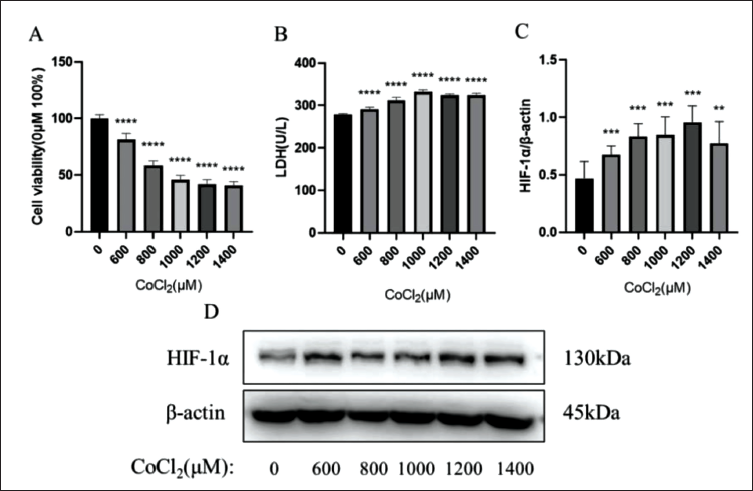
In this study, the chemical reagent CoCl2 was used to induce hypoxia injury in H9C2 cardiomyocytes. First, the effect of CoCl2 on H9C2 cardiomyocytes cultured in vitro was observed. Cell viability was assessed through the CCK-8 test following a 24-h exposure to various concentrations of CoCl2 (Figure 1A). Compared with the control group, CoCl2 significantly decreased cell viability in a dose-dependent manner. When the CoCl2 concentration was 800 µM, the cell viability of H9C2 cardiomyocytes was 58.41% ± 4.11%, which was similar to the literature (Manu et al., 2019). LDH is often used as a marker of cell damage. Detecting the LDH content in the supernatant of cell culture media can provide insight into the extent of cell damage. Compared with the control group, the LDH content in the CoCl2 group significantly increased (Figure 1B), indicating that CoCl2 can cause hypoxia injury in H9C2 cardiomyocytes. The WB results (Figure 1C) showed that the CoCl2 group significantly increased the expression of HIF-1α protein in H9C2 cardiomyocytes compared to the control group, confirming the correlation between cell viability and hypoxia. Therefore, considering the exposure time and concentration, the best condition to induce hypoxia injury in H9C2 cardiomyocytes was 800 µM CoCl2 for 24 h.
The effect of cobalt chloride (CoCl2) on H9C2 cardiomyocytes. (A) The percentage of cell viability was determined by Cell Counting Kit-8 (CCK-8) assay after H9C2 cardiomyocytes were stimulated with different concentrations of CoCl2 for 24 h. (B) The change of lactate dehydrogenase (LDH) level in cell culture medium after cells were stimulated by different concentrations of CoCl2 for 24 h. (C) and (D) After stimulating cells with different concentrations of CoCl2 for 24 h, the expression level of hypoxia-inducible factor 1 alpha (HIF-1α) protein was detected by Western blot (WB). Data are shown as the mean ± SD. **p < 0.01, ***p < 0.001, ****p < 0.0001 versus control.
