Abstract
Background
Hyperlipidemia, caused by abnormal lipid metabolism, has become a significant health concern, especially in younger populations. While statins are commonly used for treatment, they often cause severe side effects with long-term use, leading to increased interest in finding natural alternatives.
Objectives
This study aimed to investigate how Guizhi Fuling Decoction (GZFL) treats hyperlipidemia using network pharmacology and experimental validation.
Materials and Methods
Network pharmacology analyzed GZFL’s active components, targets, and treatment mechanisms for hyperlipidemia. Hyperlipidemia rat and high-fat cell models were established. Experimental validation included enzyme-linked immunosorbent assay, Western blot, quantitative polymerase chain reaction, Oil Red O staining, and other methods. Statistical analysis was performed using one-way analysis of variance followed by Tukey’s post hoc test for multiple comparisons. Pearson correlation analysis was used to identify correlations between key protein expressions and lipid levels.
Results
GZFL comprises 85 active components and targets 218 proteins. Hyperlipidemia involves 3,852 targets, with 175 overlapping targets. Key targets included estrogen receptor 1, prostaglandin-endoperoxide synthase 2, interleukin-6 (IL-6), protein kinase B (AKT) 1, tumor necrosis factor, interleukin-1 beta, peroxisome proliferator-activated receptor gamma (PPARG), tumor protein p53 (TP53), and fructo oligosaccharide (FOS). Major active components were quercetin, phytosterols, kaempferol, and baicalein. Enrichment analysis highlighted processes like lipopolysaccharide response and lipid signaling pathways. Animal studies showed GZFL significantly reduced serum total cholesterol, triglycerides, and low-density lipoprotein cholesterol levels, and increased apolipoprotein A1 (p < 0.05). Western blot indicated that GZFL influenced AKT, TP53, IL-6, PPARγ expression, and mRNA levels in the phosphatidylinositol 3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) pathway (p < 0.01). Serum pharmacology experiments demonstrated GZFL-medicated serum reduced lipid droplet formation in high-fat cell models.
Conclusion
GZFL contains multiple pharmacologically active components, such as quercetin, phytosterols, and kaempferol, which can influence lipid metabolism. It may exert its effects by regulating proteins like AKT, TP53, IL-6, and PPARγ, participating in lipid and atherosclerosis signaling pathways and the PI3K/AKT/mTOR pathway.
Introduction
Hyperlipidemia is a disorder caused by abnormal lipid metabolism or transport, defined in modern medicine as a condition where one or more lipids in plasma or serum exceed normal levels in a fasting state. It can lead to complex vascular diseases such as atherosclerosis and myocardial ischemia. The prevalence of hyperlipidemia has been rising in recent years, with an increasing trend in younger age groups, making it a significant health threat (Wiggins et al., 2018). Commonly used statins can cause severe adverse reactions, including hepatotoxicity and nephrotoxicity, with long-term use. Given the limitations of current treatments, there is a growing focus on finding supplementary or alternative natural medicines for hyperlipidemia. Traditional Chinese medicine (TCM) emphasizes holistic regulation and syndrome differentiation and treatment. Chinese herbal medicines are known for their definitive efficacy and minimal side effects, and many studies have shown that various Chinese medications can lower blood lipid levels, with some increasing serum high-density lipoprotein levels (Li et al., 2022).
The combination of network pharmacology and experimental research has yielded significant insights into the mechanisms of action of Chinese medicines, such as Chenpi, Hawthorn, Pine Pollen, and Pueraria, all of which have been shown to regulate lipid metabolism pathways and exert lipid-lowering effects (Fan et al., 2021).
