Abstract
Osteoclasts are specialized cells responsible for bone resorption, which plays a crucial role in the dynamic processes of bone remodeling and maintenance. Osteolytic skeletal disorders, such as osteoporosis, arise from an imbalance between the activities of osteoclasts and osteoblasts. The regulatory mechanisms underlying osteoclast differentiation, known as osteoclastogenesis, are governed by the receptor activator of nuclear factor kappa-B ligand (RANKL)/receptor activator of NF-κB (RANK)/osteoprotegerin (OPG) signaling system. In this system, the RANK, a type 1 transmembrane protein, acts as a central regulator of osteoclastogenesis through various signaling pathways. This review seeks to explore the influence of Traditional Chinese Medicines (TCM) on osteoclastogenesis, focusing on the NF-κB signaling pathway. This study aims to offer valuable insights into potential therapeutic strategies for managing osteolytic skeletal disorders by investigating the interaction between TCM and the regulatory mechanisms that control osteoclast function. A comprehensive literature review was conducted, concentrating on studies that examine the effects of TCM on osteoclastogenesis and the NF-κB signaling pathway. Databases such as PubMed, Web of Science, and Google Scholar were searched for peer-reviewed articles, reviews, and clinical studies. The inclusion criteria focused on studies addressing the impact of TCM on osteoclast function and the associated molecular mechanisms, especially those involving the RANKL/RANK/OPG system and the NF-κB pathway. The review identified several TCM compounds with regulatory effects on osteoclastogenesis. These compounds were found to modulate the NF-κB pathway, influencing osteoclast differentiation and activity. Key findings highlighted the discovery of specific TCM herbs and their active ingredients that can inhibit osteoclast formation and function, promoting bone health. The mechanisms of action often involved suppressing RANKL-induced NF-κB activation, a crucial process for osteoclast differentiation and bone resorption. This review underscores the potential of TCMs as therapeutic agents for osteolytic skeletal disorders through their modulation of the NF-κB signaling pathway. The findings suggest that TCM may significantly regulate osteoclastogenesis, offering new avenues for developing treatments for conditions like osteoporosis. However, further research and clinical trials are necessary to fully elucidate the therapeutic potential of TCM and translate these findings into clinical practice.
Introduction
Osteoblasts and osteoclasts are the two pivotal cell types that collaborate to maintain bone homeostasis (Shen et al., 2022; Veis & O’Brien, 2023). Osteoblasts, originating from mesenchymal progenitors, are responsible for bone formation and have the potential to differentiate into marrow stromal cells or adipocytes (Aubin, 2001). In contrast, osteoclasts derived from monocyte/macrophage lineage progenitors enter the bone via the bloodstream to resorb mineralized tissues (Niu et al., 2022). This continuous process of bone remodeling ensures structural integrity and strength, and supports hematopoiesis.
An imbalance in osteoclast activity can result in various osteolytic conditions, including osteoporosis, Paget’s disease, tumor-induced osteolysis, arthritis, and periodontal disease. These conditions are characterized by reduced bone mass and increased fracture risk, often associated with osteoblast dysfunction. The maturation of osteoclasts into multinucleated cells depends on the fusion of mononuclear precursor cells, which requires two critical factors. First, an “optimal density” of precursor cells must be reached (Ishii & Saeki, 2008). Second, two specific cytokines are necessary: macrophage colony-stimulating factor (M-CSF), which acts as a survival factor, and receptor activator of NF-κB ligand (RANKL), which functions as a differentiation factor (Ikeda & Takeshita, 2016). Both spleen cells and stromal cells secrete M-CSF and RANKL. Therefore, when bone marrow is combined with these cells, the effect is comparable to direct cytokine treatment (Tsai et al., 2023). Understanding these regulatory mechanisms is crucial for developing targeted therapies for osteolytic diseases.
The Regulatory Mechanism of Osteoclastogenesis
The differentiation of osteoclasts is governed by a signaling system known as the RANKL/receptor activator of NF-κB (RANK)/osteoprotegerin (OPG) pathway in Figure 2. Within this system, the RANK is a type 1 transmembrane protein belonging to the TNF receptor (TNFR) superfamily. RANK is primarily expressed on monocytes/macrophages, including osteoclastic precursors, activated T cells, dendritic cells, and mature osteoclasts (Hou & Tian, 2022; Lee, 2017). RANKL, also known as TRANCE (tumor necrosis factor-related activation-induced cytokine), is a type 2 membrane protein within the tumor necrosis factor (TNF) superfamily. It is synthesized by stromal cells/osteoblasts and activated T cells (Kim et al., 2002). OPG, a soluble decoy receptor related to the TNFR superfamily, is secreted by bone marrow stromal cells (BMSCs)/osteoblasts and acts as a counterbalance to RANKL (Hofbauer & Schoppet, 2001).