Effects of C18 on the H9C2 Cardiomyocytes
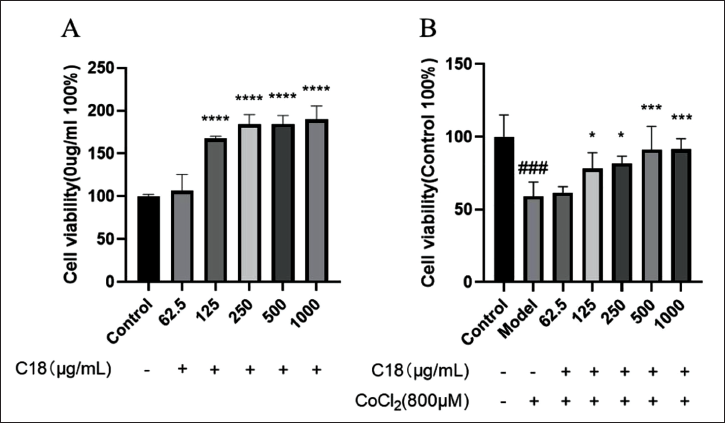
The cytotoxicity of C18 on normal H9C2 cardiomyocytes was evaluated by measuring cell viability using the CCK-8 assay (Figure 2A). Compared with the control group, C18 at concentrations of 62.5–1,000 µg/mL did not inhibit cell viability. Instead, it promotes cell proliferation. Therefore, subsequent experiments C18 could be conducted within this concentration range. In the CoCl2-induced hypoxia injury of H9C2 cardiomyocytes, results (Figure 2B) showed that C18 at concentrations of 125, 250, 500, and 1,000 µg/mL significantly improved the viability by inhibiting the H9C2 cardiomyocyte apoptosis compared with the model group.
The effect of C18 on H9C2 cardiomyocytes. (A) The percentage of cell viability was determined by Cell Counting Kit-8 (CCK-8) assay after H9C2 cardiomyocytes were stimulated with different concentrations of C18 for 24 h. ****p < 0.0001. (B) After pretreating H9C2 cardiomyocytes with different concentrations of C18 for 24 h, the cells were replaced with 800 µM cobalt chloride (CoCl2) to induce hypoxia for 24 h. The CCK-8 kit was used to detect the effect of C18 on the cell viability of CoCl2-induced hypoxic injury cells. Data are shown as the mean ± SD. ###p < 0.001, versus control; *p < 0.05, ***p < 0.001, ***p < 0.001 versus model.
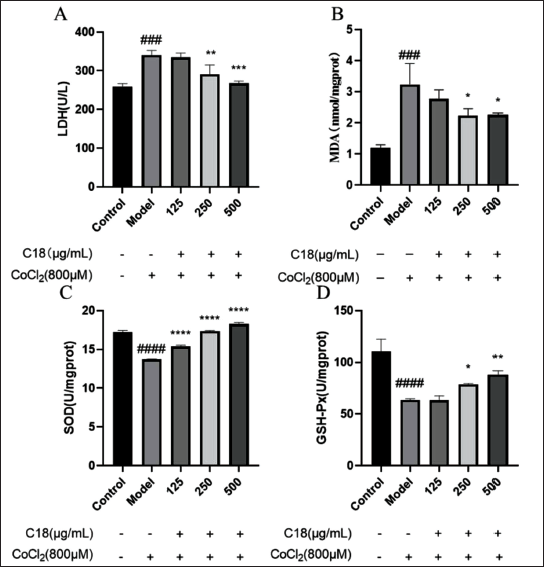
In terms of antioxidants, compared with the model group, C18 at different concentrations could significantly decrease the LDH content in the cell culture medium (250 and 500 µg/mL, Figure 3A), decrease the intracellular MDA level (500 µg/mL, Figure 3B), increase the intracellular SOD (250 and 500 µg/mL, Figure 3C) and GSH-Px level (250 and 500 µg/mL, Figure 3D). In short, C18 could significantly increase the level of antioxidant enzymes in H9C2 cells, thereby reducing CoCl2-induced hypoxia injury in H9C2 cells.
C18 reduces oxidative stress induced by cobalt chloride (CoCl2) in H9C2 cardiomyocytes. Compared with the model group, (A) C18 significantly decreased the level of lactate dehydrogenase (LDH) in the culture medium; (B) C18 significantly decreased the intracellular malondialdehyde (MDA) level; (C) C18 significantly increased the intracellular superoxide dismutase (SOD) level; (D) C18 significantly increased the intracellular glutathione peroxidase (GSH-Px) level. Data are shown as the mean ± SD. ###p < 0.0001, versus control; *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001 versus model.
