Abstract
Background
In recent years, the number of patients with myocardial ischaemia is rising year by year in China.
Objectives
The study uses network pharmacology to predict targets and pathways of action of Apocynum venetum L. (AVL) for the treatment of myocardial ischemia (MI).
Materials and Methods
The essential active compounds of AVL were obtained from the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform. The objective of selecting the target genes was achieved through the utilization of GeneCards and Swiss Target Prediction resources. The pool of MI-related targets was obtained from DisGeNET, Online Mendelian Inheritance in Man and the Therapeutic Target Database. The protein-protein interaction network was meticulously constructed using the STRING algorithm. The gene ontology (GO) analysis of AVL-MI targets was performed at Metascape. In addition, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis was conducted. Furthermore, molecular docking analysis was conducted to study the interactions of AVL components with estrogen receptor 1 (ESR1), heat shock protein 90 alpha (HSP90A), protein kinase B (Akt1), tyrosine kinase Src (SRC) and phosphatidylinositol 3-kinase (PI3K). Finally, cellular experiments were performed to validate the mechanism of action of AVL.
Results
The molecular target analysis revealed that AVL has the potential to interact with a total of 147 biomolecules. This comprises ESR1, HSP90A, epidermal growth factor receptor, protein kinase B alpha (Akt1), SRC and PI3K. As indicated by GO and KEGG analyses, AVL’s inhibitory potential against MI may be due to its action on the PI3K-Akt signalling pathway. Furthermore, docking studies conducted to assess the interactions between AVL components and their targets (ESR1, HSP90A, Akt1, SRC and PI3K) confirmed that they have robust binding affinities. Cell experiments also showed that AVL treatment significantly reduced oxidative stress indexes (p < 0.05). Additionally, the expression levels of Akt1 and PI3K proteins were significantly higher in the AVL group compared to the MI group (p < 0.05).
Conclusion
AVL therapy exhibits the potential to alleviate the symptoms of MI by mitigating oxidative stress and by orchestrating the expression of proteins such as PI3K and Akt1.
Introduction
Myocardial ischemia (MI), or heart attack, is a widespread health concern that has become increasingly important as a significant contributor to the global disease burden. Despite the wide recognition of its significant impact, the exact mechanisms leading to its development remain elusive (Ambrose, 2006). Patient survival depends heavily on the timely and effective restoration of blood supply, which remains a major challenge when dealing with MI and its complications (Li et al., 2021). In clinical studies in Xinjiang, Apocynum venetum L. (AVL) has achieved good efficacy in the field of treating cardiovascular diseases (Pratap Singh et al., 2023; Zhang et al., 2022). AVL is a subshrub plant of the Oleaceae family, a variant of Robusta, with both red and white hemp. Its Chinese name is a result of its discovery in the Lop desert, formed by the Luobu Plain, Xinjiang. It is named Luobuma due to its ability to be spun and woven (Xie et al., 2015). The AVL plant, according to contemporary scientific research, is not merely a top-notch fibre producer, but also a plant with inherent medicinal values. Fresh AVL leaves can be subjected to steaming, frying and kneading, followed by consumption as a tea. This herbal brew is effective in cooling and reducing fever, minimizing dizziness and fortifying the heart (Irie et al., 2009). AVL can yield over 100 types of alkaloids, with notable examples being Vinblastine and Vincristine, which are effective treatments for a variety of cancers and acute leukaemia in children. Additionally, AVL’s cardiac glycosides and alkaloids can widen blood vessels, reduce blood pressure, enhance myocardial contraction and improve heart function, serving as beneficial treatments and preventions for hypertension and heart disease (Guo et al., 2020).
Although AVL has been a well-known therapeutic agent for cardiovascular diseases, the mechanism of its effect on MI remains elusive. Using network pharmacology, researchers have been able to generate a comprehensive network of molecules and targets related to AVL and MI. This approach has allowed the scientific exploration of the mechanisms of AVL and has provided new avenues for the development and optimization of AVL therapy for MI.
