Abstract
Purpose
Ovarian cancer is a malignant tumor in women all over the world. Ropivacaine is identified as a potential drug for the treatment of malignant tumors, but the role and mechanism of ropivacaine in ovarian cancer remains unknown.
Materials and methods
Ovarian cancer cells were treated with different doses of ropivacaine. The function of ropivacaine in ovarian cancer was assessed using Cell Counting Kit-8 assay, flow cytometry, sphere-formation assay, Western blot, Fe2+ level analysis, and immunofluorescence. Meanwhile, the mechanism of ropivacaine in ovarian cancer was investigated by multiple molecular experiments. The protective function of ropivacaine in ovarian cancer was further confirmed by in vivo assay.
Results
The functional research data indicated that the growth and stemness of ovarian cancer cells were restrained after ropivacaine treatment, while the ferroptosis in ovarian cancer cells was facilitated. The mechanism results confirmed that ropivacaine inactivated the PI3K/AKT signaling pathway in ovarian cancer cells. Furthermore, in vivo assay demonstrated that ropivacaine repressed the proliferation of ovarian cancer cells in vivo and had a protective function in ovarian cancer.
Conclusion
Ropivacaine restrained ovarian cancer cell stemness and accelerated cell ferroptosis by inactivating PI3K/AKT signaling pathway.
Introduction
Ovarian cancer has the highest mortality rate among gynecologic tumors that threaten the health of women. 1 About 70% of the cases are diagnosed as advanced stage due to the nonspecific clinical manifestations of early ovarian cancer. 2 Despite the good progress that has been made in the drug treatment of ovarian cancer over the past few decades, the non-specificity and side effects of most drugs severely restrict the ovarian cancer therapeutic effect. 3 Therefore, discovering more effective drugs for ovarian cancer treatment and elucidating its possible mechanism are conducive to alleviating ovarian cancer.
Cancer stem cells are a subpopulation of tumor cells with the same ability of self-renewal and differentiation as stem cells, mainly participating in tumor initiation, metastasis, and resistance to chemotherapy. 4 Recent studies have confirmed that cancer stem cells are associated with the growth and metastasis of ovarian cancer. For example, Wilczyński et al. expounded that the existence of cancer stem cells is the main cause of chemotherapy resistance and recurrence in the treatment of ovarian cancer 5 ; Muñoz-Galván et al. demonstrated that the combination treatment with cisplatin and YAP inhibitors restrain MYPT1 induced resistance, and the elevated chemotherapy resistance of ovarian cancer is accompanied by the increased stemness. 6 These studies suggest that reducing ovarian cancer cell stemness improves ovarian cancer.
Ferroptosis is a novel form of cell death distinct from other known pathways of cell death. 7 Ferroptosis is caused by the accumulation of lipid ROS and is characterized by cytological changes, mainly including the reduction of mitochondrial crest and rupture of the mitochondrial outer membrane. 8 As has been reported, MAP30 protein extracted from Momordica charantia has therapeutic effects on ovarian cancer by changing metabolism and inducing ferroptosis. 9 Thus, facilitating ferroptosis is beneficial for relieving ovarian cancer.
Ropivacaine is the most commonly used local anesthetics in clinical practice and is used clinically in surgical anesthesia and acute pain management. 10 Ropivacaine has strong antitumor activity against a variety of tumor cells. For instance, ropivacaine has the function of repressing cervical cancer cell growth by suppressing cell cycle progression and accelerating cell apoptosis, prompting that ropivacaine is expected to become a therapeutic drug for clinical cancer 11 ; ropivacaine slows down the development of esophageal cancer cells by repressing the Rac1/JNK/Paxillin/FAK pathway, providing novel insights into the local anesthetics treatment for tumors. 12 However, the role of ropivacaine in ovarian cancer is not fully understood. In the current study, we preliminarily explored the ropivacaine function in ovarian cancer and confirmed that ropivacaine inhibited ovarian cancer cell growth. Aldehyde dehydrogenase (ALDH) is an enzyme that participates in the regulation of various cellular mechanisms through aldehyde detoxification. 13 Also, the ALDH activity, sphere-formation assay, and analysis of stemness marker (OCT4 and Nanog) expressions are often used to assess the stemness of tumor cells. 14 As expected, ropivacaine reduced the ALDH positive cells, weakened the sphere-formation ability of SKOV3 cells, and decreased the protein levels of OCT4 and Nanog, implying that ropivacaine restrained the stemness of ovarian cancer cells. Meanwhile, we further confirmed that ropivacaine accelerated the ferroptosis in ovarian cancer cells.
