Abstract
Background
Bone tissue undergoes continuous remodeling to maintain a steady state of bone equilibrium. During this process, osteoblasts actively stimulate bone formation, while osteoclasts continuously engage in bone resorption. The dynamic equilibrium between bone formation and bone resorption is crucial for maintaining bone structure. In a healthy human skeletal structure, the two components are constantly in a state of benign dynamic equilibrium. However, due to factors such as aging, trauma, bone diseases, and other influences, the activity of osteoblasts decreases while the activity of osteoclasts increases. This disrupts the dynamic equilibrium, leading to a decrease in bone metabolism. As a result, bone resorption gradually surpasses bone production, making it challenging to maintain a normal amount of bone mass. The effectiveness of traditional Chinese medicine in treating bone-related disorders is extraordinary, and its molecular biological mechanism has become a widely discussed subject.
Objectives
This study aims to unravel the classical signaling pathways and potential targets involved in the effects of traditional Chinese medicine on osteoclast differentiation, and to provide evidence for its clinical efficacy.
Materials and Methods
The main keywords chosen for this study were “Traditional Chinese Medicine (TCM)”, “osteoclast differentiation”, “natural plant”, and “medicinal plant”. To gather relevant literature, we utilized multiple online search engines, including PubMed, Web of Science, and CNKI, as well as other publication resources.
Results
The results indicated that Traditional Chinese Medicine (TCM) can modulate signaling pathways, including NF-κB, MAPKs, STATs, and Wnt/β-catenin pathways., to influence osteoclast differentiation. This modulation involves maintaining the balance of inflammatory interactions, inhibiting oxidative stress.
Conclusion
The impact of traditional Chinese medicine on osteoclast differentiation is reflected on multiple levels and through various pathways. Future research is envisioned to delve deeper from the perspective of precision-targeted therapy, aiming to provide insights for identifying the core targets of traditional Chinese medicine in treating orthopedic diseases.
Introduction
An osteoclast (OC) is a multinucleated giant cell that has a vital function in the growth, development, and preservation of the shape of bones. When illness-related situations such as osteomyelitis, osteoporosis, arthritis, and other diseases occur, they trigger the development of OCs, leading to excessive bone resorption. This disrupts the equilibrium between osteogenesis and OCs (Zhao et al., 2021). Recent studies have highlighted that some Chinese medicine have effects on promoting osteogenic differentiation, inhibiting osteolysis, and exhibiting anti-inflammatory, antioxidant, and immunomodulatory effects. Meanwhile, traditional Chinese medicine (TCM), whether single or compound, can restore the osteoblast (OB)–OCs balance, and the process is influenced and controlled by a series of cytokines and signaling molecules. The activation of the NF-κB receptor for nuclear factor-κB ligand (RANKL) controls the process of OC differentiation and cell death. This procedure entails the attachment to the cytosolic factor κB receptor activator of the nuclear factor kappa B (RANK) on the surface of the OC or OB. Osteoprotegerin (OPG) inhibits OC formation by preventing RANKL–RANK binding and thus induces the apoptosis of OCs (Jiang et al., 2008). Nevertheless, the cultivation of OCs poses a significant challenge for conducting pertinent research, primarily due to the complex process of OC development. The introduction of RAW264.7 cells addresses the issue of OCs being non-transferable.
Furthermore, RAW264.7 cells offer advantages such as their ability to pass through barriers and be frozen, rapid proliferation, and the potential to induce a significant number of osteoclasts (Li et al., 2014; Wang et al., 2017). Prior studies have mostly focused on improving the process of OB differentiation. These studies have shown that herbs that strengthen the kidneys play a vital role in this process by activating the MAPK and Wnt/β-catenin signaling pathways (Shihai et al., 2019; Xiao et al., 2018). However, limited attention has been given to the inhibition of OC differentiation. Hence, the objective of this article is to gather pertinent research investigating the suppressive impact of Chinese medicine on OB differentiation. The gathered data can serve as the basis for utilizing TCM to modulate the equilibrium of OB–OC differentiation and the management of bone-related disorders.
