Abstract
Background
Today, network pharmacology and molecular docking were utilized for investigation and prediction of possible mechanisms in fields such as pharmacognosy and traditional Chinese medicine formulas when treating disease.
Purpose
Chronic obstructive pulmonary disease (COPD) is caused by exposure to toxic particles and leads to chronic bronchitis and lung dysfunction. Lijing-Anfei decoction (LAD) is a traditional Chinese herbal decoction used to cure COPD. This study explored the effects of this decoction and its intrinsic mechanism in a COPD mouse model.
Materials and Methods
Possible targets were retrieved from several databases and combined using Veny. Protein-protein interaction (PPI) networks were constructed via STRING database. Gene ontology and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses were performed via the Metascape database. The COPD model was established by exposing the mice to cigarette smoke for 12 weeks. After the model was established, LAD was administered for four weeks. Enzyme-linked immunosorbent assay (ELISA) was performed, and body weights and lung wet/dry ratios were measured. Lung morphology was observed by hematoxylin-eosin staining.
Results
After the screening, 32 overlapping targets were used to generate PPI networks and the following hub genes were identified: JUN, IL6, RELA, IL1B, HIF1A, MAPK1, PPARG, PTGS2, MAPK14, and NOS2. The KEGG analysis indicated that hypoxia-inducible factor-1 (HIF-1) and other signaling pathways are possible signaling pathways. The molecular docking results demonstrated tight links between major compounds and their targets. In animal experiments, LAD attenuated body weight loss, downregulated inflammatory cell numbers, and inhibited the enhancement of Fe2+ levels. The mean linear intercept index changes indicated that LAD ameliorated the lung structural damage. ELISA revealed that it downregulated pro-inflammatory cytokine levels and inhibited excessive oxidant production, TdT-mediated dUTP-biotin nick end labeling assay demonstrated that it alleviated cell death in the lungs, and immunohistochemical staining revealed that it inhibited HIF-1α and PTGS2. ML228—an agonist of HIF-1—blocked the effect of LAD, validating the role of HIF-1-mediated ferroptosis in the pharmacological effects of this decoction.
Conclusion
This study revealed that LAD was successful in treating COPD.
Introduction
Chronic obstructive pulmonary disease (COPD) is a major health challenge experienced by the current population, especially the elderly. According to predictions, COPD is one of the top 3 leading causes of mortality worldwide (Pang et al., 2018) and imposes a marked economic and social burden worldwide (Singh et al., 2019). COPD is characterized by airflow obstruction and persistent inflammation under continuous exposure to noxious stimuli such as industrial waste gas, dust, and cigarette smoke (the major cause) (Zhang et al., 2016). Continuous exposure to cigarette smoke induces lung cellular senescence, including inflammation, excessive oxidant production, and further lung tissue damage. Most patients with COPD experience a palpable reduction in lung function and a notable loss of quality of life (Du et al., 2016). Recently, anti-inflammation and lung tissue protection have become important in COPD management; therefore, the identification of new targets and drugs is crucial for COPD therapy.
Ferroptosis is an iron-overloaded form of cell death that differs from conventional forms. Iron overload and ferroptosis are associated with COPD progression (Xia et al., 2023). Seven ferroptosis-related genes have been validated as COPD markers, indicating a relationship between COPD and ferroptosis (Lin, Xu, et al., 2022). Ferroptosis in bronchial epithelial cells results in macrophage polarization in animal models of COPD (Liu, Zhang, et al., 2022), and macrophage ferroptosis is also associated with COPD pathogenesis (Fan et al., 2023). Ferroptosis has gradually been accepted as a novel therapeutic target for COPD (Wu et al., 2021) and hydrogen sulfide inhibited airway inflammation by suppressing ferroptosis in COPD (Wang et al., 2022).
Medicinal plants have been used as natural cures for many centuries (Sharma et al., 2023). Many formulas or decoctions composed of several different medicinal plants have a long application history in various disease treatments and have proven efficacy and safety, but often lack research on modern pharmacological mechanisms (Li, He, et al., 2021; Yang et al., 2021). Network pharmacology employs genomic technology and systems biology to explore sophisticated relationships between biological systems via computational intelligent tools (Sharma, Singla, et al., 2022) and has been applied as a useful and practical research tool for mechanism and target prediction (Sharma, Jadhav, et al., 2022). Lijing-Anfei is a famous Chinese decoction composed of Hedysarum multijugum Maxim, Platycodon grandiflorum, Trichosanthes kirilowii Maxim, Salviae chinensis Herba, Radix Semiaquilegiae, Scutellaria baicalensis, and Radix Glehniae. The Lijing-Anfei decoction (LAD) has been used for COPD treatment in clinical settings for decades; however, there is a knowledge gap between clinical application and scientific evidence concerning curative effects. This study sought to answer the question: can the LAD alleviate COPD symptoms by regulating ferroptosis? Mice exposed to smoke are prone to COPD; therefore, they are the most commonly employed species in COPD models in vivo (Ghorani et al., 2017). Here, the effects and intrinsic mechanisms of LAD were probed via network pharmacology analysis, molecular docking, and animal experimental validation.