Considering the therapeutic effect of ropivacaine on ovarian cancer, this research further investigated the possible mechanism of ropivacaine protecting ovarian cancer through various molecular techniques, providing novel targets for ovarian cancer treatment in the future.
Materials and methods
Cell culture and treatment
Human ovarian cancer cell lines SKOV3 and OVCAR-3 were from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). All the cells were cultured in RPMI-1640 medium (Gibco, New York, USA) with 10% fetal bovine serum (FBS, Gibco) and 1% penicillin/streptomycin (Thermo Fisher Scientific, Waltham, USA) at 37°C, 5% CO2.
SKOV3 and OVCAR-3 cells were treated with 0, 2.5, 5, 10, 20, 40, 80 and 160 μM doses of ropivacaine (BioChemPartner, Shanghai, China) for 24 h. 15 SKOV3 and OVCAR-3 cells were treated with 40 μM ropivacaine and/or 5 mmol/l erastin (MedChemExpress, New Jersey, USA), 16 40 μM ropivacaine and/or 5 μM ferrostatin-1 (MedChemExpress) for 24 h. 17 SKOV3 cells were treated with 40 μM ropivacaine and/or 20 μM 740 Y-P (MedChemExpress) for 24 h. 18
Cell counting kit-8 (CCK-8) assay
SKOV3 and OVCAR-3 cells (2 × 103) were inoculated in 96-well plates. After 24 h of culture, 10 μl CCK-8 solution (Beyotime, Shanghai, China) was added to each well and placed in an incubator for further culture for 1 h. Afterward, the 96-well plates were placed in a microplate reader (Thermo Fisher Scientific) and the absorbance (OD) at 450 nm was detected.
Flow cytometry
The ALDH positive cells were determined using an AldefluorTM kit (Stem Cell Technologies, Shanghai, China) referring to the reagent manufacturer’s procedure. SKOV3 and OVCAR-3 cells were incubated with AldefluorTM assay buffer containing ALDH substrate and BODIPY-Aminoacetaldehyde for 50 min at 37°C. Then the ALDH+ cells were delineated by catalyzing BODIPY-Aminoacetaldehyde to its fluorescent product BODIPY-Aminoacetate; the ALDH enzymatic activity was blocked by the specific ALDH inhibitor: AldefluorTM DEAB reagent. Ultimately, the ALDH positive cells were assessed using a flow cytometer (FACSCalibur, BD Biosciences, San Jose, USA) and the BD FACSAria II System (BD Biosciences).
Sphere-formation assay
SKOV3 and OVCAR-3 (1 × 104) were inoculated in 96-well ultra-low attachment plates (Corning, NY, USA) and cultured in serum-free DMEM/F12 with the addition of 20 ng/mL human recombinant epidermal growth factor (Invitrogen, Carlsbad, USA), 10 ng/mL human recombinant basic fibroblast growth factor (Invitrogen) and antibiotics penicillin and streptomycin (Thermo Fisher Scientific) for 7 days. Then the sphere number was counted under a light microscope (Olympus, Tokyo, Japan) and Image J software (National Institutes of Health, Bethesda, USA).
Western blot
After SKOV3 and OVCAR-3 cells were harvested, the total proteins were extracted from the cells by RIPA buffer (Thermo Fisher Scientific). The protein concentrations were quantified using a BCA Assay Kit (Abcam, Cambridge, UK). The total amount of proteins (30 μg) were subjected to sodium dodecyl sulfonate-polyacrylamide gel electrophoresis (Thermo Fisher Scientific) and transferred onto polyvinylidene fluoride membranes (Millipore, Bedford, USA). After the membranes were blocked using 5% skim milk for 1 h, the membranes were incubated with the primary antibodies containing anti-OCT4 (Abcam, 1: 10,000, ab200834), anti-Nanog (Abcam, 1: 1000, ab109250), anti-GPX4 (Abcam, 1 µg/ml, ab41787), anti-SLC7A11 (xCT, Abcam, 1: 2000, ab175186), anti-p-PI3K (Abcam, 1: 500, ab182651), anti-PI3K (Abcam, 1: 1000, ab32089), anti-p-AKT (S473, Abcam, 1: 5000, ab81283), anti-p-AKT (T308, Abcam, 1: 500, ab38449), anti-AKT (Sigma-Aldrich, 1: 1000, SAB4500803), anti-p-mTOR (S2448, Abcam, 1: 1000, ab109268), anti-mTOR (Abcam, 1: 10,000, ab134,903) and anti-β-actin (Abcam, 1 µg/ml, ab8226) at 4°C overnight. The membranes were further incubated with the secondary antibody (Abcam) at room temperature for 1.5 h. The protein bands were assessed using enhanced chemiluminescence reagents (Millipore) and Image J software (National Institutes of Health).
