Abstract
Objectives
Tianxiangdan capsule (TXD) has been clinically used in the treatment of coronary heart disease angina pectoris. This study aimed to evaluate the mechanisms of TXD against myocardial ischemia and provide evidence for its subsequent clinical application.
Materials and Methods
Active components and mechanisms of action of TXD against myocardial ischemia were predicted and analyzed by network pharmacology and molecular docking. The chemical constituents in TXD were determined by HPLC-MS. The oxidative damage model was established using H2O2, which caused myocardial ischemia damage. The MTT assay and Hoechst 33342 staining were used to evaluate cell viability, while cleaved caspase-3 immunofluorescence staining was used to determine cell apoptosis. The fluorescent probe method detected ROS and intracellular Ca2+, while spectrophotometry was used to measure SOD, MDA, and NO levels in myocardial cells. Western blotting was used to detect the expression levels of ERɑ, PI3K, p-PI3K, AKT, p-AKT, and eNOS in cells.
Results
It was found that TXD plays a protective role in myocardial ischemia through the estrogen pathway. The TXD drug-containing plasma exhibited increased cell survival rates and suppressed MDA levels, elevated SOD and NO levels, and significantly suppressed ROS levels as well as intracellular Ca2+ levels. Moreover, the TXD drug-containing plasma pretreated cells had significantly suppressed PI3K, p-PI3K, AKT, and p-AKT expression levels, as well as elevated ERɑ and eNOS expression levels.
Conclusion
TXD activates estrogen receptor ERɑ, enhances NO release of cardiomyocytes through the estrogen signaling pathway, improves intracellular Ca2+ overload, oxidative stress injury, and apoptosis, and plays a protective role in myocardial ischemia injury.
Introduction
Myocardial ischemia is a pathological state that is characterized by reduced cardiac blood flow, resulting in reduced oxygen supply, abnormal myocardial energy metabolism, and inability to support heart functions (Gibbons, 2021). In China, the prevalence of myocardial ischemia has been on the rise (Li & Kronenberg, 2021). Myocardial ischemia is associated with various etiologies, including decreased aortic blood supply, coronary artery occlusion, blood viscosity changes, and myocardial lesions. Traditional Chinese Medicine (TCM) has excellent therapeutic effects and has become an important treatment modality in the treatment of myocardial ischemia (Dong et al., 2023).
In 2015, TXD received a clinical approval for the clinical approval of Class 6.1 Chinese medicine (No. 2015L03185).TXD is composed of four Chinese medicinal materials: Salvia miltiorrhiza Bge. (Labiatae), Rhodiola rosea L. (Crassulaceae), Dalbergia odorifera T. Chen (Leguminosae), and Ziziphora clinopodioides subsp. bungeana (Juz.) Rech. f. (Labiatae) (Sawuer et al., 2021). Studies have confirmed that tanshinones and phenolic acids in S. miltiorrhiza can dilate coronary arteries and improve microcirculation. Salidroside, tyrosol, and flavonoids in S. miltiorrhiza improve vascular endothelium dysfunction and promote mitochondrial biosynthesis (Li et al., 2018). Essential oils and some flavonoids are the main active components of D. odorifera and Z. clinopodioides (Lin et al., 2022). TXD has been shown to reduce the degree of microthrombosis in rats with coronary microcirculation disorder. Moreover, it has anti-inflammatory properties that inhibit inflammatory responses. Preclinical pharmacodynamic data of TXD show that it can significantly improve coronary flow, increase cardiac output in dogs with myocardial ischemia, and reduce myocardial ischemia (Zhao et al., 2022). These compounds can increase the activity of antioxidant enzymes and inhibit thrombosis (Sawuer et al., 2021). TXD potential active ingredients mediated targeting 25 target proteins and regulated arachidonic acid metabolism, calcium signaling pathways, and so on (Zhang et al., 2021). Studies on the mechanisms of anti-myocardial ischemic action of TXD have been sporadic and superficial, lacking in systematic in-depth evaluations. Its active ingredients and mechanisms of action are not precise, which has greatly limited its development and clinical use. Therefore, it is important to elucidate on the mechanisms of TXD in myocardial ischemia in order to provide a scientific basis for its clinical application.