Guizhi Fuling Pills originate from “Jingui Yaolue” and are widely used in clinical TCM practice to promote blood circulation, remove blood stasis, and resolve masses. Guizhi and Fuling are the principal herbs in this formula. Guizhi, commonly used in TCM for febrile diseases, can warm the meridians, promote blood circulation, and aid yang qi transformation. Modern research indicates that Guizhi has anti-inflammatory effects and can reduce serum levels of inflammatory cytokines, with its primary components being volatile oils (Ling et al., 2023; Wang et al., 2023). Recent research has also shown promising results for Guizhi Fuling Pills in managing cardiovascular and metabolic disorders. A 2021 study found that Guizhi Fuling Pills could significantly reduce blood lipid levels and improve endothelial function in hyperlipidemic rats by regulating the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling pathway. Another study in 2022 demonstrated that Guizhi Fuling Pills exert anti-inflammatory and lipid-lowering effects by modulating the expression of critical enzymes involved in lipid metabolism, such as HMG-CoA reductase and lipoprotein lipase. Guizhi is often combined with other Chinese herbs, and numerous studies have demonstrated its ability to regulate blood lipids, enhance antioxidant capacity, and exert lipid-lowering and antiatherosclerotic effects. For instance, Wang et al. (2020) found that Guizhi Decoction can lower serum total cholesterol (TC), triglycerides (TG), and low-density lipoprotein cholesterol (LDL-C) levels while raising high-density lipoprotein cholesterol (HDL-C) levels in hyperlipidemia rat models (Wang et al., 2020). Cheng et al. (2020) discovered that Guizhi Decoction protects vascular endothelium by lowering blood lipids and improving endothelial function through the downregulation of ET and Ang. Afify et al. (2020) found that Guizhi extract exerts antiatherosclerotic effects by regulating the Janus kinase1-signal transducer and activator of transcription 3 (JAK1/STAT3) signaling pathway to reduce levels of inflammatory cytokines.
With the advancement of TCM research, Guizhi Fuling Pills have demonstrated certain advantages in treating various diseases, including adjunctive therapy for coronary heart disease and hypertension (Teng et al., 2022). However, the active components and mechanisms by which Guizhi Fuling Pills affect cardiovascular diseases remain inadequately understood. This study aims to explore the mechanisms of GZFL in regulating blood lipids and treating hyperlipidemia through network pharmacology and experimental methods, providing new insights for the adjunctive treatment of hyperlipidemia.
Materials and Methods
Materials
Experimental Animals
Seventy-three 6-week-old specific pathogen free (SPF)-grade male Sprague-Dawley (SD) rats, each weighing 180 ± 20 g, were obtained from the Zhejiang Chinese Medical University Animal Experiment Center (License No. SYXK [Zhe] 2021-0012). This study received approval from the Experimental Animal Management and Ethics Committee of Zhejiang Chinese Medical University (Approval No. IACUC-20210816-03).
Guizhi Fuling Decoction (GZFL)
GZFL herbal granules (Guizhi 15 g, Fuling 15 g, Chishao 15 g, Danpi 15 g, Taoren 15 g) were purchased from Guangdong Yifang Pharmaceutical Co., Ltd. The granules were dissolved in purified water to prepare an aqueous solution equivalent to a crude drug concentration of 0.23 g mL–1, sterilized, and stored at 4°C. Atorvastatin calcium tablets were obtained from Pfizer Inc.
Reagents and Instruments
High-fat feed and standard rat feed were purchased from Spefo (Suzhou) Biotechnology Co., Ltd. Blood lipid and apolipoprotein assay kits were obtained from Shanghai Yaji Biotechnology Co., Ltd. DMEM high-glucose medium was purchased from Hangzhou Keyi Biotechnology Co., Ltd. Oil Red O staining kits were from Shanghai Beyotime Biotechnology Co., Ltd. Antibodies were obtained from Hangzhou Huaan Biotechnology Co., Ltd. polymerase chain reaction (PCR) kits were purchased from Jiangsu Kangwei Century Biotechnology Co., Ltd. Primers were synthesized by Sangon Biotech (Shanghai) Co., Ltd. HepG2 cells were preserved in our laboratory and purchased from the Chinese Academy of Sciences Cell Bank. Trypsin was purchased from Zhejiang Geno Biomedical Technology Co. Ltd. Fetal bovine serum was obtained from Hangzhou Sijiqing Biomedical Materials Research Institute. Instruments included a low-temperature, high-speed centrifuge (Eppendorf, Germany), SZX7 optical microscope (Olympus, Japan), and a multifunctional microplate reader (Molecular Devices, Japan, SpectraMax M3).
Databases and Methods
Traditional Chinese Medicine Database and Analysis Platform (TCMSP,
Active Components and Targets of Compound
Using the TCMSP platform, we identified all active components of Guizhi, Fuling, Danpi, Shaoyao, and Taoren. Components with oral bioavailability (OB) ≥30% and drug-likeness (DL) ≥0.18 were chosen as critical active ingredients and targets. These targets were calibrated with UniProt data, removing nonhuman genes and eliminating duplicates to obtain standardized gene names.