Detection of the Fe2+ levels
The Fe2+ levels in ovarian cancer cells with different treatments were measured using an Iron Ion Kit (Millipore) following the reagent manufacturer’s procedure. In brief, the SKOV3 and OVCAR-3 cell supernatant was collected and incubated with an iron reducer, and then labeled with an iron probe. After that, an OD value of 590 nm was measured using a microplate reader (Thermo Fisher Scientific).
Immunofluorescence assay
The SKOV3 and OVCAR-3 cells with different treatments were harvested, washed with D-Hank’s buffer, and incubated with 2’,7’-dichlorofluorescein diacetate (DCFH-DA) at 37°C for 25 min. The DCF fluorescence of cells was analyzed using a fluorospectrophotometer at an excitation wavelength of 488 nm and an emission wavelength of 535 nm. The fluorescent intensity of DCF was tested by flow cytometry.
In vivo assay
Twelve Nude female BALB/c-nu mice (5-weeks-old) were from Shanghai Lab. Animal Research Center (Shanghai, China). All mice were kept in a temperature and humidity-controlled rooms with the 12 h light/dark cycle. All animal experiments were approved by the Animal Ethics Committee of Shandong Provincial Hospital Affiliated to Shandong First Medical University.
SKOV3 cells (5 × 106) were injected subcutaneously into mice according to the previously described methods with minor changes. 19 To evaluate the effect of ropivacaine on the growth of ovarian cancer, ropivacaine (10 mg/kg) was injected intraperitoneally into mice referring to the previously reported methods with minor revisions. 15 The size of the tumor was measured every day and the tumor volumes were calculated by the formula: length × width2/2 = tumor volume (mm3). When the tumor size reached 2000 mm3, all mice were sacrificed and the excised tumor tissues were weighed to evaluate the antitumor effect.
Immunohistochemical assay
The xenografted tumor tissues were fixed with 10% formalin (Millipore) for 24 h, dehydrated and embedded in paraffin, and then made into 5 μm sections. The expressions of Ki-67, OCT4, Nanog, GPX4 and SLC7A11 were assessed using immunohistochemistry (IHC) methods with anti-Ki-67 (Abcam, 1 µg/ml, ab15580), anti-OCT4 (Abcam, 1: 250, ab200834), anti-Nanog (Abcam, 1: 100, ab109250), anti-GPX4 (Abcam, 1: 100, ab125066) and anti-SLC7A11 (Abcam, 1 µg/ml, ab37185) antibodies. Briefly, the sections were blocked by 1% BSA and were incubated with the above primary antibodies overnight at 4°C. Followed by the incubation with a secondary antibody for 1.5 h at room temperature. Subsequently, the sections were dyed with diaminobenzidine (DAB) and counterstained with hematoxylin. Ultimately, the sections were examined under a microscope (Olympus) and the percentage of positive cells was calculated.
Statistical analysis
All data were analyzed using SPSS 20.0 software (IBM, Chicago, USA). Data were presented as mean ± standard deviation. For the IC50 value of ropivacaine, the growth curve of ovarian cancer cells was drawn with different concentrations of ropivacaine as abscissa and cell viability (%) as ordinate. The IC50 value was the ropivacaine concentration of ovarian cancer cells at 50% viability. Differences between the two groups or more than two groups were assessed by Unpaired Student’s t-test or one-way ANOVA followed by Tukey’s post-test. p < .05 was considered statistically significant.
