Abstract
Background
Tanzanian communities use a herbal mixture of Euphorbia hirta and Plectranthus barbatus as a decoction to treat several diseases, including respiratory disorders, infertility, urinary tract infections (UTIs), dysmenorrhoea, and pelvic inflammatory disease. Despite its potential health benefits, there is a paucity of information regarding its safety and efficacy.
Objectives
This study examined the phytochemical profiles, acute and sub-acute oral toxicity of 1:1 (w/w) extract mixture of E. hirta and P. barbatus (EP), and evaluated the in vivo antimicrobial efficacy of the EP polyherbal formulation in albino mice and Wistar rats.
Methods
Phytochemical analysis of E. hirta and P. barbatus extracts, as well as the polyherbal formulation, was performed using Gas Chromatography-Mass Spectrometry (GC-MS), Liquid chromatography-Mass spectrometry (LC-MS), and Fourier Transform Infrared Spectroscopy (FTIR), respectively. The toxicity testing was conducted in accordance with Organisation for Economic Co-operation and Development (OECD) guidelines 423 and 407. Subsequently, the in vivo efficacy against Escherichia coli American Type culture Collection (ATCC) 25922 and Methicillin-Resistant Staphylococcus aureus (MRSA) ATCC 43300 was tested at doses of 50, 150, and 450 mg/kg in infected Wistar rats.
Results
E. hirta and P. barbatus extracts each contained 29 and 24 bioactive compounds, respectively, with known antimicrobial properties. In the acute toxicity test, the Lethal Dose 50% (LD
Conclusion
The polyherbal formulation demonstrates promising therapeutic efficacy and considered safe up to 500 mg/kg when administered repeatedly over 28 days.
Introduction
Folk medicine has been used alongside conventional remedies for centuries, particularly in developing nations with limited healthcare services. 1 Its growing popularity in healthcare emphasises the necessity of evaluating their safety and efficacy.1,2 Euphorbia hirta and Plectranthus barbatus are widely distributed and globally recognised for their therapeutic potential in treating many human illnesses. E. hirta (Euphorbiaceae family), locally known as Mziwaziwa, is traditionally utilised for treating respiratory diseases, diarrhoea, asthma, peptic ulcers, and fungal infections. 3 Several studies support its pharmacological activities, which are attributed to its rich phytochemical profile, including tannins, flavonoids, and triterpenoids. 4 Similarly, P. barbatus (Lamiaceae family), locally known as Mzungwa, has therapeutic benefits and has been traditionally used to treat hypertension, heart ailments, wounds, respiratory disorders, and abdominal pain. 5 The pharmacological benefits of P. barbatus are attributed to phytochemicals, including forskolin, glabrol and 6,8-diprenyl genistein, Coleonol B, and sugiol. 6 In Tanzania, several communities use a herbal mixture of E. hirta aerial parts and P. barbatus leaves as a decoction for managing an array of illnesses, including respiratory disorders such as coronavirus disease 2019, infertility, urinary tract infections (UTIs), dysmenorrhoea, and pelvic inflammatory diseases. The therapeutic potential of each component of this polyherbal mixture, concerning the mentioned medicinal benefits, has been described in various literature.7–10
Traditional medicine practitioners often employ combination therapy to enhance and broaden the therapeutic spectrum. 11 As such, the enhanced therapeutic outcome is achieved through synergistic and additive effects resulting from multiple interacting phytocompounds acting via various mechanisms against pathogens or certain health disorders.11,12 Despite studies revealing that many herb-herb extract combinations have demonstrated improvements in therapeutic outcomes, 11 they may also introduce the possibility of additive or new toxic effects. 13 The growing application of polyherbal remedies, therefore, necessitates toxicity evaluation to ensure their safety for human and livestock use. Currently, most toxicity studies on herbal plants primarily focus on individual extracts, while a significant portion of global herbal medicines are marketed as polyherbal mixtures. 12 In this regard, the use, registration, and commercialisation of such polyherbal medicines require validation of their safety, quality, and efficacy, which is ensured through toxicity and efficacy studies.
Several studies have evaluated the acute and sub-acute oral toxicity of E. hirta and P. barbatus extracts individually.14–17 These findings suggest that E. hirta and P. barbatus individual extracts are safe up to 5000 mg/kg as a one-time dose and up to 1000 mg/kg for repetitive, prolonged oral dose. However, evidence is scarce regarding the safety of their combined extracts, which will likely reflect their safety in polyherbal medicine formulations. Several studies disclose that certain polyherbal mixtures contain complex ingredients that may increase the likelihood of harmful effects due to the interaction of numerous phytocompounds.2,18 Thus, there is a pressing need to evaluate the safety of such polyherbal mixtures because the interactions of phytochemical constituents in the mixture may alter their toxicological profiles. On the other hand, previous studies have reported the in vitro antimicrobial efficacy of P. barbatus19,20 and E. hirta extracts21,22 against several pathogenic microbes. However, there is a lack of information on the in vivo antimicrobial efficacy of such herbal plants individually and as a polyherbal mixture in model animals.
Despite safety concerns about herb-herb combination remedies, these formulations are increasingly common in many global communities. 2 Furthermore, there is growing acceptance of herb-herb combination therapy, especially in treating chronic diseases such as diabetes, cancer, and Human Immunodeficiency Virus/Acquired Immunodeficiency Syndrome. 23 In this context, ensuring the safety, efficacy, and quality of such polyherbal formulations is therefore quite demanding for their acceptance in modern healthcare and regulatory approval systems. Thus, the current study evaluated the toxicity of 1:1 (w/w) extract mixture of E. hirta and P. barbatus (EP) in model animals, as well as the in vivo antimicrobial efficacy of their polyherbal formulation (granulated capsules). The study further characterised the phytochemical profiles of E. hirta and P. barbatus.
Materials and Methods
Authentication, Gathering, and Processing of Plant Samples
Plant materials from E. hirta and P. barbatus were collected in February and March 2024 at Nambala, Arusha, Tanzania. A botanist authenticated the samples and assigned voucher numbers MO03 and MO05 for E. hirta and P. barbatus, respectively. After processing, the voucher samples were stored at the National Herbarium of the Tanzania Plant Health and Pesticides Authority, Tanzania. The collected plant samples were cleaned and dried in the shade for two weeks. The dried samples were then ground into powder separately using an electric blender. The pulverised materials were extracted with 80% ethanol at a 1:5 w/v ratio of plant powder to solvent for 72 h, using the maceration method. 24 Before freeze-drying the extract, the remaining solvent was removed by using a rotary evaporator set at 50 ◦C.