Hyperlipidemia-related Target Prediction
Keyword “hyperlipidemia” was searched in GeneCards, OMIM, and CTD databases to obtain disease-related targets. All targets were integrated and calibrated using the UniProt database to obtain disease-target gene information.
Drug-Component-Disease-Target Prediction
The targets of the drug components and disease were mapped, and a Venn diagram was generated to identify intersecting genes. Cytoscape 3.7.0 software was utilized to construct a “Drug-Component-Disease-Action Target” network and conduct topological analysis.
Protein–Protein Interaction (PPI) Network Construction
The intersecting target genes were imported into the String database for PPI predictions and then visualized using Cytoscape software to create the PPI network.
Gene Ontology (GO) Enrichment Analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathway Analysis
Core genes were analyzed using R software packages for GO enrichment, annotating target proteins’ roles in biological processes (BP), cellular components (CC), and molecular functions (MF). KEGG pathway enrichment analysis was performed to investigate the biological functions of intersecting genes. Pathways with corrected p < 0.05 were visualized and illustrated with bubble plots, predicting GZFL’s mechanisms in treating hyperlipidemia.
In vivo Experimental Verification
Model Construction and Administration
After one week of adaptive feeding, 53 rats were selected, with 8 as the control group and the rest fed a high-fat diet. After 4 weeks, fasting blood samples were taken, and rats meeting hyperlipidemia model criteria (significantly higher TC, TG, and LDL-C levels and substantially lower HDL-C levels compared to the control group, p < 0.05) were selected and randomly divided into models, positive control (atorvastatin calcium), and treatment groups (high, medium, and low doses of GZFL). Administration was conducted via gavage for 2 weeks. Dosages were calculated based on equivalent doses between rats and humans (70 kg adult human, rat dose is 6.3 times human dose), with high, medium, and low doses being 2.268, 1.512, and 0.756 g kg–1, respectively. The positive control group received 0.06 g kg–1 atorvastatin calcium tablets. The model and control groups received saline.
Indicator Monitoring
Blood samples were collected, and serum was separated. Serum lipid and apolipoprotein levels were detected using an enzyme-labeled method, and liver wet weight was measured.
Western Blot Analysis of Protein Expression
Rat serum was filtered and sterilized, inactivated at 56°C, and total protein concentration was measured. Sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) electrophoresis was performed, followed by blocking with 1% bovine serum albumin (BSA) in tris-buffered saline Tween (TBST), incubation with primary antibodies overnight at 4°C at the following concentrations: protein kinase B (AKT) 1 (1:1,000); interleukin-6 (IL-6) (1:500); peroxisome proliferator-activated receptor gamma (PPARγ) (1:1,000); tumor protein p53 (TP53) (1:1,000). After washing, the membranes were incubated with secondary antibodies at a concentration of 1:2,000 for 1 hour at room temperature. Membranes were developed and detected using a protein gel imaging system, with experiments independently repeated three times.
PCR Detection of Phosphatidylinositol 3 (PI3), AKT, and Mammalian Target of Rapamycin (mTOR) mRNA Levels in Rat Liver
Liver tissues were collected from each group, lysed, and total RNA extracted. RNA concentration was measured, and total RNA was reverse-transcribed to complementary deoxyribose nucleic acid (cDNA) using a kit. Fluorescent quantitative PCR was performed with β-actin as the internal reference, using the 2–∇∇Ct method for calculation. Primer sequences were: β-actin forward: ATCATGTTTGAGACCTTCAACA, reverse: CATCTCTTGCTCGAAGTCCA; phosphatidylinositol 3-kinase (PI3K) forward: GCTCTCCTGGTCATGTCTCAGC, reverse: TGTAGCCCTCCCTAACACTTTC; AKT forward: TTTACTTCCAATGCTAGCTGTGA, reverse: TTTACTTCCAATGCTAGCTGTGA; mTOR forward: GATGTAAGAGGTCGGGTTTTCC, reverse: ACTGGGCAGGGGTCACG. The specific conditions of the PCR cycles were as follows: initial denaturation at 95°C for 5 minutes, followed by 35 cycles of denaturation at 95°C for 30 seconds, annealing at 60°C for 30 seconds, and extension at 72°C for 45 seconds, with a final extension at 72°C for 5 minutes. The relative expression levels of the target genes were normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and calculated using the 2–∆∆Ct method.
