Abstract
Objectives
The objective of this investigation was to assess the subacute oral toxicity of the Pankajakasthuri Orthoherb Tablets, a polyherbal formulation used to manage pain and inflammation due to various pathological reasons, including osteoarthrosis, polyarthritis, rheumatoid arthritis, and other chronic degenerative disorders.
Materials and Methods
The subacute toxicity studies of Pankajakasthuri Orthoherb Tablets were conducted using Wistar albino rats of both sexes, according to Organization for Economic Cooperation and Development-approved procedures. In this study, rats were orally administered Pankajakasthuri Orthoherb Tablets daily at doses of 250, 500, and 1000 mg/kg body weight for 28 days. Throughout the study period, general behavior, mortality, and adverse effects were recorded. Body weight, selected hematological and clinical chemistry parameters were determined at the end of the investigation period. Finally, histopathological analysis was performed to determine whether the study drug was inducing any alternations in the vital organs.
Results
Daily administration of Pankajakasthuri Orthoherb Tablets for a continuous 28 days did not record any major toxicity indications. Additionally, no significant variation in food and water consumption, body weight, relative organs and hematology and clinical chemistry parameters was recorded in both sexes compared to the control. Histopathological investigation of the vital organs also revealed normal architecture, which suggests no morphological alterations after the administration of Pankajakasthuri Orthoherb Tablets.
Conclusion
The outcomes of this investigation confirmed that Pankajakasthuri Orthoherb Tablets did not have the potential to induce toxicity signs in the experimental rats. The No observed Adverse Effect Level (NOAEL) for female and male rats of Pankajakasthuri Orthoherb Tablets under these experimental conditions was 500 mg/kg body weight. Based on the NOAEL, the human equivalent dose of the Pankajakasthuri Orthoherb Tablet was calculated at 5.195 g/day.
Keywords
Introduction
Polyherbal formulations have gained widespread acceptance and popularity in the treatment of various diseases and disorders due to their provenance and the fact that polyherbals possess some benefits not found in a single herb. Even if a herbal medicine is safe or low toxic for short-term use, repeated or long-term use may cause some adverse side effects. Furthermore, different polyherbal formulations have earlier been reported to cause several adverse effects, which include hematological and biochemical toxicity, organ toxicity, and teratogenicity (Roome et al., 2021; Teshome et al., 2021; Ugwah-Oguejiofor et al., 2019). As a result, toxicological studies at the preclinical stage are a required step for every industrialized drug in the pharmaceutical industry. When conducted in a suitable animal model, these studies allow for the identification of drugs’ toxic potential. Toxicological investigations include acute, subacute, and chronic drug exposure, and they provide scientific evidence of the drug’s safety spectrum as well as the drug’s toxic response to specific organs of the body (Parasuraman, 2011). The laboratory animals used in these studies are typically low cost, simple to care for and produce accurate and reliable results. The usage of herbal medicines among people is increasing in most countries every year, owing to the mistaken belief that ayurvedic preparations are free of side effects, despite cases reported in Pharmacovigilance (Leenaars et al., 2019). To ensure the safety of medicinal herbs within a scientific framework before they are used as therapeutics, toxicity studies are essential.
Pankajakasthuri Orthoherb Tablets, a polyherbal Ayurvedic formulation, was formulated by Herbals India Pvt. Ltd. This tablet was proven to have a major role in managing the pain and inflammation in joints due to osteoarthritis, polyarthritis, rheumatoid arthritis, and other chronic degenerative disorders. This polyherbal formulation contains active herbal ingredients like Ricinus communis, Adathoda vasica, Aegle marmelos, Azadirachta indica, Sida retusa, Tragia involucrata, Abrus precatorius, Cyclea peltata, Scoparia dulcis, etc. A clinical trial of Pankajakasthuri Orthoherb Tablets revealed a significant role in joint pain and inflammation management. Continuous, long-term administration of this formulation is required to manage the pain and inflammation associated with joints and this may raise questions regarding the safety profile. Despite the presence of several therapeutically beneficial secondary metabolites, plants contain a number of toxic phytochemical constituents. Continuous use of these plant-based therapeutic agents in humans may result in chronic adverse/toxic effects, primarily renal and hepatic toxicities. Emerging evidence suggests that taking certain plant remedies used in traditional medicine on a regular basis may have negative effects, particularly at higher doses than the therapeutic dose (Liyanagamage et al., 2020). As a result, the current study aims to assess the toxicity profile of this polyherbal formulation in Wistar albino rats. This study’s findings may fill a gap in the literature regarding the long-term safety profile of this herbal product.
Materials and Methods
Reagents and Chemicals
For this study, ethanol (99.9%) was purchased from Sigma Aldrich Ltd., India. Ethylene diamine tetra acetic acid (EDTA) was obtained from Thermo Fisher Scientific India Pvt. Ltd., Mumbai, India. The other chemicals and reagents used in this study were all of analytical grade.