Network pharmacology elucidates on the interactions between drugs and organisms from the perspectives of system biology and biological network’s balance (Nogales et al., 2022; Zhang et al., 2021). In this study, network pharmacology and molecular docking methods were used to predict the anti-myocardial ischemia mechanisms of TXD as well as its active components.
Materials and Methods
Experimental Reagents and Instruments
Tianxiangdan Capsule was obtained from The Fourth Clinical Medical College of Xinjiang Medical University (20190901). Salvianolic acid B, cryptotanshinone, tanshinone ⅡA, salidroside, kaempferol, hyperoside, naringenin, and salvianic acid A sodium were purchased from the China Institute for Food and Drug Control. Fetal bovine serum and pancreatin were purchased from Gibco, USA. D-Hanks buffer was purchased from Boster, China; collagenase II was purchased from Worthing-BioChem, USA; MTT and DMSO were purchased from MP Biomedicals, USA; DMEM/high glucose was bought from HyClone, USA; hydrogen peroxide was purchased from Tianjin Hongyan Chemical Reagent Factory, China; β-estradiol and DAPI were purchased from Sigma, USA; superoxide dismutase (SOD), malondialdehyde (MDA), and nitric oxide (NO) kit were purchased from Beyotime, China; Hoechst 33342, reactive oxygen species assay kit, Triton X-100, and Fluo-3/AM were purchased from Solarbio, China; anti-ESR1 (Boster), cleaved caspase-3, anti-beta actin, anti-PI3K kinase p85 alpha, anti-PI3K kinase p85 (phospho), anti-AKT, anti-AKT (phospho), anti-eNOS, and anti-rabbit IgG (HRP-linked antibody) were purchased from Cell Signaling Technology; anti-alpha Smooth Muscle Actin and FITC antibodies against Mouse IgG H&L (Alexa Fluor 488) were purchased from Abcam, UK. The inverted microscope and Enzyme standard instrument were purchased from Olympus, Japan; the Western blot full set was purchased from Bio-Rad, USA; flow cytometer was purchased from BD, USA; and laser confocal microscope was purchased from Nikon, Japan.
HPLC-MS Analysis
The plasma samples were dissolved in methanol and a protein precipitate was formed. Then, it was filtered using a 0.22-µm filter, making up the missing weight with methanol.
Separation was performed on a BEH C18 column (100 mm × 2.1 mm) at 30°C with a flow rate of 0.3 mL/min. The mobile phase was a mixture of acetonitrile (A) and 0.1% formic acid water (B). The gradient program of the mobile phase was as follows: 0–1 min, 5%–20% A; 1–3 min, 20%–90% A; and 3–6.1 min, 90%–95% A. The injection volume was 3 µL. The quasi-molecular ions [M−H]− and [M+H]+ were selected as precursor ions and subjected to target-MS/MS analyses. Using MRM scanning mode, the acquisition parameters were as follows: ion source temperature 150°C, electrospray voltage 2.8 kV, solvent removal gas: 800 L/h, taper voltage ESI+: 26 V; ESI–: 5 V; while the mass range was recorded from m/z 100 to 800. The Masslynx V4.1 software was used for quantitative analysis.
Preliminary Prediction of Network Pharmacology
The Tianxiangdan compound data were obtained from the TCMSP database (
Experimental Grouping
Cells were treated in groups when they were in good condition. Control group: only DMEM high glucose culture medium was added; Model: 100 µmol/L H2O2 solution prepared by DMEM high glucose culture medium was added for 2 h; Blank plasma (Bp): 15% blank plasma prepared with DMEM high glucose culture medium was added for 24 h, and then 100 µmol/L H2O2 solution was added for 2 h; Tianxiangdan capsule drug-containing plasma low-concentration, medium-concentration and high-concentration (5% Dp, 10% Dp, and 15% Dp): 5%, 10%, and 15% drug-containing plasma prepared with DMEM high-glucose culture medium was added to treat for 24 h, and then 100 µmol/L H2O2 solution was added to damage for 2 h; E2: 10−8 mol/L E2 prepared by DMEM high glucose culture medium was added for 24 h, and then 100 µmol/L H2O2 solution was added for 2 h damage.