Effect of Drug-containing Serum on HepG2 High-fat Cell Model
HepG2 cell model and control groups were established. The model group was cultured with 1 mmol/L free fatty acid (FFA) (a mixture of oleic acid and palmitic acid in a 2:1 ratio) for 24 hours, while the control group was cultured normally. Following the drug-containing serum experiment protocol (Xiaobing et al., 2020), rats were divided into high, medium, and low dose groups (10 times, 5 times, and normal dose) and positive control group (10 nm atorvastatin calcium), with 5 rats per group. Serum was collected 2 hours post-gavage, centrifuged at 3,000 rpm, and filtered through a 0.22 µm membrane. When cell density reached 70%–80%; high-fat cells were randomly divided into high, medium, low dose, positive control, and model groups, with each group having three replicates. Drug-containing serum at concentrations of 2%, 5%, and 10% was added to the culture medium in the high-, medium-, and low-dose groups, respectively. The model and control groups received saline. Oil Red O staining was performed 24 hours later.
Statistical Analysis
Statistical analyses were performed using Statistic Package for Social Science (SPSS) 26 (IBM, Armonk, NY, USA) and GraphPad Prism 10.2.3 software (La Jolla, CA, USA). Data are presented as mean ± standard deviation (x̅ ± s). One-way analysis of variance (ANOVA) was applied for comparisons among multiple groups, with a significance level set at p < 0.05. Post hoc comparisons were conducted using Tukey’s test to determine specific group differences.
Results
Network Pharmacology Analysis
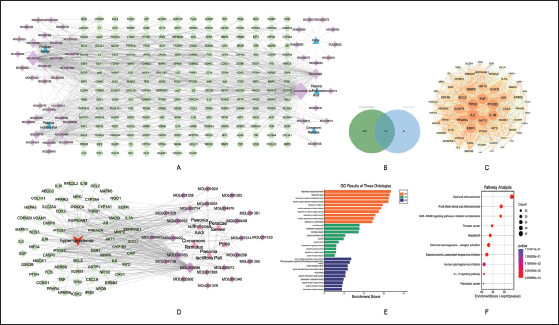
A total of 85 active ingredients were collected from the TCMSP database, including 7 from Cinnamomum cassia, 15 from Poria cocos, 11 from Moutan Cortex, 29 from Paeonia lactiflora, and 23 from Prunus persica, corresponding to 218 targets. The drug-ingredient-target network is shown in Figure 1A. From the GeneCards, OMIM, and CTD databases, 2,106, 292, and 4,471 hyperlipidemia-related targets were obtained, respectively, and after removing duplicates, 3,852 targets were identified and corrected using the UniProt database. The intersection of drug active ingredient and hyperlipidemia targets resulted in 175 intersecting genes (Figure 1B), serving as potential pharmacological targets. A PPI network was constructed with 175 nodes and 3695 edges using the STRING database (version 11.0) and visualized with Cytoscape (version 3.8.2), and topological analysis (degree > 103.67, betweenness > 0.0023, and closeness > 0.73) identified 64 key targets (Figure 1C). The visual regulatory network “Drug-Ingredient Targets-Disease-Disease Targets” (Figure 1D) predicted the main active ingredients, including quercetin, beta-sitosterol, kaempferol, and baicalein, with critical targets such as estrogen receptor 1 (ESR1), prostaglandin-endoperoxide synthase 2 (PTGS2), interleukin-6 (IL-6), AKT1, tumor necrosis factor (TNF), interleukin-1 beta (IL-1B), peroxisome proliferator-activated receptor gamma (PPARG), tumor protein p53 (TP53), fructo oligosaccharide (FOS), and jun proto-oncogene (JUN). GO analysis revealed that critical genes are involved in BP like response to lipopolysaccharide and reactive oxygen species metabolism, as well as MF related to membrane rafts and plasma membrane rafts (Figure 1E). KEGG pathway analysis identified 10 pathways (p < 0.01) related to the disease, highlighting the biological functions and signal pathways of intersecting genes, with main enriched pathways including lipid and atherosclerosis and fluid shear stress (Figure 1F). The lipid and atherosclerosis pathways contained the most core targets (Figure 2).