The Study Drug: Pankajakasthuri Orthoherb Tablets
The study drug is Pankajakasthuri Orthoherb Tablets (500 mg), a polyherbal Ayurvedic preparation in tablet form for oral usage. Pankajakasthuri Herbals India Pvt. Ltd. (Poovachal, Thiruvananthapuram, Kerala, India), a GMP-approved company, manufactured this tablet.
Animals
Sixty healthy Wistar albino rats (30 males and 30 females) were obtained from the CARe KERALAM Ltd. in-house breeding centre of the small animal house laboratory. The study strictly followed laboratory animal care rules and prior approval was obtained from the IAEC of CARe KERALAM Ltd. for performing the experiments (Study No: CKL/TOX/IAEC/2021-3/164).
Subacute Toxicity Studies of Pankajakasthuri Orthoherb Tablets
A subacute toxicity investigation of the Pankajakasthuri Orthoherb Tablet was carried out on Wistar albino rats strictly based on the procedures depicted in OECD Guidelines 407 (OECD, 2008).
Grouping of Animals for the Subacute Toxicity Study and Dosing Regimen
For the current study, 60 healthy Wistar albino rats, aged 8−10 weeks, with average body weights of 200−280 g for males and 155−205 g for females, were used. These rats were allocated into six groups (five/sex/group) based on body weight categorization and randomization (Table 1). The rats were grouped so that the average body weight variation did not surpass 25% of the mean body weight of each sex.
Animal Grouping for the Experiment.
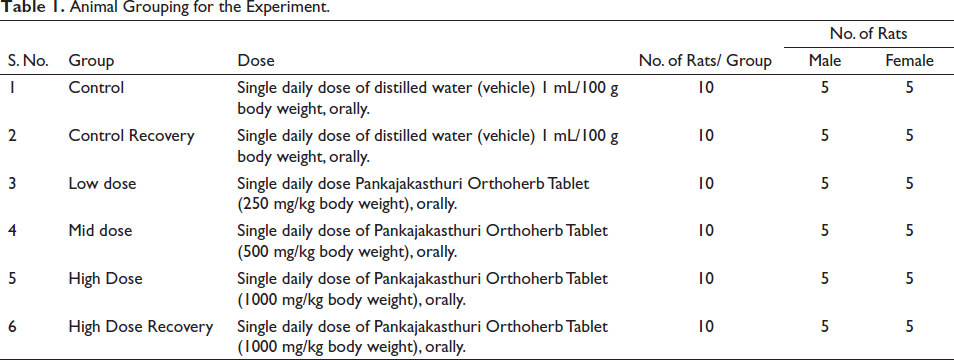
Throughout the study period, all rats were allowed to access food and water freely until the day before scheduled euthanasia.
Observations of Various In-Life Clinical Parameters
Throughout the study period, the mortality of the rats was observed twice every day. The observation of clinical signs in the rats of both control and treatment groups was recorded during the time of drug administration and in the open field on the 0th, 7th, 14th, 21st, and 28th day. Rats from the satellite group were noted further on the 35th and 42nd days after the initiation of the study. The major clinical signs noted were variations in sensory organs (skin, eyes, and nose), secretion of the body, autonomic activities (respiratory rate, pupil size, and piloerections), walking pattern, muscle tremors and other behaviors, and so on (Anderson & Meade, 2014).
Body Weights
Before the initiation of the investigation, the body weight of each rat was recorded. Subsequently, body weight was recorded at weekly intervals until the termination of the study.
Consumption of Food and Water
The consumption of weekly food and water by the rats was noted by calculating the disparity between offered and left-over food and water in subsequent weeks.
Hematology Analysis
After finishing the treatment period, that is, on the 28th day and 42nd day (recovery group), rats fasted overnight and the blood samples were carefully collected by cardiac puncture with EDTA anticoagulant to determine the following hematological parameters according to good laboratory practices by the Medonic M series hematology analyzer. The following hematological parameters were examined: hemoglobin (Hb), leukocyte count (total WBC), erythrocyte count (total RBC) and platelet count.
Serum Analysis
The serum was collected by centrifuging the blood samples (without anticoagulant) at 1500× g for 10 min. Thus, the obtained serum was stored at −20°C for further investigation. Total protein (TP) and albumin, glucose, aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), total cholesterol, triglycerides (TGs), bilirubin (total and direct), urea, and creatinine. These parameters were evaluated using a Vital Scientific − Selectra Junior and standard diagnostic test kits (Quality Labs).
Histopathological Observation
On the 29th day, a necropsy was performed on both the control and treatment groups, whereas in the satellite groups, this was performed on the 43rd day. Following blood collection, rats were sacrificed via cervical dislocation, and vital organs including the liver, lungs, kidney, heart, spleen, and brain were removed. The organs were meticulously cleaned to remove the fat content and blotted with clean tissue paper to remove the water content. Following that, the weight of the organs was recorded using a digital balance. The relative organ’s weight (ROW) was calculated and recorded in proportion to the body weight based on the following equation:
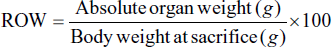
The tissue section obtained from the vital organs was subjected to histopathological analysis. First, the tissue was fixed in 10% buffered formalin, followed by habitual processing, and then embedded in paraffin wax. With the assistance of an experienced pathologist, paraffin sections (5 µm) were cut on glass slides and stained with hematoxylin and eosin. The slides were observed under a light microscope (Nikon E50i, Nikon Corporation, Japan), as mentioned elsewhere, to determine the toxicity signs.