Results
Ropivacaine restrains the ovarian cancer cell growth
The ovarian cancer cell growth was assessed after the SKOV3 and OVCAR-3 cells were treated with different doses of ropivacaine. The results demonstrated that the SKOV3 cell growth began to be repressed at a 10 μM dose of ropivacaine in comparison with the control group, and the IC50 was 18.64 μM (Figure 1(a)). Meanwhile, the growth of OVCAR-3 cells was also inhibited from the 10 μM dose of ropivacaine compared with the control group, and the IC50 was 24.86 μM ((Figure 1(b)). The 10, 20, and 40 μM doses of ropivacaine were selected for subsequent experiments referring to the experimental data of the CCK-8 assay. In general, ropivacaine repressed ovarian cancer cell growth. Effect of ropivacaine on ovarian cancer cell growth. Ovarian cancer cells SKOV3 and OVCAR-3 were treated with different concentrations (0, 2.5, 5, 10, 20, 40, 80, and 160 μM) of ropivacaine for 24 h. (A–B) The growth of SKOV3 and OVCAR-3 was evaluated by a Cell Counting Kit-8 (CCK-8) assay. *p < .05, **p < 0.01 vs. control (0 μM). The independent assays were repeated three times. Rop: ropivacaine.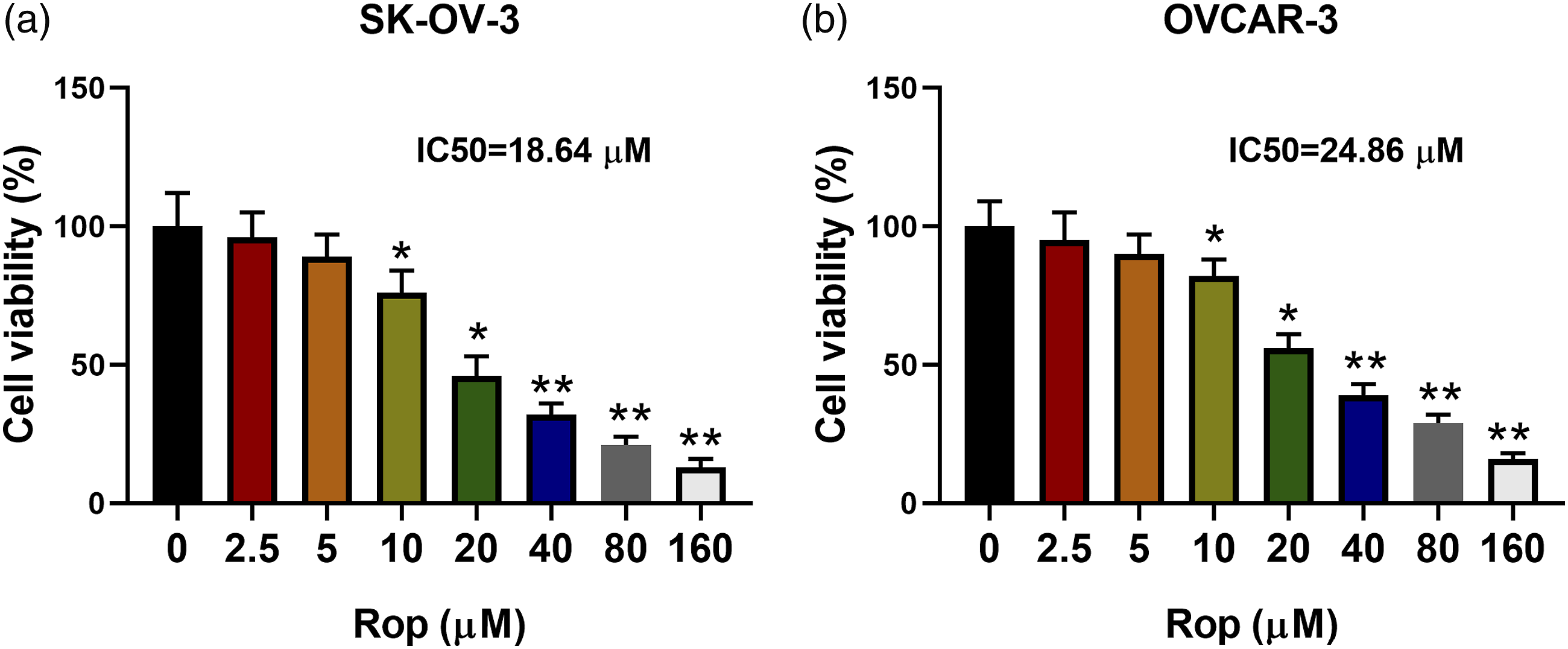
Ropivacaine represses the stemness of ovarian cancer cells
To further investigate the ropivacaine function on the ovarian cancer cell stemness, the ALDH positive cells were assessed after the ovarian cancer cells were treated with different concentrations of ropivacaine. As displayed in Figure 2(a), the ALDH positive cells were decreased gradually with the increased ropivacaine concentration, and the ALDH positive cells were reduced by nearly half at the 20 μM dose. As we all know, the sphere-formation ability is a pivotal method for the identification of tumor stem cells in vitro. Furthermore, the results of the sphere-formation assay confirmed that the sphere-formation ability of SKOV3 cells was weakened from the 10 μM dose of ropivacaine, and the sphere-formation ability of OVCAR-3 cells began to be reduced at a 20 μM dose of ropivacaine (Figure 2(b)). As presented in Figure 2(c), the protein levels of markers for stem cells OCT4 and Nanog protein levels began to be decreased in SKOV3 cells at a 10 μM dose of ropivacaine, and the OCT4 and Nanog protein levels in OVCAR-3 cells were down-regulated from the 20 μM dose of ropivacaine. The protein quantification results were displayed in Figure 2(c). To sum up, ropivacaine restrained the ovarian cancer cell stemness. Influence of ropivacaine on the stemness of ovarian cancer cells. Ovarian cancer cells SKOV3 and OVCAR-3 were treated with different contents (0, 10, 20, and 40 μM) of ropivacaine for 24 h. (A) The Aldehyde dehydrogenase (ALDH) positive cells were determined by flow cytometry. (B) The sphere-formation ability of ovarian cancer cells was analyzed using a sphere-formation assay. (C) The protein levels of OCT4 and Nanog were measured by Western blot. *p < .05, **p < .01 vs. control (0 μM). The independent assays were repeated three times.