Experimental Animals
Based on the random number table, rats were labeled and divided into four groups (20 in each group). Based on the surface area of human and rat bodies, the rats received 6.3 times the dose of healthy human adults (0.378 g/kg/day). On the evening of the third day, rats were fasted overnight and only provided drinking water. Starting from the morning of the fourth day, rats received a day dosage. Rats in the control group were treated similarly, except that they were given physiological saline. Rats were weighed and anesthetized by intraperitoneal injection of pentobarbital sodium (40 mg−1·kg−1). Surgical anesthetization was performed 1 h after the last drug administration, and blood was collected from the abdominal aorta of rats. The collected drug-containing plasma was centrifuged at 4°C for 3000 rpm for 10 min. Plasma samples were inactivated at 56°C for 30 min, and samples were filtered using a 0.22-µm filter and stored at −80°C for later use.
Cell Viability Assay
Cell viability was determined through the MTT assay. Myocardial cell suspensions (100 µL) were inoculated in a 96-well plate at a density of 1 × 106/well and cultured for 3 days. Cells were treated in groups when they were in good condition. Then, the original medium was discarded and replaced with 100 µL of DMEM/High Glucose solution medium and 10 µL MTT (5 mg/mL) followed by 4 h of culture. A volume of 150 µL DMSO was added to each well. Samples absorbance was measured at 490 nm with an enzyme standard instrument. Cell viability was determined using the following formula: Cell viability (%) = (drug group − blank group)/(normal group − blank group) × 100%.
Hoechst 33342 Ataining
Cardiomyocytes were inoculated in a six-well plate coated with polylysine-coated cover glasses at a density of 1 × 106/well. After treatment, they fixed in 4% paraformaldehyde at room temperature for 20 min. Cells were stained with 10 mg/mL Hoechst 33342 at 37°C for 10 min. Cell apoptotic morphologies were observed under a fluorescence microscope. Normal cells had oval nuclei with homogeneous chromatin. The nuclei of apoptotic cells exhibited a shrunk morphology, their chromatin was aggregated, and apoptotic bodies were formed.
Immunofluorescence Staining of Cleaved Caspase-3 Cells
Cardiomyocytes were seeded in a 35-mm laser confocal dish coated with polylysine at a density of 1 × 106/well. After treatment, fixed in 4% paraformaldehyde at room temperature for 20 min, after which they were permeated with 0.5% Triton X-100 for 15 min. Next, goat serum was added and sealed for 30 min. Cells were incubated with cleaved caspase-3 antibody (1:400) at 4°C overnight. Next, they were incubated with a secondary antibody (FITC) at room temperature for 2 h and then subjected to PI staining for 10 min. The fluorescence intensity of cardiomyocytes was imaged by LSCM. Cells showing green fluorescence represented cleaved caspase-3 expression, red fluorescence represented cardiac myocytes in the field of view, and cell apoptosis was determined using the formula: Cell apoptosis rate = (green fluorescent cell number/red fluorescent cell number) × 100%.
Measurement of Intracellular ROS Levels
Cardiomyocytes were inoculated in a 35-mm laser confocal dish and six-well plate at a density of 1 × 106/well. After treatment, they were washed three times using PBS, incubated with DCF-HA in a 37°C incubator for 30 min, and washed three times using PBS. The fluorescence intensity of DCF-HA was imaged by LSCM. Cells in a six-well plate were digested with trypsin, and the mean fluorescence density of DCF was measured by flow cytometry.
Determination of SOD, MDA, Ca2+, and NO Levels in Myocardial Cells
Cardiomyocytes were inoculated in a six-well plate at a density of 1 × 106/well. After treatment, they were collected and washed three times using 4°C PBS. Then, cells were lysed using the cell lysis buffer and centrifuged at 4°C. Cell supernatants were collected and used in the measurement of SOD, MDA, Ca2+, and NO levels in myocardial cells following the kit instructions.