Statistical Analysis
The data on body weight, food intake, water intake, hematology, and clinical chemistry generated from the present study were subjected to computer statistical analysis using GraphPad Prism software, Version 5.00, USA, 2007. A one-way analysis of variance (ANOVA) was executed for the treatment groups and compared with the data of the control group. All analyses and comparisons were assessed at a 5% significance level.
Results
Clinical Observation
After the administration of the Pankajakasthuri Orthoherb Tablets, the rats were subjected to general observations on a daily basis. All the rats were subjected to clinical observation, which was conducted outside the cage. Individual rats were observed 30 min after the study drug was administered, then every 24 h and then daily until the study was completed. In the present study, none of the rats recorded any observable toxicity signs, such as variations in skin and fur, eyes or mucus membranes, when compared with the control. In addition to this, the rats did not record any behavioral changes, including respiratory function, tremors, convulsions, salivation, diarrhea, lethargy, sleep or coma, until the termination of the experiment.
Furthermore, in this study, no mortality occurred in the rats administered orally with Pankajakasthuri Orthoherb Tablets daily for 28 days at doses of 250 mg/kg, 500 mg/kg, and 1000 mg/kg body weight.
Body Weights and Body Weight Changes
The effect of Pankajakasthuri Orthoherb Tablets on the mean body weight of the experimental rats in all treatment groups is provided in Figure 1. From the outcomes, it was clear that a steady upsurge in the mean body weight was recorded in both the control and treated groups during the 28-day study period. In addition to this, no statistical difference in the mean body weight was recorded for rats in the drug-treated as well as the control groups (p > 0.05). At the end of 7, 14, 21, and 28 days, there was no noteworthy difference in the percentage of body weight gain between the treated group and the control group.
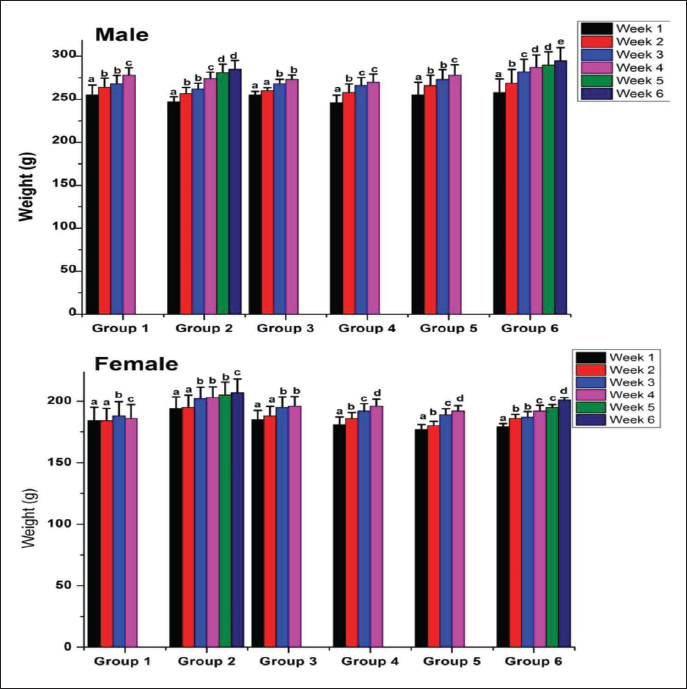
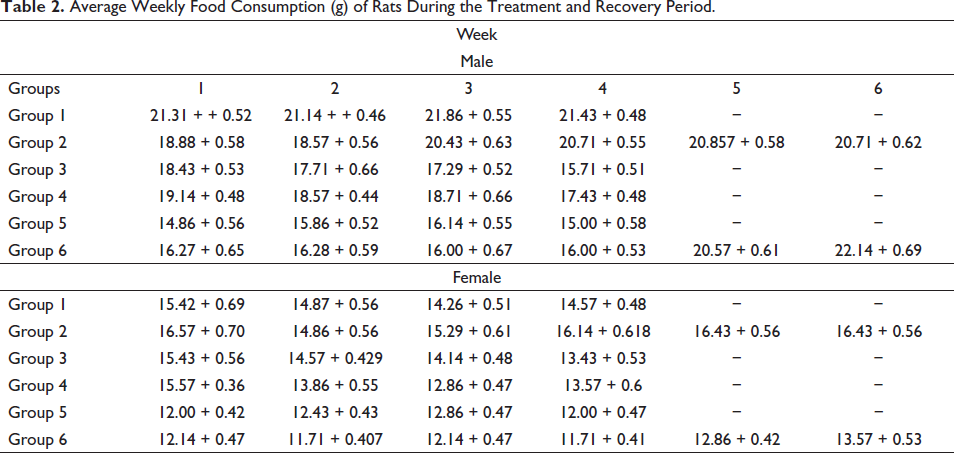
During the entire study period, no variation in the weekly food consumption of drug treated and control group rats was recorded (p > 0.05) (Table 2). No noteworthy decrease in food consumption was recorded in the drug-treated, control, or satellite groups. Throughout the study period, the water consumption of rats in all treated groups was comparable with that of control groups.