Screening of Secondary Metabolites
The ethanolic extracts from E. hirta and P. barbatus were screened qualitatively for the main classes of secondary metabolites, including saponnins, alkaloids, flavonoids, tannins, terpenoids, steroids, and anthraquinone using procedures described by Gul et al 25 and Maria et al 26
Gas Chromatography-Mass Spectrometry (GC-MS) Analysis of Extracts
The EP extract, prepared at 0.4 mg/mL in ethanol, was then filtered. Afterward, 1 µL was injected into the Agilent GC-MS 7980, coupled with the Mass Selective Detector 5790 and 7860 auto-sampler (Agilent Technologies, Little Falls, United States of America), operated under helium gas (99.999%) at 1.2 flow/minute in split mode using an Agilent High-Performance 5% Phenyl Methyl Siloxane column at 68.3 kPa for analysis. The oven temperature was set to 60 °C with a 2-min hold, then ramped at 10 °C/minute to 150 °C with a 3-min hold. The temperature was then gradually raised to 250 °C at a rate of 5 °C/minute, with a final hold of 6 min. Concurrently, the injection temperature was maintained at 250 °C. The total run time was approximately 40 min. Detection was done using an electron ionisation (70 eV) mass spectrometer with a mass-to-charge ratio scan range of 50-500 atomic mass unit. The resulting mass spectra were compared to the National Institute of Standards and Technology Spectral Library 2021 for compound identification.
Liquid Chromatography-Mass Spectrometry (LC-MS) Analysis of Extracts
The analysis of plant extracts was conducted using a Thermo-Scientific Vanguish Flex Ultra-High Performance Liquid Chromatography coupled with a tandem TSQ Quantis MS instrument. Dried crude extracts were dissolved in methanol to make 0.25 mg/mL, then filtered through a 0.22 µm polytetrafluoroethylene syringe filter. A 2 µL aliquot of the solution was injected into the LC-MS/MS system. Chromatographic separation was performed using a Thermo-Scientific Zorbax Eclipse Plus reversed-phase column (100 mm × 2.1 mm, 1.7 µm particle size), maintained at 40 °C. A binary solvent system was used: mobile phase A consisted of water with 0.1% formic acid, and mobile phase B contained acetonitrile with 0.1% formic acid. The elution gradient started at 5% B, held for 2 min, then increased linearly to 95% B over 10 min, remained at 95% B until 16 min, and then re-equilibrated to the initial conditions from 16 to 20 min. The flow rate was set at 0.3 mL/min. During mass spectrometry, the electrospray ionisation source operated in both positive and negative ionisation modes with automatic switching. The source parameters were set at: First quadrupole mass analyser (Q1) resolution 0.7; spray voltage 3700 V (positive) and 2500 V (negative); ion transfer tube temperature 325 °C; vaporiser temperature 350 °C; sheath gas flow 10 L/min; and auxiliary gas flow 5 L/min. Data acquisition was performed using Thermo Scientific TraceFinder 7.2 software. MS/MS utilised the automatic Multiple Reaction Monitoring mode at a collision energy of 6 eV. Compound identification was based on comparison with the National Institute of Standards and Technology Library 2021 spectral database.
Fourier Transform Infrared Spectroscopy (FTIR) Analysis of the Polyherbal Formulation
To identify the functional groups of the phytochemicals in the formulated granulated capsules, FTIR analysis was performed to obtain a spectrum. The scanning range was from 400 to 4400 cm−1, with 32 scans per minute and a resolution of 4 cm−1.
Test Animals and Dose Preparation
An acute toxicity test was conducted on 12 female Swiss albino mice aged between 7 and 9 weeks (weighing 24 ± 2 g), which were procured from the Institute of Traditional Medicine, Tanzania. The sub-acute toxicity test used forty (40) Wistar rats (20 females and 20 males) aged between 8 and 10 weeks, weighing 108 ± 14 g for females and 135 ± 21 g for males. The rats were obtained from the Faculty of Veterinary Medicine at Sokoine University of Agriculture, Tanzania. All animals were kept in cages and administered with a continuous supply of standard food pellets and water. The standard food pellets were composed of 15% of crude protein, 5% of crude fibre, 4% of crude fat, 12% of moisture, and 64% of carbohydrate supplied by Salibaba Pellet Co. Ltd, Tanzania. Before the tests, the animals were acclimated to the laboratory setting for a week at 23 °C, following typical day and night cycles. The handling of animals complied with international guidelines for respecting animal rights, and Tanzania Animal Welfare Act on care and use of laboratory animals.27,28 Notably, all experimental protocols used in this study were approved by the Kibong’oto-Nelson Mandela-Cedha Health Research Ethical Committee (KNCHREC), and given an ethical approval NO. KNCHREC 00014/01/2024, Tanzania Republic. Furthermore, test doses were prepared from the dried EP extract mixture (1:1w/w) by dissolving it in 5% dimethyl sulfoxide (DMSO) to achieve concentrations of 2000 mg/Kg for acute toxicity testing and 250, 500, and 1000 mg/kg for sub-acute toxicity testing.
Acute Oral Toxicity Test
During experimentation, 6 female Swiss albino mice were given a single dose of 2000 mg/kg of EP extract via gavage. Simultaneously, another 6 mice, serving as the control group, received 5% DMSO (vehicle). The dose volume was calculated based on a 1 mL/100 g body weight ratio, as specified by Organisation for Economic Co-operation and Development (OECD) guideline 423. 29 Subsequently, all animals were observed for mortality, clinical signs, behavioural changes, body weights, and food intake over 14 days. On day fifteen, after an overnight fast of 12 h, 2% pentobarbital sodium solution (60 mg/kg) was injected intraperitoneally to anesthetise the mice. Then, the animals were dissected, and their kidneys and livers were collected for histological analysis.
Sub-Acute Oral Toxicity Test
During the investigation, Wistar rats were divided into 4 main groups of ten rats, which were further divided into male and female subgroups as directed by OECD guideline 407. 30 The first three groups were supplied with respective doses of 250, 500, and 1000 mg/kg of EP extract, administered daily via gavage for 28 days. Concurrently, the fourth bunch was administered with vehicle (5% DMSO) as the control group. The dose volume was calculated based on a 1 mL/100 g body weight ratio, as specified by OECD guideline 407. Afterward, the animals’ general behaviours, clinical signs, body weights, and dietary intake were monitored for the entire test period. Next to the 28th day, animals were anaesthetised with 2% pentobarbital sodium solution (60 mg/kg) after an overnight fast of 12 h. Later, about 5 mL of blood samples were obtained from each animal by heart puncture using disposable syringes. After that, the sacrificed animals were dissected to obtain the kidneys and liver organs for histological assessment.
Haematology Analysis
2.5 ml of each collected blood sample from each Wistar rat was stored in a Dipotassium ethylenediaminetetraacetic acid vacutainer. After which, they were examined using an Automatic haematology analyser (DH76, Shenzhen Dymind Biotechnology Co., Ltd). The parameters investigated were Red blood cells (RBC), Haemoglobin (HGB), Haematocrit (HCT), Mean Corpuscular Volume (MCV), Mean Corpuscular Haemoglobin (MCH), Mean Corpuscular Haemoglobin Concentration (MCHC), Platelets (PLT), White Blood Cells (WBC), Neutrophils (Neu), Lymphocytes (Lym), Monocytes (Mon), Eosinophils (Eos), and Basophils (Bas).