Materials and Methods
Prediction and Combination of Targets
Targets related to each herb in the LAD were identified based on traditional Chinese medicine systems pharmacology and analysis platform database (Ru et al., 2014). In addition, Therapeutic Targets Disease (Zhou et al., 2022), GeneCards (Rebhan et al., 1997), Pharmacogenomics Knowledgebase (Whirl-Carrillo et al., 2012, Online Mendelian Inheritance in Man (Amberger & Hamosh, 2017), and DisGeNet databases (Piñero et al., 2017) were used to search for COPD-related targets by entering keywords of “COPD” or “chronic obstructive pulmonary disease.” Ferroptosis-related genes were downloaded from the FerrDb database (Zhou & Bao, 2020). After removing duplicate targets, the full protein names were converted into gene abbreviations using the UniProt database (The UniProt Consortium, 2018). A Venn diagram was automatically generated on the Venny 2.1.0 website (
Network Construction and Analysis
The protein-protein interaction (PPI) network (Athanasios et al., 2017), when the input of LAD-COPD-ferroptosis overlapping genes was automatically generated using the STRING database (Szklarczyk et al., 2019), with a threshold of species ‘Homo sapiens’, and the cutoff confidence score, was high with a threshold of >0.7. Further, the result was input into Cytoscape 3.9.1 to explore the hub genes (Shannon et al., 2003).
Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment Analysis
GO analysis which consists of molecular function (MF), biological processes (BP), and cellular components (CC), and KEGG analysis to predict possible intrinsic mechanisms were performed via the Metascape database (Zhou et al., 2019).
Molecular Docking of LAD and Targets of Interest
The 3D-structure files of major hub genes were obtained from the protein data bank. The main compound structures in the LAD were obtained from the PubChem database, and ChemBio3D software (v. 20.0.0.41) was used for binding energy minimum and file conversion. The AutoDock tool (v. 1.5.7) was employed to generate the ligand and perform protein hydrogenation, and the structure was saved in the pdbqt file. AutoDock Vina (Trott & Olson, 2010) was used for molecular docking. PyMOL software (PyMOL Molecular Graphics System, v. 2.0 Schrödinger, LLC) based on Python (Mooers, 2020) was used to visualize the binding amino acids and distances.
Reagents and Chemicals
Antibodies were obtained from Abcam (MA, USA). Enzyme-linked immunosorbent assay (ELISA) kits for cytokines and hematoxylin-eosin (HE) reagents were provided by Beyotime Corporation (Nantong, Jiangsu, China). The agonist was purchased from Selleck Chemicals (MA, USA). Biochemical kits for determining oxidative stress levels were obtained from Nanjing Jiancheng Bioengineering Institute (Nanjing, Jiangsu, China). The Fe2+ content determination kit was provided by Pulilai Corporation (Beijing, China).
Animal Study Ethics Approval and Group Assignment
Ethical approval was obtained from the Laboratory Animal Ethics Committee of the Yongxinkangtai Institute (YXKT2022L019), China. Male C57 mice (19–21 g) were provided by the Jiesijie Laboratory Animal Co., Ltd. and housed in the Yongxinkangtai Experimental Animal Center. They were kept following the routine of raising experimental animals. After a few days of acclimation, 42 mice were randomly assigned to 7 groups (n = 6): (a) control (control), (b) COPD model (COPD), (c)–(f) LAD at a 7.5, 15, 30, and 30 mg/kg combined with a specific hypoxia-inducible factor-1 (HIF-1) agonist ML228 to validate the role of the intrinsic pathway (30 mg/kg + ML228), respectively. The positive drug group (g) was given 2 mg/kg dexamethasone.
Animal Model Establishment and Drug Treatment
COPD was induced using 3R4F cigarettes (University of Kentucky, USA). The COPD model was established according to a method with minor differents (Xu et al., 2018). Briefly, the mice were exposed to cigarette smoke in an exposure chamber maintained by a ventilation and diffusion machine (Yuan-sen-kai-de Technology Corp., Beijing, China) for 60 min, 500 mg/m3 total particulate matter twice daily (8–9
Among groups 3–6, LAD granules dissolved in PBS and administered orally at 7.5, 15, and 30 mg/kg/day once daily. In group 6, 0.03 mg ML228 dissolved in 1 mL DMSO was injected intraperitoneally (0.1 mL) into each mouse once daily (Endo & Panayi, 2020), while 30 mg/kg LAD was administered orally. In group 7, dexamethasone (2 mg/kg) was intraperitoneally injected (Luo et al., 2017). In the COPD group, equivalent volumes were administered orally once daily. All drugs were administered for four weeks.
Body Weight Assessment and Collection of Bronchoalveolar Lavage Flush
Body weight was recorded after the last administration. The mice were sacrificed, and the lungs were harvested immediately. The bronchoalveolar lavage flush (BALF) from the right lung was collected (Fan et al., 2018). A syringe was inserted into the trachea, and precooled PBS was used to lavage the lungs three times. The BALF was moved to tubes and centrifuged at 3,000 rpm, 2,664 g, and 4°C for 10 min. Supernatants were collected in tubes, and inflammatory cells were stained using Wright-Giemsa and counted by an independent pathologist who was blinded to the animal experiments (Peng et al., 2018). The wet/dry weight ratio was assessed (Huang et al., 2019). For the left lungs, the tissue weights were recorded under wet conditions. After drying at 65°C for two continuous days, the left lungs were reweighed under dry conditions, and the wet/dry weight ratios were further quantified.