Western Blotting Analysis
Cardiomyocytes were inoculated in a six-well plate at a density of 1 × 106/well. After treatment, they were washed using PBS and treated with the RIPA cell lysis buffer containing PMSF for 30 min on ice. After lysis, samples were centrifuged, and the supernatant was used to measure protein concentrations using the BCA quantitative protein kit. Equal amounts of protein samples were separated by SDS-PAGE and transferred onto the PVDF membranes. Membranes were blocked with 5% skim milk powder at room temperature for 1 h. The membranes were incubated with primary antibodies: β-actin (1:1000), ERɑ (1:500), PI3K (1:1000), p-PI3K (1:1500), AKT (1:1000), p-AKT (1:2000), and eNOS (1:1000) overnight at 4°C. Then, membranes were washed three times using Tris-buffered saline Tween (TBST) and incubated with an HRP-conjugated secondary antibody for 1 h at room temperature in a shaker. Then, protein bands were detected using enhanced chemiluminescence (ECL kit) and quantified by the ImageJ analysis software. β-actin was used as the internal control.
Statistical Analysis
Data were analyzed using the SPSS 21.0 statistical software. Measurement data are expressed as the mean ± standard deviation (SD). One-way ANOVA was used for comparisons among groups. p < 0.05 was set as the threshold for statistical significance.
Results
Chemical Compounds in Tianxiangdan Capsule
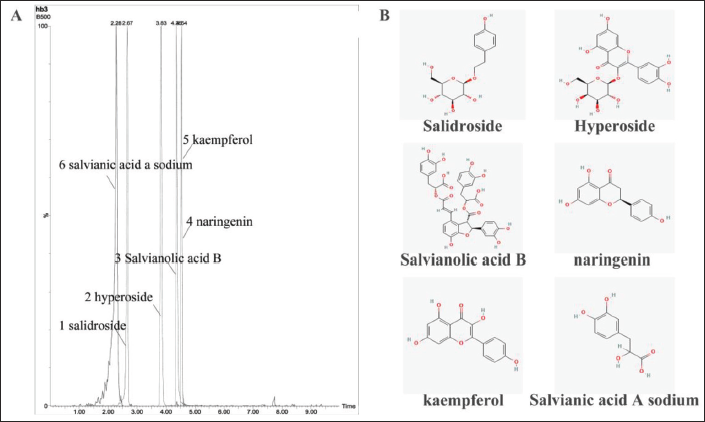
The components were quantitatively analyzed by HPLC-MS, and the results showed as follows: With reference to the standard material composition, the six characteristic components in the drug-containing plasma of TXD were identified to be completely consistent. In the HPLC-MS [M-H]− model, six components were identified: (1) salidroside, (2) hyperoside, (3) salvianolic acid B, (4) naringenin, (5) kaempferol, and (6) salvianic acid A sodium (Figure 1).
(A) The Component of Drug-containing Plasma Reference Substance was Detected by HPLC-MS. (B) Molecular Structure of Chemical Component.
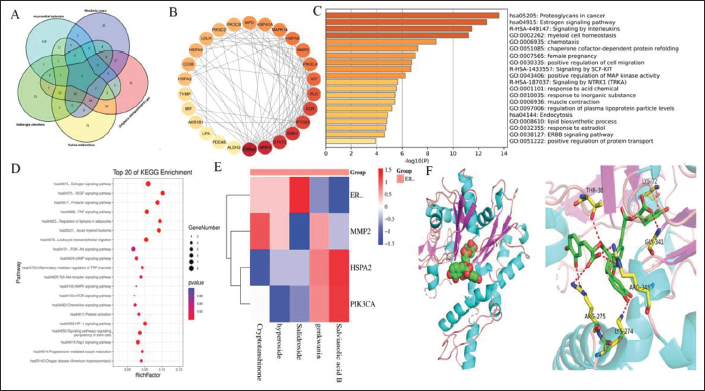
Predicting the Core Target of TXD for the Treatment of Myocardial Ischemia
Compounds of TXD were searched, and 51 species of S. miltiorrhiza, 31 species of D. odorifera, 13 species of Rhodiola rosea, and 23 species of Z. clinopodioides Lam were obtained. The venn diagram was used to map the target genes of the TXD active ingredients with the target genes of myocardial ischemia, and 29 overlapping genes were obtained. We find that estrogen receptor alpha (ERα), heat shock protein (HSP) group (HSPA2 and HSPA1A), matrix metalloproteinase (MMPS) group (MMP9 and MMP2) and phosphatidylinositol 3-hydroxyl kinase (PI3K) group (PIK3CA, PIK3CB and PIK3CD) are the key targets. Molecular docking visualization was performed between ERα, the core target protein of the estrogen signaling pathway, and salidroside with the highest content of blood component. Interactions between salidroside and ERα formed seven hydrogen bonds in the Glu385, Arg515, Leu479, and Thr483 positions of the A chain. Besides, hydrophobic contacts formed between salidroside and the surrounding residues (Asn455, Ser512, Leu508, Ile451, Leu511, and Ser456) significantly promoted the interactions between salidroside and ERα (Figure 2).