Expression of HIF-1α Protein
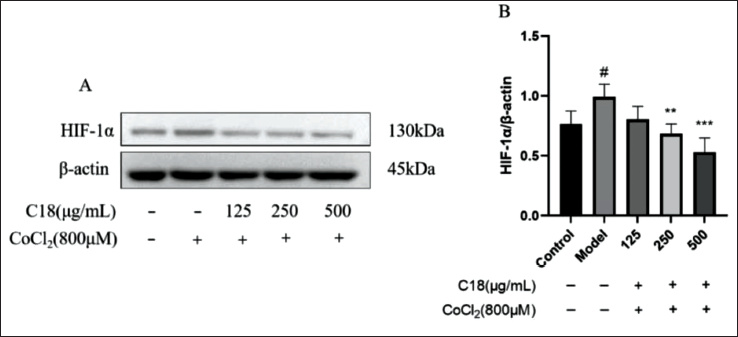
The WB experiment results showed that C18 could affect the expression of HIF-1α protein (Figure 4A). Compared with the model group, the C18 at concentrations of 250 and 500 µg/mL could significantly downregulate the expression of HIF-1α protein in cells. C18 pretreatment could significantly improve the survivability of hypoxic H9C2 cardiomyocytes induced by CoCl2, which may have a relation to the downregulation of intracellular HIF-1α protein expression.
C18 affects the expression of hypoxia-inducible factor 1 alpha (HIF1-α) protein in H9C2 cardiomyocytes induced by cobalt chloride (CoCl2). Compared with the model group, (A) and (B) C18 significantly reduced the expression level of HIF1-α protein in cells induced by CoCl2. Data are shown as the mean ± SD. #p < 0.05, versus control; **p < 0.01, ***p < 0.001, versus model.
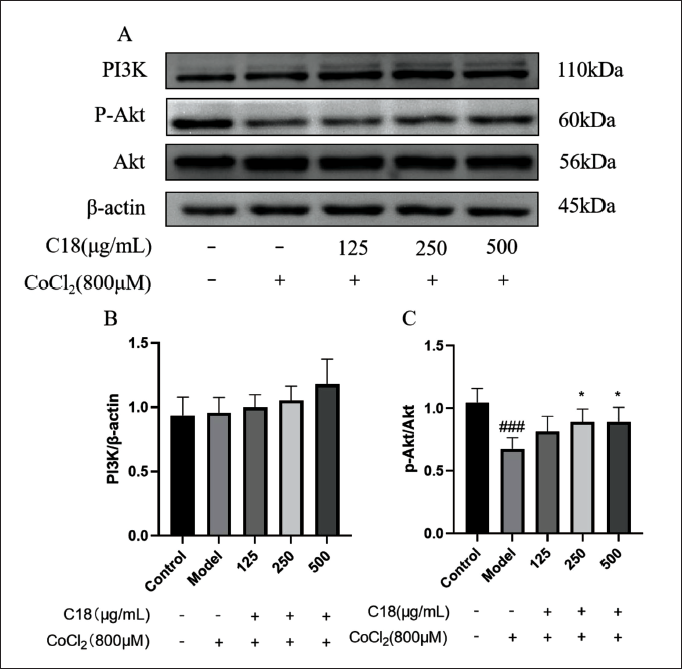
Regulation of PI3K/AKT Signaling Pathway
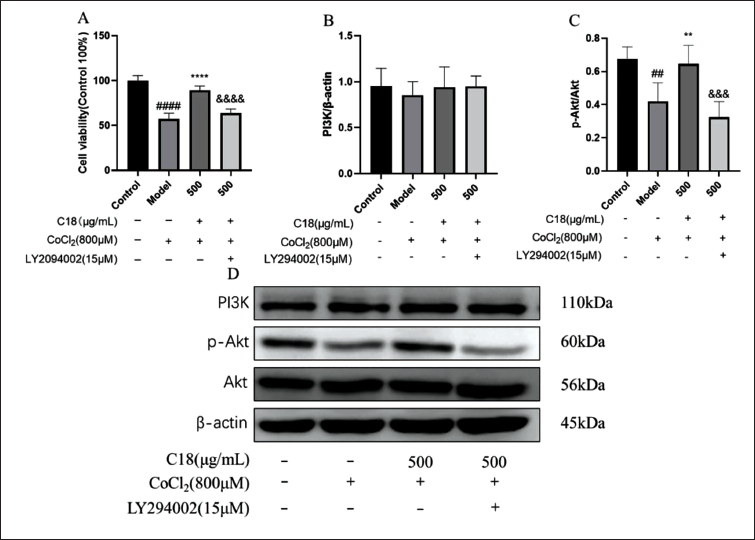
WB results showed that C18 had no significant effect on the expression of PI3K protein in H9C2 cardiomyocytes compared with the model group (Figure 5B). But the C18 at concentrations of 250 and 500 µg/mL significantly upregulated the expression of p-AKT/AKT protein (Figure 5C). We speculate that C18 may increase the viability of H9C2 cardiomyocytes by activating the PI3K/AKT signaling pathway and reduce the hypoxia-induced injury induced by CoCl2. To further verify the experimental hypothesis, we used the PI3K/AKT specific inhibitor (LY294002) and a concentration of 500 µg/mL of C18 for subsequent experimental studies. In this experiment, we chose a concentration of 15 µg/mL of PI3K/AKT specific inhibitor (LY294002) to intervene in H9C2 cardiomyocytes for 2 hours, while other experimental conditions remained unchanged. As shown in Figure 6A and C, compared with the model group, C18 at 500 µg/mL could significantly increase cell viability and the expression of p-AKT protein. After being blocked by LY294002, cell viability and the expression of p-AKT protein decreased drastically and C18 lost its protective effect. Therefore, the PI3K/AKT signaling pathway plays a big part in the increase of the viability of hypoxic H9C2 cardiomyocytes by C18.