Common Factors That Affect Osteoclast Differentiation
The Function of Cytokines and Chemokines in the Process of Osteoclast Differentiation
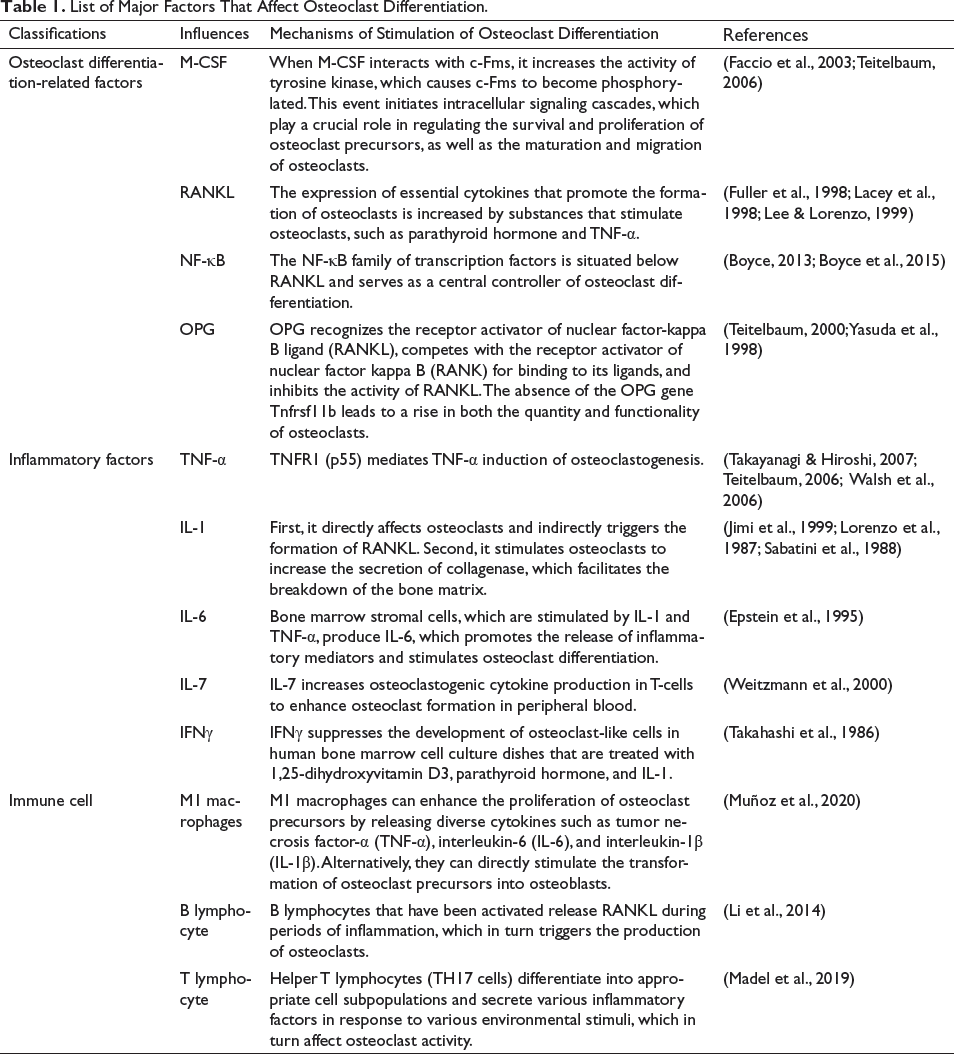
The current factors that influence OC differentiation primarily include cytokines, inflammatory factors, and immune cells, as detailed in Table 1. The binding of M-CSF to the c-Fms receptor initiates tyrosine kinase activity, resulting in the phosphorylation of c-Fms. This phosphorylation then stimulates intracellular signaling cascades, which play a role in regulating the survival and proliferation of OC precursor cells, as well as the maturation and migration of OCs (Faccio et al., 2003; Teitelbaum, 2006). Animal experiments have demonstrated impaired OC differentiation following the knockdown of the M-CSF gene, resulting in congenital osteosclerosis (Fleisch et al., 1993). RANKL is a crucial cytokine for the formation of OCs, and its expression is upregulated by OC-stimulating substances such as parathyroid hormone and tumor necrosis factor-α (TNF-α) (Cesar & Bogado, 2011; Fuller et al., 1998; Lee & Lorenzo, 1999). The NF-κB family of transcription factors is located after RANKL and serves as a key controller of OC differentiation (Boyce, 2013; Boyce et al., 2015). The transcription factor NF-κB consists of homologous or heterodimers of RelA/p65, cRel, RelB, p52, and p50. These components have a significant role in various physiological and pathological processes, including cellular energy metabolism, angiogenesis, and inflammatory responses (Courtois & Gilmore, 2006). TNF-α exerts its influence by binding to two receptors, TNFR1 and TNFR2. TNFR1 helps TNF-α-induced osteoclastogenesis to happen. This increases the production of OCs and starts bone resorption by these cells. Within a living organism (Takayanagi & Hiroshi, 2007; Teitelbaum, 2000; Walsh et al., 2006), Interleukin-1 (IL-1) is the strongest activator of bone breakdown under laboratory conditions. It directly affects OCs and indirectly increases the synthesis of RANKL, which leads to a higher release of collagenase and greater destruction of the bone matrix (Jimi et al., 1999; Lorenzo et al., 1987; Sabatini et al., 1988). Stimulation of bone marrow stromal cells by IL-1 and TNF-α induces the production of IL-6. IL-6, in turn, enhances the release of inflammatory mediators and promotes the development of OCs (Manolagas & Jilka, 1995; Sims, 2021). IL-7 is essential for the generation of B- and T-cells and has an impact on the maintenance of bone stability. Administration of IL-7 throughout the body improves the production of cytokines, thus, promoting the formation of bone breakdown cells in the blood outside the bone (Weitzmann et al., 2000). In addition, the study by Toraldo et al. (2003) found that IL-7 did not cause bone loss in mice that lacked T-cells, known as naked mice. IFNγ suppresses the formation of OC-like cells in human bone marrow cell culture when stimulated with 1,25-dihydroxy vitamin D3, parathyroid hormone, and IL-1. Studies conducted on rats have shown that injecting IFNγ for a duration of eight days leads to bone loss (Mann et al., 1994; Takahashi et al., 1986).