Enzyme-linked Immunoassays for Pro-inflammatory Mediator Levels in BALF and Oxidant Levels in the Lung
ELISA was used to assess the cytokines of pro-inflammatory mediators in BALF of six mice in six groups, except for the positive drug group. The Th1 type inflammatory mediator levels regarding tumor necrosis factor-alpha (TNF-α), interleukin-2 (IL-2), and interferon-γ (INF-γ) were quantified by ELISA kits. The average optical density was calculated using a microplate reader (PT-3502; Ponetov, Beijing, China).
Oxidative stress parameters were quantified using commercial kits following the manufacturer’s protocol. The upper right lung samples were homogenized in pre-cooled PBS. The levels of glutathione (GSH) and reactive oxygen species (ROS) in lung tissue homogenates were quantified. Fe2+ levels in the lungs were measured (Bai et al., 2020).
Lung Morphometry
The lower right mice lungs were collected, fixed, and washed. Subsequently, they were embedded in paraffin wax and cut into 5 µm sections. HE staining was conducted to visualize the lung structural changes using a light microscope (MF31, Mshot Light, Guangzhou, China). The mean linear intercept (MLI) represents the average alveolar size and is the gold standard for observing COPD morphometry. Several equally distributed lines were laid on lung sections under a light microscope, the length of each grid line was separated by the number of alveolar intercepts, and the calculation was performed (Fujioka et al., 2021).
TdT-mediated dUTP-biotin Nick End Labeling (TUNEL) Assay
After the mice in the six groups (except the positive drug group) were executed, the lower right lungs were subjected to TUNEL experiments to investigate the apoptosis ratio (Bi et al., 2021; Zhang et al., 2016). Briefly, lung tissue was placed in liquid nitrogen and stored in a −80°C freezer. The mouse lung specimens were incubated successively with proteinase K, Triton X-100, and TINEL action solution. Before photographing, the slices were stained with DAPI and sealed using anti-fluorescence quenching. The specimens were then imaged via a fluorescence microscope (3D Histech Ltd., Budapest, Hungary). Apoptotic cell numbers were determined by green fluorescence, and the relative apoptotic ratio (number of apoptotic cells/total cells × 100%) was analyzed using ImageJ software (v. 6.0; National Institutes of Health, USA).
Lung Immunohistochemistry (IHC)
After execution, the lower right lungs of the COPD mice were subjected to IHC analysis to investigate the related protein expression (Du et al., 2016). The left lower lung sections were cut into 5 µm sections and then blocked with primary antibodies (anti-HIF-1 alpha (HIF-1α), 1:150, ab237544, Abcam, anti-PTGS2 (COX2), 1:150, ab179800, Abcam). Subsequently, mice lung sections were incubated with secondary antibodies. In last, mice lung sections were photographed using a microscope (MF31, Mshot, Guangzhou, China). The main indicators for comparison include integrated optical density and area of immunostaining which are assessed using NIH Image-Pro Plus 6.0 image analysis software (Media Cybernetics Co., MD, USA).
Statistical Analysis
Data from animal experiments are expressed as mean ± SD. Statistical differences between groups were considered significant at p < 0.05. Statistical analysis was performed using one-way ANOVA with the statistical software SPSS (v. 27.0; IBM, Chicago, IL, US) and GraphPad Prism (v. 6.0; GraphPad Software Inc., San Diego, CA, USA).
Results
Screening of Active Targets Between LAD, Ferroptosis, and COPD
After duplicates were removed, 1,327 LAD potential targets were obtained from the abovementioned databases. Furthermore, 1995 and 728 related targets of COPD and ferroptosis, respectively, were obtained from the above-mentioned databases. Finally, 32 overlapping targets of LAD–COPD-mediated ferroptosis were identified (Figure 1A). Information on the 32 overlapping targets is detailed in Supplementary Table 1.