Results of Network Pharmacological Analysis. (A) Network of Drug-active Ingredients-targets. Each Green Node Represents an Active Ingredient from Guizhi Fuling Decoction (GZFL), and Each Purple Node Represents a Target Protein; (B) Venn Diagram Showing Intersecting Targets between Drug Components and Disease; (C) Protein–Protein Interaction (PPI) Network of Intersecting and Core Targets; (D) “Drug-Active Ingredients-Disease-Targets” Network. Note: Blue Triangles Indicate Traditional Chinese Medicines, Purple Diamonds Represent Active Ingredients, and Green Circles Denote Related Targets; (E) Gene Ontology (GO) Enrichment Analysis; (F) Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathway Enrichment Analysis. The Size of Each Dot Represents the Number of Genes Involved in the Pathway, and the Color Indicates the Level of Significance (p Value).
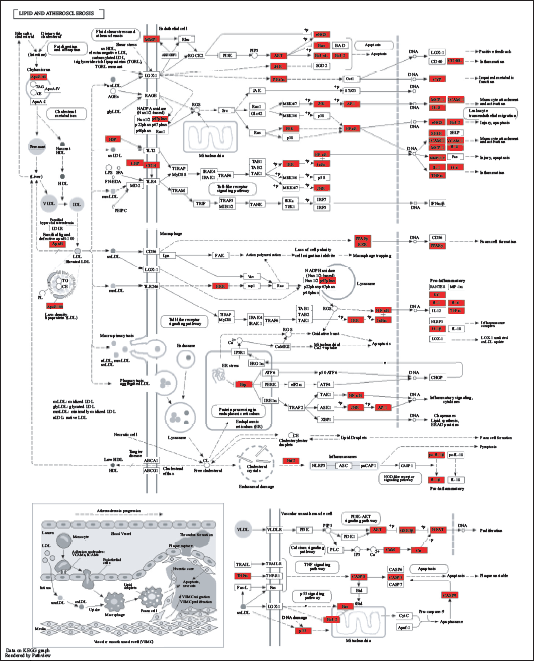
Lipid and Atherosclerosis Signaling Pathways. The Detailed Pathway Diagram Shows the Involvement of Core Targets in Lipid and Atherosclerosis Signaling Pathways. The Red Nodes Represent the Key Proteins Involved, and the Edges Show the Interactions and Regulatory Relationships between Them.
In vivo Experimental Validation
Analysis of serum indices and liver weight in rats revealed that the model group exhibited significantly higher levels of TC, TG, LDL, and liver weight than the blank group (p < 0.01). There were no significant differences in TC levels between the blank and positive control groups and the low-, medium-, and high-dose GZFL groups. However, the liver weight in the high-dose GZFL group was significantly lower (p < 0.01) than in the model group and comparable to the blank group. Both the medium- and high-dose GZFL groups significantly reduced serum TC, TG, and LDL levels (p < 0.01) and markedly increased apolipoprotein A1 (APOA1) levels (p < 0.01) without substantially affecting apolipoprotein B (APOB) and HDL (Figure 3a).
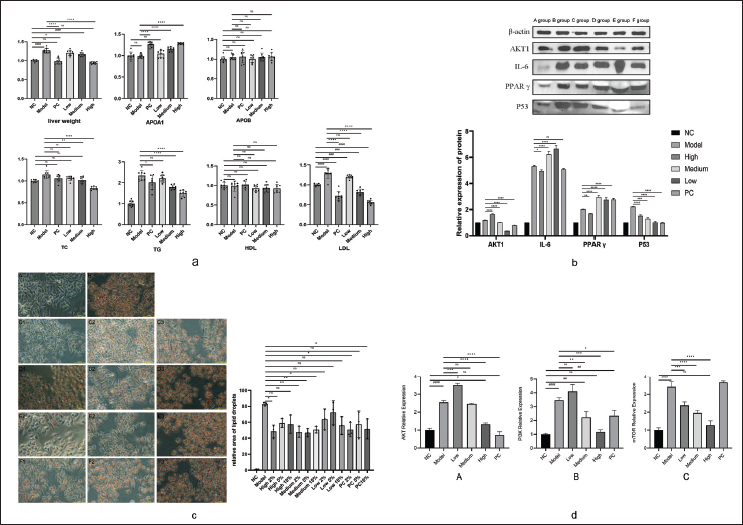
Protein expression analysis in rat serum indicated that the model group exhibited significantly higher levels of AKT1, IL-6, PPARγ, and TP53 than the blank group (p < 0.05, p < 0.01). In contrast, the high-dose GZFL group showed significantly increased AKT1 levels (p < 0.01), reduced IL-6 and PPARγ levels (p < 0.05, p < 0.01), and elevated TP53 levels compared to the model group (p < 0.01) (Figure 3b). mRNA expression levels (Figure 3d) demonstrated that the model group had significantly elevated PI3K, AKT, and mTOR mRNA levels compared to the blank group (p < 0.01). However, compared to the model group, both the medium- and high-dose GZFL groups exhibited significantly reduced PI3K levels, while the high-dose group had substantially lower AKT levels (p < 0.01). Moreover, all GZFL treatment groups showed decreased considerably mTOR levels (p < 0.01).
