Abstract
Objectives
To investigate the mechanism of Cistanche deserticola Ma (CDA) in the treatment of myocardial ischemia-reperfusion (I/R) injury by network pharmacology and cell experiments.
Materials and Methods
The main active components of CDA were collected from the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP). I/R-related targets were identified from DisGeNET, OMIMD, and TTD; the I/R protein–protein interaction (PPI) network was constructed using the STRING input. The targets of CDA that inhibit I/R injury in Matescape and Microshengxin were subjected to Gene Ontology (GO) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis. Cell viability, levels of lactate dehydrogenase (LDH), superoxide dismutase (SOD), and malondialdehyde (MDA), and protein expression of phosphatidylinositol 3-kinase (PI3K) and serine/threonine kinase 1 (Akt1) were determined.
Results
A total of 236 targets were identified, with PI3K, Akt, epithelial growth factor receptor (EGFR), and another kinase being the major targets, and according to GO and KEGG analysis, CDA was most likely to inhibit I/R through the PI3K-Akt pathway. The optimal concentration of 10% medicated serum of CDA was determined to be the most effective concentration. The levels of LDH and MDA were significantly decreased in the CDA and BEZ23 groups, but the levels of SOD were significantly increased, thereby alleviating cell damage. In addition, the expression of PI3K, Akt, and p-AKT proteins was significantly reduced in the CDA group.
Conclusion
CDA alleviates I/R injury through antioxidation and inhibition of the PI3K/Akt signaling pathway.
Introduction
Cistanche deserticola Ma (CDA) is a valuable Chinese medicinal material, known as “desert ginseng,” with certain economic value (Wang et al., 2020). CDA has a high medicinal value according to the “Compendium of Materia Medica” and “Rihua Zi Bencao,” CDA can be used to treat kidney yang deficiency, loss of semen and blood, and other diseases (Bai et al., 2023). Recently, CDA has also been proven to have certain curative effects in treating myocardial infarction (Yu et al., 2016).
According to the World Health Organization, myocardial ischemia is the leading cause of death worldwide (Pagliaro et al., 2020). At present, reperfusion is the definitive treatment for acute ischemic heart disease. However, there is no effective treatment to prevent heart injury caused by myocardial ischemia–reperfusion injury (I/R) (Zhu et al., 2021). Reperfusion may lead to a large number of myocardial cell deaths, which is the main cause of cardiac insufficiency after ischemia, manifested by arrhythmia and heart failure. The cell death mode in the process of I/R has been widely noticed (Cai et al., 2022). This study explored the pathway of CDA to reduce myocardial injury through the phosphatidylinositol 3-kinase (PI3K)-serine/threonine kinase 1 (Akt1) pathway through network pharmacology prediction and detected the key role of CDA in regulating PI3K and Akt proteins in the occurrence of I/R through experiments, providing a theoretical basis for the resource development of CDA and the treatment of I/R.
Materials and Methods
Experimental Reagents and Instruments
Ten SD rats and H9c2 cells; CDA; Penicillin–Streptomycin Solution (Yeasen, Product No. 60162ES76); PI3K inhibitors (BEZ23, BEZ, Selleck, S10095); dimethyl sulfoxide (DMSO, source leaf, product number: R21950); cell counting kit 8 (CCK-8, Abbkine, item number: KTC011001); fetal bovine serum (Gibco, product number: 16000044); bicinchoninic acid (BCA, Pulai, product No. P1511-1/2/3); PIK3 antibody (Abcam, product number: ab154678); Akt1 antibody (Abcam, product number: ab148547); goat anti-rabbit IgG (Abcam, product number: ab150077); pancreatin (Yeasen, product number: 40126ES60); three-gas incubator (Thermo Fisher, USA); microplate reader (Bio-Rad, USA); inverted fluorescence microscope (Penetrating instrument); high-speed centrifuge (Shanghai Anting Scientific Instrument Factory).
Obtaining CDA and I/R Targets
According to the oral bioavailability ≥30% and drug-likeness ≥0.18 thresholds, the compounds were collected via TCMSP. The SMILES files of the compounds were obtained from PubChem. The SMILES structures were entered into Swiss Target Prediction to obtain target information related to CDA. Using the disease keyword “myocardial ischemia reperfusion injury,” the DisGeNET database, TTD database, and OMIM database were used to retrieve the above targets, and the I/R-related disease targets were obtained by combining the above targets.