C18 affects the expression of phosphoinositide 3-kinase/protein kinase B (PI3K/AKT) signaling pathway proteins in H9C2 cardiomyocytes induced by cobalt chloride (CoCl2). (A) The expression level of PI3K and p-AKT protein was detected by Western blot (WB). Compared with the model group, (B) C18 had no significant effect on the expression of PI3K protein in H9C2 cells; (C) C18 can significantly increase the ratio of phosphorylated protein kinase B (p-AKT)/AKT protein expression. Data are shown as the mean ± SD. ###p < 0.001, versus control; *p < 0.05, versus model.
Phosphoinositide 3-kinase (PI3K) inhibitor (LY294002) could inhibit the expression of phosphorylated protein kinase B (p-AKT) protein in H9C2 cells treated with C18. (A) LY294002 significantly inhibited the effect of C18 on the viability of H9C2 cardiomyocytes. (B) LY294002 had no significant effect on the expression of PI3K protein in H9C2 cardiomyocytes treated with C18. (C) LY294002 significantly inhibited the expression of p-AKT protein in H9C2 cardiomyocytes treated with C18. (D) The expression level of PI3K and p-AKT protein was detected by Western blot (WB). The PI3K/AKT signaling pathway plays an important role in reducing cobalt chloride (CoCl2)-induced hypoxia injury in H9C2 cardiomyocytes by C18. Data are shown as the mean ± SD. ##p < 0.01, ####p < 0.0001, versus control; **p < 0.01, ****p < 0.01 versus model. &&&p < 0.001, &&&&p < 0.0001 versus C18 (500 µg/ML) group.
Discussion
With the continuous hypoxic condition, high altitude reaction is often prone to occur during tourism or work in high altitude areas. When cardiomyocytes are in a state of hypoxia, cellular energy metabolism changes, inducing cell injury, and further development can lead to the development and exacerbation of cardiovascular disease (Manu et al., 2019).