Materials and Methods
Experimental Reagents and Instruments
10 Sprague-Dawley (SD) rats and H9c2 cells from the Animal Experimental Center of Xinjiang Medical University (SCXK: 20220003); AVL from the Xinjiang Medical University; Taselisib (phosphatidylinositol 3-kinase [PI3K] inhibitors, Taselisib (PI3K inhibitors), Selleck, CAS: GDC 0032); penicillin/streptomycin double antibody (Yeasen, CAS: 60162ES76); DMEM medium (Yeasen, CAS: 41420ES76); foetal bovine serum (Gibco, CAS: 160,00044); trypsin (Yeasen, CAS: 40126ES60); DMSO (source leaf, CAS: R21950); CCK-8 kit (Abbkine, CAS: KTC011001); BCA protein quantification kit (Prilosec, CAS: P1511-1/2/3); PI3K antibody (Abcam, CAS: ab302959); Akt1 Antibody (Abcam, CAS: ab183556); goat anti-rabbit secondary antibody (Abcam, CAS: ab150077); enzyme labeller (Bio-Rad, USA); high-speed centrifuge (Shanghai Anting Scientific Instrument Factory); inverted fluorescence microscope (SungKan Instruments); three-gas incubator (Thermo Fisher, USA).
HPLC Detection of AVL Components
To prepare the AVL solution, meticulously weigh 0.5 g of AVL and transfer it into a glass vial. Add 20 mL of deionized water to the vial, securely cap it, weigh the contents of the vial again, and immerse it in a water bath for reflux extraction for 60 minutes. Once the extraction has concluded, allow the vial to cool, and add an appropriate volume of deionized water to compensate for any weight loss incurred during the extraction process. Finally, filter the solution through a 0.45-micron membrane filter to ensure a pristine product. The analytical technique used in this study involved the use of a Waters XSelect HSS T3 (4.6 × 250 mm, 5 µm) chromatographic column. The mobile phase employed was a mixture of methanol (mobile phase A) and a 0.05% phosphoric acid solution (mobile phase B). In this analysis, the column temperature was maintained at 35°C, and the detection wavelength was set at 265 nm. The gradient elution process was carefully designed and executed, targeting a theoretical plate count of not less than 2,000 for the peak. The sample injection volume was precisely measured at 10 µL.
Access to AVL Active Ingredients
Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform collect and compile data on active ingredients and reference relevant literature to identify, correct and confirm the identities of each active ingredient. These include (Z)-caryophyllene, sitosterol, Procyanidin B1, kaempferol, (+)-catechin, (−)-Caryophyllene oxide, Phytodolor, luteolin, Morkit and ent-Epicatechin.
Get AVL-MI Common Targets
The compounds (Z)-caryophyllene, sitosterol, Procyanidin B1, kaempferol, (+)-catechin, (−)-Caryophyllene oxide, Phytodolor, luteolin, Morkit, ent-Epicatechin can be obtained from the SMILES format file. This Smiles structure can then be input into the Swiss Target Prediction tool to identify potential targets. Through this tool, the potential targets of AVL were identified. The keywords ‘myocardial ischemia’ were used in DisGeNET, Therapeutic Target Database and OMIM to identify potential targets for MI. OMIM was also used to identify potential targets for MI. A Venn diagram was generated using Venny software to screen for AVL-MI common targets.
Obtaining a Protein-protein Interaction (PPI) for AVL-MI
To construct the PPI network model, String software was employed to establish the requisite criteria for the target input, which encompassed ‘Homo sapiens’ and ‘highest confidence (>0.9)’. Subsequently, the MI map of AVL was retrieved utilizing the Cytoscape software.
Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathway Analysis in AVL-MI
We further analysed the targets through Metascape, a tool that identifies and generates these targets, mapping them to specific GO biofunctions and KEGG signalling pathways using Weishengxin.