In vitro Experiments
Compared to the blank group, the model group exhibited a significant increase in lipid droplets in cells (p < 0.01). In contrast to the model group, adding 2% and 10% positive control medicated serum led to a significant reduction in lipid droplets (p < 0.05), while 5% positive control medicated serum showed no significant effect. Addition of 2% high-dose medicated serum also significantly reduced lipid droplets (p < 0.05), whereas 5% and 10% high-dose medicated serum did not produce significant effects. Moreover, 2% and 5% medium-dose medicated serum significantly reduced lipid droplets (p < 0.01), and 10% medium-dose medicated serum showed a significant reduction (p < 0.05). However, 2%, 5%, and 10% low-dose medicated serum did not significantly affect lipid droplets in cells (Figure 3c).
Discussion
In TCM, hyperlipidemia is a condition of blood stasis and phlegm dampness, closely related to imbalances in Qi and blood. Some symptoms also manifest as thickened blood. GZFL (Guizhi Fuling Formula) is known for its properties of warming and activating the blood, promoting Yang, invigorating blood circulation, and resolving masses. Clinically, it is used to treat syndromes such as phlegm obstruction, spleen and kidney Yang deficiency, Qi stagnation, and blood stasis. It has been applied as an adjunctive treatment for hyperlipidemia.
This study used a network pharmacology approach to identify 85 bioactive ingredients and 218 corresponding targets in GZFL, alongside 3,852 hyperlipidemia-related targets. We identified 175 intersecting targets between the drug and the disease. Key active ingredients included quercetin, beta-sitosterol, kaempferol, catechin, and ellagic acid. Quercetin is widely found in various plants and has multiple biological properties (Xiaobing et al., 2020), including tumor inhibition, free radical scavenging, lipid lowering, blood pressure reduction, antioxidation, antibacterial, and immunomodulation functions. It is significant in treating and preventing cancer, cardiovascular diseases, and aging (Wang et al., 2018). Both quercetin and catechin can be used in cancer treatment (Fujiki et al., 2015). Kaempferol has been shown to reduce cancer risk, possess anti-inflammatory properties, and prevent atherosclerosis (Wang et al., 2021). β-Sitosterol can inhibit cholesterol absorption, thereby reducing blood cholesterol levels, and has strong antioxidative, antitumor, and tissue repair properties (Zhang et al., 2024). Studies by Duan et al. (2018) found that paeoniflorin can inhibit SREBP-1c, FAS, and ACCa, suppressing lipid synthesis, downregulating PPARY, HMGCR, and LDLR, and reducing liver lipid synthesis. It also enhances PPARa, ACOX, UCP2, and CPT-1a expression, promoting liver lipid oxidation.
Constructing a PPI network revealed that GZFL’s mechanism for treating hyperlipidemia is mainly associated with 64 target proteins. Core targets include ESR1, PTGS2, IL-6, AKT1, TNF, IL-1B, PPARG, TP53, FOS, JUN, EGFR, CASP3, HIF1A, MYC, and GSK3B. These targets are involved in BP like response to lipopolysaccharides and reactive oxygen species metabolism, with mechanisms possibly related to lipid and atherosclerosis signaling pathways.
Research indicates that GZFL can modulate inflammatory factors and exhibit antitumor properties, showing efficacy in treating uterine fibroids (Wang et al., 2022). It also has vasodilatory and antiplatelet aggregation effects, adjusting blood circulation and reducing blood viscosity. Studies by Zhou et al. (2022) suggest that the anti-inflammatory mechanism of Guizhi Fuling capsules may be related to reduced expression of IL-1β and mPGES-1. Wei et al. (2021) found that certain active ingredients can inhibit the production of inflammatory factors IL-1β, IL-6, and TNF-α, exerting anti-inflammatory effects. IL-6 is a crucial inflammatory cytokine that plays a significant role in lipid metabolism (Delgado-Lista et al., 2011; Hashizume et al., 2010). High expression of IL-6 can promote endothelial and smooth muscle cell degeneration and necrosis, leading to atherosclerosis formation and rupture.