PPI Mapping of CDA-I/R
The Venny diagram was generated by Venny and the intersection of CDA-I/R targets was taken to screen out the CDA-I/R co-target. The same target was input into String and the standard was set as Homo sapiens, “highest confidence (>0.9),” to construct the protein–protein interaction (PPI) network I/R.
Gene Ontology (GO) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analyses of CDA-I/R targets were performed.
The GO-biological function and KEGG signaling pathways of the same targets were screened in Matescape to analyze the mechanism of CDA treatment for I/R.
Drug-containing
CDA was continuously administered at 2 g−1·kg−1·d−1 for 4 days. Ten SD rats were anesthetized by intraperitoneal injection of pentobarbital sodium (40 mg·kg−1). Anesthesia was given for 1 h after the last administration; abdominal aorta blood collection was performed and left at room temperature for 20 min. Then, the supernatant was centrifuged at 4℃ at 1500 r·min−1 for 10 min. The complement was inactivated in a water bath for 30 min, filtered and packaged with a 0.22 µM filter, and stored at −80℃ for future use.
Experimental Design
Con Group: cells were cultured in 10% fetal bovine serum and 1% Dulbecco’s Modified Eagle Medium and incubated in a humidified incubator containing 5% carbon dioxide (CO2) at 37°C.
I/R Group: cells were cultured in sugar-free medium and incubated in a hypoxic environment (1% O2, 5% CO2, and 94% N2) at 37°C for 8 h, and then reoxygenated for 4 h (Yu et al., 2019).
CDA-H Group: cells were cultured in a sugar-free medium containing 10% drug-containing serum and incubated in an anaerobic environment at 37°C for 8 h, and then reoxygenated for 4 h.
CDA-L Group: cells were cultured in a sugar-free medium containing 5% drug-containing serum and incubated in an anaerobic environment at 37°C for 8 h, and then reoxygenated for 4 h.
BEZ Group: cells were cultured in a sugar-free medium containing 10% drug-containing serum and 5 nM BEZ, incubated in an anaerobic environment at 37°C for 8 h, and then reoxygenated for 4 h (Qiu et al., 2019).
Cell Viability was Determined by CCK-8
The cells were seeded into gelatin-coated 96-well culture plates at a density of 1×105/cm and incubated for 12 h. H9c2 cells were then placed in a three-gas incubator for 8 h of hypoxia, followed by 4 h of reoxygenation. The medium was then replaced with a fresh assay medium. CCK-8 reagent was added to each well of the plate and the plates were incubated for 1 h. Absorbance was measured at 450 nm using a microplate reader.
DCFH-DA Detecting ROS
Cell spreading was performed 1 day before the assay to ensure that the cell density reached 50%–70% at the time of the assay. Remove the cell culture medium, add an appropriate amount of the drug diluted to the working concentration in a suitable buffer or serum-free medium, and incubate for 20 min at 37℃ in a cell culture incubator protected from light. Dilute dichloro-dihydro-fluorescein diacetate (DCFH-DA) with serum-free medium at 1:1000 to a final concentration of 10 µM. Aspirate off the inducing drug and add an appropriate volume of diluted DCFH-DA working solution to cover the cells. Wash the cells with serum-free medium 1–2 times to fully remove the DCFH-DA that did not enter the cells. Direct observation by laser confocal microscope.
Measurement of Biochemical Parameters
The cells were inoculated into 96-well plates at a density of 1×104 and each group had wells. When the confluence rate was 80%–90%, experimental groupings were conducted. The levels of lactate dehydrogenase (LDH), superoxide dismutase (SOD), and malondialdehyde (MDA) were determined by a microplate reader.
Western Blotting was used to Detect the Expression of PI3K, Akt1, and p-Akt1 Proteins
Proteins from H9C2 cells were extracted according to the Solarbio procedure, followed by electrophoresis for 70 min and membrane transfer for 60 min. Sealed with 5% milk on a shaker for 2 h, the cells were incubated overnight at 4°C with diluted PI3K (1:1000), Akt1 (1:1000), p-Akt1 (1:1000), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (1:500) antibodies in a 4°C refrigerator. The membranes were washed with Tris-buffered saline with Tween 20 (TBST) for 10 min and then incubated with immunoglobulin G (1:500) for 2 h at room temperature. The membranes were washed three more times at 10 min for color exposure, and the obtained protein band images were quantified using BIO-RAD software, with GAPDH as a control.
Statistical Treatment
SPSS 23.0 statistical software was used for data analysis. All results are expressed as mean values ± standard deviation and used t-test. A p-value of <0.05 was considered statistically significant.