Traditional Chinese Medicines (TCMs) Therapeutic Promise in Osteoclastogenesis. TCMs Consist of Diverse Constituents that Proficiently Influence Osteoblasts, Offering a Preventive Measure against Complications like Osteoporosis.
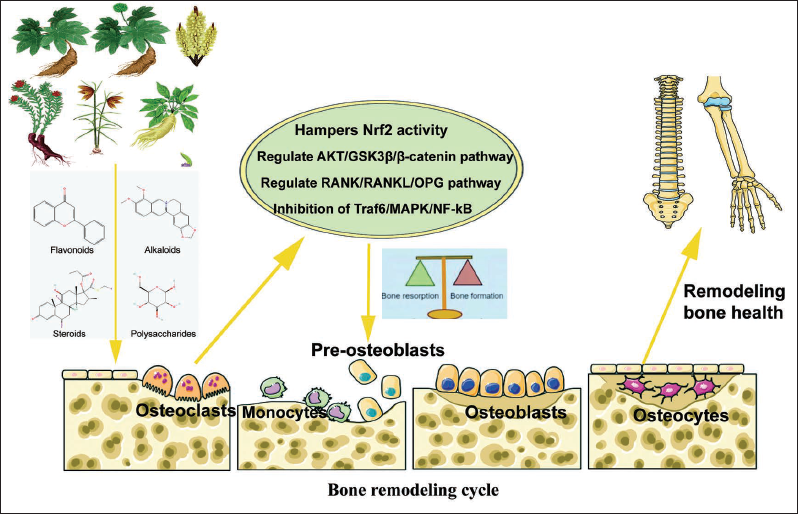
Traditional Chinese Medicine (TCM) can Enhance Osteoblast and Bone Mesenchymal Stem Cell Growth, Augment Osteogenic Abilities, Suppress Oxidative Stress-induced Apoptosis in Osteoblasts, and Positively Impact by Inhibiting Osteoclastogenesis and the Bone-resorption Function of Osteoclasts.
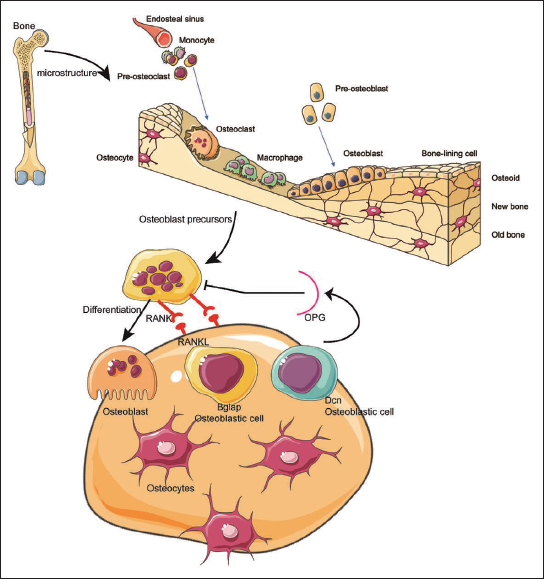
The interaction between RANKL and RANK facilitates the differentiation of osteoclastic precursors into osteoclasts by inducing the expression and activation of osteoclastogenesis-specific transcription factors. Advances in mouse genetics have significantly enhanced our understanding of osteoclast functions by identifying the transcription factors that regulate osteoclast differentiation. RANK’s ability to trigger nuclear factor kappa-B (NF-κB) activation is recognized as a hallmark of its signaling pathway and is one of the initial responses mediated by RANK (Yang et al., 2019). While tumor necrosis factor receptor-associated factor 6 (TRAF6) is crucial for mediating RANK-induced NF-κB activation, it is unlikely that NF-κB is the sole downstream molecule responsible for TRAF6’s complex functions. Although TRAF6 activates various molecules, genetic evidence underscores explicitly the indispensable role of NF-κB, rather than other molecules like mitogen-activated protein kinases (MAPKs), in osteoclastogenesis.