Molecular Docking Validation
Large-degree structures of estrogen receptor 1 (ESR1), heat shock protein 90 alpha (HSP90A), epidermal growth factor receptor (EGFR), Akt1, SRC and PI3K were first obtained from the RCSB PDB in pdb format, followed by salidroside SDF format acquisition from PubChem. These datasets were subsequently used for molecular docking using DockThor.
Drug-containing Serum of AVL
Ten SD rats were maintained in the rearing room. The rats were fed 0.5 g/kg AVL twice daily for a period of five days. One hour after the last administration, the rats were anaesthetized intraperitoneally with pentobarbital. Blood samples were collected from the abdominal aorta and centrifuged at 4,000 r/min for 15 minutes at 4°C. The serum was isolated and filtered through a 0.22 µm filter, subsequently heat-inactivated at 56°C for 30 minutes, and stored at −80°C until required.
Cell Grouping and Treatment
The following experimental groups were established: Con group: Culturing the cells in DMEM supplemented with 10% foetal bovine serum and 1% penicillin/streptomycin under conditions of a humidified incubator at 37°C with 5% CO2, MI group: Culturing the cells in a sugar-free medium under conditions of an anaerobic environment at 37°C (1% O2, 5% CO2 and 94% N2) for 24 hours, AVL-H group: Culturing the cells in a sugar-free medium supplemented with 20% AVL Drug-containing serum under conditions of an anaerobic environment at 37°C for 24 hours, AVL-L group: Culturing the cells in a sugar-free medium supplemented with 15% AVL Drug-containing serum under conditions of an anaerobic environment at 37°C for 24 hours, Taselisib (PI3K inhibitors) (TAB) group: Incubating the cells in a sugar-free medium supplemented with 100 nM TAB under conditions of an anaerobic environment at 37°C for 24 hours. The administration time and concentration of the AVL group were set by referring to the related literature (Zhang et al., 2022).
Effect of AVL Concentration on the Viability of Hypoxia Injured H9c2 Cells
Ninety-six-well plates were prepared with three replicate wells per group. A low-sugar medium supplemented with AVL was then added and the wells were incubated for a total of 12 hours. Next, H9c2 cells were transferred to a triple-air incubator, where they were exposed to hypoxic conditions for 24 hours. Subsequently, the CCK8 reagent was added to the wells, and the cells were further incubated for 40 minutes. The optical density value of each well was subsequently measured using a 450 nm enzyme-based spectrophotometer. Cell activity = (Atreatment − Ablank)/(Acontrol − Ablank) × 100%.
Determination of Malondialdehyde (MDA), Superoxide Dismutase (SOD) and Lactate Dehydrogenase (LDH) in H9c2 Cells Damaged by Hypoxia by AVL
Cells of the H9c2 cardiomyocyte strain in the logarithmic growth phase were selected and their number was adjusted to 104/well, being then inoculated in 96-well plates. The experimental group was subjected to the same treatments, with MDA, SOD and LDH being quantified in the cell culture medium, utilizing kit instructions as a guide.
Western Blot Detection of PI3K, Akt1 Protein Expression
H9c2 cells were subjected to trypsin digestion and enumeration, followed by inoculation with a cell suspension of 2.5 × 105 cells/mL in a six-well plate. The cells were then segregated and incubated for a total of 12 hours. Subsequently, the cells were transferred to a triple-gas incubator for 24 hours in anoxic conditions. Protein was extracted employing the BCA protein quantification kit, electrophoresed for a total duration of 80 minutes, and the membrane was transferred for a further 60 minutes. The membranes were sealed for 2 hours on a shaking platform, and incubated overnight in a refrigerator at 4°C in a 1:1,000 dilution of PI3K, Akt1 and GAPDH antibodies. The membrane was then washed for 10 minutes, three times with TBST, and subsequently incubated with a 1:5,000 dilution of goat anti-rabbit IgG at room temperature for 2 hours. The membrane was further washed for 10 minutes, three times, and then exposed to the colourimetric reagent for 5 minutes. The protein band images were captured and quantified using BIO-RAD software, with GAPDH serving as the internal control.