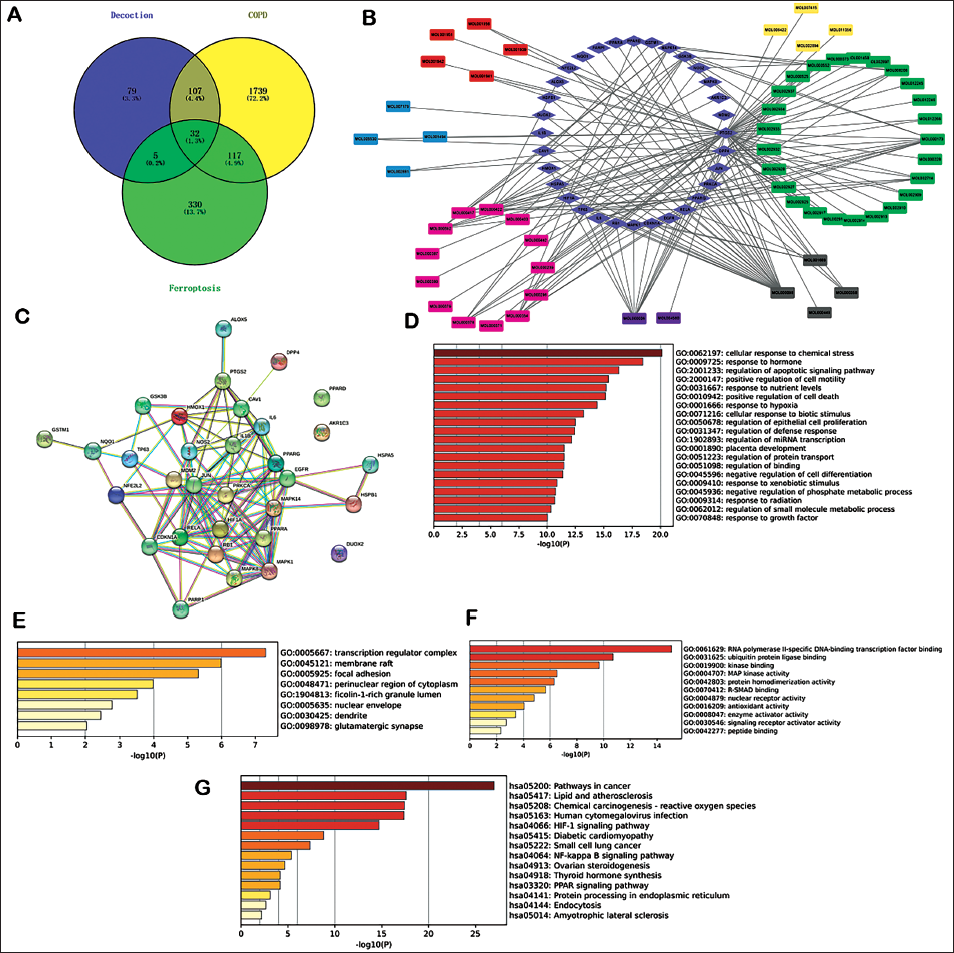
Compound-gene Network, PPI Network, and Hub Genes
The compound-gene network diagram is shown in Figure 1B. The compound information from related medicinal plants is indicated in Supplementary Table 2. There were many interactions between compounds of LAD and genes. The top 10 highlighted compounds and targets with centralities in the network and the compound-target network ranked by the degree method, closeness method, and betweenness are indicated in Supplementary Table 3. The PPI network is illustrated in Figure 1C. There were 32 nodes and 102 edges. The average node degree was 6.38. The top 10 core target genes using the cytoHubba plugin were JUN at a maximal clique centrality score of 625, IL6 (534), RELA (361), IL1B (336), HIF1A (278), MAPK1 (278), PPARG (272), PTGS2 (157), MAPK14 (152), and NOS2 (152), ranked by MCC scores.
GO and KEGG Pathway Enrichment Analysis
The overlapping 32 common targets were used for GO-BP, GO-MF, and GO-CC analyses. The top 10 items in each analysis according to −log10 (p values) are shown in Figure 1D–F. The top 15 KEGG prediction results were selected according to −log10 (p values) (Figure 1G).
Molecular Docking
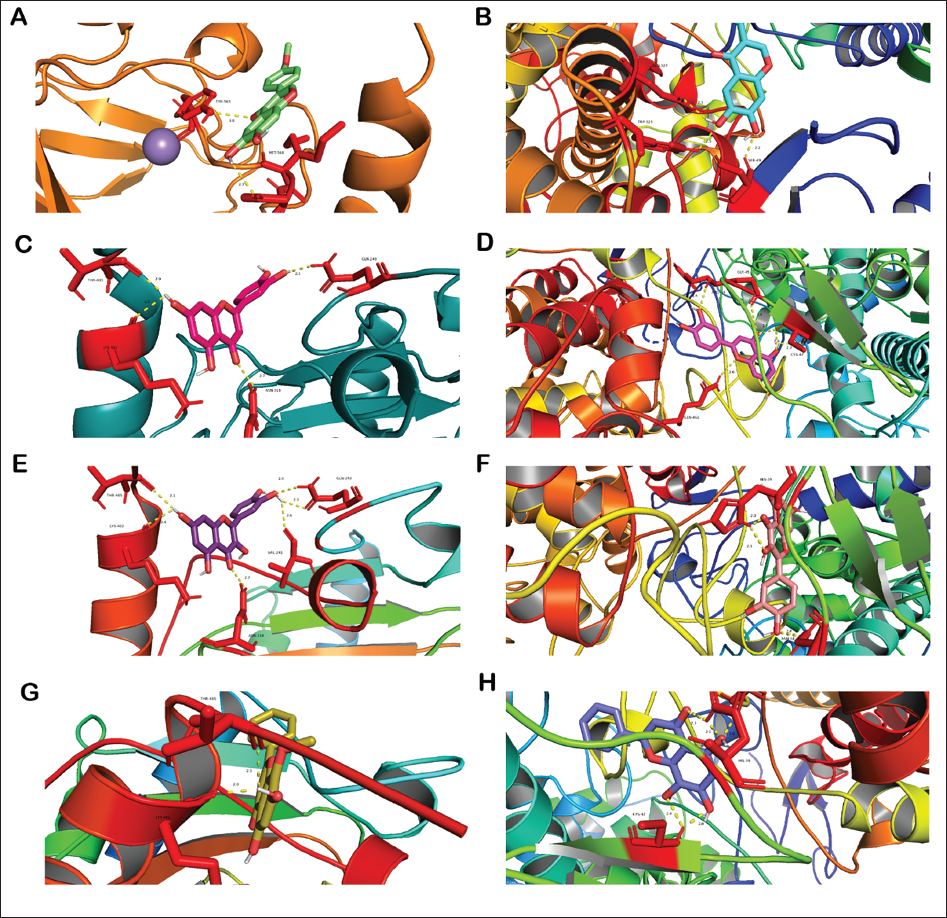
The ferroptosis marker proteins (PTGS2) and HIF-1 protein were used as receptors. Several interested main active compounds of the LAD were used as ligands. The docking results are shown in Figure 2A–H, and the docking energies of the major active compounds of the LAD to HIF-1 and PTGS2 are shown in Supplementary Table 4. All the docking energies were <−5 kcal/mol, indicating the relative stability between proteins and small-molecule compounds following the previous criteria (Lin, Gu, et al., 2022).