Researchers have established a series of hypoxic injury cellular models in vitro, by liquid paraffin closure method, anaerobic bag gas production method, mixed gas culture method, and other methods (Zhang et al., 2023). CoCl2-induced hypoxia injury is a classic chemical method. In this study, after being conducted with 800 µM CoCl2, H9C2 cardiomyocytes were apoptotic and their viability decreased. At the same time, the level of cellular oxidation changed sharply with the LDH and MDA increase, SOD and GSH-Px decrease. Moreover, the HIF-1α protein significantly enhanced. When pretreated with C18, the cellular oxidation and HIF-1α protein expression are down-regulated. HIF-1α is considered to be the main regulator of hypoxia and plays a big part in regulating the response to hypoxia homeostasis. Regulation of HIF-1α by PI3K/AKT/mTOR was demonstrated in previous studies (Xiao et al., 2017; Zhong et al., 2000) and PI3K/AKT/FRAP (Laughner et al., 2001) signaling pathways. When human mesenchymal stem cells are hypoxic, the levels of p-AKT and HIF-1α proteins in cells are significantly upregulated. Therefore, in the further mechanism experiment, C18 could develop the p-AKT level, but the LY294002 significantly inhibited this effect as a PI3K/AKT-specific inhibitor, which also suggests that C18 may reduce the hypoxia-induced injury caused by CoCl2 by activating the PI3K/AKT signaling pathway.
The PI3K/AKT signaling pathway plays a big part in regulating cellular metabolism, growth, proliferation, and survival (Joshi et al., 2015; Sayhan et al., 2012). PI3K and the downstream effector AKT, as members of a family of conserved signal transduction enzymes, are involved in regulating several cellular activities, including cell activation, apoptosis, and inflammatory responses (Zhang et al., 2018). Therefore, by activating the PI3K/AKT signaling pathway, C18 could protect the H9C2 cardiomyocytes against hypoxia injury.
Tibetan medicine occupies an extremely crucial part of traditional Chinese medicine. Tibetan medicine is a medical theory formed in the course of people’s long-term struggle against nature and adaptation to the natural environment. C18 is a compound prescription including four edible medicines that are grown in this oxygen-deprived environment. The major component, R. crenulatae radix et rhizome, could supplement Qi and activate blood, clear pulse, and relieve asthma. Salidroside, the main active component of R. crenulatae radix et rhizome, played a critical role in protecting cardiomyocyte against oxidative injury by restoring the tricarboxylic acid (TCA) cycle, increasing the ∇Ψm, inhibiting the excessive consumption of adenosine triphosphate (ATP), reducing the mitochondrial Ca2+ overload and ROS production, and attenuating the activation members of the caspase family (Chen, 2015). Compounds known as anthocyanins, which are derived from the plant L. ruthenicum Murry, have the ability to ameliorate the effects of hypoxia on apoptosis and cell proliferation in H9c2 cells (Li et al., 2023). Polyphenols from Hippophae species have antioxidant activity. The compound combination may enhance their antihypoxic activity. Due to the local economic and scientific level, many Tibetan medicines have not been thoroughly studied. Consequently, a thorough investigation into the chemical foundations and mechanism of Tibetan medicine’s antihypoxic effects is merited.
Conclusion
C18 can alleviate CoCl2-induced hypoxia injury in H9C2 cardiomyocytes mainly by increasing antioxidant levels, decreasing HIF-1α protein expression, and regulating the PI3K/AKT signaling pathway. This suggests that C18 has the possibility to be developed into a prescription formulation for anti-hypoxic medicinal foods. Moreover, in the future, we should pay more attention to the research of active ingredients in antihypoxic Tibetan medicine.
Abbreviations
MDA: Malondialdehyde; SOD: Superoxide dismutase; LDH: Lactate dehydrogenase; GSH-Px: Glutathione peroxidase; HIF-1α: Hypoxia-inducible factor 1 alpha; PI3K: Phosphoinositide 3-kinase; AKT: Protein kinase B; ROS: Reactive oxygen species; FBS: Fetal bovine serum; DMEM: Dulbecco’s modified Eagle medium; PBS: Phosphate buffer saline.
Author Contribution
Guoxin Chang and Hongyi Xie contributed equally as the first author.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Guangdong Basic and Applied Basic Research Fund Project (No. 2021A1515110640), the Provincial Enterprise Joint Fund General Project of Guangdong Basic and Applied Basic Research Fund (2022A1515220132), the Special Fund for Key Fields of Higher Education of Guangdong Province (2023ZDZX2058), and the Guangzhou Science and Technology Project (No. 202201010086).