NF-κB is a critical transcription factor essential for osteoclast formation. Comprising five members—including Rel A (p65), Rel B, c-Rel (Rel), NF-κB1 (p105/p50), and NF-κB2 (p100/p52)—these proteins undergo post-translational processing to form shorter versions (p50 and p52) and create dimers with Rel family members (Boyce et al., 2015). p50-/- and p52-/- mice exhibit altered immune responses without developmental issues (Soysa & Alles, 2009). However, NF-κB p50/p52 double knockout (dKO) mice display osteopetrosis, growth retardation, and craniofacial abnormalities, including unerupted incisors (Soysa & Alles, 2009). Osteoclast formation is absent in NF-κB p50/p52 dKO mice, and fluorescence-activated cell sorting (FACS) analysis shows a threefold increase in RANK-expressing splenocytes. This indicates that NF-κB p50 and p52 are not essential for RANK-expressing progenitor formation but are crucial for RANKL-RANK-induced osteoclastogenesis (Callewaert et al., 2010). RANKL or TNF fails to induce c-Fos (cellular proto-oncogene Fos) in M-CSF-treated NF-κB p50/p52 dKO splenocytes. However, exogenous c-Fos rescues the defect in osteoclast formation in the absence of RANKL, suggesting that c-Fos operates downstream of NF-κB (Kodama & Kaito, 2020; Yao et al., 2008).
Upon cell stimulation, including by RANKL, NF-κB proteins, initially located in the cytoplasm, swiftly translocate into the nucleus (Cavey et al., 2005; Takayanagi et al., 2000). NF-κB activation involves two distinct pathways: the classical pathway, which activates the IKK complex leading to IκB degradation, and the alternative pathway, which processes the p100–RelB complex to activate p52–RelB dimers (Ruocco et al., 2005). Studies on p50/p52-deficient mice indicate that both pathways are crucial for osteoclastogenesis, suggesting they regulate common target genes. Recent findings reveal more intricate mechanisms, highlighting the role of NF-κB-inducing kinase (NIK) in controlling p100 processing in the alternative pathway (Ghosh & Karin, 2002; Oeckinghaus et al., 2011). NIK’s involvement underscores its significance in NF-κB activation and osteoclastogenesis. In vitro, differentiation studies show that osteoclast precursors deficient in NIK fail to respond to RANKL. Interestingly, NIK-deficient mice do not display osteopetrosis, suggesting that other signals may be involved in the regulation (Paloneva et al., 2000).
Disruption of IκB kinase alpha (IKKα), a key catalytic component in the alternative pathway, impairs osteoclastogenesis in vitro but not in vivo. Conversely, disruption of IκB kinase beta (IKKβ), a catalytic component in the classical pathway, inhibits osteoclastogenesis both in vitro and in vivo (Ruocco et al., 2005). Furthermore, inhibiting the classical pathway using a cell-permeable peptide effectively prevents osteoclast formation in vitro and mitigates inflammatory bone destruction in vivo. These findings indicate that the classical pathway, rather than the alternative pathway, is indispensable for osteoclastogenesis.
However, this conclusion seems somewhat contradictory to observations in p50-/- and p50/p52-deficient mice, as the former exhibit no bone abnormalities. Therefore, the specific functions of both NF-κB activation pathways and the roles of individual NF-κB components in osteoclastogenesis remain partially ambiguous. There is a suggestion of intercommunication between the classical and alternative pathways involving the inhibition of p50–RelA dimers by p100. Comprehensive investigations are essential to thoroughly and precisely understand the intricate regulatory mechanisms involved in osteoclastogenesis.
Role of NF-κB in Disorders Affecting Bone Metabolism and Osteolysis
NF-κB is a pivotal player in the development of osteolysis within inflammatory conditions such as rheumatoid arthritis, periprosthetic osteolysis, periodontitis, low-grade systemic inflammation, Paget’s disease of bone (PDB), and various bacterial infections (Abu-Amer, 2009; Baker et al., 2011). This transcription factor is also crucial in mediating the effects of multiple cytokines implicated in postmenopausal osteoporosis. Its central role in these pathological processes underscores the potential of targeting NF-κB for therapeutic interventions in bone metabolic disorders and osteolytic conditions shown in Figure 3.