Biochemical Analysis
2.5 mL of each collected blood sample from each Wistar rat was stored in a plain Vacutainer, followed by centrifugation at 2000 rpm for 10 min to obtain serum. Thereafter, sera were analysed for alanine aminotransferase (ALT), alkaline phosphatase (ALP), aspartate aminotransferase (AST), albumin, bilirubin direct, creatinine, urea, total bilirubin, and total protein using an Automated Chemistry analyser (XL 200, Erba Diagnostics Mannheim GmbH, Biotechnology Company).
Histopathological Assessment
Once the liver and kidneys were obtained from mice and rats, they were immediately stored in 5% neutral buffered formalin and fixed for 72 h. The organ sections were then sequentially dehydrated in 70%, 80%, 95%, and 100% ethanol, followed by clearing in xylene. These sections were embedded in paraffin wax and sliced to a thickness of 5 μm. Afterward, they were mounted on microtome slides and stained with haematoxylin (H) and eosin (E) for analysis. 31 Finally, tissues were visualised at ×40, ×100 and ×400 magnifications, using a light microscope (LAB 40 Series Microscope, OPTA-TECH Ltd, Poland). The slides were analysed by two independent pathologists.
In Vivo Antimicrobial Efficacy Assessment of Polyherbal Formulation (Capsules) from EP Extracts (1:1) w/w Against Escherichia coli ATCC 25922 and Methicillin-Resistant Staphylococcus aureus (MRSA) ATCC 43300
Before the test, granulated capsules containing 50 g of extract, 20 g of starch, 700 mg of magnesium stearate, and 70 mg of sodium methylparaben were prepared using the wet granulation method described in previous studies.32,33 An In vivo susceptibility test was conducted on E. coli using 20 Wistar rats weighing between 180 and 220 g. After 6 days of acclimatisation to the laboratory setting, the animals were sorted into five groups of four rats each. All animals received an intraperitoneal injection of 1 mL of E. coli inoculum, approximately 1.5 × 108 CFU/mL. After 48 h, the development of infection was confirmed, followed by the initiation of treatment. Groups A, B, and C received 1 mL of extract at doses of 50, 150, and 450 mg/kg, respectively. Groups D and E were treated with 20 mg/kg of ciprofloxacin and distilled water, serving as positive and negative controls, respectively. All doses were administered orally via gavage every 12 h until the infection cleared from the blood. Similarly, 1 mL of 3 × 108 CFU/mL MRSA ATCC 43300 was injected intraperitoneally into Wistar rats, which were then tested against the polyherbal capsules at comparable doses for 14 days. Doses were chosen not to exceed the No-Observed-Adverse-Effect Level (NOAEL of 500 mg/kg body weight) established in the sub-acute toxicity study. To explore a wider efficacy range for the polyherbal formulation, doses were set at threefold intervals based on OECD 423 and 407 guidelines, to identify both the minimum and optimal effective doses. Additionally, the selected doses are comparable to those reported in previous in vivo antibacterial studies using animal models.34,35 Enumeration of E. coli and MRSA was done using standard microbiological procedures. Blood samples (100 µL) from each animal were collected every two days from the lateral tail vein, serially diluted in peptone water, cultured, and colony counts performed. Culturing was carried out on Crystal violet MacConkey agar (for E. coli) and mannitol salt agar (for MRSA) plates, by plating 100 µL of the diluted sample. Plates were then incubated at 35 ◦C overnight. Bacterial colonies were initially identified based on morphology and colour, and subsequently confirmed through Gram staining and Indole tests as biochemical assays. Presumed E. coli and MRSA colonies from the incubated MacConkey and Mannitol salt agar plates, respectively, were picked and further cultured in Sulphide Indole Motility (SIM) medium for 24 h. Afterwards, a few drops of Kovacs reagent were added, and the formation of a purple or pink ring at the surface confirmed the presence of E. coli. Additionally, motility and sulphur production tests were performed in SIM medium further to confirm the presence of E. coli and MRSA. Finally, all animals were treated with gentamycin.
Statistical Analysis
In this study, the values for body weight gains, haematological and biochemical indices are expressed as means and standard deviations. The significant difference was ascertained by one-way Analysis of Variance and Tukey's post hoc test using Genstat 12th edition. The p-value < 0.05 at a 95% confidence level indicated that the differences in results were deemed significant.
Results
Phytochemical Screening Results
The screening of secondary metabolites confirmed that saponnins, steroids, flavonoids, tannins, terpenoids, and alkaloids were present in both E. hirta and P. barabatus ethanolic extracts, except for anthraquinone.
GC-MS Compound Identification
The analysis unveiled 45 and 39 phytocompounds from E. hirta and P. barbatus, respectively (Supplementary Tables 1 & 2). 1,2,3-Benzenetriol (28.29%), 9, 12, 15-Octadecatrienoic acid, (Z,Z,Z)- (22.72%), n-Hexadecanoic acid (9.18%), 9-Octadecenamide (Z)- (6.31%), and Phytol (3.01%) were confirmed in E. hirta extract, whereas, 1,2,3-Benzenetriol (71.10%), 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- (4.32%), Benzeneacetaldehyde (4.12%) and 2-Furancarboxaldehyde, 5-methyl- (1.87%) in P. barbatus extract, as most abundant phytocompounds.
LC-MS Compound Identification
The analysis identified 27 and 6 compounds in P. barbatus and E. hirta extracts respectively (Supplementary Tables 3 & 4). 3-Quinolinecarboxylic acid, Emodin, Gymnemagenin, Azadirachtin A, Gambogic acid, Azadirachtin B, and Acetamide are among the identified bioactive compounds which have been documented to possess antimicrobial activities.
FTIR Results
The analysis revealed functional groups that indicate the presence of various phytocompounds in the granulated capsules. It established absorption between 3282.94 cm−1 and 419.34 cm−1 (Supplementary Figure 1). The spectrum displayed a broad, smooth band peak at 3282.94 cm−1 (O–H stretching), signifying the presence of alcohol and hydroxyl groups, including compounds such as 9,12,15-Octadecatrien-1-ol, phenol, catechol, and 12,3-benzenetriol. A pronounced peak at 2923.92 cm−1 (C–H bending) is attributed to alkanes, including cyclohexane. The peak at 1708.97 cm−1 (C = O stretching) indicates aliphatic ketones, including cyclohexanone, butyrolactone, and 3-methyl-2-cyclopenten-1-one. Further, the peak at 1604.13 cm−1 (C = C stretching) is associated with conjugated alkenes, including 1-silacyclohexa-2,5-diene and 1-silacyclo-2,4-hexadiene.
Behavioural and Clinical Toxicity Observations
No deaths, unusual behaviours, or signs of lethality were noticed in mice and rats during the investigation of the toxicity tests. Moreover, all animals had normal food and water consumption.
Animal Body Weight Tracking
All animals in the acute toxicity test exhibited positive growth at the end of the test, with percentage body weight gains of 22.8% and 27.41% for the control and 2000 mg/kg groups, respectively. In the sub-acute toxicity test, the percentage body weight gains ranged from 23% to 25% for females and 37% to 40% for male rat groups (Supplementary Figures 2 & 3). Furthermore, animals in the EP extract-treated groups showed no significant difference in body weight gain compared to those in the control groups for both acute and sub-acute toxicity assessments (Acute toxicity in female mice: p = 0.138; sub-acute toxicity in males and females: p = 0.950 and 0.993, respectively).