Statistical Treatment
For the data analysis, we utilized the SPSS23.0 statistical software. All results are reported as mean values ± standard deviation and analysed using the t-test. p < 0.05 was considered statistically significant.
Results
Detection of the Main Components of AVL by HPLC
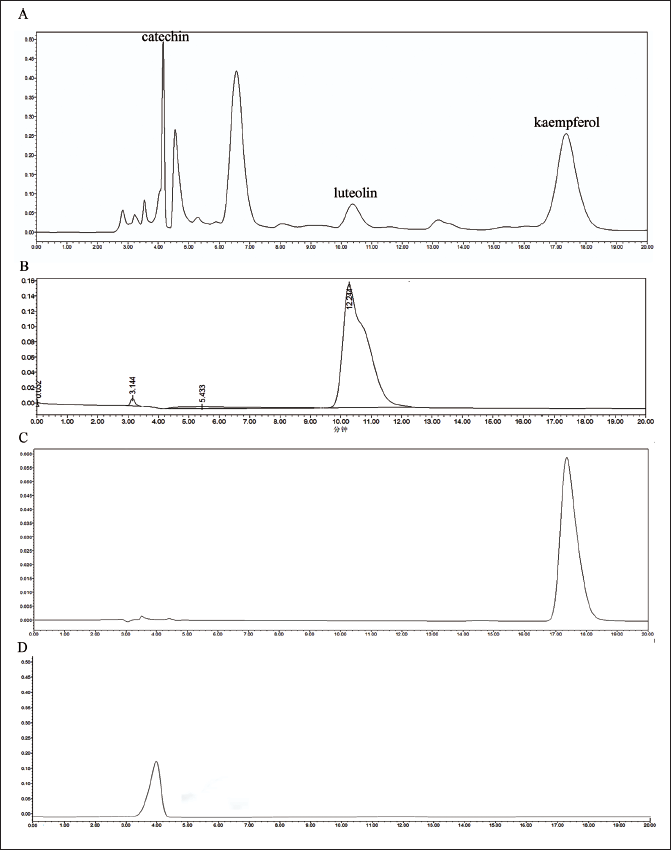
The shape and separation of the peaks of AVL were good and consistent with the standard control, and the detection of AVL could be completed within a retention time of about 20 minutes. The main components of AVL are luteolin, kaempferol and catechin (Figure 1).
Detection of the Main Components of AVL by HPLC. (A) Chromatogram of AVL. (B) Chromatogram of Luteolin. (C) Chromatogram of Kaempferol. (D) Chromatogram of Catechin.
Chemical Components of AVL
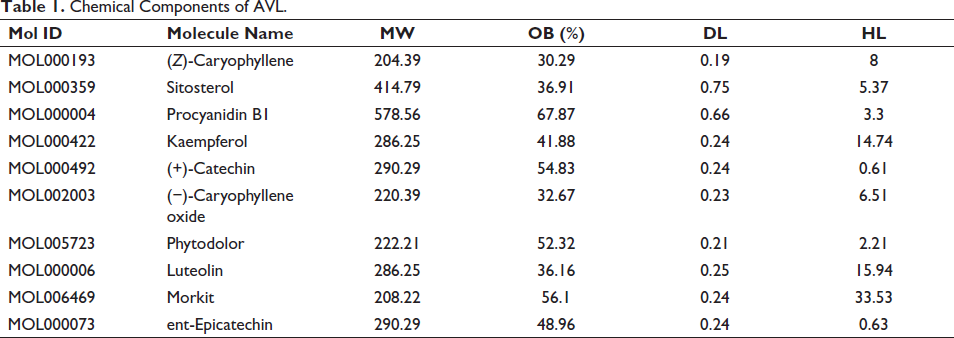
The active ingredients retrieved were subjected to rigorous screening based on Lipinski’s five principles, resulting in the identification of 10 chemical components. These key active ingredients encompass (Z)-caryophyllene, sitosterol, Procyanidin B1, kaempferol, (+)-catechin, (−)-Caryophyllene oxide, Phytodolor, luteolin, Morkit and ent-Epicatechin (Table 1, Figure 2).