Ropivacaine accelerates ferroptosis in ovarian cancer cells
Considering the importance of Fe2+ levels in ferroptosis (the novel mechanisms that regulate cell death and repress ovarian cancer cell growth), the Fe2+ levels were assessed in SKOV3 and OVCAR-3 cells and the results demonstrated that the Fe2+ levels were gradually elevated with the increased ropivacaine concentration and the Fe2+ levels increased nearly twofold when the ropivacaine dose was 20 μM (Figure 3(a)). The SKOV3 and OVCAR-3 cells were treated with ropivacaine and/or erastin (an agonist of ferroptosis), ropivacaine and/or ferrostatin-1 (inhibitor of ferroptosis). As displayed in Figure 3(b), the Fe2+ levels were further elevated after the erastin treatment, while the ferrostatin-1 treatment decreased the Fe2+ levels. Meanwhile, the ROS levels of SKOV3 cells were elevated from the 10 μM dose of ropivacaine, and the ROS levels of OVCAR-3 cells began to be increased at a 20 μM dose of ropivacaine (Figure 3(c)). Western blot data indicated that the protein levels of ferroptosis inhibitors GPX4 and SLC7A11 began to be decreased in SKOV3 cells at a 10 μM dose of ropivacaine, and the GPX4 and SLC7A11 protein levels in OVCAR-3 cells were reduced from the 20 μM dose of ropivacaine (Figure 3(d)). The above data suggested that ropivacaine accelerated the ferroptosis in ovarian cancer cells. Regulation of ropivacaine on ferroptosis in ovarian cancer cells. (A) Ovarian cancer cells SKOV3 and OVCAR-3 were treated with different contents (0, 10, 20, and 40 μM) of ropivacaine for 24 h. The Fe2+ levels were detected by the Iron ion kit. (B) SKOV3 and OVCAR-3 cells were treated with 40 μM ropivacaine and/or 5 mmol/l erastin, 40 μM ropivacaine and/or 5 μM ferrostatin-1 for 24 h. Analysis of the Fe2+ levels using an Iron ion kit. (C) Ovarian cancer cells SKOV3 and OVCAR-3 were treated with different contents (0, 10, 20, and 40 μM) of ropivacaine for 24 h. The ROS levels of ovarian cancer cells were assessed by immunofluorescence assay (scale bar, 50 μM). (D) The GPX4 and SLC7A11 protein levels were measured using Western blot. *p < .05, **p < .01 vs. control (0 μM) or 40 μM ropivacaine. The independent assays were repeated three times.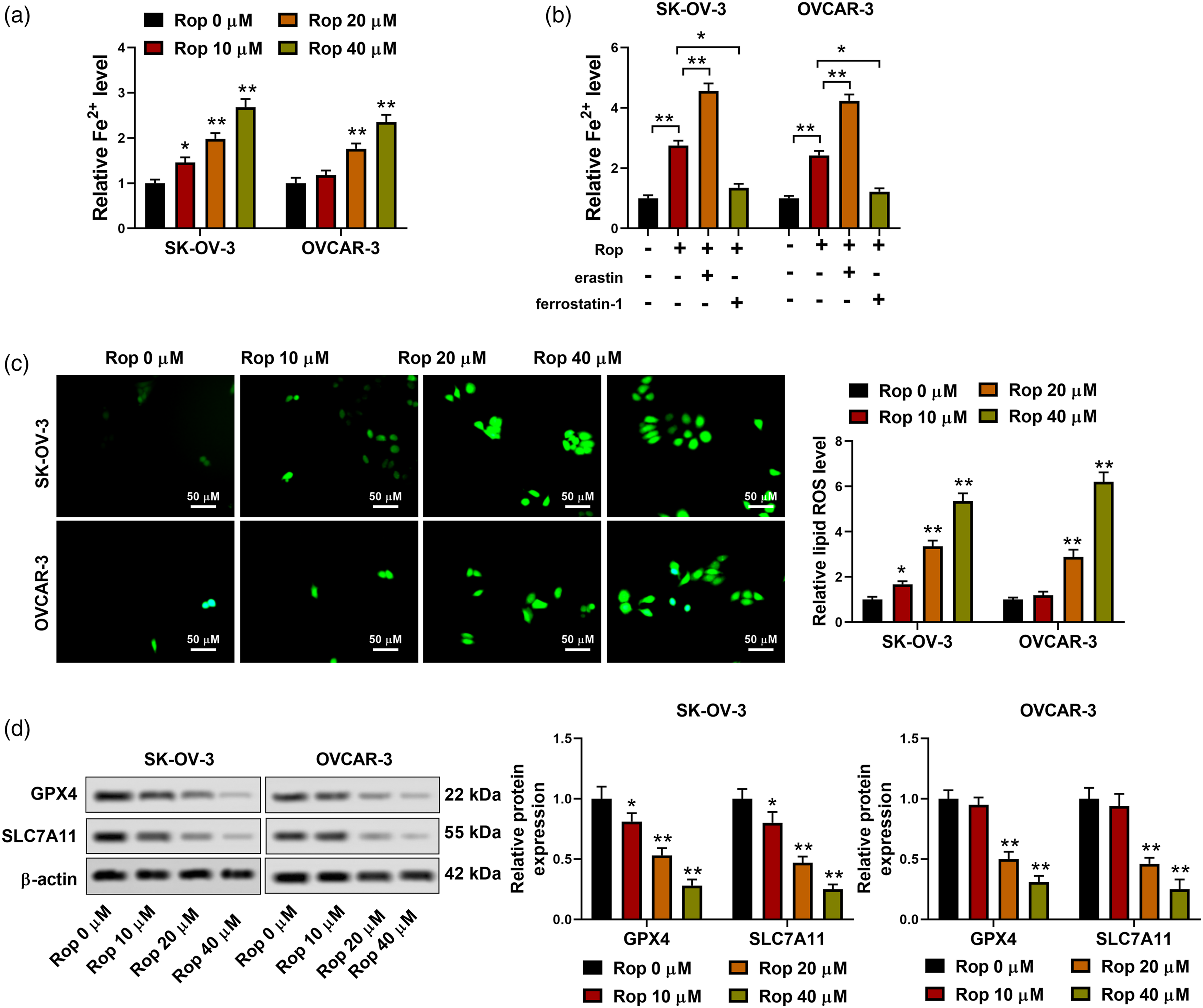
Ropivacaine inactivates the PI3K/AKT signaling pathway in ovarian cancer cells
As reported, the abnormal activation of the PI3K/AKT signaling pathway accelerates ovarian cancer development.
20