Average Weekly Food Consumption (g) of Rats During the Treatment and Recovery Period.
Effect of Pankajakasthuri Orthoherb Tablets on Hematological Parameters
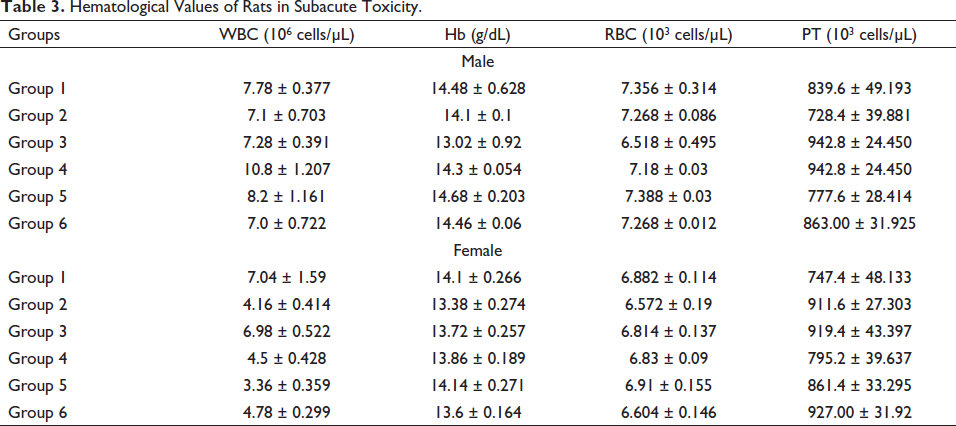
The consequence of Pankajakasthuri Orthoherb Tablets on hematological values (Table 3) of the treatment and control groups recorded that all the hematological values remained within the physiological range during the 28-day treatment period.
Hematological Values of Rats in Subacute Toxicity.
Outcome of Pankajakasthuri Orthoherb Tablets on Serum Marker Enzymes
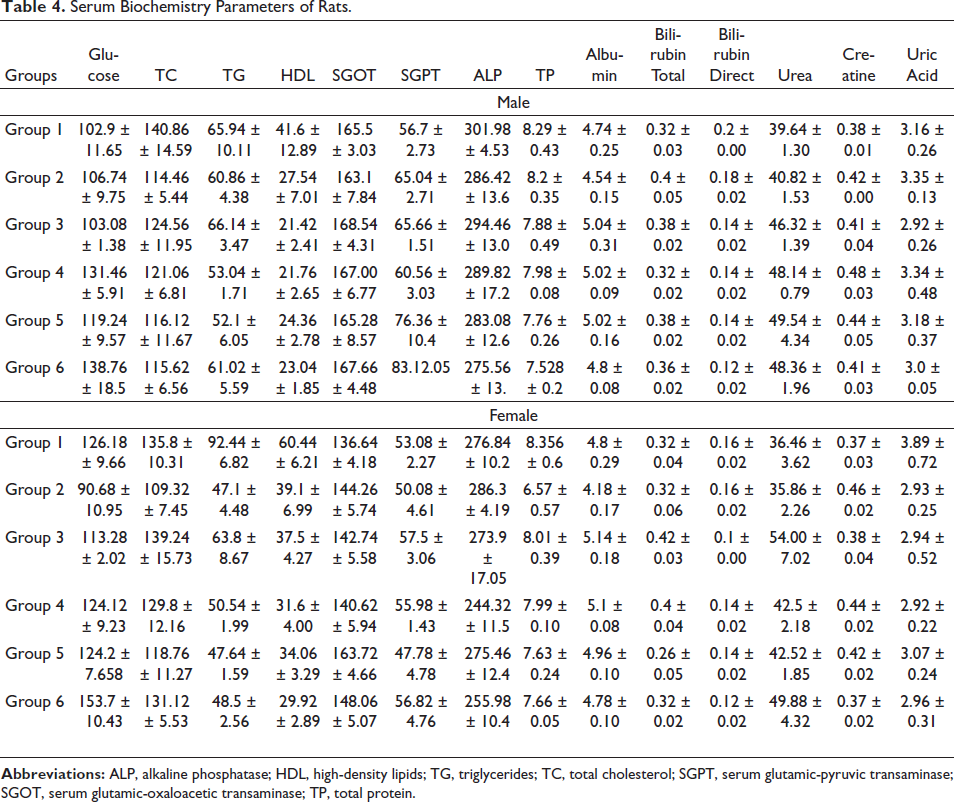
The outcome of serum biochemistry studies (Table 4) displayed that there was no noteworthy variation in the levels of serum glucose, ALT, AST, and ALP in both male and female rats treated with the study drug when compared to the control group.