Chemical Components of AVL.
Molecular Diagram of the Chemical Constituents of AVL. AVL and Myocardial Ischemia are Common Targets.
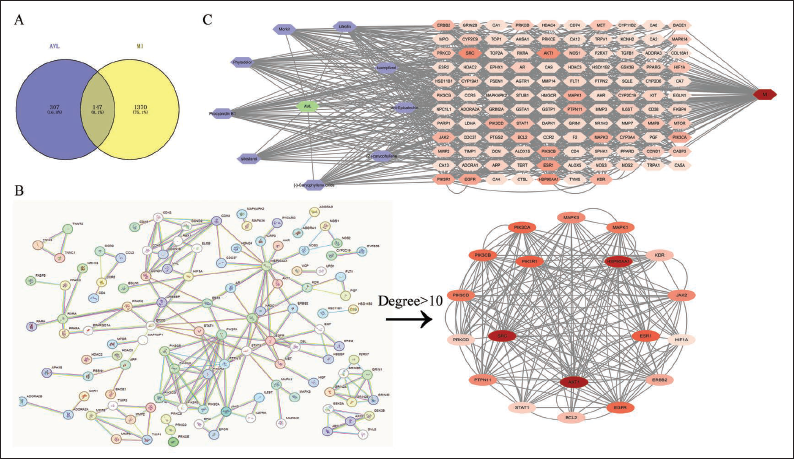
We have collected 454 targets of AVL and 1,517 disease targets related to MI, as well as 147 co-interacting targets of AVL-MI. The targets related to AVL-MI were further analysed and converted to a PPI network. The network contained 147 nodes and 964 edges. From the PPI network, we have identified ESR1, HSP90A, EGFR, Akt1, SRC and PI3K as the targets with the largest connectivity and potential influence on AVL-MI. These genes may act as key targets for the treatment of MI (Figure 3).
(A) Venny of AVL-myocardial Ischemia. (B) AVL-myocardial Ischemia PPI Network. (C) AVL-Components-targets-myocardial Ischemia.
GO Functional Enrichment Analysis and KEGG Signalling Pathway Analysis
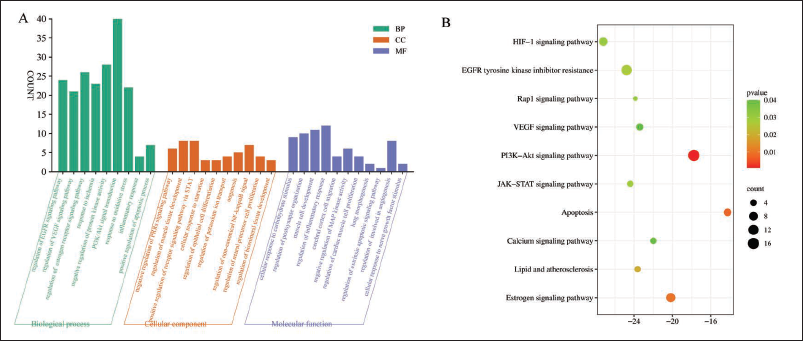
As a result of our research, a total of 1,025 GO function enrichment analysis entries were identified in the Metascape platform, including 845 BP (biological process), 101 CC (cellular component) and 79 MF (molecular function) data. Among these, the PI3K-Akt signalling pathway, exhibiting the highest number of targets and robust correlation, was selected for further analysis using GO. Additionally, 29 KEGG signalling pathways, which demonstrated a strong correlation with MI, were identified, with AVL likely directly regulating the PI3K-Akt and Apoptosis signalling pathways (Figure 4).