Haematological Findings from sub-Acute Toxicity Test
The results (Table 1) of the haematological parameters for animals in the treatment and control groups were not statistically different, except for basophils in male test subjects from all EP extract-treated groups, which were significantly lower compared to the control group (p < .001). However, all haematological values fall within the acceptable range for all groups.
Haematological Indices of Animals in Sub-Acute Toxicity Test.

Key: RBC = Red blood cells, HGB = Haemoglobin, HCT = Haematocrit, MCV = Mean Corpuscular Volume, MCH = Mean Corpuscular Haemoglobin, MCHC = Mean Corpuscular Haemoglobin Concentration, PLT = Platelets, WBC = White Blood Cells, Neu = Neutrophils, Lym = Lymphocytes, Mon = Monocytes, Eos = Eosinophils, Bas = Basophils. Haematological indices are expressed as means ± standard deviations,
Serum Biochemical Findings from sub-Acute Toxicity Test
Table 2 presents the biochemical indices results for animals in the sub-acute toxicity study. The 250 and 500 mg/kg male dose groups had significantly higher urea levels (p = 0.015) compared to the control group. Similarly, the 500 and 1000 mg/kg male dose groups showed increased levels of ALP (p = 0.017) relative to the control. For the other parameters, the analysis indicated no significant difference between the EP extract-treated and control groups (p > 0.05). Furthermore, aside from ALP, all biochemical values stay within acceptable ranges across all groups.
Biochemical Indices of Animals in a Sub-Acute Toxicity Test.

Key: ALT = Alanine aminotransferase, ALP = Alkaline phosphatase, AST = Aspartate aminotransferase. Biochemical indices are expressed as means ± standard deviations;
Histopathological Evaluation in Acute and sub-Acute Toxicity Tests
In the acute oral toxicity test (Figure 1), microscopic examination of the liver and kidneys of all mice showed no significant morphological lesions. The analysis of these organs revealed no notable effects in mice treated with EP extract compared to the control group. The typical renal architecture in all animals was preserved, including normal Bowman's capsules, glomeruli, distal convoluted tubules, proximal convoluted tubules, loops of Henle, and normal cellularity at the cortex and medullary parts in the kidneys. Meanwhile, their livers showed normal endothelial cells, bile ducts, central veins, sinusoids, portal veins, and hepatocytes.
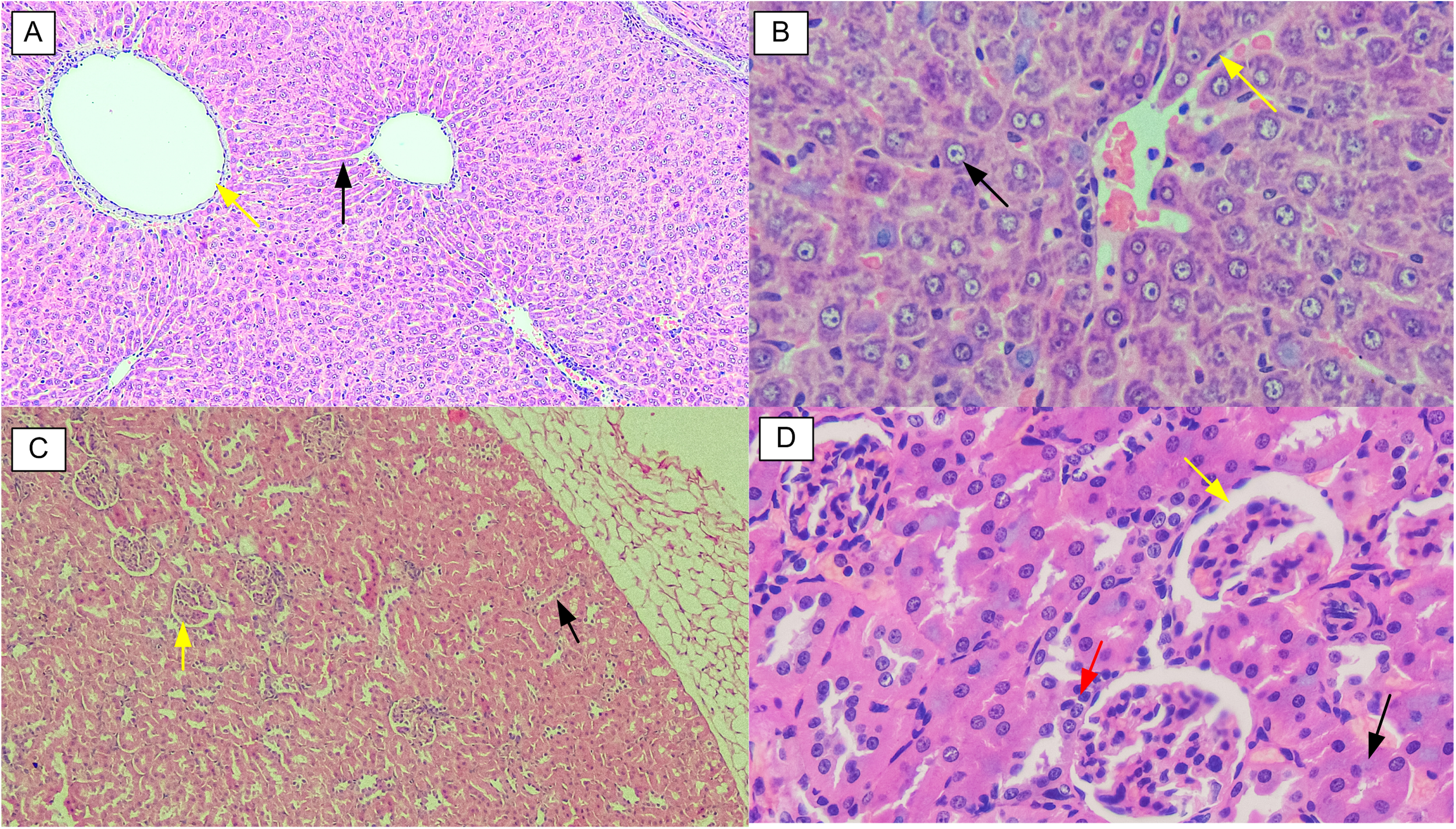
Liver and Kidney Sections from Female Swiss Mice (Haematoxylin & Eosin Stained) in the Acute Oral Toxicity Assessment. A – Liver (Control Group): Shows a Normal Central Vein (Yellow Arrow) with Surrounding Sinusoids (Black Arrow); B – Liver (2000 mg/kg Dose Group): Displays Normal Hepatocytes (Black Arrow) and Kupffer Cells (Yellow Arrow); C – Kidney (Control Group): Shows Normal Glomerulus (Yellow Arrow) and Renal Tubules (Black Arrow); D – Kidney (2000 mg/kg Dose Group): Shows Normal Bowman's Capsule (Yellow Arrow), Proximal Convoluted Tubules (Black Arrow), and Macula Densa (Red Arrow).