List of Major Factors That Affect Osteoclast Differentiation.
Macrophages serve as precursors for OCs, undergoing differentiation into OCs upon stimulation by M-CSF and RANKL (Jin et al., 2023).
M1 facilitates the proliferation of OC precursors by releasing a variety of cytokines such as TNF-α, IL-6, and IL-1β. Additionally, M1 directly induces the differentiation of OC precursors into OCs (Muñoz et al., 2020). Under inflammatory conditions, activated B lymphocytes secrete RANKL, thereby activating OC formation (Li, Zhang, 2014). Among T lymphocytes, helper T-cells (TH17 cells) differentiate into corresponding cell subpopulations and secrete a variety of inflammatory factors in response to a variety of environmental stimuli, which in turn affect OC activity (Madel et al., 2019).
Signaling Pathways Involved in Osteoclast Differentiation
The OPG/RANKL/RANK signaling pathway is crucial in regulating the development of OBs and OCs as well as sustaining the processes of bone production and resorption (Figure 1) (Kushlinskiĭ et al., 2014). Zhang et al. (2020) demonstrated that the modulation of this signaling pathway can effectively control the balance between OB and OC, and promote OB while inhibiting OC differentiation. This regulatory mechanism preserves the bone mass equilibrium, offering essential support for bone disease healing.
Traditional Chinese Medicine Acts on the Body Through Signaling Pathways to Treat Organism Damage.
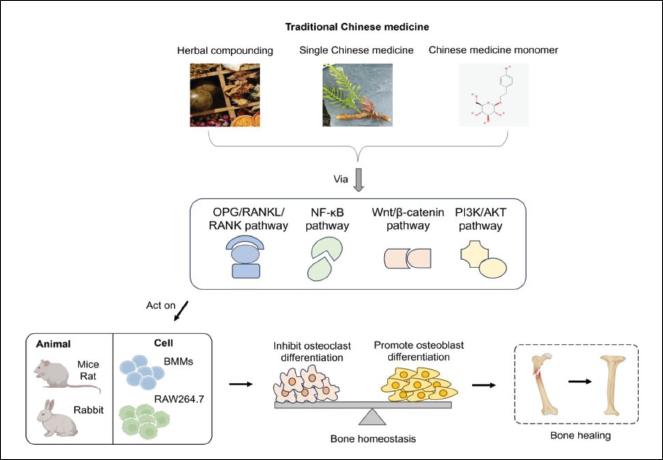
In addition, the rat model was subjected to testing with the Zuogui pill. The results revealed a significant increase in OPG mRNA expression and a decrease in VPAC2 and RANKL mRNA expression as a result of the Zuogui pill. OCs play a crucial role in regulating the balanced stability of bone, where NF-κB facilitates the process of differentiation, maturation, and resorption of bone tissue. Furthermore, NF-κB also has a significant impact on the growth, specialization, and programmed cell death of OBs (Lei et al., 2010). Meng et al. (2021) provided evidence that junceellolide D (JD) effectively suppresses the process of OC development in bone marrow macrophages (BMMs) triggered by RANKL. JD also suppresses the activation of NF-κB and MAPK signaling pathways induced by RANKL (Meng et al., 2021). These findings indicate that JD has the ability to impede the formation of OCs by activating Nrf2 and suppressing NF-κB and MAPK signaling pathways. As a result, it can prevent bone-destructive disorders caused by OCs. Du et al. (2016) discovered that suppressing β-catenin led to the presence of tartrate-resistant acid phosphatase (TRAP), an elevation in the quantity of OCs, and an enhancement in the expression of matrix metalloproteinase-9 (MMP-9) protein (Du et al., 2016).