(A) AVL-myocardial Ischemia GO Biological Function. (B) AVL-myocardial Ischemia KEGG Pathway Analysis.
Molecular Docking Results
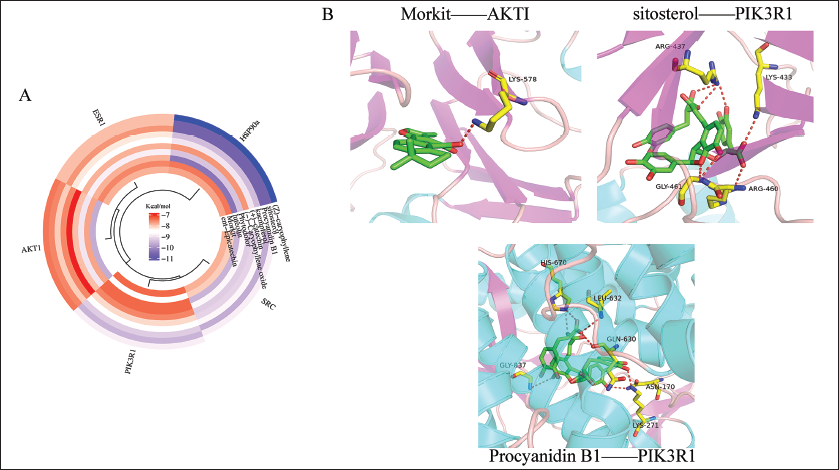
Molecular docking results showed that (Z)-caryophyllene, sitosterol, Procyanidin B1, kaempferol, (+)-catechin, (−)-Caryophyllene oxide, Phytodolor, luteolin, Morkit, ent-Epicatechin and ESR1, HSP90A, Akt1, SRC and PI3K binding energies were all less than −7 kcal/mol, suggesting that the components of AVL bind tightly to key targets (Figure 5).
(A) Chemical Composition and Target Docking Heat Map. (B) Docking Diagram of the Part of the Docking Site with Tight Binding Energy.
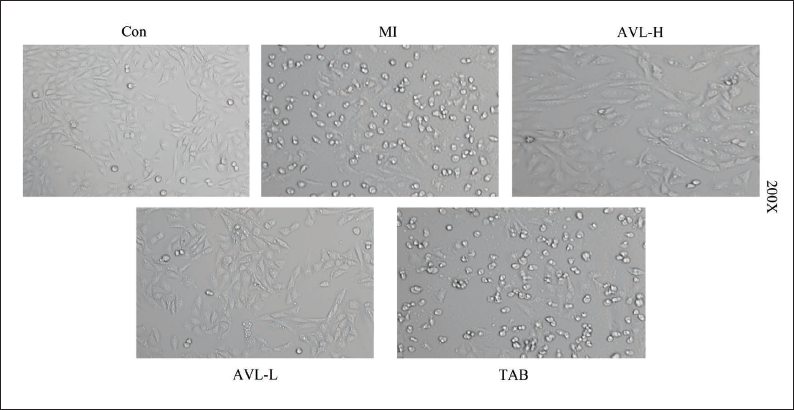
Effect of AVL on the Morphology of the H9c2 Cell Model
Microscopic examination of the H9c2 cells in the Con group revealed that they exhibited clustered growth patterns, adhering firmly to the wall. The majority of these cells exhibited spindle-shaped morphologies with distinct edges and robust refractive index. In contrast, cells in the I/R and TAB groups exhibited a large number of rounded morphologies, accompanied by floating cell death, compromised refractive index and poor adherence to the wall. The AVL group was found to significantly reduce the occurrence of cell death, indicating a positive therapeutic effect (Figure 6).