Serum Biochemistry Parameters of Rats.
Outcome of Pankajakasthuri Orthoherb Tablets on Serum Albumin, Urea, Uric Acid, Creatinine, and Total Protein
The outcome of the use of Pankajakasthuri Orthoherb Tablets on serum total protein, albumin, urea, and creatinine is shown in Table 4. The rats administered with various doses of Pankajakasthuri Orthoherb Tablets did not record any significant difference in the levels of serum total protein, albumin, serum urea, and creatinine in both sexes when compared to the control group.
The Outcome of Pankajakasthuri Orthoherb Tablets on the Lipid Profile
The outcomes of Pankajakasthuri Orthoherb Tablets on the levels of serum TG and TC are shown in Table 4. A substantial decrease in serum TG levels was recorded in the rats administered with Pankajakasthuri Orthoherb tablets when compared to the control group. Furthermore, no significant variation in serum TC was recorded in the rats treated with Pankajakasthuri Orthoherb Tablets at all test doses when compared to their respective control groups.
Outcome of Pankajakasthuri Orthoherb Tablets Serum Electrolytes Level
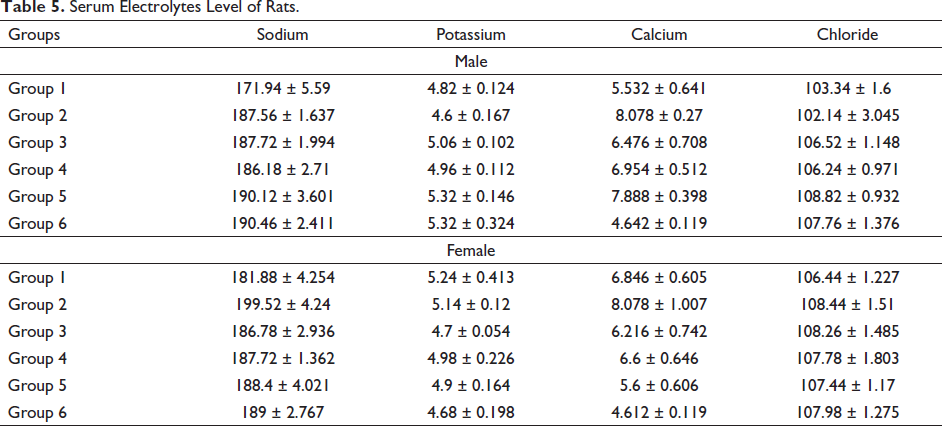
The analysis of serum electrolytes after treatment with Pankajakasthuri Orthoherb Tablets did not show any difference between the treatment and control groups (Table 5).
Serum Electrolytes Level of Rats.
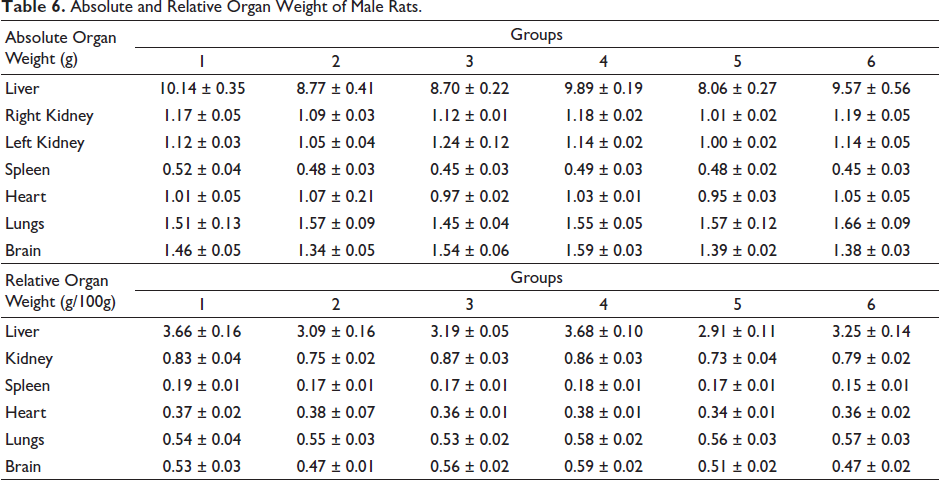
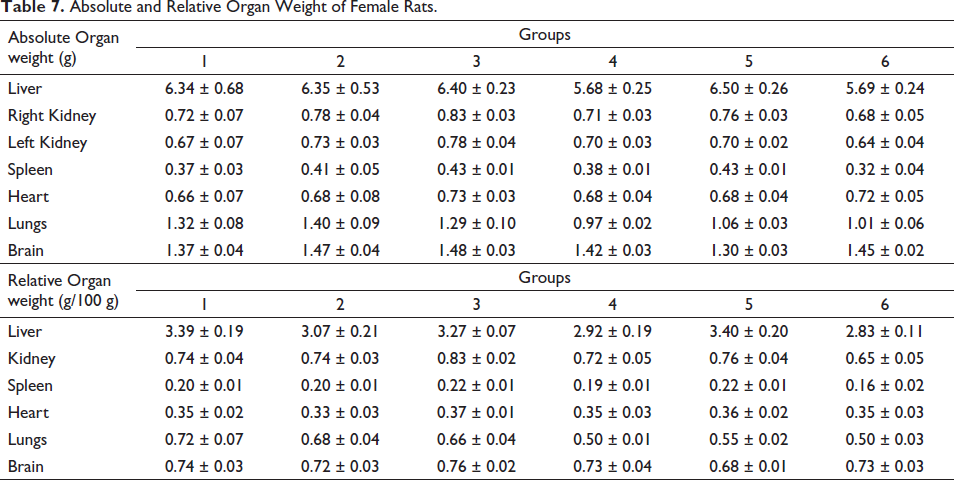
Effect of Pankajakasthuri Orthoherb Tablets on Relative Organ Weight
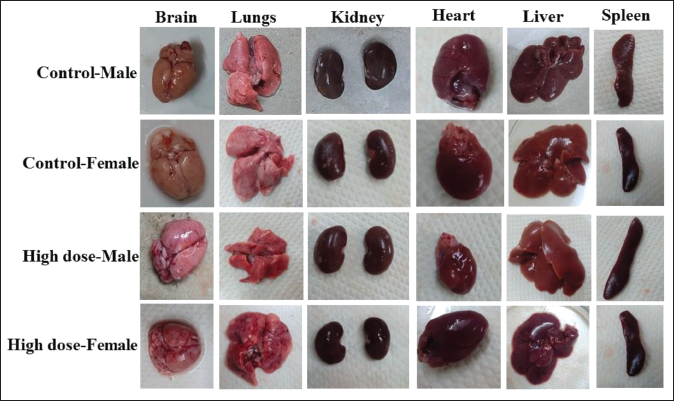