In this context, Han et al. (2021) have discovered that TCMs commonly used in the treatment of osteoporosis, such as Eucommia ulmoides, Cuscuta chinensis, and Rhizoma Drynariae, have been demonstrated to modulate the PI3K/Akt signaling pathway, a certain substance that can impede the development of OCs and decrease the loss of calcium and phosphorus in bones (Han et al., 2021).
Experimental Study on Inhibition of Osteoclast Differentiation by Chinese Medicine
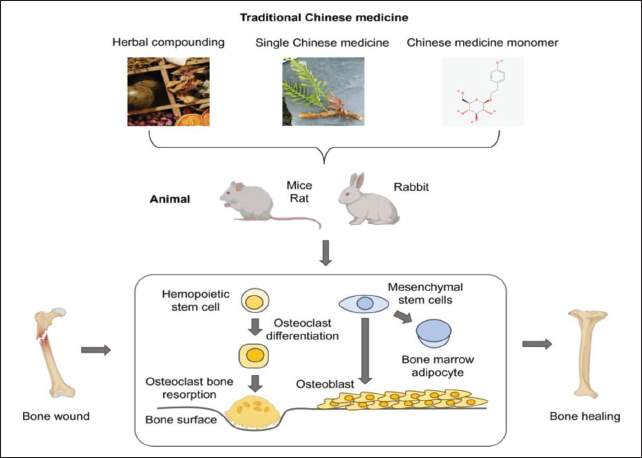
Previous experiments have revealed that single herbs and compound formulas within traditional Chinese medicine can inhibit OC differentiation and promote the repair of damaged bones, primarily by regulating the balance between osteogenesis and osteoclastogenesis (Figure 2).
Traditional Chinese Medicine to Maintain Bone Homeostasis and Heal Bone Injuries.
Single-flavored Herbs and Herbal Ingredients
Salidroside (SAL) is an active constituent derived from the TCM Rhodiola rosea and was examined for its impact on OS differentiation by Dandan Gao (Manolagas & Jilka, 1995). The investigation focused on SAL’s influence under hypoxic conditions, utilizing a TRAP assay to observe the differentiation of mature OCs induced by RANKL and M-CSF. In settings with low oxygen levels, SAL was found to have a suppressive impact on HIF-1α and the genes it regulates (VEGF, ANGPTL4, and IL-6), as well as the production of associated proteins in mature OCs generated by RAW264.7. The production of HIF-1α and its downstream target genes VEGF, ANGPTL4, and IL-6 was considerably elevated when different doses of SAL (10 and 100 nM) were combined with CoCl2, compared to the group treated with CoCl2 alone (p < 0.05). Moreover, there was a notable reduction in the expression of upstream regulated gene pVHL and downstream target gene miR-20a of HIF-1α in response to SAL, emphasizing its regulatory role in this pathway (p < 0.05) (Pan et al., 2021).
Curculigoside (Cur) is a phenolic component found in Curculigo orchioides, a TCM. Studies indicate that Cur may function as an inhibitor of osteoclastogenesis by reducing the generation of reactive oxygen species (ROS) through suppressing the activity of oxidative enzymes. This is achieved by stimulating the Nrf2 pathway and suppressing the NF-κB signaling pathway. When Cur and N-Acetyl-
Astragalus total brassica is an extract of the TCM Astragalus, which was investigated by Liu et al. (2021) in vivo. Their research uncovered that this extract has a suppressive impact on the synthesis of TNF-α, IL-1, and RANKL in the bloodstream of rats. Simultaneously, it demonstrated a promotional effect on the production of OPG in serum. Manipulating the RANKL/RANK/OPG pathway has the potential to impede the differentiation of OCs, reduce bone erosion caused by excessive OC production, and improve normal bone development (Chong et al., 2016).