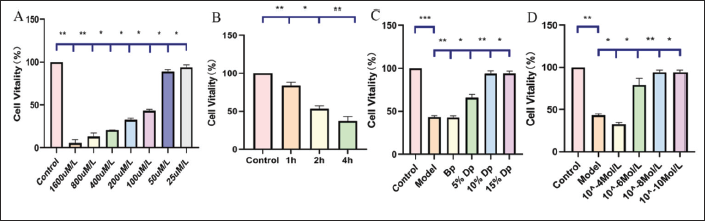
Effects of TXD and E2 on the Activity of H2O2-induced Myocardial Cells
Compared with the control group, H2O2-induced cell injury resulted in a significant decrease in cell viability (p < 0.05). We found that after the concentration of H2O2 was 100 µmol/L and cultured cells for 2 h, the survival rate of cells was reduced to 50%–60%, the degree of cell damage was moderate, and the method had good reproducibility. Therefore, 100 µmol/L H2O2 damage to cells for 2 h was chosen as the oxidative damage modelling method in this experiment. In contrast, the survival rate of cells treated with the TXD drug-containing plasma was improved. Besides, the 10−8 mol/L E2-treated cells also exhibited an improved cell survival rate. The TXD drug-containing plasma and E2-treated cells exhibited a protective effect on H2O2-induced injured myocardial cells (Figure 3).
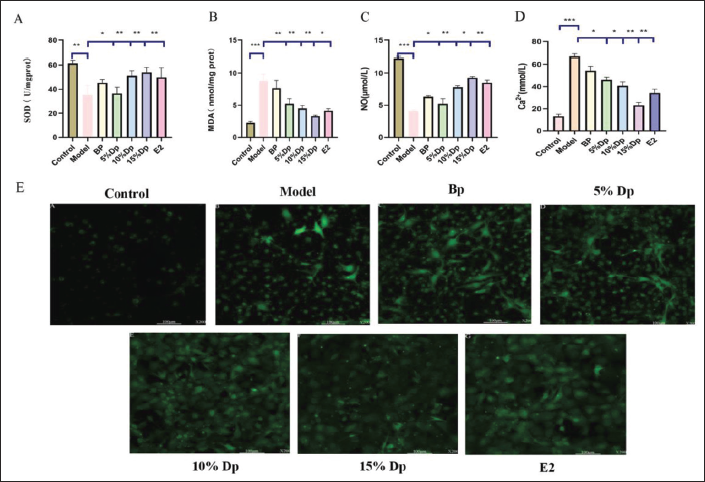
Effects of TXD Drug-containing Plasma and E2 on SOD, MDA, Ca2+, and NO in H2O2-induced Myocardial Cells
After H2O2 treatment, SOD(A) and NO(C) levels in the myocardial cells were suppressed, while MDA(B) and Ca2+(D) levels were elevated (p < 0.05). Pretreatment with TXD drug-containing plasma group and E2 group enhanced SOD and NO levels in myocardial cells following oxidative damage and suppressed MDA and Ca2+ levels (p < 0.05). These results suggest that TXD drug-containing plasma and E2 can improve endogenous antioxidant capacities of myocardial cells, inhibit the production of oxygen free radicals, and reduce lipid peroxidation damage (Figure 4).
Effects of TXD and E2 on the Level of ROS in H2O2-induced Myocardial Cells
After H2O2 treatment, oxidative damage occurred in cells, and ROS level in cardiomyocytes was significantly elevated. In this study, a DCFH-DA fluorescent probe was used to detect the effects of TXD drug-containing plasma and E2 on the level of ROS in myocardial cells after oxidative damage. Experimental results showed that compared with the model group, the TXD drug-containing plasma group and E2 group significantly reduced the fluorescence intensity of DCF in myocardial cells after H2O2 treatment (p < 0.01). These findings imply that TXD drug-containing plasma and E2 inhibited the production of ROS in myocardial cells after oxidative stress injury (Figure 5).