In the sub-acute toxicity test (Figures 2 and 3), microscopic analysis revealed typical kidney and liver morphology in the control, 250, and 500 mg/kg dose groups. Tissues from animals in these groups displayed normal central veins with sinusoids, normal hepatocytes with round nuclei, evenly distributed reticuloendothelial cells, spindle-shaped Kupffer cells, and normal portal triads with bile ducts in their livers. Similarly, these animals had normal glomeruli with a substantial distribution of podocytes and mesangial cells, Bowman's capsules with double layers of epithelial visceral and parietal tissues, distal convoluted tubules with clear lumens and dense cuboidal epithelium, and proximal convoluted tubules with some larger vacuolated cytoplasm and fuzzy lumens in their kidneys. Conversely, some animals in the highest dose group exhibited mild interstitial haemorrhage, oedematous interstitial tissue, proximal tubular vacuolation, and mild haemorrhagic glomeruli (slightly infiltrated with RBCs) of their kidneys compared to controls. At the same time, some animals had central veins with disrupted endothelium (loss of cellular structure), and hepatocytes that exhibited mild karyorrhesis and vacuolation due to slight degeneration of the cytoplasm (necrosis) in their livers relative to the control group.
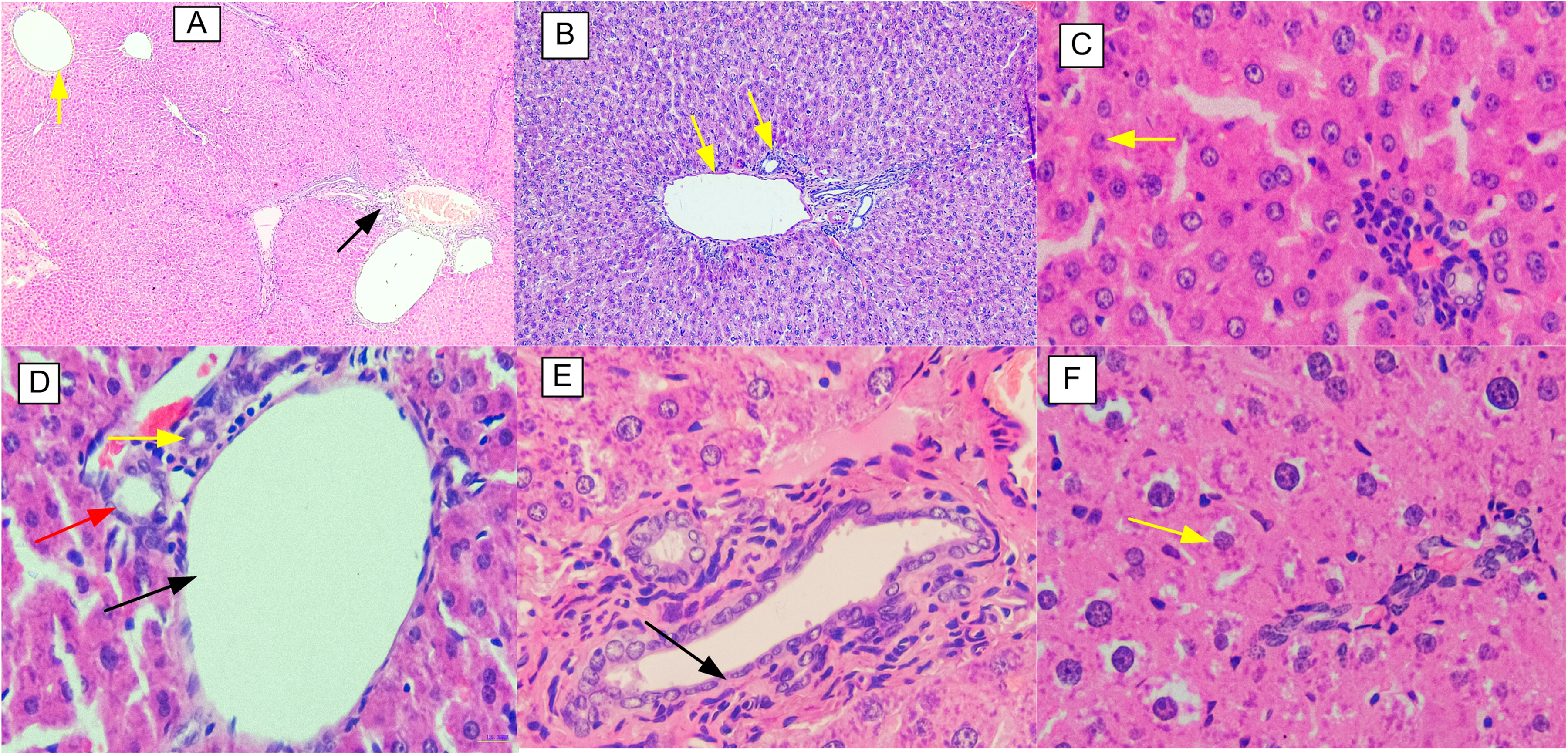
Liver Sections from Male and Female Wistar Rats (Haematoxylin & Eosin Stained) in the sub-Acute Toxicity Assessment. A-Female (Control Group): Shows Normal Central Vein Surrounding Sinusoids (Yellow Arrow) and Portal Triad (Black Arrow); B-Female (250 mg/kg Dose Group): Shows Normal Portal Triad (Yellow Arrow); C- Female (1000 mg/kg Dose Group): Shows Hepatocytes Undergoing Slight Degeneration of Cytoplasm (Yellow Arrow); D-Male (Control Group): Shows Hepatic Artery (Yellow Arrow), Portal Vein (Black Arrow), and Bile Duct (red Arrow) of Normal Portal Triad; E-Male (250 mg/kg Dose Group): Shows Normal Endothelium of Portal Vein (Black Arrow); and F- (Male 1000 mg/kg Dose Group): Shows Hepatocytes Undergoing Slight Degeneration of Cytoplasm (Yellow Arrow).