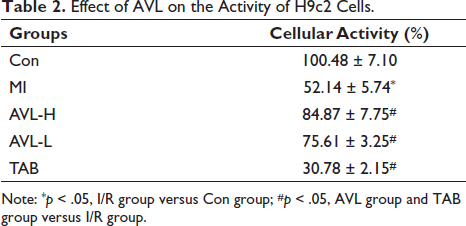
Effect of AVL on the Activity of H9c2 Cells
Compared to the control group, the MI group exhibited a significantly decreased cell activity, with a statistically significant difference (p < 0.05). Contrarily, the AVL group exhibited a significantly higher cell activity compared to the MI group, while the TAB group exhibited a significantly lower cell activity (p < 0.05) (Table 2).
Effect of AVL on the Activity of H9c2 Cells.
Effect of AVL on Biochemical Indices of Model Cells
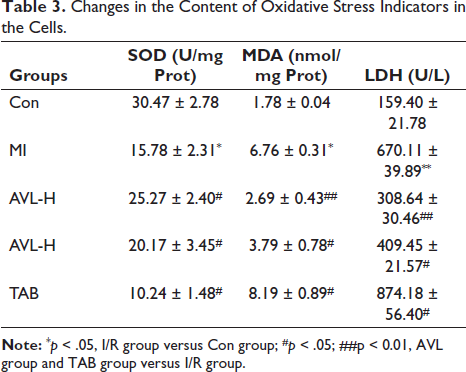
The levels of MDA, LDH in the MI group were significantly elevated when compared with those in the control group (p < 0.05). Conversely the levels of SOD in the MI group were lower when compared with those in the control group (p < 0.05). Similarly, the AVL group demonstrated a significant reduction in MDA, LDH levels and an increase in SOD levels compared to the MI group (p < 0.05). Similarly, the TAB group demonstrated an increase in MDA, LDH levels and a decrease in SOD levels compared to the MI group (p < 0.05) (Table 3).
Changes in the Content of Oxidative Stress Indicators in the Cells.
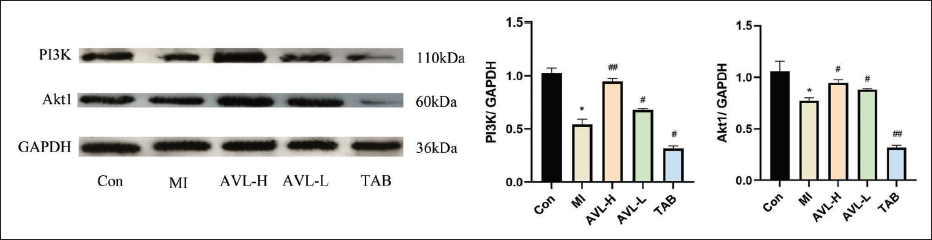
Effect of AVL on H9c2 Cells on PI3K, Akt1 Expression
The findings indicated that the expression of PI3K and Akt1 proteins was significantly lower in the MI group in comparison to the Con group (p < 0.05). Conversely when compared to the MI group, the expression of PI3K and Akt1 proteins was significantly elevated in the AVL group, and was noticeably decreased in the TAB group (p < 0.05) (Figure 7).
Discussion
Literature has recently reported that AVL has significant efficacy in treating a variety of conditions, including hypertension, chronic congestive heart failure and hyperlipidaemia. In addition, it is believed to possess several healthcare functions, including anti-allergy, anti-cancer, anti-radiation and delayed ageing (Chang et al., 2020). AVL demonstrated a significant reduction in serum total cholesterol and triglyceride levels in rats with hyperlipidaemia induced by Triton. AVL also normalized electrocardiographic changes in atherosclerotic rabbits, but the constrictive effect on coronary arteries was particularly noticeable.