Traditional Chinese Medicine (TCM) plays a Role in Modulating Various Signaling Molecules Associated with Osteoblastogenesis.
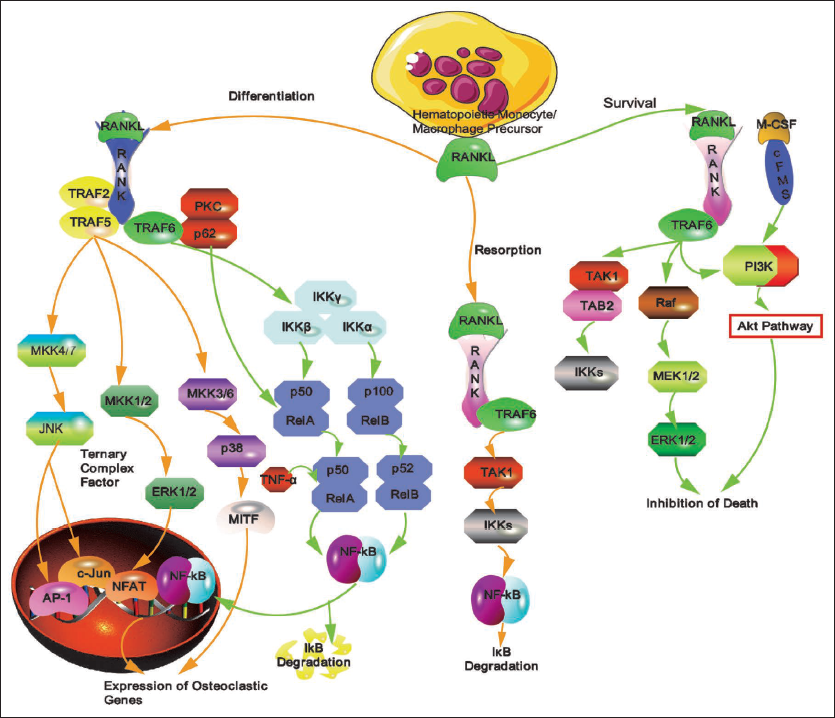
Inflammatory Arthritis
Inflammatory arthritis is an autoimmune disease characterized by the activation of immune cells, including T and B cells, macrophages, and neutrophils, within the joint spaces in Figure 4 (Findlay & Haynes, 2005; Haque et al., 2021; Kondo et al., 2021). This activation triggers an inflammatory response marked by elevated levels of cytokines such as TNF, IL-1β, IL-17, and IL-6, detectable in synovial fluids and serum. These cytokines activate NF-κB signaling and often synergize with RANKL to exacerbate osteoclastogenesis and osteolysis. Therapies targeting TNF, IL-1, and IL-6 have shown promise in treating rheumatoid arthritis and improving bone health (Chindamo et al., 2020; Tanaka, 2019; Zamri & de Vries, 2020; Zhang, 2021).
The Role of Traditional Chinese Medicines (TCMs) Involves Modulating Receptor Activator of NF-κB Ligand (RANKL) Signaling Molecules to Influence Osteoclastogenesis and Osteolysis. TCMs are Crucial in Addressing Inflammatory Arthritis-related Disorders by Regulating Activated Immune Cells such as T and B Cells, Macrophages, and Neutrophils within the Joint.