General Situation and Body Weights
After the mice were exposed to the cigarette smoke, their fur turned yellow, especially in the COPD group, suggesting that cigarette smoke induced lung insufficiency and, consequently, low nutrition. All mice survived the last LAD administration during the 16th week. As shown in Figure 3A. There was a statistically significant difference between the control group and the other six groups in the 12th week. However, no statistically significant difference was observed among the other six groups in the 12th week. In the 16th week, the COPD mice body weights were lower than those of the control. The body weights of the LAD group increased in a dose-dependent manner compared to those of the COPD group demonstrating that the LAD alleviated the weight loss caused by lung insufficiency.
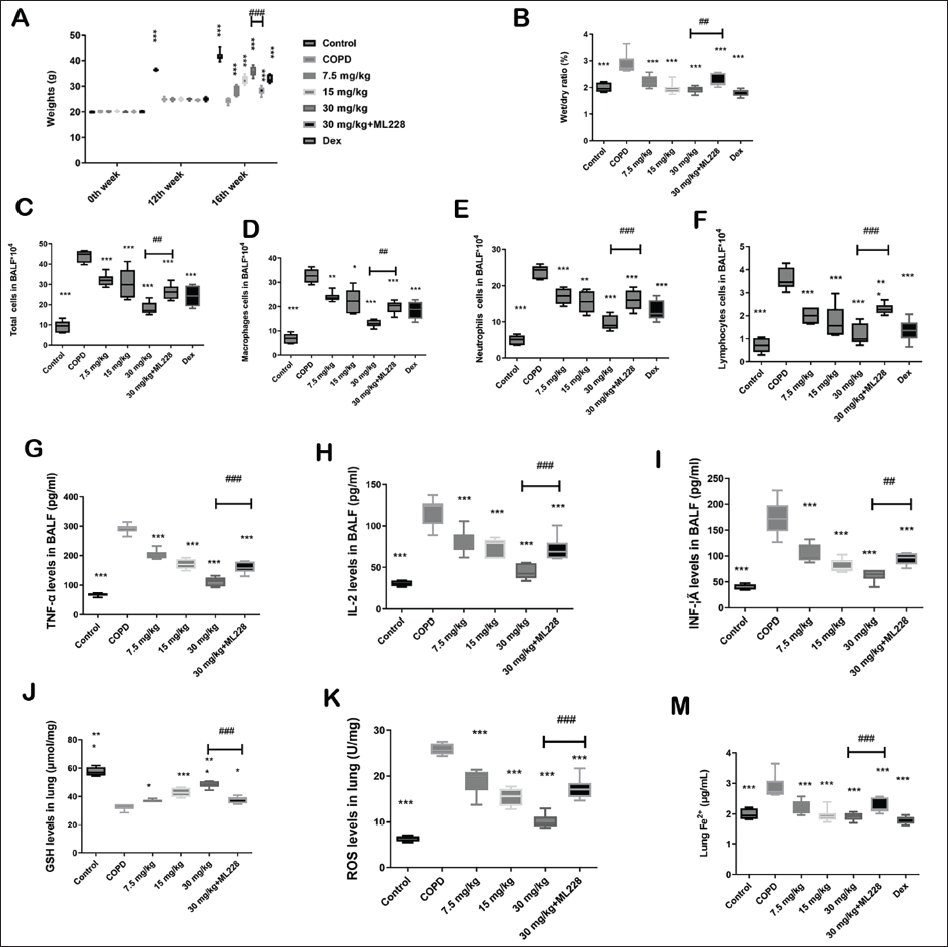
Lung Wet/Dry Weight Ratio and BALF Cell Counts
The lung wet/dry weight ratio is another index that reflects inflammatory responses. The lung wet/dry weight ratio in the COPD group was higher than that in the control, whereas the weight ratios in the LAD-treated groups were lower than those in the COPD group (Figure 3B). The total macrophage, neutrophil, and lymphocyte BALF cell numbers were maintained at low levels in the control (Figure 3C–F). After continuous cigarette smoke exposure, they increased in the COPD group. Conversely, LAD treatment attenuated this increase after four weeks.