H9c2 cell model by establishing a hypoxia-reoxygenation-induced MI experimental, which is technically reliable, simple and reproducible. Myocardial ischaemia Biomarkers of haemorrhage LDH are most commonly used to detect myocardial injury (Liao et al., 2023). SOD specifically targets superoxide radicals, functioning as a protective mechanism against superoxide radical damage and oxidative stress by catalysing the reduction reaction of superoxide radicals and converting them into oxygen molecules and hydrogen peroxide (Lin et al., 2023). MDA, on the other hand, is a lipid peroxide, which is usually considered one of the hallmarks of the free radical-induced lipid oxidation process (Hu, et al., 2023). In this study, we found that the model group exhibited significant increases in LDH and MDA content and a significant decrease in SOD content, indicating the effects of hypoxia on cardiomyocytes. After administration of AVL, there was a significant increase in the levels of LDH, TNF and MDA, while the level of SOD was significantly decreased. However, after AVL administration, the levels of LDH and MDA significantly decreased, while the level of SOD significantly increased, indicating that AVL could reduce the degree of cellular hypoxia damage by regulating the content of oxidative stress.
The PI3K-Akt signalling pathway is a crucial intracellular mechanism that responds to external signals to enhance the cell’s metabolic activity, stimulate cell proliferation and survival, encourage cell growth and promote angiogenesis (Ghafouri-Fard et al., 2022). PI3K phosphorylates phosphatidylinositol to produce PIP3 (Qin et al., 2021). Once activated, Akt can be translocated from the plasma membrane to the cytoplasm and nucleus, where many of its substrates reside (Liu et al., 2023). By phosphorylating Akt, thereby regulating many types of downstream pathways (Liu et al., 2023). The downstream pathways of the PI3K-Akt pathway include mTOR, VEGF, MAPK, glycolysis/gluconeogenesis, FoxO, cell cycle, Apoptosis, NFκB, P53 and many others, reflecting its significant role in cell function and regulation (Li et al., 2023). Through the application of network pharmacology, several potential targets were identified for the treatment of MI with Avil: IL-6, Akt1, PI3K and HIF-1. It was found that in the TAB group, by increasing the inhibition of the PI3K/akt1 signalling pathway, cell activity was significantly reduced. This result was consistent with the predicted primary targets. When comparing the AVL group to the MI group, it was observed that the expression of PI3K and akt1 was elevated in the AVL group, whereas it was decreased in the TAB group. These findings suggest that Avil can modulate the PI3K/akt1 signalling pathway to alleviate hypoxic damage.
The results of this study demonstrate that AVL can effectively suppress MI by reducing oxidative stress and regulating the PI3K/Akt1 signalling pathway. Furthermore, AVL has the potential to serve as a therapeutic target, offering a theoretical basis for its use in the treatment of MI.
Abbreviations
Akt1: Protein kinase B alpha; AVL: Apocynum venetum L.;
Footnotes
Acknowledgments
The authors thank the Xinjiang Medical University for providing the experimental platform.
Authors’ Contributions
Dr Yue YJ participated in performed experiments and contributed to manuscript writing. Dr Wang HM and Dr Li Y also participated in experimental work. Dr Ji Z and Dr Rong X performed the data analysis. Dr Jiang L provided financial support for manuscript writing and gave final approval for publication. All authors have read and concurred with the manuscript for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval and Informed consent
This experiment was approved by the Experimental Animal Ethics Committee of Xinjiang Medical University (Ethics Batch Number: IACUC-20210326-13) and the date (26-3-2021) of this approval. Experimental compliance (The Animal Research: Reporting of in vivo Experiments [ARRIVE] guidelines).
Funding
This research project has been funded by the National Natural Science Foundation of China (Grant No. 8196140154), the Natural Science Foundation of Xinjiang Uygur Autonomous Region (Grants No. 2023D01C139, 2023D01C63) and the State Key Laboratory of Pathogenesis, Prevention and Treatment of High Incidence Diseases in Central Asia (Grant No. SKL-HIDCA-2020-8).