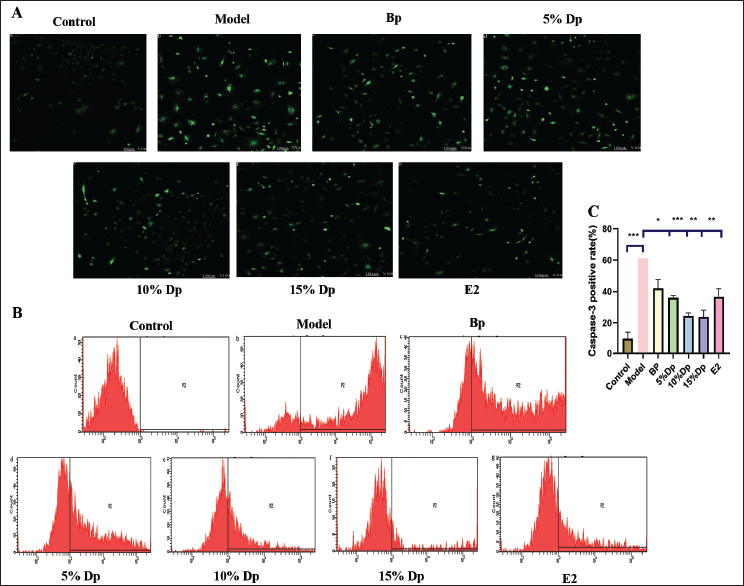
TXD Inhibits Apoptosis and Attenuates H2O2 Damage
Hoechst 33342 staining revealed that after H2O2 treatment, cells exhibited morphological features associated with apoptosis, including nuclear contraction, nuclear chromatin condensation, and apoptotic body formation. Compared with the control group, immunofluorescence staining of cleaved caspase-3 revealed that the model group was significantly increased after H2O2 treatment (p < 0.05). However, in the TXD drug-containing plasma group and E2 group, there was a significant reduction in apoptosis. Therefore, TXD drug-containing plasma and E2 can improve oxidatively damaged myocardial cells and enhance viability (Figure 6).
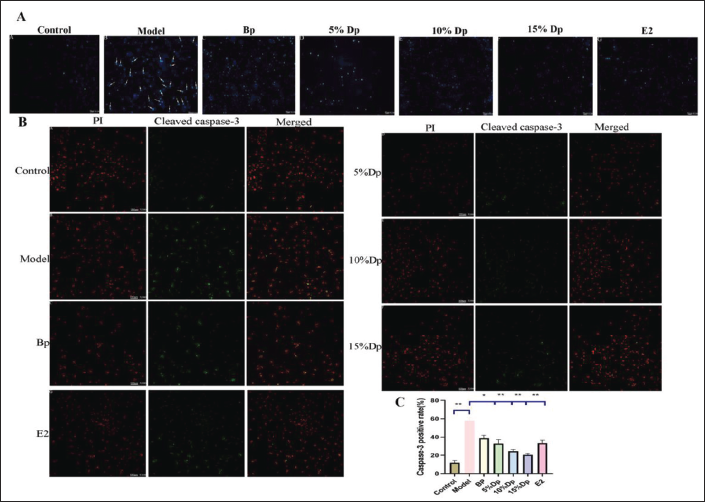
Effects of TXD Drug-containing Plasma and E2 on NO Release in Estrogen Pathway
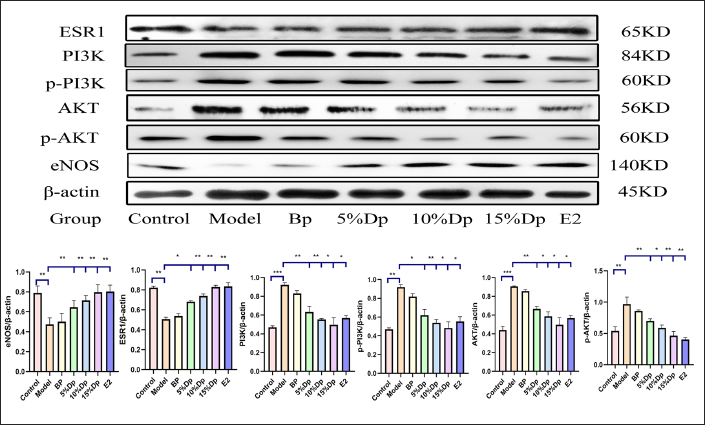
To verify the anti-myocardial ischemia effect of TXD through the regulation of the estrogen signaling pathway, We performed Western blotting on proteins related to NO release in the estrogen pathway analyzed online by KEGG. The focus was on the effect of TXD-containing plasma and E2 on the expression levels of ERɑ, PI3K, p-PI3K, AKT, p-AKT, and eNOS in myocardial cells after oxidative injury.