Inflammatory Cytokines in BALF and Oxidative Stress Parameters in the Lung
Inflammatory cytokine level increase is another COPD characteristic. The TNF-α (Figure 3G), IL-2 (Figure 3H), and INF-γ (Figure 3I) were maintained at low levels in control. After smoke exposure, the IL-2, TNF-α, and INF-γ levels were increased in the COPD group. Conversely, LAD treatment attenuated the increase in IL-2, TNF-α, and INF-γ levels after four weeks.
LAD regulates oxidative stress parameters. Exposure to cigarette smoke decreased the GSH levels (Figure 3J) and increased ROS levels (Figure 3K) in the lungs of mice in the COPD group compared to those in the control. However, LAD treatment attenuated GSH levels in the treatment groups compared to the COPD group. However, ROS levels showed the opposite trend.
Fe2+ levels remained low (Figure 3M) but were higher in the model group than in the control under smoke stimulation; however, LAD downregulated Fe2+ levels in a dose-dependent manner.
Histopathologic Analysis
Cigarette smoke induced a rise in MLI in the COPD group compared to the control (Figure 4A). The LAD decreased this rise and attenuated the mouse lung air space size. Chronic cigarette smoke exposure resulted in a marked inflammatory cell accumulation, especially around the trachea. Moreover, some alveolar walls were broken and the integrity of the alveoli was damaged. LAD treatment ameliorated lung tissue injury recovered alveolar integrity, reduced tracheal epithelium thickness, and reduced inflammatory cell numbers.
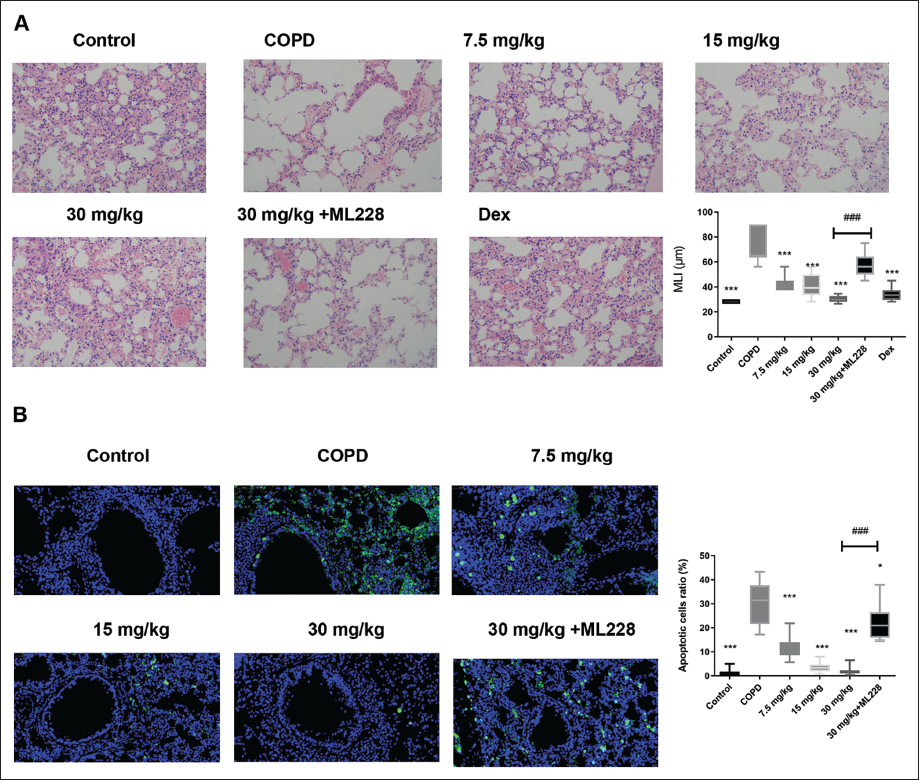
Cell Death Regulation, HIF-1, Ferroptosis Marker Proteins, and Mechanism Validation by LAD
First, we investigated cell death using a TUNEL assay. Cigarette smoke exposure enhanced the HIF-1 protein expression levels (Figure 4B); however, LAD administration inhibited cell death compared with that in the COPD group.
Second, the IHC results demonstrated that cigarette smoke exposure enhanced the HIF-1 protein expression levels (Figure 5A); however, LAD administration downregulated this compared with those in the COPD group. Furthermore, cigarette smoke exposure increased PTGS2 protein levels in the model compared to control (Figure 5B) but the LAD downregulated these levels.