Therefore, we attempted to clarify whether ropivacaine participated in regulating the activation of the PI3K/AKT signaling pathway in ovarian cancer. Total-PI3K, total-AKT, and total-mTOR were internal controls for the calculation of relative expressions of p-PI3K, p-AKT (S473), p-AKT (T308) and p-mTOR (S2448), respectively. As displayed in Figure 4(a), the protein levels of p-PI3K and p-AKT (S473), p-AKT (T308), and p-mTOR (S2448) in cells were gradually decreased with the increase of ropivacaine concentration, while the total-PI3K, total-AKT and total-mTOR protein levels did not change significantly. After the SKOV3 cells were treated with ropivacaine and/or 740 Y-P (a commonly used PI3K agonist), the protein levels of p-PI3K and p-AKT (S473), p-AKT (T308), and p-mTOR (S2448) in cells were down-regulated after the ropivacaine treatment, while the 740 Y-P treatment partially reversed this down-regulation; the total-PI3K, total-AKT, and total-mTOR protein levels were unchanged between all groups (Figure 4(b)). Meanwhile, the CCK-8 results corroborated that the ropivacaine treatment reduced cell viability, and this reduction was partially reversed by the 740 Y-P treatment (Figure 4(c)). As displayed in Figure 4(d), the sphere-formation ability of ovarian cancer cells was weakened after the ropivacaine treatment, and this trend was partially reversed by the 740 Y-P treatment. Furthermore, the treatment of ropivacaine increased the Fe2+ levels, and the Fe2+ levels were increased nearly threefold, while the treatment of 740 Y-P partially reversed this increase (Figure 4(e)). Similarly, the ROS levels of ovarian cancer cells were elevated after the ropivacaine treatment, and this effect was partially reversed by the 740 Y-P treatment (Figure 4(f)). In summary, ropivacaine inactivated the PI3K/AKT signaling pathway in ovarian cancer cells. Effect of ropivacaine on the PI3K/AKT signaling pathway in ovarian cancer cells. (A) Ovarian cancer cells SKOV3 were treated with different contents (0, 10, 20, and 40 μM) of ropivacaine for 24 h. Analysis of the p-PI3K, total-PI3K, p-AKT (S473), p-AKT (T308), total-AKT, p-mTOR (S2448), and total-mTOR protein levels in cells by Western blot. (B) The SKOV3 cells were treated with 40 μM ropivacaine and/or 20 μM 740 Y-P for 24 h. Detection of the p-PI3K, total-PI3K, p-AKT (S473), p-AKT (T308), total-AKT, p-mTOR (S2448), and total-mTOR protein levels in cells using Western blot. (C) The viability of SKOV3 cells was assessed by CCK-8 assay. (D) The sphere-formation ability of SKOV3 cells was evaluated using a sphere-formation assay. (E) The Fe2+ levels were measured by an Iron ion kit. (F) Analysis of the ROS levels of SKOV3 cells using immunofluorescence assay (scale bar, 50 μM). *p < .05, **p < .01 vs 0 μM ropivacaine, control or 40 μM ropivacaine. Con: control. The independent assays were repeated three times.