Zhang et al. (2020) performed tests in which puerarin was administered either alone or in combination with RANKL on osteoclast precursors (OCPs) generated from bone marrow macrophages (BMMs). They assessed autophagic parameters of OCPs such as autophagy protein, LC3 conversion, autophagic vesicles, or LC3-puncta using Western blotting.
CCK-8 assay was used to find OCP proliferation, and the tartaric acid–resistant acid phosphatase (TRAP) staining was used to check for OC differentiation. The findings show that gerberoside has the ability to impede OCP autophagy regardless of the presence or absence of RANKL. Additionally, it can hinder OCP proliferation and OC development, hence mitigating bone loss (Zhang et al., 2019).
Zhang et al. (2019) investigated Drynaria’s impact on the Wnt/β-catenin pathway in BMSCs. The research results revealed that the activated Wnt/β-catenin signaling pathway in Drynaria regulates OPG and RANKL recreation in BMSCs, suppressing the OPG/RANKL/RANK pathway in OCs. This inhibition impedes the process of OC formation and maturation, resulting in a decrease in bone resorption. Additionally, the study observes that Drynaria restrains OC differentiation while promoting OB differentiation, achieving a balance between osteogenesis and OCs. Overall, Drynaria contributes to bone protection, as demonstrated in this study (Zhang, 2019).
Sun et al. (2019) confirmed that concentrations of cajaninstilbene at 2.5 µmol/L and above, devoid of cytotoxic effects, effectively impede RANKL-induced OC differentiation—this conclusion is based on comprehensive investigations encompassing cell experiments, animal studies, and clinical trials. Cajaninstilbene exhibits the capacity to hinder the expression of genes associated with OCs, consequently suppressing their differentiation and mitigating bone erosions (Sun et al., 2019).
Zeng et al. (2016) found that the viability of RAW264.7 cells was not affected by aconitine at doses of 0.125 and 0.25 µmol/L. Nevertheless, they demonstrated a correlation between the dosage and the suppression of OC production and bone resorption activity mediated by RANKL. In addition, aconitine reduced the activation of NF-κB and NFATc1 in RAW264.7 cells in a dose-dependent manner when stimulated by RANKL. Consequently, there was a decrease in the manifestation of genes that are particular to OCs (c-Src, β3-Integrin, cathepsin K, and MMP-9) and a transmembrane protein specific to dendritic cells (DC-STAMP). Therefore, the process of OC development in RAW264.7 cells, triggered by RANKL, leads to a decrease in the resorption of bone tissue (Xiang et al., 2021). Table 2 displays numerous other factors that impede the process of OC differentiation.