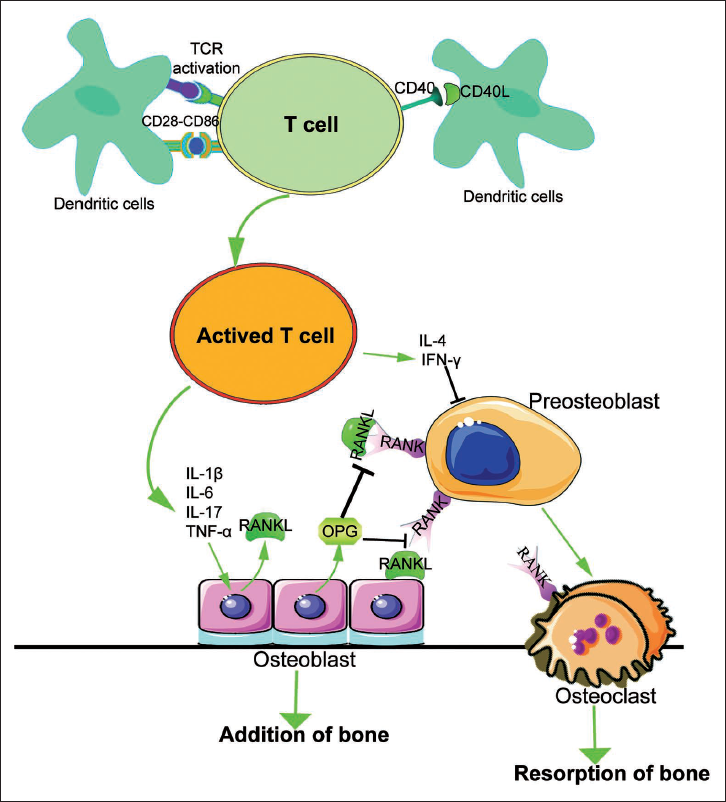
Direct inhibition of NF-κB, using strategies such as NEMO-binding peptides (NBD), super-repressor IκB, IKK2 inhibitors, and agents blocking NF-κB nuclear activation, has demonstrated significant anti-inflammatory and anti-osteolytic benefits (Abu-Amer & Faccio, 2006). Studies in mouse models of rheumatoid arthritis have provided valuable insights. In collagen-induced arthritis (CIA), T cell activation produces TNF and interferon gamma (IFNγ), regulated by NF-κB. Intervening at the level of NF-κB is beneficial; transgenic mice expressing super-repressor IκB exhibit milder CIA symptoms and lower circulating levels of IFNγ and TNF (Simmonds & Foxwell, 2008). Another mouse model showed elevated NF-κB in synovial tissue. Blocking NF-κB using virally expressed and cell-permeable peptide-conjugated super-repressor IκB and NBD ameliorated rheumatoid arthritis (Herrington et al., 2016).
Periprosthetic Osteolysis
Inflammatory osteolysis in orthopedic implant failure is primarily caused by wear debris activating macrophages and osteoclasts around the implant–host interface. This triggers inflammatory and osteolytic responses, leading to implant loosening and failure, which is particularly challenging in elderly patients with compromised bones. The pathological response involves recruiting various cell types, including macrophages and osteoclasts, that are responsive to immune cues. Recent research, including our own, has identified crucial cellular components contributing to inflammatory osteolysis. Orthopedic particles such as polyethylene, titanium alloy, and polymethylmethacrylate (PMMA) stimulate monocytes/macrophages through NF-κB and MAPK pathways (Jones & Buckle, 2020; Yin et al., 2023).
PMMA particles activate TAK1, a key signal transduction regulator, leading to NF-κB and activator protein (AP-1) activation. PMMA induces ubiquitination of NEMO, likely mediated by TRAF6, TAK1, and UBC13. Using a NEMO-K392R mouse model, where lysine 392 was replaced, it was demonstrated that polyubiquitination is crucial for mediating inflammatory osteolysis. Mice expressing NEMO-K392R resist orthopedic particle-induced calvarial osteolysis (Adapala et al., 2020). Inhibiting NF-κB and MAPK pathways with inhibitors and NEMO decoy molecules effectively hinder PMMA-induced osteolysis in experimental calvarial osteolysis mouse models.
PDB
Patients with PDB experience osteolytic lesions due to localized osteoclast hyperactivity, leading to poorly remodeled, fracture-prone bones (Gennari et al., 2022). PDB involves abnormal TRAF6-distal signaling, mainly through the activation of NF-κB. Polyubiquitinations regulate NF-κB signaling and osteoclastogenesis, with p62 (sequestosome 1) recruiting deubiquitinating enzymes to TRAF6 for pathway regulation. Deletions or mutations in p62 or deubiquitinating enzymes result in sustained NF-κB activity, causing inflammatory osteolysis. Most p62 mutations occur in the ubiquitin-binding domain, highlighting the significance of abnormal polyubiquitination in the pathogenesis of PDB (Schimmack et al., 2017).