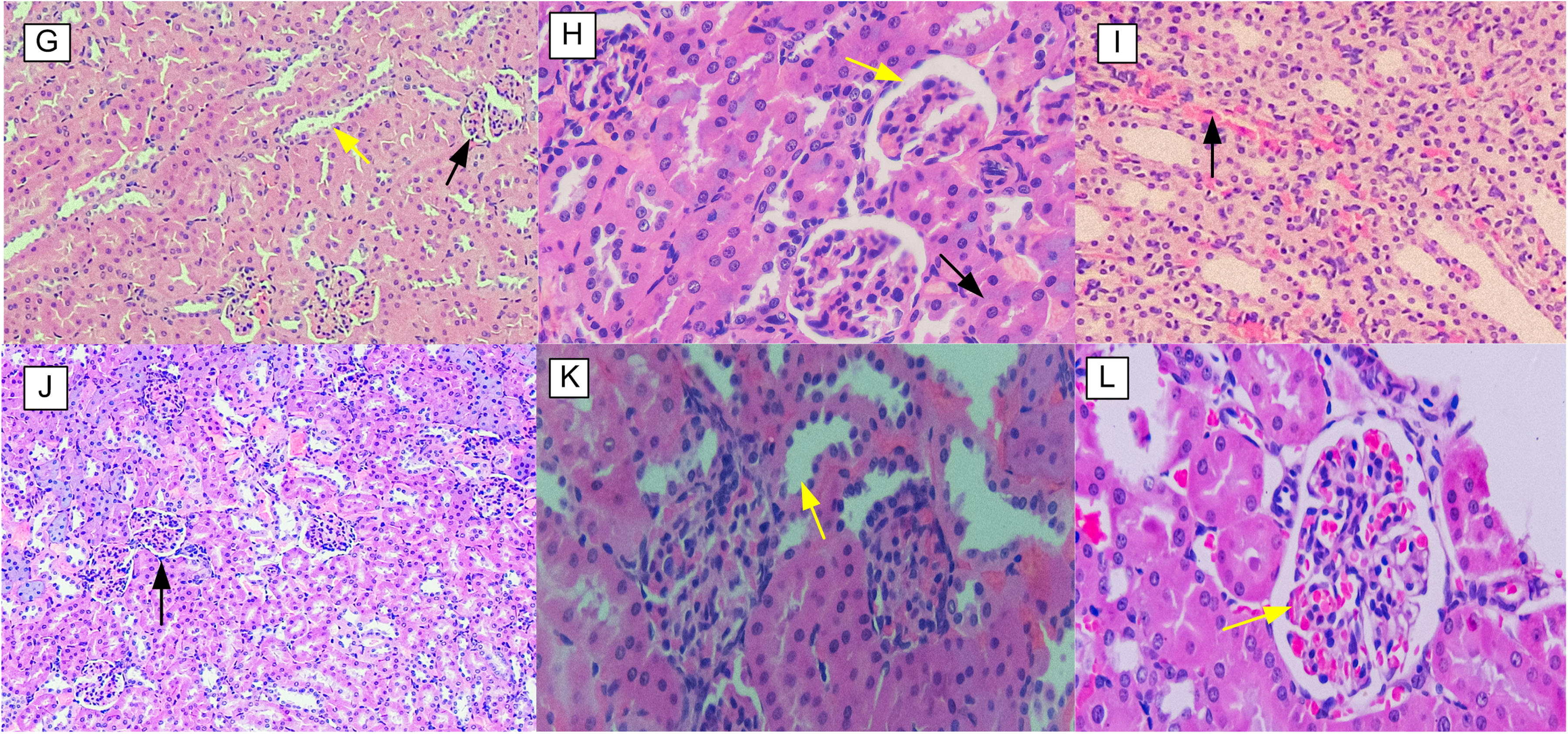
Kidney Sections from Male and Female Wistar Rats (Haematoxylin & Eosin Stained) in the Sub-Acute Toxicity Assessment. G-Female (Control Group): Displays Normal Glomerulus (Black Arrow) and Renal Tubules (Yellow Arrow); H-Female (250 mg/kg Dose Group): Shows Normal Bowman's Capsule (Yellow Arrow) and Proximal Convoluted Tubule (Black Arrow); I-Female (1000 mg/kg Dose Group): Shows Mild Interstitial Haemorrhage (Black Arrow); J-Male (Control Group): Shows Normal Glomerulus (Black Arrow); K-Male (250 mg/kg Dose Group): Shows Distal Convoluted Tubule with Normal Cellularity (Yellow Arrow); and L-Male (1000 mg/kg Dose Group): Shows Mild Haemorrhage in Glomerulus (Yellow Arrow).
In Vivo Antimicrobial Efficacy Results
The results (Supplementary Figure 4) show that doses of 150 and 450 mg/kg cleared the E.coli loads by the eighth day of the test. Similarly, the lower dose of 50 mg/kg cleared it by day 10. In comparison, ciprofloxacin cleared the load by day 6. On the other hand, the negative control group exhibited an initial increase in E. coli load and a decrease towards the end of the test. During the test, one rat in the 150 mg/kg dose group died on day 2, although the cause was not determined, whether it was due to E.coli infection or mechanical injury during oral gavage. On the other hand, doses of 450 and 150 mg/kg of the formulation cleared the MRSA infection by the 10th and 14th days, respectively (Supplementary Figure 5). At the same time, a 20 mg/kg dose of ciprofloxacin, used as a positive control, cleared the MRSA by day 8 (Supplementary Figure 5). All untreated animals developed some clinical signs such as lethargy, tremors, and decreased eating habits. Of the four rats, two from the untreated group died on day 9.
Discussion
The use of herbal remedies in healthcare is gaining world-wide recognition.1 However, evidence to support the safety of most polyherbal formulations for humans use remains limited. Therefore, this study investigated the toxicity of the EP extract (1:1 w/w) using Swiss albino mice and Wistar rats. In this acute oral toxicity study, the EP extract showed no immediate toxic effects in female Swiss albino mice, with a Lethal Dose 50% (LD50) greater than 2000 mg/kg. These findings suggest that the phytochemicals in the EP extract are non-toxic at doses up to 2000 mg/kg. Additionally, the results of the histopathological analysis (Figure 1) revealed that all mice treated with EP extract exhibited no substantial changes in the normal morphology of the liver and kidneys compared to the control group. This indicates that the phytochemicals in the tested EP extract dose were non-toxic to the animal's liver and kidneys. Therefore, its LD50 exceeds 2000 mg/kg, although in some cases, the manifestation of acute toxicity from the orally administered extract can be hindered by its low dissolution and absorption. 31 The acute oral toxicity results for the combined EP extract in this study are consistent with other research, which is based on toxicity profiles of individual extracts from E. hirta and P. barbatus.15,17
The information on acute toxicity has limited therapeutic significance unless supported by long-term toxicity tests. In this context, the current study investigated the sub-acute toxicity effects of the combined EP extract in rats. Concerning body weight changes, the results showed that all rats experienced normal growth throughout the entire test period. According to OECD guidelines, if the tested animals do not show a loss of 20% or more of their body weights over 7 days or longer, 36 then, such an observation indicates that, the doses of EP extract and vehicle (5% DMSO) used in the study had no harmful effects on the growth of the test animals. In this regard, the findings suggest that the phytochemicals in the extracts do not interfere with the biological processes or cells involved in rat growth.