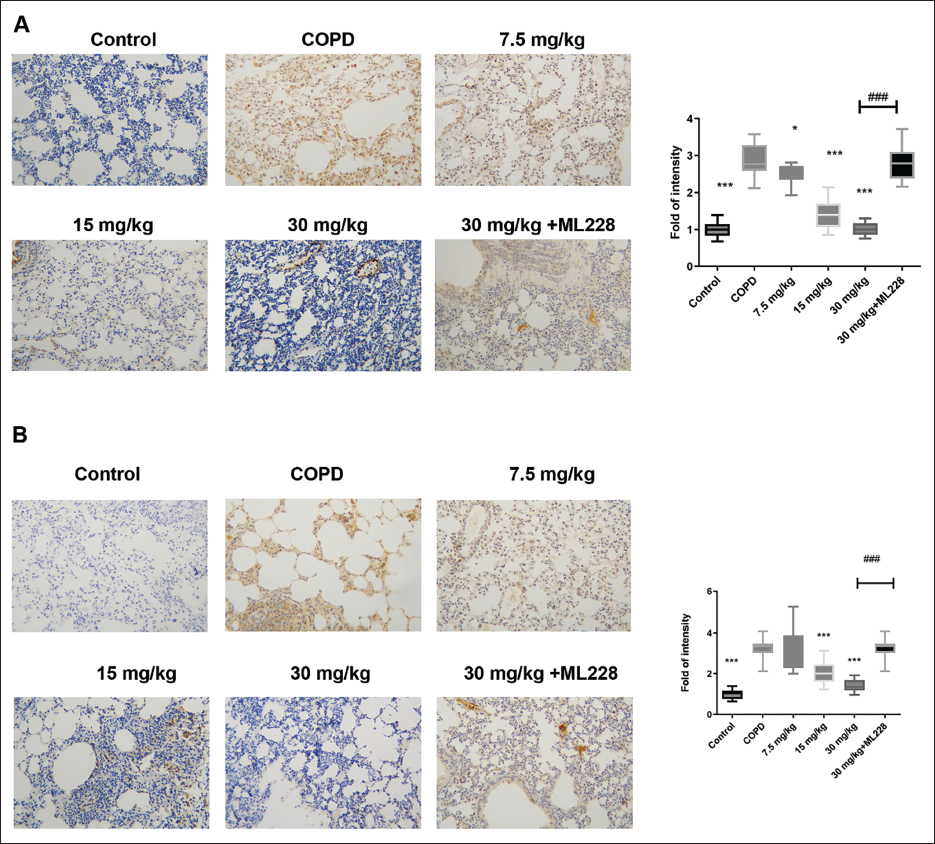
Third, the IHC staining results demonstrated that ML228, an agonist of HIF-1, reactivated the HIF-1 signaling pathway. Moreover, ML228 blocked the LAD effects on body weight (Figure 3A), lung weight ratio (Figure 3B), anti-inflammatory cytokine production (Figure 3G–I), antioxidant stress (Figure 3J and K), lung injury alleviation (Figure 4A), and cell death (Figure 4B). These results further validate the LAD’s role in mediating HIF-1 in COPD mice.
Discussion
COPD is a severe chronic respiratory condition with a high and growing prevalence in Asia and other regions worldwide (Adeloye et al., 2015). The LAD protective effect was tested in a COPD mouse model, demonstrating that it inhibited inflammatory responses and lung injury.
Herbs and medicinal plants contain various phytochemicals. Decoctions—consisting of several medicinal plants—contain more phytochemicals, and their mixed effects are more difficult to predict. Network pharmacology is leading research in drug discovery and development (Choudhary & Singh, 2018, 2022) and is a poly-pharmacological approach to construct a multi-targeting framework of various small molecules based on the concept of multiple compounds in medicinal plants—multiple targets (Choudhary et al., 2021; Choudhary & Singh, 2019). An outstanding prospect is drug discovery and prediction of the intrinsic mechanism of medicinal plants used in traditional medicine in Asian countries (Choudhary & Singh, 2018). Here, network pharmacology analysis was used to predict the LAD effect on COPD and the intrinsic mechanism of the HIF-1 signaling pathway. The top highlighted compounds in the compound-target network ranked by the degree centrality, closeness, and betweenness methods were MOL000098, MOL000006, MOL000173, and MOL000422; MOL000098, MOL000006, MOL000173, MOL000422, MOL000354, MOL000392, and MOL000417; and MOL000098, MOL000006, MOL000422, and MOL000173, respectively. PTGS was the top 1 target in the network ranked by degree, closeness, and betweenness. The high degree of centrality value in this compound-target network represents the overall importance of these phytochemicals. The betweenness and closeness centralities revealed the relative importance of the compound and gene nodes in the LAD-COPD network. Furthermore, we used MOL000098, MOL000006, MOL000173, MOL000422, and MOL000392 in the molecular docking analysis which demonstrated a strong link between these compounds. Previous reports have revealed the effects of certain compounds on COPD. Quercetin improves pulmonary function and ameliorates emphysema in mice. Dihydroquercetin, a compound structurally like quercetin, inhibits ferroptosis in animal models (Liu, Ma, et al., 2022). Formononetin alleviates COPD by inhibiting the inflammation and apoptosis of bronchial epithelial cells in animals (Li et al., 2024). Luteolin, the main agent in the Bu-Fei formula for COPD treatment, modulates apoptosis (Zhang et al., 2022). Bioinformatics analysis has shown the role of Wogonin in the Sangbaipi decoction for COPD treatment (Wang et al., 2023). Therefore, these highlighted compounds constitute the basis for the COPD improvement effect of the LAD. The luteolin, wogonin, quercetin and formononetin were all flavones. Medical plants are often rich in flavonoids, which are the basis of their pharmacological effects (Costine et al., 2022).