Osteoporosis
Osteoporosis, a prevalent metabolic skeletal disorder, results from hormonal imbalances, aging, and inflammation. Postmenopausal osteoporosis, primarily due to sex hormone deficiency, affects skeletal metabolism by regulating osteoblasts and osteoclasts. Estrogen, an essential hormone, acts anabolically by recruiting osteoblasts, promoting their differentiation, and inhibiting apoptosis (Callewaert et al., 2010; Chandra & Rajawat, 2021; Kim et al., 2020). It also modulates osteoclast activity by regulating cytokines such as TNFα, IL-1β, IL-6, M-CSF, GM-CSF, and prostaglandins, whose elevated levels in postmenopausal individuals contribute to bone loss (Oka et al., 2008; Stein & Yang, 1995). NF-κB, a transcription factor, mediates the osteolytic effects of these cytokines. Estrogen receptors non-genomically inhibit NF-κB, attenuating its binding to deoxyribonucleic acid (DNA) elements in osteoclasts (Lencel & Magne, 2011).
Osteoporosis can also result from low-grade inflammation, inflammatory bowel disease, and the presence of reactive oxygen species (ROS) due to aging. NF-κB activation associated with these conditions enhances osteoclastogenesis, leading to bone loss (Lencel & Magne, 2011; Romas & Gillespie, 2006). Studies suggest that ROS inhibitors such as simvastatin and α-tocopherol can inhibit NF-κB and osteoclast activity, offering potential pharmacological interventions for mitigating osteoporosis (Agidigbi & Kim, 2019; Moon et al., 2014; Nazrun et al., 2012).
Traditional Chinese Medicine (TCM)
The Nobel Prize was awarded to Tu Youyou for her discovery of artemisinin, which has significantly heightened global interest in TCM, especially in herbal treatments. TCM research is advancing at a remarkable pace both in China and internationally. Notable examples include herbal extracts such as Astragalus polysaccharides (derived from Astragalus propinquus), which have shown promise in lung cancer immunotherapy (Agidigbi & Kim, 2019), and Ginsenoside Rg-1 (extracted from Panax ginseng), which has been shown to promote angiogenesis and aid in diabetic wound healing (Bamodu et al., 2019; Cai et al., 2019).
TCM theory originated over 700 years before Christ and is grounded in ancient Chinese practices designed to combat diseases. It evolved through principles of simple materialism and spontaneous dialectics, and in 2018, was formally recognized by the World Health Organization as part of the International Statistical Classification of Diseases. Unlike modern medicine, TCM adopts a macroscopic perspective on physiology and pathology, viewing the human body as an interconnected system of physical structures, emotions, and spirit. This holistic approach has effectively managed the health of large populations for millennia (World Health Organization, 1994).
A fundamental tenet of TCM is the belief that humans are integral to the natural environment and mirror its ecosystem. The human body functions similarly to an ecological system, with the circulation of qi (the essential substance that maintains vital activities), blood, and jing (organ secretions and bodily fluids). The extensive theoretical framework of TCM aligns with various modern medical perspectives. Contemporary theories on immunity, hormones, psychology, and intestinal flora increasingly resonate with TCM, particularly evident in the concept of the “microenvironment” (Spill et al., 2020). Integrating the globally dominant modern medicine with TCM—a cornerstone of the Chinese healthcare system—presents an opportunity for effective synergy. This integration promises to enhance the efficacy of treatments by leveraging the strengths of both medical paradigms, ultimately improving health outcomes worldwide.
TCM on Osteoclastogenesis
Various TCMs have been widely employed to treat bone disorders, demonstrating remarkable efficacy in regulating both osteoclasts and osteoblasts. The dynamic balance between osteoblast-mediated bone formation and osteoclast-driven bone resorption primarily influences the physiological and pathological aspects of bone volume fraction (Figure 1). In several pathological conditions, such as osteoporosis, the primary cause of bone loss is the excessive bone resorption caused by overactive osteoclasts. Current treatment strategies for osteoporosis can be categorized into three main types of drugs: anti-resorptive agents (inhibiting osteoclast function), bone-forming agents (enhancing osteoblast activity), and dual-effect drugs (simultaneously promoting bone formation and inhibiting osteoclasts) in Table 1.
List of Traditional Chinese Medicines (TCMs) that Act on the Osteoclastogenesis by Nuclear Factor Kappa-B (NF-κB) Pathway.