Results
Active Ingredients of CAD
The retrieved active ingredients were screened according to Linpinski’s five principles, resulting in seven chemical components (Table 1).
Chemical Components of CDA.
The Targets of CDA-I/R
The planar structure of yangambin, suchilactone, quercetin, arachidonate, marckine, beta-sitosterol, fenvalerate in CDA is shown in Figure 1A.
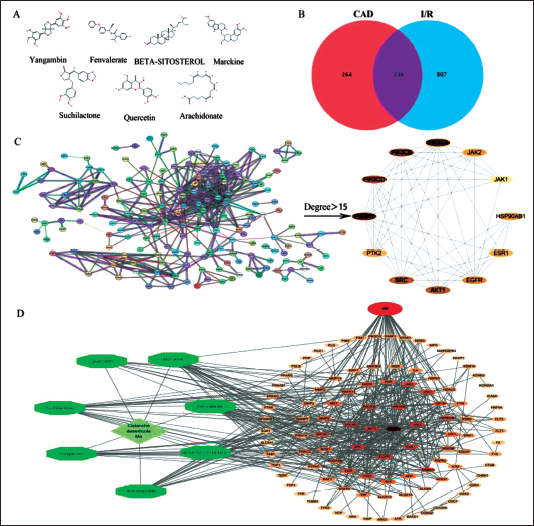
CDA had 500 targets and I/R had 1043 related disease targets. Venny 2.1 was used to make a Venny diagram, and the overlapping part was the common targets of CDA-I/R, with a total of 236 targets (Figure 1B).
The common targets of CDA-I/R were sorted by connection degree and the targets with a large connection degree were PI3K, Akt, epithelial growth factor receptor (EGFR), and just another kinase. The above targets may be the key targets of CDA in the treatment of I/R (Figure 1C).
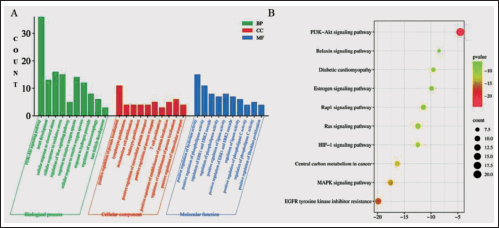
One thousand four hundred and thirty-two GO entries were obtained through GO function enrichment analysis. Among them, CDA was mainly through PI3K-AKT, cellular response to chemical stress, and cellular response to oxidative stress (Figure 2A).
The KEGG pathway enrichment analysis result showed that 96 signaling pathways were obtained. There were 46 pathways with a high correlation with I/R and the CDA targets may be the PI3K-Akt signaling pathway, EGFR tyrosine kinase inhibitor resistance, and MAPK signaling pathway. The most important signaling pathway was PI3K-AKT (Figure 2B).
CDA Alleviates I/R Injury
After treatment with H9c2 cells at gradient concentrations, it was shown that the cardiomyocyte viability of each concentration group of CDA-containing plasma was higher than that of the Con group and 10% was the optimal concentration (Figure 3A).
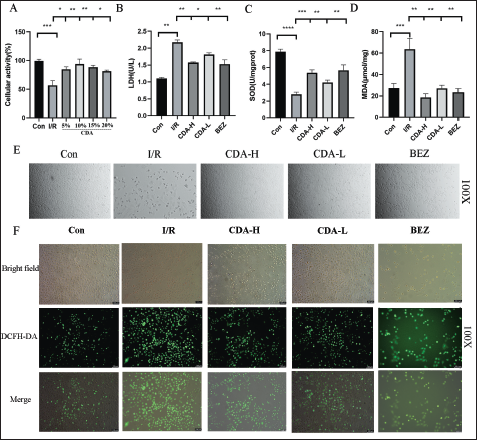
Compared with the blank group, the contents of LDH and MDA in the I/R group were significantly increased, and the content of SOD was significantly decreased; the contents of LDH and MDA in the CDA and BEZ groups were significantly decreased, and the content of SOD was significantly increased compared with the I/R group, and the effect of the CDA-H group was stronger than that of the CDA-L group (p < 0.05) (Figure 3B–D).
H9C2 cells in the Con group grew in clusters and adherent to the wall, most of which were fusiform cells with clearly visible edges and strong refractive power; in the I/R group, a large number of cells became round, died, and floated after 24 h of hypoxia, with weakened refractive power and poor adherent to the wall; the damage was reduced after administration of CDA and BEZ (Figure 3E).