Compared with the control group, protein levels of ERɑ and eNOS in the model group were significantly decreased, while protein levels of PI3K, p-PI3K, AKT, and p-AKT were significantly elevated (p < 0.01). Compared with the model group, protein levels of PI3K, p-PI3K, AKT, and p-AKT in the TXD drug-containing plasma group and E2 group were significantly decreased. In contrast, protein levels of ERɑ and eNOS were significantly elevated (p < 0.01) (Figure 7).
Discussion
When myocardial injury occurs, oxidative stress, cell apoptosis, and other conditions result in irreversible damage to myocardial cells (Xiao et al., 2022). Myocardial ischemic injury is a complex phenomenon associated with several independent etiologies, such as genetic factors, obesity, environmental factors, and gender factors (Regitz-Zagrosek & Gebhard, 2023). Clinical studies have found that the incidence of cardiovascular diseases in males is significantly higher than that in females, and there is no significant difference between the incidence of cardiovascular diseases in postmenopausal females and males (Vaccarino et al., 2018). Low estrogen levels and postmenopausal women are at higher risk of cardiovascular disease than normal women. The gender differences in cardiovascular disease presentation are consistent with the network pharmacological prediction of the TXD against myocardial ischemia, which focuses our study on the mechanism of the TXD’s anti-myocardial ischemia action on the estrogen signaling pathway. The results of molecular docking between TXD and estrogen receptor ERɑ indicated that salidroside may be the main active component of TXD against myocardial ischemia.
Cell death occurs after ischemic injury, resulting in an energy crisis in cells, swelling, and rupture of organelles, which is then followed by cell apoptosis that is characterized by chromatin concentration and caspase activation. Caspases are central regulators of apoptosis, and a member of this family (Caspase-3) is the critical mediator of neuronal cell apoptosis (Zhao & Vinten-Johansen, 2002). After Caspase-3 activation, apoptosis cannot be prevented. Activation of the apoptotic pathway and an imbalance in cell survival factors determine cell survival and death, and oxidative stress is the main stimulus that triggers apoptosis and death of necrotic cardiomyocytes. Oxidative stress also disrupts intracellular signals associated with ROS generation and activates apoptotic regulatory proteins to mediate a series of cellular responses causing increased rates of apoptosis (Bugger & Pfeil, 2020). After H2O2-induced cell injury, cell apoptosis rate and ROS level in the model group were significantly elevated. In contrast, the TXD drug-containing plasma and E2 pretreatment group significantly suppressed cell apoptosis rate and ROS level. The results show that the TXD drug-containing plasma enhanced the activity of cardiomyocytes, improved the morphology of apoptosis, and reduced the rate of apoptosis and intracellular ROS levels against H2O2-induced oxidative stress injury.
Regardless of external or internal factors, Ca2+ accumulates following damage to myocardial cells (Larsson et al., 2017). Intracellular Ca2+ is a crucial second messenger for excitation-contraction and excitation-transcriptional coupling of coronary smooth muscles and regulates the proliferation, migration, and calcification of coronary smooth muscles (Yu et al., 2022). Moreover, Ca2+ induces intercellular decoupling during myocardial ischemia, which marks the transition from reversible ischemic injury to irreversible ischemic injury (Razavi et al., 2022). This leads to excessive intracellular and mitochondrial Ca2+ levels, which causes cell contracture, protein lysis, and mitochondrial failure, and ultimately to necrosis or apoptotic death. The fluorescence intensity of Ca2+ in the model group was significantly increased, while that of TXD drug-containing plasma and E2 pretreated cells was significantly decreased. The results revealed that TXD drug-containing plasma suppressed intracellular Ca2+ levels and reduced damage to myocardial cells. SOD plays an essential role in oxidation balance in the body. It catalyzes the reaction involved in reducing cell damage attributed to free radicals (Mei et al., 2022). MDA is one of the lipid peroxidation products used to measure oxidative stress in vivo (Nie et al., 2021). NO is a significant regulatory factor in the cardiovascular system that protects cardiomyocytes by regulating downstream protein cascade reactions (Fioranelli et al., 2018). We found that SOD content and NO level in the model group were suppressed, while MDA level was elevated. After TXD drug-containing plasma and E2 pretreatment, SOD content and NO level were found to be elevated, and MDA content was greatly suppressed. Therefore, TXD drug-containing plasma has antioxidant capacities and it can increase the release of NO and relax the vascular smooth muscle, thus relieving angina.