After the study period, we meticulously conducted a macroscopic examination, which comprises the observation of body systems (gastrointestinal, respiratory, excretory, and reproductive) and organs (lungs, brain, pancreas, sciatic nerve, skeletal muscles, adrenal gland, heart, liver, and spleen). In the present study, macroscopic examination after treatment with Pankajakasthuri Orthoherb Tablets recorded no major alteration in the gross anatomical structures of major organs of both male and female rats as compared to control group rats (Figure 2). Furthermore, rats in the satellite group also noted no major changes in the anatomical features of the major organs.
Photograph of Major Organs from the Subacute Toxicity Study.
No significant decrease in relative organ weights was recorded in the treatment or control groups (Tables 6 and 7).
Absolute and Relative Organ Weight of Male Rats.
Absolute and Relative Organ Weight of Female Rats.
Histopathological Analysis
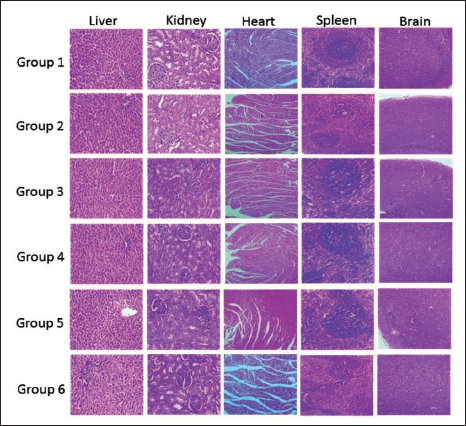
A histopathological investigation was conducted to verify the structural variations that occurred due to the treatment with the study drug. No gross pathological lesions were observed in the liver, kidney, heart, brain, and spleen of rats treated with the study drug when compared with the control groups (Figures 3 and 4).
Photomicrograph of Vital Organs of Male Rats from Different Groups After Treatment with Pankajakasthuri Orthoherb Tablets.
Photomicrograph of Vital Organs of Female Rats from Different Groups After Treatment with Pankajakasthuri Orthoherb Tablets.