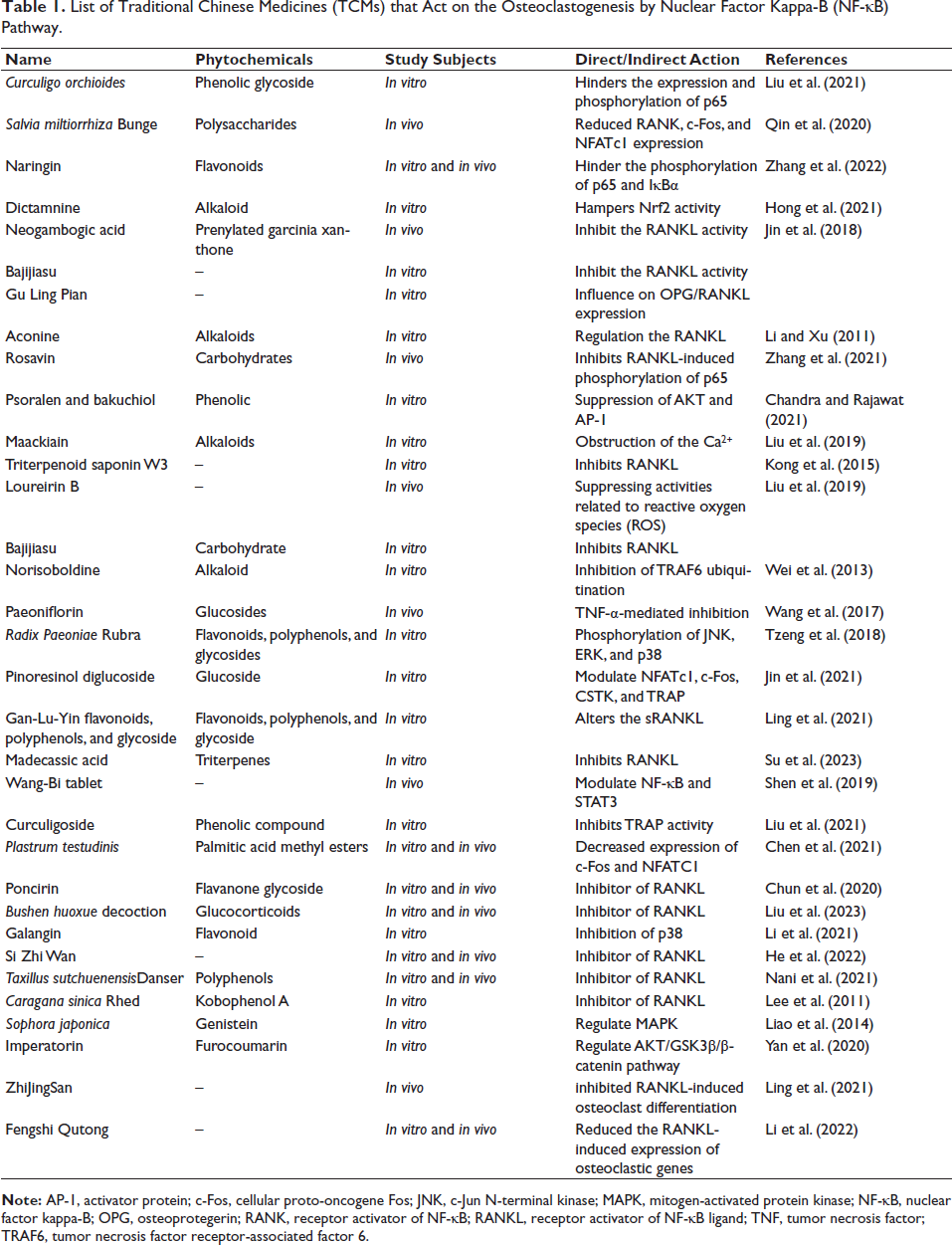
Inhibitors of Osteoclastogenesis in TCM
For instance, Curculigoside (Cur), a natural phenolic glycoside metabolite derived from Curculigo orchioides, is widely used in TCM. Studies have shown that Cur effectively inhibits the expression and phosphorylation of p65, thus blocking the NF-κB signaling pathway. This mechanism further reduces the excessive production of ROS induced by hydrogen peroxide (H2O2), as well as osteoclast differentiation. By inhibiting the NF-κB signaling pathway, Cur significantly reduces osteoclast formation (Liu et al., 2021).
Similarly, a combination of Salvia miltiorrhiza (Danshen) and Pueraria montana var. lobata (Gegen) (DG) has been shown in animal studies to regulate the NF-κB pathway, significantly reducing RANK, c-Fos, and NFATc1 expression. This leads to reduced NF-κBp65 phosphorylation and ROS production, effectively suppressing osteoclast differentiation (Qin et al., 2020). Additionally, Naringin has been found to inhibit the phosphorylation of p65 and IκBα, mitigating oxidative stress and reducing osteoclast formation in intervertebral discs (Zhang et al., 2022). These studies underscore the efficacy of TCM components in inhibiting osteoclastogenesis by modulating the NF-κB signaling pathway.