Ropivacaine restrains the proliferation of ovarian cancer cells in vivo
Subsequently, we further evaluated the function of ropivacaine in ovarian cancer in vivo. The ropivacaine treatment reduced the tumor volume in comparison with the sham group (Figure 5(a)) and the representative macroscopic figures of tumors were provided in Supplementary Figure 1. Also, the treatment of ropivacaine had no significant changes in the weight of mice in comparison with the sham group (Figure 5(b)). Meanwhile, the ropivacaine treatment decreased the tumor weight compared with the sham group (Figure 5(c)). Besides, the expressions of Ki-67 (proliferating marker), OCT4, Nanog, GPX4, and SLC7A11 in subcutaneous tumor tissues of mice were measured and the results confirmed that the ropivacaine treatment reduced the Ki-67, OCT4, Nanog, GPX4 and SLC7A11 expressions in tissues (Figure 5(d)). The above data suggested that ropivacaine weakened the proliferation of ovarian cancer cells in vivo. Ropivacaine regulates the proliferation of ovarian cancer cells in vivo. SKOV3 cells (5 × 106) were injected subcutaneously into mice and ropivacaine (10 mg/kg) was injected intraperitoneally into mice. (A–C) Analysis of the tumor volume, mice weight, and tumor weight between different groups. (D) The expressions of Ki-67 (proliferating marker), OCT4, Nanog, GPX4, and SLC7A11 in subcutaneous tumor tissues of mice were assessed using an immunohistochemical assay (scale bar, 50 μM). **p < .01 vs. sham. The independent assays were repeated three times.
Discussion
Ovarian cancer seriously affects the physical and mental health of women, so it is urgent to explore effective treatment strategies. 21 Ropivacaine has an antitumor role in various human tumors. 22 However, the ropivacaine role in ovarian cancer remained uncertain. In this study, we treated ovarian cancer cell lines with different doses of ropivacaine and demonstrated that ropivacaine inhibited ovarian cancer cell growth, implying that ropivacaine might exert a protective role in ovarian cancer. This study is the first to investigate the ropivacaine function in ovarian cancer.
Recently, the important function of cancer stem cells in ovarian cancer has been widely recognized with the deepening of research. LncRNA HOX transcript antisense intergenic RNA maintains the stemness of ovarian cancer stem cells through regulating the miR-206/TBX3 axis 23 ; platelet-activating factor accelerates the spheroid formation and increases the stemness genes expressions, thus aggravating ovarian cancer, 24 which implies that reducing the ovarian cancer cell stemness is expected to relieve ovarian cancer. Considering that cell subsets with high ALDH activity in tumors are closely related to the stemness of cancer stem cells, 25 this study further confirmed that the ALDH positive cells were decreased gradually with the increased ropivacaine concentration. Also, we demonstrated that the ropivacaine treatment repressed the ovarian cancer cell stemness by sphere-formation assay. Meanwhile, as the markers for stemness, OCT4 and Nanog are commonly used to evaluate the stemness of tumor cells. 26 This study further indicated that ropivacaine down-regulated the OCT4 and Nanog protein levels. The above findings revealed that ropivacaine repressed the ovarian cancer cell stemness.