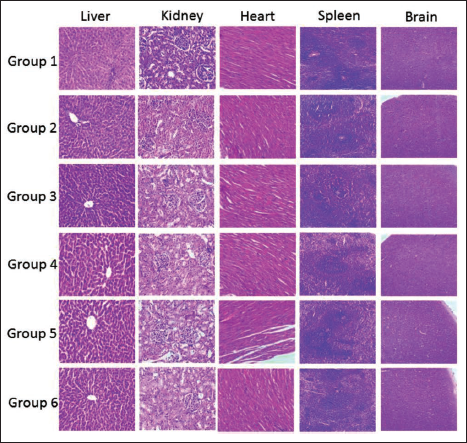
The microscopic images of the liver and kidney of the male and female rats in the control and drug-treated groups recorded regular morphological features. Further rats in the satellite group also recorded regular morphological features. The histopathological examination of the liver of the study drug-treated rats recorded normal features, including binucleation. Moreover, regular cellular architecture without any distortions, similar to the control groups, was also observed. Additionally, signs of injury, necrosis, congestion, accumulation of fatty acids, or hemorrhagic regions around the central vein or sinusoids of the liver were not recorded in our study. In the case of the histopathological analysis of kidneys, there was no morphological difference observed in all rat groups treated with Pankajakasthuri Orthoherb Tablets. The glomerular architecture of the treated and control rats appeared to be normal. The glomeruli, distal, and proximal tubules in the kidneys of both treated and control groups recorded normal structural features. In both groups, all the nephron cells recorded normal and evidently visible nucleoli with no degeneration, bleeding, or necrosis in the sub-acute oral toxicity study of Pankajakasthuri Orthoherb Tablets.
In our study, cardiac muscle fibers were recorded as normal in both control and Pankajakasthuri Orthoherb Tablets-treated rats of both sexes. Likewise, the spleen also recorded normal structure and histology in all rats in the subacute oral toxicity study. To conclude, histopathological assessments of the vital organs did not disclose any major morphological irregularities due to 28 days of continuous oral administration of Pankajakasthuri Orthoherb Tablets to the rats.
Discussion
The use of complementary and alternative medicine dates back many years. Additionally, it is an evidence-based medicine due to its popularity and extensive usage for several years (Catarino et al., 2016). A factual approach and a lack of regulation are frequent and have a negative effect. This might be brought on by a dearth of relevant scientific research data and research methodologies for drug evaluation (Mazumder et al., 2016). As a result, a number of herbs and their preparations have been found to be toxic globally (Agarwal et al., 2012; De Smet, 2004; Skalicka-Woźniak et al., 2017). Concerning the potential toxic effects of numerous herbal remedies and/or commonly consumed herbal preparations, the Food and Drug Administration has issued warnings (De Smet, 2004). It has been established that medicinal plants used around the world carry toxicological risks (Paré et al., 2019). To increase market acceptance, standardization, and/or regulation of the herbal medicine market as it is currently practiced, safety tests are required. According to accepted standards, acute and subacute toxicity must be evaluated before any pharmacological validation or the development of a phytomedicine from any medicinal plant (El-Gazayerly et al., 2014). Hence, the present study was conducted to study the repeated-dose subacute toxicity of Pankajakasthuri Orthoherb Tablets in Wistar rats.
In this investigation, no treatment-related morbidity, mortality, or clinical symptoms of local or systemic toxic effects of the study drug were recorded during the entire investigation period. The general behavior of the rats was noted daily (overall health and various clinical signs or symbols of toxicity), and the results clearly demonstrated that no changes were recorded during the entire study period (Kharchoufa et al., 2020). Thus, it can be concluded that the study drug does not impart any treatment-related behavioral change in the experimental animals.
Generally, variation in body weight is one of the most significant indicators of adverse side effects following exposure to any toxic substance in animals. Usually, animals cannot survive when a loss of more than 10% of their initial body weight occurs (Féres et al., 2006). Moreover, the health status of experimental animals is directly associated with variations in body weight. In this investigation, the rats from neither the treatment nor the recovery groups recorded any statistically significant changes in their body weights compared to their respective control groups.
In any toxicity investigation, hematology also plays a vital role in assessing the probable toxic side effects caused by the oral administration of the test drug (Nair & Sasidharan, 2021; Rao, 2006; Yakubu et al., 2007). It is considered that bone marrow is one of the major subtle targets of toxic agents that enter the body. The health status of the bone marrow and intravascular system of the experimental rats can be closely observed by their hematological values (Ekanayake et al., 2019). Furthermore, variations in the hematological parameters of drug-administered rats have an advanced extrapolative value for developing toxicity in human beings (Ekanayake et al., 2019). In our subacute oral toxicity investigation, all hematological values in rats administrated with Pankajakasthuri Orthoherb Tablets at a dose level of 500 mg/kg body weight were comparable with the values of control group rats.
In addition to this, evaluating the biochemical parameters of blood and exploring toxic effects on vital organs, especially the liver and the kidneys, in experimental animals may deliver useful data concerning the mode of toxicity of the test agents (Yamthe et al., 2012). Hence, the clinical biochemistry parameters of the treatment as well as recovery groups were linked with the particular control groups to know whether the study drug was inducing any kind of toxicity or not.