Dual-mechanism Modulators of Both Osteoclast and Osteoblast Activity
Certain TCM components not only inhibit osteoclastogenesis but also promote osteoblast activity, providing dual modulation of bone metabolism. For example, dictamnine (DIC), an innovative furanquinoline alkaloid, demonstrates specific binding to RANKL, effectively inhibiting Nrf2 activity and reducing ROS production. Studies have shown that DIC inhibits NFATc1 expression and the activation of the NF-κB signaling pathway, significantly reducing osteoclast formation and bone resorption. This dual action suggests its potential in regulating osteoclastogenesis and overall bone metabolism (Hong et al., 2021). Neogambogic Acid (NGA), another TCM component, modulates c-Jun N-terminal kinase (JNK) expression and NFATc1 activation, inhibiting osteoclast differentiation and reducing bone resorption. The monomeric nature of NGA makes it a promising alternative treatment for osteoporosis.
TCM Components that Promote Osteoblast Activity through Cellular Signaling Pathways
Some TCM components enhance bone formation by promoting osteoblast activity by regulating cellular signaling pathways. For instance, Gu Ling Pian (GLP) regulates OPG/RANKL expression via the p38 MAPK pathway, promoting osteoblast differentiation, maturation, and enhancing bone formation (Hong et al., 2021). This dual-action mechanism makes GLP a promising candidate for preventing osteoporosis. Similarly, Rosavin inhibits osteoclastogenesis by suppressing RANKL-induced p65 and IκBα phosphorylation while promoting bone formation in BMSCs. This dual functionality positions Rosavin as a potential treatment for osteoporosis by inhibiting bone resorption and enhancing bone formation (Zhang et al., 2021).
Through the regulation of key signaling pathways such as NF-κB, NFATc1, and ROS, TCM components exhibit broad effects in modulating osteoclastogenesis and bone metabolism. These components not only inhibit osteoclast differentiation and function, but many also enhance osteoblast activity, contributing to increased bone formation. This dual regulation highlights the potential of TCM for treating degenerative bone diseases such as osteoporosis. The mechanisms through which these components act not only improve the control of bone metabolism but also offer promising avenues for developing new therapeutic strategies for osteoporosis and other bone resorptive diseases.
Conclusion
In conclusion, the impact of TCM on osteoclastogenesis through the NF-κB pathway represents a promising area of research. Various TCM herbs and formulations have demonstrated anti-osteoclastogenic effects by modulating key signaling pathways, providing a foundation for developing novel therapeutic approaches for bone disorders. As research in this field progresses, the integration of TCM and conventional medicine may offer more comprehensive and effective strategies for preventing and treating osteoclast-related pathologies.
Footnotes
Abbreviations
AP-1: Activator protein; c-Fos: Cellular proto-oncogene Fos; CIA: Collagen-induced arthritis; FACS: Fluorescence-activated cell sorting; IKKα/IKKβ: IκB kinase alpha/beta; MAPK: Mitogen-activated protein kinase; M-CSF: Macrophage colony-stimulating factor; NF-κB: Nuclear factor kappa-B; NIK: NF-κB-inducing kinase; OPG: Osteoprotegerin; PDB: Paget’s disease of bone; PMMA: Polymethylmethacrylate; RANK: Receptor activator of NF-κB; RANKL: Receptor activator of NF-κB ligand; ROS: Reactive oxygen species; TCM: Traditional Chinese Medicine; TNF: Tumor necrosis factor; TRAF6: Tumor necrosis factor receptor-associated factor 6.
Authors Contribution
JJH designed the study; DW collected and analyzed the data; YZ drafted the initial manuscript; YX and JQH reviewed the article; BY are the correspondence authors. All authors approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
None.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported financially by Research Program on Sports Issues in Henan Province (Grant number: 202320); Henan Province Traditional Chinese Medicine scientific research projects (Grant number: 2019JDZX095); National TCM Clinical Characteristics Technology Inheritance Talent Program [Letter of Humanistic Teaching on Chinese Medicine (2019) No. 96].