DCFH-DA showed that, compared with the I/R group, the reactive oxygen species (ROS) level in the CDA group was significantly reduced and the ROS level was significantly reduced after BEZ pretreatment. CDA can prevent I/R-induced myocardial injury by inhibiting ROS levels (Figure 3F).
In summary, CDA can alleviate I/R injuries. BEZ, an inhibitor of PI3K, can also alleviate I/R injury, indicating that I/R injury is related to the PI3K signaling pathway.
CDA Inhibited the PI3K-Akt Signaling Pathway
Compared with the Con group, the protein expressions of PI3K, Akt, and p-AKT were significantly increased in the I/R group (p < 0.05). Compared with group I, the expression of PI3K, Akt, and p-AKT protein in group CDA was significantly decreased, and the trend in group CDA-H was stronger than that in group CDA-L (p < 0.05) (Figure 4).
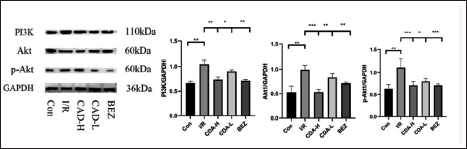
Discussion
In this study, we constructed a bioactive-target network using network pharmacology to explore the mechanism of CDA in treating hypoxic injury. The contents of yangambin, suchilactone, quercetin, arachidonate, marckine, beta-sitosterol, and fenvalerate, were among the top contents in CDA. Yangambin proved to be an effective pharmacological agent against cardiovascular collapse and mortality in endotoxin shock (Araújo et al., 2001). Quercetin and its derivatives lead to an enhancement in heart features, indicating the prospective for quercetin to be used therapeutically in the treatment of cardiac diseases (Patel et al., 2018).
I/R injury occurs when the blood supply to an organ is interrupted and then restored, and it underlies many diseases, particularly heart attack and stroke. Although reperfusion of ischemic tissue is essential for survival, it also triggers oxidative damage, cell death, and abnormal immune responses through mitochondrial ROS production. Although mitochondrial ROS production in I/R is well established, it is usually considered a non-specific response to reperfusion (Chouchani et al., 2014). LDH is highly sensitive for the early detection of cardiomyocyte damage in patients with chest pain (Rossello et al., 2016). MDA is a major metabolite of oxygen-free radicals, reflecting the degree of peroxidation in myocardial tissue (Liu et al., 2023). And SOD, as an important antioxidant enzyme, can ameliorate myocardial oxidative stress injury by scavenging oxygen-free radicals (Kondo f Duan, J. (2022). Modulating., 1999). The levels of LDH, ROS, SOD, and MDA were detected, which confirmed that the hypoxic H9c2 cells were significantly decreased after CDA treatment, proving that CDA can reduce the degree of cell damage by antioxidants (Ren et al., 2020).
The PI3K/AKT pathway is an intracellular signal transduction pathway that responds to extracellular signals to promote cell survival, growth, and angiogenesis (Xie et al., 2019). It was found that myocardial injury activates PI3K, maintains the production of proinflammatory cytokines, and promotes the growth of cardiomyocytes (Wang et al., 2019). PI3K activation results in the production of the second messenger PIP3 at the plasma membrane, which binds to Akt, an intracellular PH domain-containing signaling protein, to regulate cell proliferation, differentiation, apoptosis, and migration. The results of this study showed that the I/R group significantly downregulated the expression of PI3K and Akt, and CDA and BEZ reversed these changes. These findings provide strong evidence that CDA helps inhibit the PI3K-Akt pathway in H9C2 cells. Since PI3K-Akt plays an important role in myocardial I/R, inhibition of the signaling pathway may have clinical significance.
To sum up, CDA alleviates I/R injury through antioxidation and inhibition of the PI3K-Akt signaling pathway.
Footnotes
Acknowledgments
The authors thank Xinjiang Medical University for providing the experimental platform.
Authors’ Contributions
YJ Yue and L Jiang participated in conceptual design, conducting experiments, data analysis and manuscript writing; HM Wang and Y Li conducted experiments; Z Ji and X Rong conducts data analysis; L Jiang provided financial support for manuscript writing and final approval. All authors have read and agree to the published manuscript version
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This experiment was approved by the Experimental Animal Ethics Committee of Xinjiang Medical University (Ethics Batch Number: IACUC-20210326-13) and the date (26-3-2021) of this approval. Experimental compliance (The Animal Research: Reporting of in vivo Experiments [ARRIVE] guidelines).
Funding
This study was supported by the Natural Scientific Foundations of China (8196140154).