Inflammation is the primary component of COPD pathogenesis (Churg et al, 2008) where, macrophages serve as the first line of defense. Persistent smoke elicits pulmonary macrophages and neutrophils, leading to their infiltration into lung tissue (Shin et al., 2018). Macrophages are activated by smoke to defend themselves against noxious substances (Liu et al., 2018). Neutrophils are increased in airspaces and lung secretions during innate immune responses (Chung & Adcock, 2008). Moreover, T- and B-cell infiltration elicit adaptive immunity, leading to lung tissue necrosis and airway remodeling (Lambers et al., 2009). Therefore, anti-inflammatory activity is the primary action mechanism for most anti-COPD drugs (Li, Han, et al., 2021). Some pro-inflammatory cytokines are created by macrophages and neutrophils during COPD pathogenesis, such as TNF-α, IL-2, and IFN-γ. Here, LAD administration downregulated the increase in pro-inflammatory cytokines.
Previously, pathological mechanisms of airway inflammation and oxidative stress have been commonly found in COPD. A characteristic of late-stage COPD is the coexistence of airway inflammation and high levels of oxidation, aggravating airway injury. In addition, GSH is a crucial oxidative stress marker in patients with COPD. It clears free radicals from the human body and downregulates oxidative stress during cellular responses. Regulation of the oxidative stress capacity is a mechanism by which COPD is inhibited. Here, the LAD attenuated the increase in ROS and the reduction in GSH levels.
Iron plays crucial roles in the ocean of BP including oxygen transport, DNA synthesis, nutrition transmission, immune function, cell respiration, and electron transfer (Abbaspour et al., 2014). Iron in the human body exists as Fe2+ and Fe3+. Excessive iron elicits Fe2+-dependent lipid peroxidation in the cell membranes, the main ferroptosis characteristic (Cao & Dixon, 2016). An imbalance in tissue oxidative stress leads to increased ferroptosis, and excessive ROS exacerbates ferroptosis via Fenton and Haber–Weiss reactions (Valko et al., 2016). Cigarette smoke and PM2.5 result in lipid peroxidation and ferroptosis in COPD pathogenesis (Meng et al., 2022), and ferroptosis—elicited by abnormal amino acid metabolism—further links to abnormal GSH metabolism. Here, PTGS2 (COX2) was ranked eighth among the PPI network hub genes, and increased PTGS2 expression is tightly linked to ferroptosis in the brains of mice with intracerebral hemorrhage (Li et al., 2017), ulcerative colitis (Cui et al., 2021), and sepsis (Frazier et al., 2012). Here, PTGS2 levels were downregulated after the LAD administration.
The HIF-1α-related oxidative stress signaling pathway is involved in COPD and ferroptosis. HIF-1α is highly expressed in COPD pathogenesis due to hypoxia in blocked airways and resultant lack of air (Raguso et al., 2004). In addition, HIF-1α drives the SLC7A11/GPx4 axis and furthers ferroptosis in liver injury (Liu et al., 2023). The HIF-1/iron regulatory protein 1 axis drives ferroptosis in lung cancer (Cai et al., 2023), while the HIF-1/HEME oxygenase (HO-1) signaling pathway rescues ferroptosis in mice (Feng et al., 2021; Wu et al., 2022). Here, LAD administration downregulated the HIF-1/HO-1 signaling pathway, thereby inhibiting ferroptosis in COPD.
Although many compounds can be predicted using network pharmacology methods, the gold standard for the quantification and standardized analysis of compounds in decoctions is HPLC, especially the MS-HPLC method, which provides a complete physicochemical profile. This study focused only on the compounds and their mechanisms predicted by network pharmacology, a primary limitation. Furthermore, there are many compounds in the LAD, and other pathways may also be underlying mechanisms. Our future research will focus on the study of other pathways while searching for the main effective LAD agents for COPD treatment to bridge the gap between basic research and clinical application.
Conclusion
This study investigated the key targets and possible signaling pathways that exert the LAD effects on COPD and performed an in vivo experiment for validation. The LAD exerted anti-inflammatory, antioxidant, and lung tissue damage-alleviating effects. Most importantly, LAD alleviated ferroptosis in mice with COPD.
Abbreviations
BP: Biological processes; CC: Cellular components; COPD: Chronic obstructive pulmonary disease; GO: Gene ontology; GSH: Glutathione; HIF-1: Hypoxia-inducible factor-1; HO-1: HEME oxygenase-1; IL-2: Interleukin-2; INF-γ: Interferon-γ; KEGG: Kyoto Encyclopedia of Genes and Genomes; LAD: Lijing-Anfei decoction; MF: Molecular function; MLI: Mean linear intercept; PPI: Protein-protein interaction; ROS: Reactive oxygen species; TNF-α: Tumor necrosis factor-alpha; TUNEL: TdT-mediated dUTP-biotin nick end labeling.
Author Contributions
Haizhu Xu designed the animal study and supervised it. Jiajia Zhu and Jie Liu completed the data collection and analysis. Wei Jia and Guoqiang Shao help the data collection and visualization. Jiajia Zhu and Jie Liu contributed equally to this work.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethics approval was obtained from the Laboratory Animal Ethics Committee of Yongxinkangtai (YKXT2022L019).
Funding
This work was financially supported by the Shanghai Hongkou District Health Commission Project (Fund No.: HKGYQYXM-2022-11) and Hongkou District Clinical Key Support Specialist (Fund No.: HKZK2020A04).
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