Through establishing a hyperlipidemia rat model, we found that medium and high doses of GZFL could reduce serum levels of TC, TG, and LDL in rats and increase APOA1 levels, improving lipid profiles in hyperlipidemic rats. GZFL also affected the expression of AKT, IL-6, PPARγ, and TP53 proteins, reducing the mRNA levels of PI3, AKT, and mTOR. The serum-containing drug experiments indicated that specific doses of the serum could reduce lipid droplet formation in cells. The PI3K/AKT/mTOR signaling pathway is critical in regulating lipid metabolism (Krycer et al., 2010). mTOR is a necessary pathway for alcohol-regulated ALD lipid metabolism (Chen et al., 2018), involving complex signaling networks that promote lipid uptake and adipogenesis, ultimately reducing circulating triglycerides and FFA levels, thus lowering blood lipid levels. Previous studies have shown that activation of the PI3K/AKT/mTOR pathway is associated with increased lipid synthesis and storage. For instance, Zhuang et al. demonstrated that inhibiting the PI3K/AKT/mTOR pathway could significantly reduce lipid accumulation in hepatocytes, indicating its potential as a target for lipid-lowering therapies. By influencing the PI3K/AKT/mTOR pathway, GZFL may exert its lipid-lowering effects through multiple mechanisms, including reducing lipid synthesis, enhancing lipid oxidation, and modulating inflammatory responses. These findings provide a basis for further investigation into the molecular mechanisms of GZFL and its potential as a complementary therapy for hyperlipidemia.
Conclusion
In summary, this study preliminarily explored the mechanisms of GZFL in hyperlipidemia. Network pharmacology predicted critical active ingredients and pharmacological targets with lipid-lowering effects and potential targets and signaling pathways for treating hyperlipidemia. Animal experiments validated GZFL’s improvement of lipid profiles in rats, affecting the expression of AKT1, IL-6, PPARγ, and TP53 proteins and genes in the PI3K/AKT/mTOR signaling pathway, reducing inflammation associated with hyperlipidemia and lipid accumulation in cells. These findings suggest that GZFL can ameliorate hyperlipidemia through multiple components, targets, and pathways, preliminarily validating the feasibility of using network pharmacology as an auxiliary research tool to predict GZFL’s therapeutic potential. However, clinical application feasibility requires further in-depth research considering individual differences, dosage, and treatment duration.
Abbreviations
GZFL: Guizhi Fuling Decoction; TCM: Traditional Chinese medicine; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; TC: Total cholesterol; TG: Triglycerides; HDL-C: High-density lipoprotein cholesterol; LDL-C: Low-density lipoprotein cholesterol; JAK1/STAT3: Janus kinase1-signal transducer and activator of transcription 3; SPF: Specific Pathogen Free; SD: Sprague-Dawley; OB: Oral bioavailability; DL: Drug-likeness; PPI: Protein–protein interaction; GO: Gene ontology; BP: Biological processes; CC: Cellular components; MF: Molecular functions; SDS-PAGE: Sodium dodecyl sulphate-polyacrylamide gel electrophoresis; BSA: Bovine serum albumin; TBST: Tris-buffered saline Tween; PCR: Polymerase chain reaction; PI3K: Phosphatidylinositol 3-kinase; AKT: Protein kinase B; mTOR: Mammalian target of rapamycin; cDNA: Complementary deoxyribose nucleic acid; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; FFA: Free fatty acid; SPSS: Statistic Package for Social Science; ESR1: Estrogen Receptor 1; PTGS2: Prostaglandin-endoperoxide synthase 2; IL-6: Interleukin-6; TNF: Tumor necrosis factor; IL-1B: Interleukin-1 beta; PPARG: Peroxisome proliferator-activated receptor gamma; TP53: Tumor protein p53; FOS: Fructo oligosaccharide; JUN: Jun proto-oncogene; APOB: Apolipoprotein B.
Footnotes
Acknowledgments
None.
Authors’ Contributions
Mi Zhang and Chunlei Fan designed the study and performed the experiments, Yuntian Zhang collected the data, Hongyun Wang analyzed the data, Mi Zhang and Chunlei Fan prepared the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study received approval from the Experimental Animal Management and Ethics Committee of Zhejiang Chinese Medical University (Approval No. IACUC-20210816-03).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