The enzymes AST, ALT, ALP, and serum bilirubin are measured as vital markers for knowing liver function (Arneson & Brickell, 2007). The damage to the liver cells is characterized by a noteworthy upsurge in the levels of serum AST and ALT. Furthermore, serum total protein and albumin are also considered non-enzymatic markers of liver toxicity. In male rats, the administration of Pankajakasthuri Orthoherb Tablet does not induce any toxicologically noteworthy variations in the serum levels of AST, ALT, ALP, bilirubin, TP, or albumin, and this clearly demonstrates that liver function was preserved during the study period. However, in female animals, those treated with a high dose (1000 mg/kg body weight) of Pankajakasthuri Orthoherb Tablet recorded a mild elevation in the AST levels when compared to normal control rats, and this was apparently normalized during the treatment withdrawal, as evident from the AST values of the animals treated with a high dose during the recovery period. Besides, in high dose-treated rats, we observed a significant elevation in total protein and a marked increase in albumin levels in recovery groups, suggesting improvement in liver function. Serum ALT, ALP, and bilirubin levels were equivalent to those of the respective normal controls in the female animals during treatment and recovery periods. A higher AST level without a consistent increase in ALT may rule out liver damage since AST is known to occur in large concentrations in the heart as well as liver tissues (Cheesebrough, 1981) with a small amount present in other tissues (Tan et al., 2007).
Kidney function was evaluated by means of serum urea, uric acid, creatinine, and electrolyte (Na, K, Ca, and Cl) values. In preclinical toxicity studies, changes in renal values are predominantly likely to occur because of the administration of high doses. Furthermore, the kidneys are the major excretory organs that eliminate many drugs and their toxic metabolites from the body (Schreiner & Maher, 1965). In the present investigation, a noteworthy elevation in serum urea concentration was noted in both sexes administered with 1000 mg/kg body weight, but a similar variation was not recorded in rats administrated with a higher dose (1000 mg/kg body weight) in the recovery period. However, serum uric acid and creatinine levels in the male and female treatment groups were comparable to those in the normal control groups. In male and female rats, electrolytes (K+, Na+, Ca2+, and Cl−) in serum were evaluated and did not show any statistically remarkable change within the treatment and recovery groups in comparison with the respective control groups. The outcome of serum biochemical analysis in our study indicates that the Pankajakasthuri Orthoherb Tablet does not have any important toxic effect on serum parameters up to a dose of 500 mg/kg body weight.
There is a very high possibility that some herbal medications may cause severe toxicity to vital organs because of their various roles in the human body. The assessment of histopathological alterations in organs continues to serve as the foundation for determining the safety of any substance (Coussanes et al., 2022). In the present study, administration of Pankajakasthuri Orthoherb Tablet for 28 days did not record any pathologically relevant lesions in the gross appearance of organs such as the liver, heart, lung, brain, kidney, spleen, stomach, ovary, or testis when compared to the control experimental rats. Another crucial marker of an animal’s physiological and pathological status is organ weight. The ROW is critical in determining whether or not the organ was injured. The primary organs affected by toxicant-induced metabolic reactions are the heart, liver, kidney, spleen, and lungs (Dybing et al., 2002). In the present investigation, no noteworthy variation in the absolute and relative liver weight was noted in the treated rats when compared to the control rats. Also, no significant variations were noted in the absolute and relative weights of the vital organs of rats in the treatment groups compared to the respective control groups. Hence, it can be concluded that the treatment with the Pankajakasthuri Orthoherb Tablet does not induce any change in the vital organs.
Conclusion
The outcome of this investigation clearly indicates that Pankajakasthuri Orthoherb Tablet, up to a dose of 500 mg/kg body weight (No Observed Adverse Effect Level [NOAEL]), does not record any kind of toxicity in rats when administered orally. This conclusion is presumed based on the fact that subacute oral treatment with Pankajakasthuri Orthoherb Tablet did not record any mortality or adverse side effects. Besides, the study drug did not induce noteworthy changes in any of the hematological, serum biochemical or histopathological values examined in this study. This is important evidence for the application of the Pankajakasthuri Orthoherb Tablet to patients. Based on the NOAEL, the HED of Pankajakasthuri Orthoherb Tablet was calculated at 5.195 g/day. More research into its therapeutic efficacy and biopharmaceutical applications is needed.
Abbreviations
NOAEL: No observed adverse effect level; HED: Human equivalent dose; GMP: Good manufacturing practices; IAEC: Institute animal ethics committee; ROW: Relative organ’s weight; Bwt: Body weight.
Footnotes
Acknowledgements
The authors express their gratitude to “Aadhyathma Chinthalayesan” of Chinthalaya Ashram, Pothencode, Trivandrum, Kerala, India, for his compassion and blessings. We are grateful to Pankajakasthuri Herbal Research Foundation, Kattakada, Thiruvananthapuram, Kerala, India, and Pankajakasthuri Herbals India Pvt. Ltd Poovachal, Kattakada, Thiruvananthapuram, Kerala, India, for helping us with the research. We would also like to thank all of the Directors and Employees of Pankajakasthuri Herbal Research Foundation and Pankajakasthuri Herbals India Pvt. Ltd for their assistance in the completion of this project. The authors would like to thank CARe KERALA in Koratty, Thrissur, Kerala, India, for providing the necessary infrastructure for the animal experiments.
Authors’ Contribution
HNJSS: Conceived and designed the experiments; SMSKNA: Performed the experiments; analyzed and interpreted the data; prepared the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