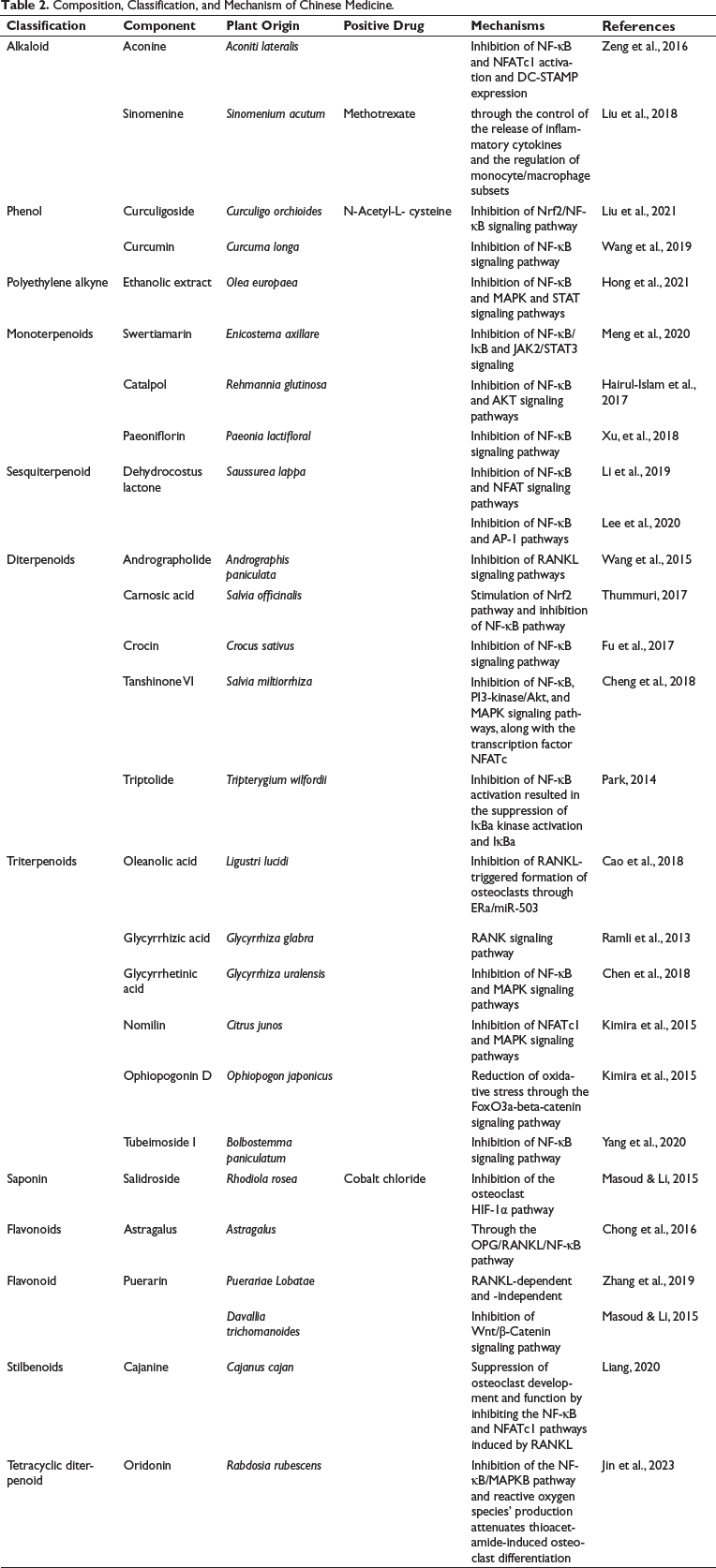
Composition, Classification, and Mechanism of Chinese Medicine.
Herbal Compounding
Jinkui Shenqi Pill, derived from the book Synopsis of Golden Chamber, stands as the greatest TCM for tonifying the kidney and strengthening Yang. Based on the concept of “the kidney generating marrow and dominating bone,” a recent clinical investigation has shown that taking Jinkui Shenqi Pill can significantly improve the bone density of patients. In a foundational experiment, the medication demonstrated a significantly reduced IL-6 and increased OPG concentration in the serum of rats. Consequently, this hinders the growth and specialization of OCs as well as the occurrence of bone disease (Zhang et al., 2020).
Song (2020) investigated the impact of varying concentrations of Duzheng decoction extract on the RAW264.7 cell proliferation using the CCK-8 method. The researcher investigated the generation of OC cells using the tartrate-resistant acid phosphatase test. He identified the presence of RANK, RANKL, TRAF-6, and NF-κB proteins by Western blot analysis. The results showed that doses of 50, 100, and 200 µg/ml did not hurt RAW264.7 cells and stopped OCs from differentiating.
Notably, at 200 µg/ml concentration, OC differentiation is potentially inhibited by the downregulation of the expression of RANK, NF-κB, TRAF6, PI3K, and AKT proteins. In a separate study, Wang et al. (2019) used Jiangsu granules extracted with ethyl acetate to RAW264.7 cells induced by RANKL and found that the activity of OC differentiation maturation and bone resorption was significantly inhibited relative to that without Jiangu granules. This inhibitory effect is primarily attributed to the suppression of SrcmRNA expression and related proteins in OC cells. Consequently, downstream PI3k, Akt, NFATc1 mRNA, and proteins were affected by controlling OC differentiation and maturation, inhibiting bone resorption erosion, and reducing bone loss (Song, 2020).
Through a comparative analysis of varying dosages of Longzuantongbi prescription, a compound in Zhuang medicine, and Western medicine, Huang (2019) observed that the high-dose groups of both TCM and Western medicine effectively suppressed OC differentiation. This observation was made while assessing the anti-rheumatoid arthritis effects of Longzuantongbi decoction and methotrexate across different dosage levels (Huang, 2019).