Haematological data serve as essential biomarkers for assessing the toxicity caused by various toxic substances.37,38 In this study, analysis of blood parameters revealed that the tested extract doses had no substantial effect on haematopoiesis. There were no notable changes in haematological parameters between the EP extract-treated and control groups, except for basophils, which were significantly lower in all EP extract-treated groups in male animals (p ˂ 0.001). Notably, all animals maintained haematological values within acceptable ranges. The normal range for WBC count in male Wistar rats is 3.20-10.50; the WBC count in male rats was on the lower end of this range in both the control and EP extract-treated groups. This suggests that the reduced WBC count or production could be due to environmental factors such as diet or the vehicle (5% DMSO) used, rather than the administered extract. Therefore, the lowered WBC count warrants further investigation. The haematological parameters reported in this study are consistent with data reported elsewhere.39,40
The damage to the liver and kidneys caused by diseases or exposure to toxic substances, including plant extracts, can be revealed through analysis of biochemical parameters.41,42 In this current sub-acute toxicity assessment, biochemical analysis of urea and creatinine was carried out to evaluate the effect of the EP extract on kidney function. The results from the biochemical indices revealed no remarkable changes in creatinine levels between the EP extract-treated and control groups. In contrast, however, urea levels in the 250 and 500 mg/kg EP extract-treated male rats were significantly higher compared to the control group (p ˂ 0.015). These elevated urea levels were dose-independent, indicating the observed changes could be due to random variation rather than the effect of the EP extract. Although, the 250 and 500 mg/kg dose groups showed urea levels of 26.24 and 26.25 mg/dL, respectively, compared to 23.34 mg/dL of the control group, the levels in both female and male rats remained within the normal lower levels, as the maximum acceptable levels are 48.2 and 60.74 mg/dL for male and females Wistar rats, respectively. Overall, since, urea and creatinine levels stayed within the acceptable range across all groups, this suggests that the tested EP extract did not produce significant toxicity or did not substantially impair kidney excretory function of the kidneys in the experimental animals.
On the other hand, liver enzymes and proteins play a crucial role as important biomarkers for assessing liver health. No substantial changes were observed in the levels of total protein, albumin, AST, and ALT between the EP extract and control groups, except for ALP in the 500 and 1000 mg/kg male treatment groups. The male rats in these groups showed notably higher ALP levels compared to the reference group (p ˂ 0.017). Additionally, the ALP values in both the EP extract and control groups were outside the acceptable range. In this context, this difference alone does not indicate that these doses cause toxicity to the liver tissues, unless the control group's ALP values are within the acceptable limits while the treatment groups’ values are outside. Therefore, this study cannot confirm that the EP extract has a toxic effect on ALP production, since both groups’ values fell outside the acceptable limits. However, the slight, dose-dependent trend of increasing liver enzyme levels suggests that at higher doses, the EP extract could influence hepatic secretory function. The abnormally high ALP levels observed cannot be attributed to the tested EP extract, as both control and treatment groups had values beyond acceptable ranges; instead, they may be caused by other factors, such as diet or the vehicle used to prepare the doses. Nonetheless, the observed difference between the control and treatment groups is attributed to the effect of the EP extract. This observation has been reported elsewhere.43,44 Moreover, the biochemical data presented in this study align with findings from other studies.31,45 In summary, urea and albumin levels were at lower normal limits, AST and total protein at intermediate levels, and creatinine and ALT within the upper limits of acceptable ranges in both groups. Therefore, these results suggest that the EP extract did not have a significant impact on the kidney or liver tissues.
The results from histopathological analysis show that there were no significant differences in the morphology of kidney and liver tissues among the control, 250, and 500 mg/kg dose groups. This aligns with the results from haematological and biochemical analyses at similar doses, indicating that the EP extract did not cause significant damage to liver, kidney, spleen, or bone marrow cells involved in the production of RBC, WBC, and platelet cells (haematopoiesis). Contrary, the results demonstrate that some animals from the highest dose group exhibited mild morphological changes in both liver and kidney tissues compared to the control. This suggests that, at doses above 500 mg/kg the phytochemicals in the EP extract may have deleterious effect on vital organs such as the liver and kidneys. Usually, changes in biochemical indices are reflected in the histopathological analysis of these organs. Unexpectedly, the histopathological analysis results in the highest dose group are inconsistent with those of haematological and biochemical parameters at similar dose. This finding indicates that the morphological changes observed in the liver and kidneys were not substantial enough to induce changes in biochemical parameters. Similar observations have been reported in previous studies.45,46 In this context, since the results from haematological, biochemical and histopathological analyses consistently show that the EP extract has no toxic effect at doses up to 500 mg/kg, these findings suggest that the NOAEL for this extract is 500 mg/kg body weight.
The notable morphological changes detected in the kidneys and liver tissues of rats in the highest dose indicate the toxic effects of the combined EP extracts at dose of 1000 mg/kg and above. Contrary to previous findings, based on individual extracts,14,15 the current study demonstrates relatively higher toxicity of the EP extract. Therefore, the higher toxicity of the EP extract observed in this study, compared to individual extracts, may be ascribed to either the additive toxic effects of the interacting phytocompounds in the combined extracts or environmental factors that influence both the phytoconstituents and biological factors. 47
E.coli and MRSA are among the major causes of UTIs. 48 Considering that such infections are among those treated with a decoction from the E. hirta and P. barbatus polyherbal mixture, the EP extract's polyherbal formulation (capsulated granules) was tested in vivo against them. This polyherbal formulation demonstrated promising therapeutic outcomes against E. coli and MRSA infections in Wistar rats. For E. coli, test doses of 150 and 450 mg/kg resolved the infection by the eighth day of treatment. In comparison, ciprofloxacin (20 mg/kg) cleared the infection by day 6. Concurrently, the 150 and 450 mg/kg doses of the formulation successfully cleared the MRSA infection by the 14th and 10th days of treatment, respectively. In contrast, the 50 mg/kg dose failed to eliminate the infection within 14 days, suggesting that the concentration of bioactive compounds was too low to eradicate all microbes. Regarding the E. coli test, no deaths were reported among the rats in the negative control group during the study, as the E. coli ATCC 25922 strain used does not produce verotoxin, which can cause severe health complications. Meanwhile, two rats died on day 9 after showing clinical signs, indicating the virulence of the pathogenic MRSA ATCC 43300 strain.
The extrapolation of animal doses for a specific herbal formulation to therapeutic use in humans depend on the established toxicity profiles and in vivo efficacy doses determined in model animals, including rats. In the present study, the NOAEL for the EP extract determined from the sub-acute toxicity study is 500 mg/kg body weight. The in vivo efficacy doses of its polyherbal formulation were 50 and 150 mg/kg/day for E.coli and MRSA, respectively. In this context, the Human Equivalent Dose (HED) was calculated using the formula provided below, as described by Wojcikowski and Gobe,
49
based on adult individuals weighing 60 kg and more. The formula shown below is recommended by the U.S Food and Drug Administration regulation for converting doses from preclinical studies using model animals.