The role of the estrogen signaling pathway is to activate relevant receptors, producing physiological effects. The heart has three estrogen receptors: ERɑ, ERß, and GPER. ESR1 is the editing gene of ERɑ, which plays a more important role in cardiovascular maintenance than ERß (Grinshpun et al., 2023). Estrogen receptor activation can induce multiple signaling pathways, including regulation of NO and Ca2+ release pathways, apoptosis pathways, antioxidant stress pathways, anti-inflammatory pathways, nuclear effects, and so on, change the expression or activity of ion channels, contractile proteins, and ROS production, and regulate vascular function, inflammation, metabolism, and cardiomyocyte survival. Protein kinases are part of signal transduction and are involved in the transfer of extracellular information from the cytoplasm to the nucleus (Adachi et al., 2022). In addition, estrogen can also participate in the regulation of complex diseases. This study investigated the pathway of the estrogen signaling pathway that activates ERɑ, regulates PI3K/Akt/eNOS, and releases NO. Compared with the model group, ERɑ, eNOS, and NO levels in TXD and E2 groups were significantly increased. This suggests that giving TXD activates ERɑ, increases the release of NO, and relieves angina. However, the expression levels of PI3K, P-PI3K, AKT, and P-Akt were significantly decreased in this signaling system. Considering that PI3K and AKT may be involved in the expression of multiple signaling pathways, the overall level of superposition masked the effect of a single pathway.
Conclusion
It is shown that TXD exerts its anti-myocardial ischemic effect through the estrogen pathway. Experiments of H2O2-induced ischemia injury in cardiomyocytes confirmed that TXD activates estrogen receptor ERɑ, enhances NO release of cardiomyocytes through estrogen signaling pathway, improves intracellular Ca2+ overload, oxidative stress injury, and apoptosis, and plays a protective role in myocardial ischemia injury.
Abbreviations
TXD: Tianxiangdan capsule; E2: β-estradiol; TCM: Traditional Chinese Medicine; TCMSP: Traditional Chinese Medicine system pharmacology analysis platform; CNKI: Chinese National Knowledge Infrastructure; ADME: Absorption, distribution, metabolism, excretion; OB: Oral bioavailability; DL: Drug-likeness; MW: Molecule weight; Alogp: Water partition coefficient; Hdon/Hacc: Hydrogen-bond donors/acceptors; Bp: Blank plasma; Dp: Drug-containing plasma; SOD: Superoxide dismutase; MDA: Malondiadease; NO: Nitric oxide; ESR1: Estrogen receptor 1; PI3K: Phosphatidylinositol-3-hydroxykinase; AKT: Serine protein kinase; eNOS: Endothelial nitric oxide synthase.
Footnotes
Acknowledgments
The authors thank Xinjiang Medical University for providing the experimental platform.
Authors’ Contributions
LJ and Y-JY designed the research. MT, XL, and YL performed the research. Y-CW contributed the analytic tools. LJ and MT wrote the manuscript.
Consent for Publication
All authors agree to publish.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article (and/or its supplementary materials).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the State Key Laboratory of Pathogenesis, Prevention and Treatment of High Incidence Diseases in Central Asia Fund (SKL-HIDCA-2020-8).
Statement of Informed Consent and Ethics Approval
This experiment was approved by the Experimental Animal Ethics Committee of Xinjiang Medical University (Ethics Batch Number: IACUC-20210326-13; dated 26-3-2021). The experiments were performed in compliance with the Animal Research: Reporting of in vivo Experiments (ARRIVE) guidelines.