Zhuang medicine effectively diminishes TLR4 and Myd88 protein expression in OC cells and blocks the TLR4/NF-κB signaling pathway. Consequently, it downregulates TNF-a, NFAT-c1, and c-Fos gene expression in OC cells; this process effectively hinders the development and specialization of OCs. Wang et al. (2014) conducted a study to investigate the impact of varying dosages of Qing’e prescription on MMP-2, MMP-9, MMP-13, OPG, and RANKL in the tissues of osteoporotic rats with decreased bone mass and then studied the effects on OC cell maturation and differentiation and bone resorption erosion, and the findings demonstrated that varying concentrations (10%, 15%, and 20%) of Qing’e prescription had an impact on OC cell differentiation. Moreover, the effects were more pronounced as the concentration increased, indicating a dose-dependent relationship (Wang et al., 2014).
Clinical Study on the Inhibition of Osteoclast Differentiation by Chinese Medicine
Zhao (2019) conducted a clinical trial that included 60 patients diagnosed with rheumatoid arthritis, with 30 patients assigned to the treatment group and 30 patients assigned to the control group. The treatment group was supplied the wenjing-qingluo-yishen prescription in addition to the basic treatment, whereas the control group received Leigongteng multi-glucoside pills along with the basic treatment. Both groups were treated for a duration of 12 weeks. Changes in clinical symptoms, inflammatory indexes, and immunoglobulins were monitored before and after treatment in both groups. The study utilized ELISA to examine the expression of serum OC cell-related receptors, inflammatory factors, and TNFs. The results demonstrated that both clinical and experimental investigations supported the efficacy of Chinese medicine in treating rheumatoid arthritis. Moreover, animal studies have shown that the recommended treatment increased the levels of OSCAR in the blood and decreased the expression of ESR, CRP, IL-1, and TNF-a. Consequently, this hindered the process of OC differentiation and maturation, resulting in a decrease in bone resorption and erosion. The treatment demonstrated favorable results in managing rheumatoid arthritis and other illnesses associated with bone loss (Zhao, 2019).
Conclusion
Bone remodeling refers to the mechanism via which OCs eliminate an older or impaired bone and substitute it with a newly created bone by OBs (Schwendich et al., 2022). OCs originate from hematopoietic stem cells (HSCs) (;Xu & Teitelbaum, 2013; Zhu et al., 2020) and possess the ability to break down bone tissue through the secretion of acids and proteolytic enzymes. These enzymes have the capacity to dissolve collagen and matrix proteins, facilitating the process of osteoclastic bone resorption (Charles & Aliprantis, 2014; Kim et al., 2020). Macrophage colony-stimulating factor (M-CSF) and RANKL are very important in the process of OC differentiation (Kim et al., 2014; Xiang et al., 2021). RANKL and M-CSF can induce the transformation of OC precursor cells into OBs, which stimulates the expression of RANK (Gal et al., 2022), and the binding of RANK to RANKL activates OC-associated NF-κB ligand. The combination of RANK and RANKL activates the OC-associated NF-κB and MAPK signaling pathways, which stimulates the expression of genes and proteins upstream and downstream of the pathways (Kim et al., 2020). In recent years, the culture of TCM has been strongly promoted, the experimental research of TCM has become more and more standardized, and a large number of studies have found that the components of TCM have a variety of substances that can inhibit OC differentiation, which provides a new way of thinking for the treatment of bone-related diseases by TCM. While numerous studies have been conducted on the efficacy of TCM in treating osteoporosis, there is a scarcity of information regarding its effectiveness in treating osteomyelitis and other bone illnesses. More clinical and scientific researchers must practice and explore whether the destruction of bone mass in osteomyelitis and other bone diseases can also control the overall situation by inhibiting OC differentiation.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Not applicable
Funding
This study was supported by the National Natural Science Foundation Project (82104833), Special Project for Research on Traditional Chinese Medicine in Henan Province (2023ZY1008), and Special Project for Research on Traditional Chinese Medicine in Henan Province (2019ZYBJ15).
Authors’ Contributions
YJC and CLY designed the study; YNQ collected and analyzed the data; YNQ drafted the initial manuscript; SMC and CLY revised the article critically; SMC reviewed the article; YNQ and SMC are co-first authors. All authors approved the final manuscript.