50
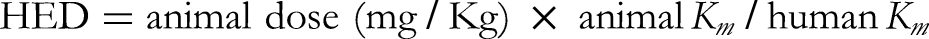
Moreover, the current study revealed the presence of saponnins, steroids, flavonoids, tannins, terpenoids, and alkaloids in the ethanolic extracts of E. hirta and P. barbatus. These secondary metabolites are often attributed to the therapeutic benefits of the herbal plants, including their potential as antimicrobial agents. Notably, GC-MS and LC-MS results showed the presence of numerous compounds in both E. hirta and P. barbatus ethanolic extracts. Among those, 29 and 24 from E. hirta and P. barbatus, respectively, possess antimicrobial activity (Supplementary Tables 1, 2, 3, & 4). The findings from GC-MS and LC-MS correlate with known bioactive compounds reported in the literature. Specifically, this study identified phytocompounds, such as 2-methoxy-4-vinylphenol, phenol, 1,2,3-benzenetriol, eugenol, phytol, quercetin, catechol, salicylic acid, oleic acid 5-hydroxymethylfurfural, orcinol, scopoletin, and furfural,51–55 which have previously been reported to exhibit antimicrobial activity. These compounds were detected via GC-MS analysis and may be responsible for the observed in vivo antimicrobial effects of the formulation. These phytochemicals exert their antimicrobial effects through several mechanisms: thymol, 1,2,3-benzenetriol, catechol, eugenol, and orcinol disrupt microbial cell membranes, causing leakage of cellular contents 56 ; 2-methoxy-4-vinylphenol inhibits cell wall synthesis 56 ; phenol, furfural, and scopoletin inactivate key metabolic enzymes 56 ; 1,2,3-benzenetriol and quercetin interfere with nucleic acid and protein synthesis 57 ; and salicylic acid, oleic acid, and phytol inhibit efflux pumps. 58 Furthermore, the preliminary phytochemical analysis and FTIR spectral peaks suggest the presence of phenolics, flavonoids, tannins, steroids, and terpenoids, which are widely documented for their antibacterial properties. 56 Hence, this further supports that these phytochemicals are responsible for the observed in vivo efficacy of the polyherbal formulation.
Despite their pharmacological benefits, some groups of phytochemicals, including alkaloids and terpenoids are reported to have toxic effects in humans and other animals.59,60 The GC-MS analysis of EP extracts revealed the presence of alkaloids, terpenoids and other compounds known to be toxic to humans and animals, such as phytol, catechol, cyclohexanone, phenol, 1,2,3-benzenetriol, furfural, and 5-hydroxymethylfurfural.61,62 The toxicity level caused by these phytochemicals mainly depends on the amount consumed by the animal. Therefore, among other factors, these compounds may be responsible for the sub-acute oral toxicity observed in animals at the highest EP extract dose (1000 mg/kg) used in the current study.
In this study, the polyherbal capsules were tested against a limited number of microbial strains, including a Gram-negative bacterium (E. coli) and a Gram-positive, antimicrobial-resistant strain (MRSA). This results in a narrow scope of microbial testing over a broader range of UTI pathogens. Concurrently, the in vivo antimicrobial efficacy was evaluated in Wistar rats at doses of 50, 150, and 450 mg/kg, conducted at threefold intervals following OECD guidelines 423 and 407, to explore a broader range of effective doses. However, the study did not test short dose intervals, including at least five dose levels, such as 50, 100, 150, 200, 250, 300, 350, and 400 mg/kg, which would allow for the determination of the half maximal inhibitory concentration (IC50) for comparability and pharmacological insight. Additionally, the study assessed only the acute and sub-acute toxicity of the ethanolic EP extract. To determine its long-term toxicity profiles, sub-chronic and chronic toxicity studies are necessary.
Conclusion and Recommendations
Based on an acute toxicity study, the EP extract was found to be non-toxic when given as a single oral dose. It was classified as category 5 in the Globally Harmonised System of Chemicals Classification, indicating low toxicity. In a sub-acute toxicity study, doses up to 500 mg/kg, showed that both haematological, biochemical, and histopathological indices consistently demonstrated that the EP extract was non-toxic. This suggests that the NOAEL for this extract is 500 mg/kg body weight. For human therapeutic use, the minimum effective in vivo doses of the polyherbal capsules are estimated at 50 mg/kg and 150 mg/kg/day for E. coli and MRSA, respectively, which correspond to HEDs of 8 mg/kg and 24.32 mg/kg /day against these infections, respectively. These doses are considered safe, as they do not exceed the NOAEL. The study recommends further research involving a broader range of clinically relevant uropathogens at short-dose intervals, as well as sub-chronic and chronic toxicity studies, to enhance clinical relevance.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251388330 - Supplemental material for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation
Supplemental material, sj-docx-1-npx-10.1177_1934578X251388330 for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation by Optatus Mwalongo, Lydia Kisula, David Credo and Elingarami Sauli in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251388330 - Supplemental material for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation
Supplemental material, sj-docx-2-npx-10.1177_1934578X251388330 for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation by Optatus Mwalongo, Lydia Kisula, David Credo and Elingarami Sauli in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X251388330 - Supplemental material for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation
Supplemental material, sj-docx-3-npx-10.1177_1934578X251388330 for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation by Optatus Mwalongo, Lydia Kisula, David Credo and Elingarami Sauli in Natural Product Communications
Supplemental Material
sj-docx-4-npx-10.1177_1934578X251388330 - Supplemental material for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation
Supplemental material, sj-docx-4-npx-10.1177_1934578X251388330 for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation by Optatus Mwalongo, Lydia Kisula, David Credo and Elingarami Sauli in Natural Product Communications
Supplemental Material
sj-docx-5-npx-10.1177_1934578X251388330 - Supplemental material for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation
Supplemental material, sj-docx-5-npx-10.1177_1934578X251388330 for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation by Optatus Mwalongo, Lydia Kisula, David Credo and Elingarami Sauli in Natural Product Communications
Supplemental Material
sj-docx-6-npx-10.1177_1934578X251388330 - Supplemental material for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation
Supplemental material, sj-docx-6-npx-10.1177_1934578X251388330 for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation by Optatus Mwalongo, Lydia Kisula, David Credo and Elingarami Sauli in Natural Product Communications
Supplemental Material
sj-docx-7-npx-10.1177_1934578X251388330 - Supplemental material for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation
Supplemental material, sj-docx-7-npx-10.1177_1934578X251388330 for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation by Optatus Mwalongo, Lydia Kisula, David Credo and Elingarami Sauli in Natural Product Communications
Supplemental Material
sj-docx-8-npx-10.1177_1934578X251388330 - Supplemental material for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation
Supplemental material, sj-docx-8-npx-10.1177_1934578X251388330 for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation by Optatus Mwalongo, Lydia Kisula, David Credo and Elingarami Sauli in Natural Product Communications
Supplemental Material
sj-docx-9-npx-10.1177_1934578X251388330 - Supplemental material for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation
Supplemental material, sj-docx-9-npx-10.1177_1934578X251388330 for Phytochemical Composition, Toxicity, and in vivo Antimicrobial Properties of Euphorbia hirta and Plectranthus barbatus Ethanolic Extracts’ Polyherbal Formulation by Optatus Mwalongo, Lydia Kisula, David Credo and Elingarami Sauli in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to acknowledge the management and staff of the Institute of Traditional Medicine, Muhimbili University of Health and Allied Sciences (MUHAS), for technical support and the use of laboratories.
Ethical Approval
This study was approved by the Kibong’oto-Nelson Mandela-Cedha Health Research Ethical Committee (KNCHREC), Tanzania Republic, and given an ethical approval NO. KNCHREC 00014/01/2024.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the animal care guidelines approved by KNCHREC, Tanzania Republic.
CRediT Authorship Contribution Statement
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Mbeya University of Science and Technology through High Education for Economic Transformation (HEET) project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