Ferroptosis is a novel cell death pathway first proposed by Dixon et al 27 and is characterized by the accumulation of iron-dependent lipid peroxides. 28 Various studies have been conducted on the mechanism of ferroptosis since the concept of ferroptosis is proposed. To date, several regulatory mechanisms and signaling pathways of ferroptosis have been identified. For instance, Zhang et al. confirmed that the attenuation of the PKCβII-ACSL4 pathway reduces the accumulation of lipid peroxidation, thereby effectively blocking ferroptosis 29 ; Yi et al. indicated that the abnormal activation of the PI3K-AKT-mTOR signaling pathway protects cancer cells from oxidative stress and ferroptosis through SREBP1/SCD1-mediated lipogenesis. 30 In recent years, the ferroptosis function in ovarian cancer has been gradually confirmed. As reported, ferroptosis promotes the occurrence and development of ovarian cancer and that ferroptosis might aggravate ovarian cancer by mediating tumor metastasis. 31 A recent study reveals that erastin and cisplatin induce ferroptosis by increasing the level of reactive oxygen species, thereby inhibiting ovarian cancer cell growth and alleviating ovarian cancer. 32 Considering the inhibitory effect of ropivacaine on ovarian cancer cell growth, we attempted to clarify whether ropivacaine mediated the ferroptosis in ovarian cancer cells. As expected, our data revealed that the different doses of ropivacaine facilitated ferroptosis in ovarian cancer cells, which was similar to the above conclusions.
In recent decades, the molecular mechanisms of various genetic factors in cancer have been revealed, including Phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT). 33 PI3K is a lipid kinase and an important cause of chemotherapy resistance in cancer treatment. 34 AKT is a critical downstream effector of the PI3K signaling pathway and regulates various pathways, including restraining cell apoptosis, stimulating cell growth, and regulating cell metabolism. 35 Accumulated studies corroborate that the PI3K/AKT signaling pathway is involved in the regulation of the occurrence and development of multiple tumors. For example, the carcinogenic function of IGF2BP2 in pancreatic cancer is achieved through the activation of the PI3K/AKT signaling pathway to accelerate tumor proliferation 36 ; microRNA functions in the growth, and metastasis of breast cancer cells by targeting the PI3K/AKT carcinogenic signaling pathway. 37 Emerging evidence suggests that the PI3K/AKT signaling pathway participates in ovarian cancer progression. Ediriweera et al. confirmed the importance of the PI3K/AKT signaling pathway in ovarian cancer progression and comprehensively analyze the clinical application of the PI3K/AKT signaling pathway inhibitors in ovarian cancer 38 ; Deng et al. expounded that inactivating the PI3K/AKT signaling pathway reduces ovarian cancer chemoresistance by decreasing the cancer stem cell markers levels, 39 implying that repressing the abnormal activation of the PI3K/AKT signaling pathway slowed down the occurrence and development of ovarian cancer. Based on our previous experimental results that ropivacaine repressed the ovarian cancer cell growth, we attempted to clarify whether ropivacaine mediated the ovarian cancer progression through the PI3K/AKT signaling pathway. Interestingly, our results confirmed that the ropivacaine treatment inactivated the PI3K/AKT signaling pathway in ovarian cancer cells. After confirming the function and molecular mechanism of ropivacaine in ovarian cancer in vitro, we further verified the protective function of ropivacaine in ovarian cancer in vivo. As expected, ropivacaine reduced the proliferation of ovarian cancer cells in vivo, which was similar to the in vitro results.
In conclusion, we confirmed the ropivacaine function in ovarian cancer from the following multiple perspectives: (1) ropivacaine treatment repressed the ovarian cancer cell growth; (2) ropivacaine inhibited the stemness of ovarian cancer cells; (3) ropivacaine accelerated the ferroptosis in ovarian cancer cells. After confirming that ropivacaine had a protective function against ovarian cancer, we further investigated its potential molecular mechanism. Our data corroborated that ropivacaine inactivated the PI3K/AKT signaling pathway in ovarian cancer cells. Meanwhile, the in vivo assay indicated that ropivacaine restrained the proliferation of ovarian cancer cells in vivo. This research might provide novel drugs and potential biomarkers for ovarian cancer treatment. Furthermore, a recent research has indicated that Frizzled-7 restrains ferroptosis and promotes stemness both in vitro and in vivo models of ovarian cancer, thereby exacerbating ovarian cancer. 40 Thus, the limitation of this study mainly lies in that whether ropivacaine repressed the proliferation of ovarian cancer cells by activating ferroptosis was not investigated in vivo. We would further explore in vivo in the future to enrich the content of this study. Moreover, in view of the important role of ropivacaine in ovarian cancer models in vivo and in vitro, we would verify the ropivacaine function in clinical tissue samples to further enrich the content of this study.
Supplemental Material
Supplemental Material - Ropivacaine represses the ovarian cancer cell stemness and facilitates cell ferroptosis through inactivating the PI3K/AKT signaling pathway
Supplemental Material for Ropivacaine represses the ovarian cancer cell stemness and facilitates cell ferroptosis through inactivating the PI3K/AKT signaling pathway by Yi Lu, Jinbao Mao, Yanbing Xu, Hao Pan, Yu Wang and Wei Li in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
